GO OD MO RN ING DENTURE BASE RESINS

GO OD MO RN ING …
DENTURE BASE RESINS…

Introduction: Syenthetic resins/plastics are used In variety of dental applications such as; -dentures (bases, artificial teeth). -cavity filling materials (composites). -impression materials. -equipments (mixing bowels). -resin based cements.

History: Before 1937, materials used were -vulcanite -nitrocellulose -phenol formaldehyde - vinyl plastics - porcelain
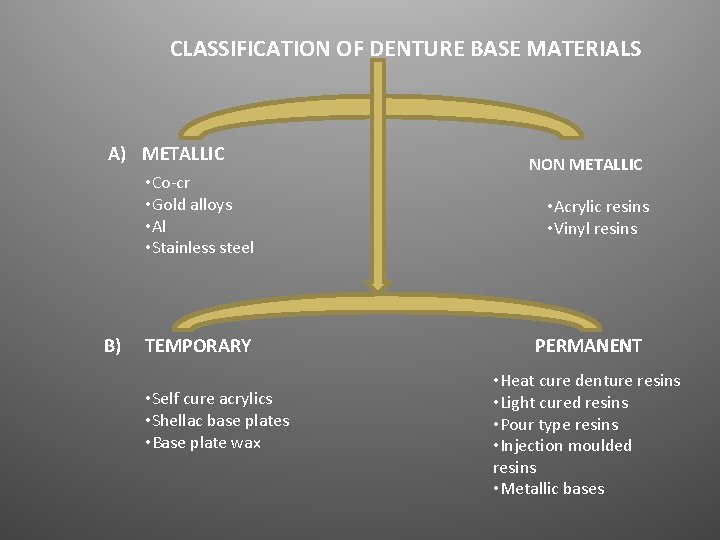
CLASSIFICATION OF DENTURE BASE MATERIALS A) METALLIC • Co-cr • Gold alloys • Al • Stainless steel B) TEMPORARY • Self cure acrylics • Shellac base plates • Base plate wax NON METALLIC • Acrylic resins • Vinyl resins PERMANENT • Heat cure denture resins • Light cured resins • Pour type resins • Injection moulded resins • Metallic bases
SYNTHETIC RESINS: -These are non metallic compounds which are moulded into various forms and then hardened for commercial use -These materials are composed of polymers or complex molecules of high molecular weight.

CLASSIFICATION OF RESINS (Based on thermal behavior) THERMOPLASTIC Ø Can be repeatedly softened, moulded under heat and pressure without any chemical changes. Ø Fusible Ø Soluble in organic solvent THERMOSET Ø Can be moulded once

IDEAL REQUIREMENTS : 1. TASTLESS, ODOURLESS, NON TOXIC, NON IRRITANT TO ORAL TISSUES. 2. ESTHETICALLY SATISFACTORY. 3. DIMENSIONALLY STABLE. 4. SHOULD HAVE ENOUGH STRENGTH, RESILIENCE &ABRASION RESISTANCE. 5. INSOLUBLE &IMPERMEABLE TO ORAL FLUIDS. 6. LOW SPECIFIC GRAVITY. 7. SHOULD ABLE TO TOLERATE TEMPARATURE. 8. EASY TO FABRICATE AND REPAIR. 9. GOOD THERMAL CONDUCTIVITY. 10. RADIO OPAQUE. 11. LOW COST.
USES: - 1. FABRICATION OF DENTURE. 2. ARTIFICAL TEETH. 3. TOOTH RESTORATION. 4. CEMENTATION OF ORTHODONTIC BRACKETS, CROWN & BRIDGES. 5. ORTHODONTIC & PEDO DONTIC APPLIANCES. 6. MAXILLOFACIAL PROSTHESIS. 7. INLAY & POST CORE PATTERNS. 8. TEMPORARY CROWNS AND BRIDGES. 9. ENDODONTIC AND CORE FILLING MATERIALS. 10. ATHELITIC MOUTH PROTECTOR. 11. SPLINTS AND STENTS.



