Glycoproteins and Mucins B Sopko Content Glycoproteins Structures

Glycoproteins and Mucins B. Sopko

Content � Glycoproteins: Structures and Linkages � Interconversions � Other and activation of dietary sugars pathways of sugar nucleotide metabolism � Biosynthesis � Functions of oligosaccharides of the oligosaccharide chains of glycoproteins � Mucins � Pathological glycosylation
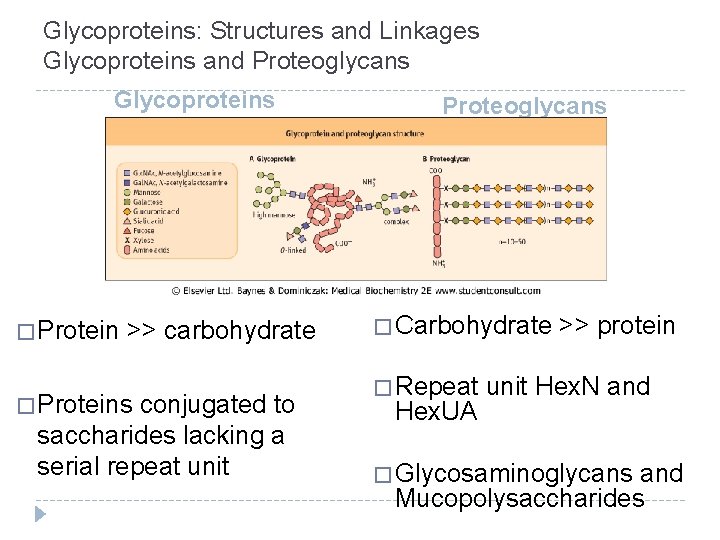
Glycoproteins: Structures and Linkages Glycoproteins and Proteoglycans Glycoproteins � Protein >> carbohydrate � Proteins conjugated to saccharides lacking a serial repeat unit Proteoglycans � Carbohydrate � Repeat Hex. UA >> protein unit Hex. N and � Glycosaminoglycans and Mucopolysaccharides
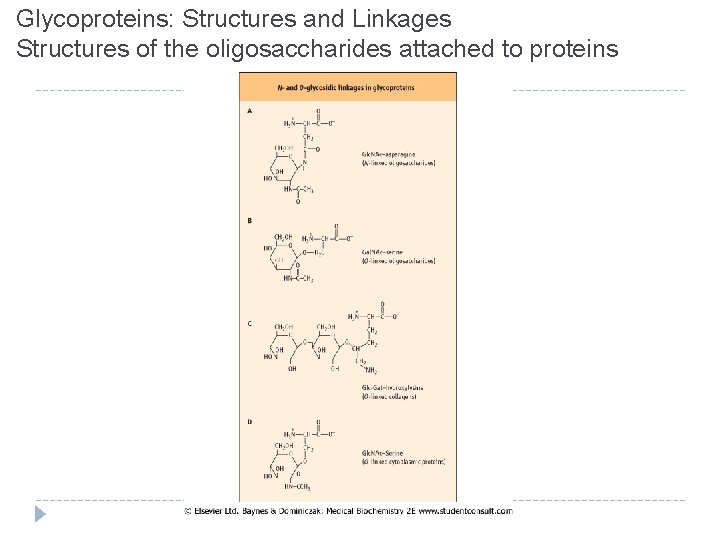
Glycoproteins: Structures and Linkages Structures of the oligosaccharides attached to proteins
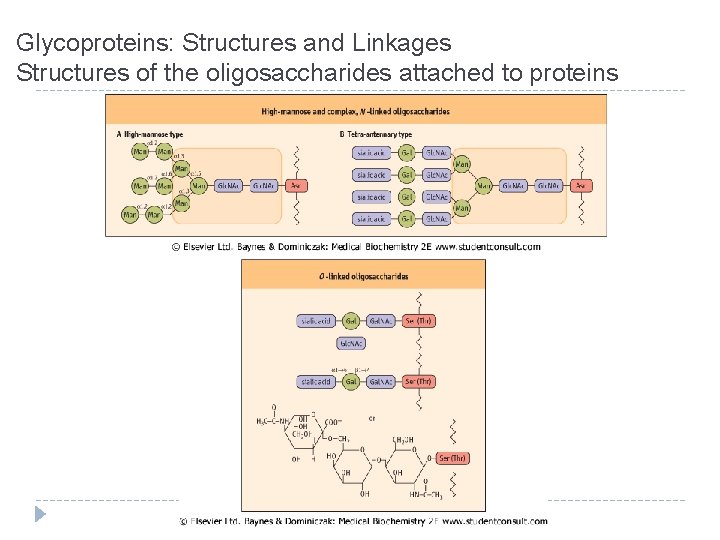
Glycoproteins: Structures and Linkages Structures of the oligosaccharides attached to proteins
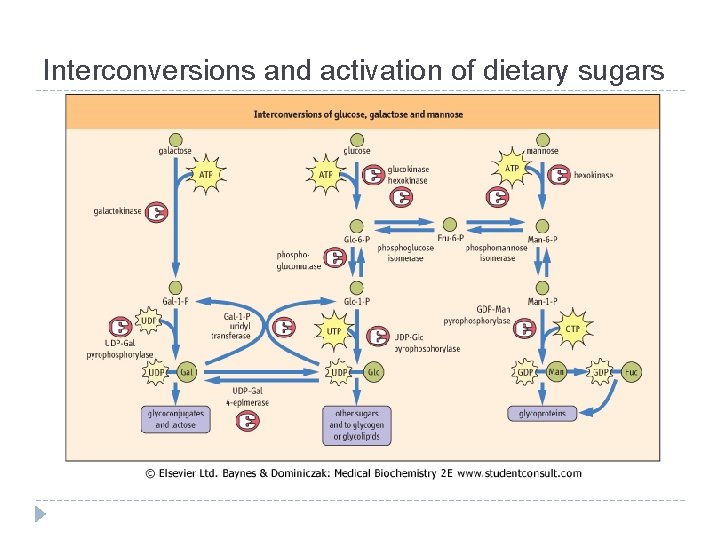
Interconversions and activation of dietary sugars