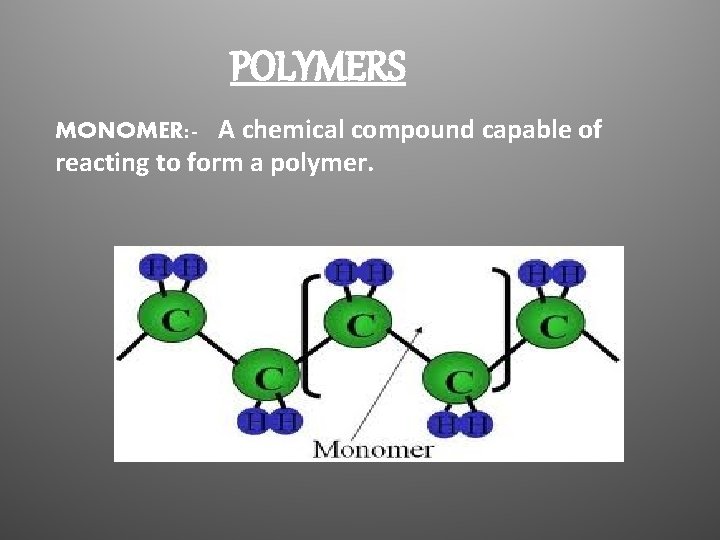
POLYMERS MONOMER: - A chemical compound capable of reacting to form a polymer.

Polymer: - chemical compound consisting of large organic molecules formed by the union of many repeating smaller monomer unit.
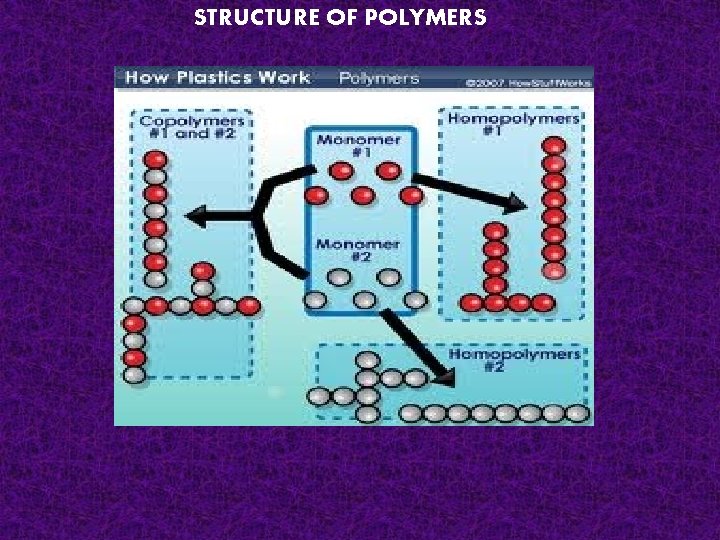
STRUCTURE OF POLYMERS

1. Linear polymer; in this the ‘mer’ units are connected to each other in a linear sequence.

LINEAR HOMOPOLYMER: - -A-A-ARANDOM COPOLYMER LINEAR TYPE: - -A-A-B-B-A-A-BBLOCK COPOLYMER : - -A-A-A-B-B-B-
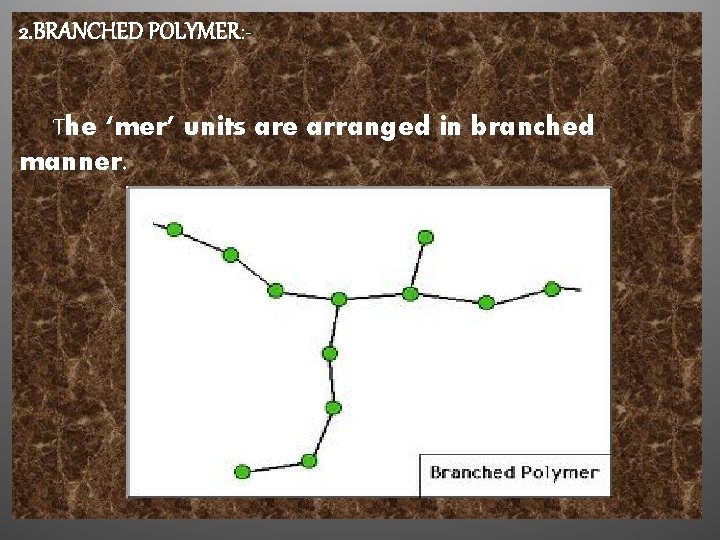
2. BRANCHED POLYMER: The ‘mer’ units are arranged in branched manner.
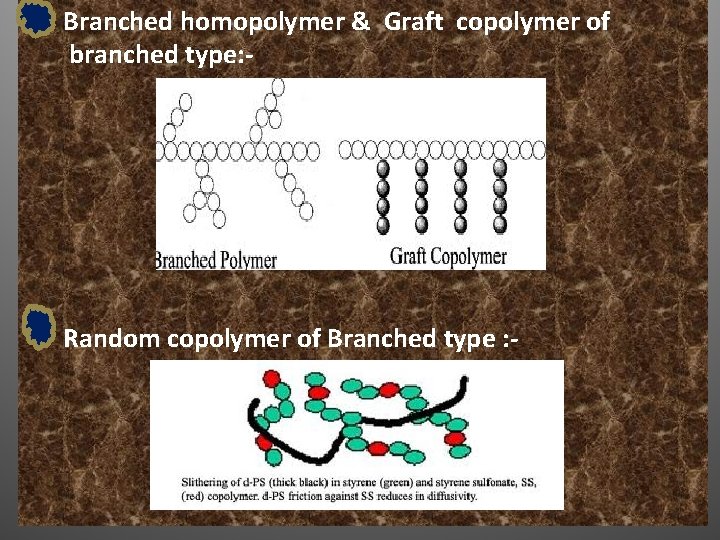
Branched homopolymer & Graft copolymer of branched type: - Random copolymer of Branched type : -
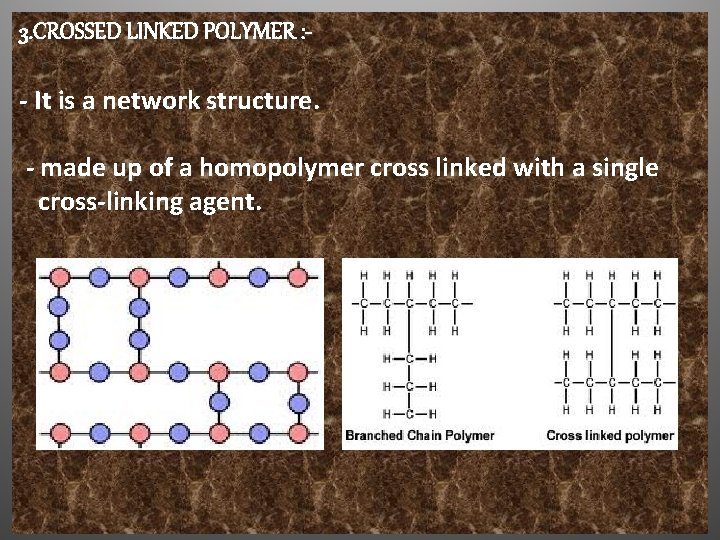
3. CROSSED LINKED POLYMER : - It is a network structure. - made up of a homopolymer cross linked with a single cross-linking agent.
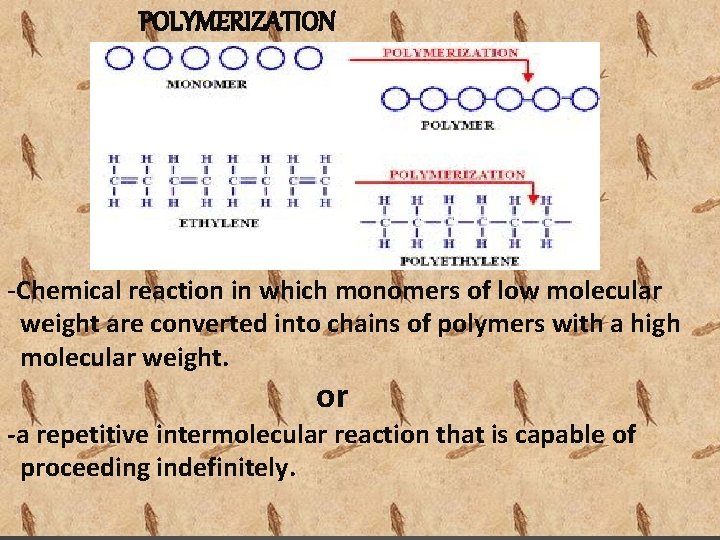
POLYMERIZATION -Chemical reaction in which monomers of low molecular weight are converted into chains of polymers with a high molecular weight. or -a repetitive intermolecular reaction that is capable of proceeding indefinitely.
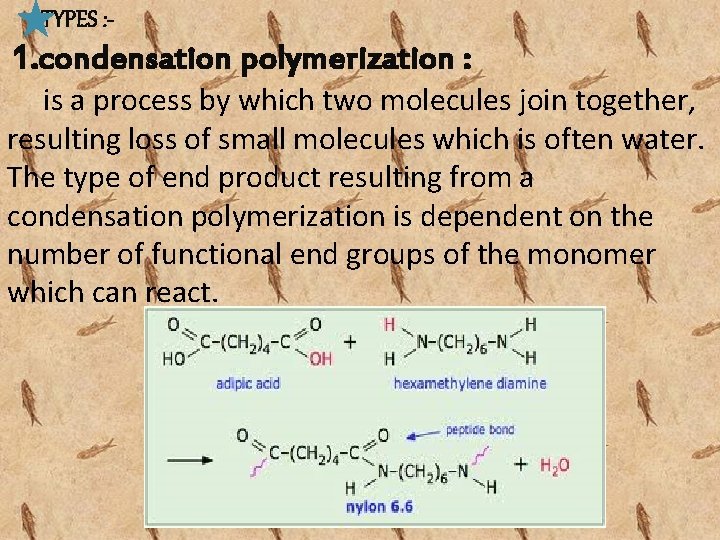
TYPES : - 1. condensation polymerization : is a process by which two molecules join together, resulting loss of small molecules which is often water. The type of end product resulting from a condensation polymerization is dependent on the number of functional end groups of the monomer which can react.
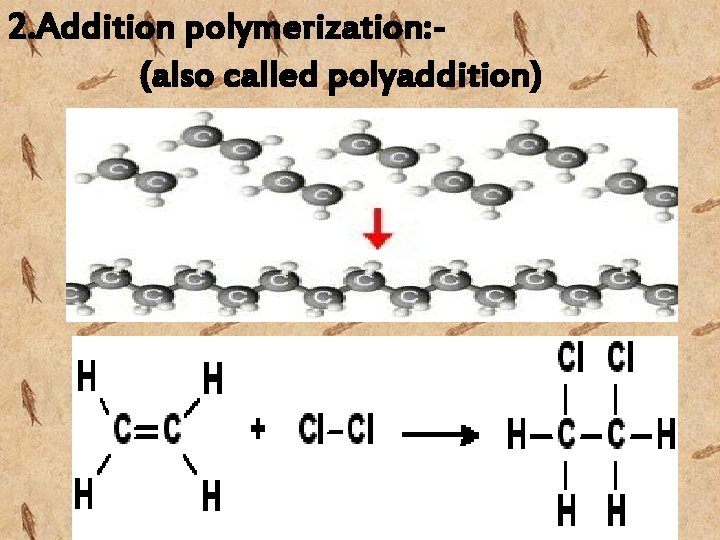
2. Addition polymerization: (also called polyaddition)

-in these polymer is formed by an addition reaction, where many monomers bond together via rearrangement of bonds without the loss of any atom or molecule. all resins employed mainly in dental procedures are produced by addition polymerization. -

ss

IT OCCURS IN 4 STAGES: - 1. Induction 2. Propagation 3. Termination 4. Chain transfer

1. induction; it is the time period during which the initiator molecules becomes energized and breaks down in free radicals, these radicals reacts with the monomer molecules to initiate the chain growth. - impurity - temperature induction -INIATION ENERGY =16000 -29000 CAL/MOL

3 induction systems for dental resins: üHeat activationthe free radicals are liberated by application of heat. eg; benzoyl peroxide on heating liberates free radicals to initiate polymerization of methyl methacrylate monomer.

üChemical activation- - it consists of at least two reactants, on mixing they liberate free radicals. üLight activation- in this the photons activate the initiator to generate free radicals.

2. Propagation: -As reaction has started it continuous with evolution of heat until all the monomer is converted to polymer. -now energy required is 5000 -8000 cal/mol.
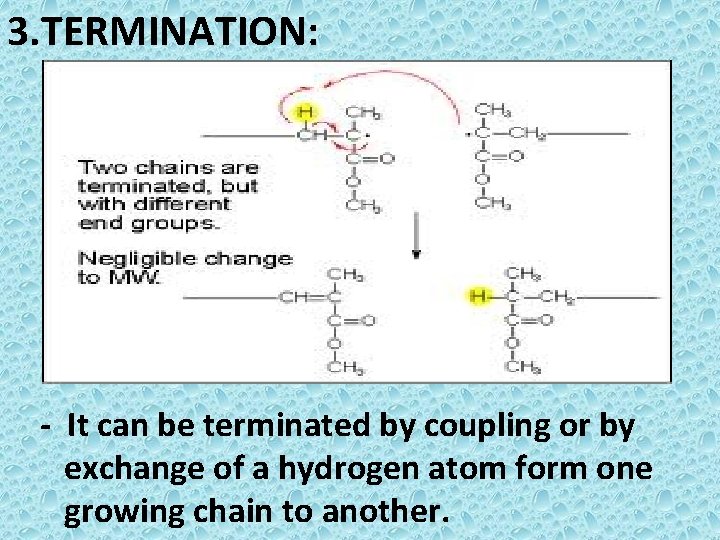
3. TERMINATION: - It can be terminated by coupling or by exchange of a hydrogen atom form one growing chain to another.
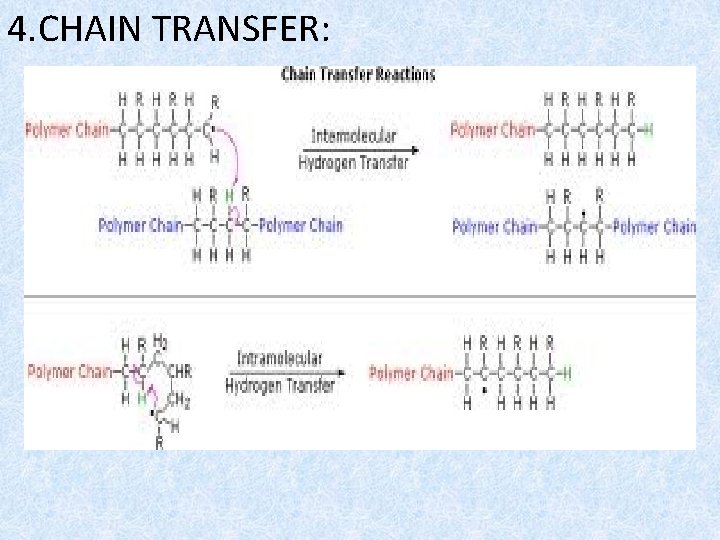
4. CHAIN TRANSFER:

- in these the active state is transferred from an activated radical to an inactive molecule and new nucleus is created.

THANK YOU. .
- Slides: 34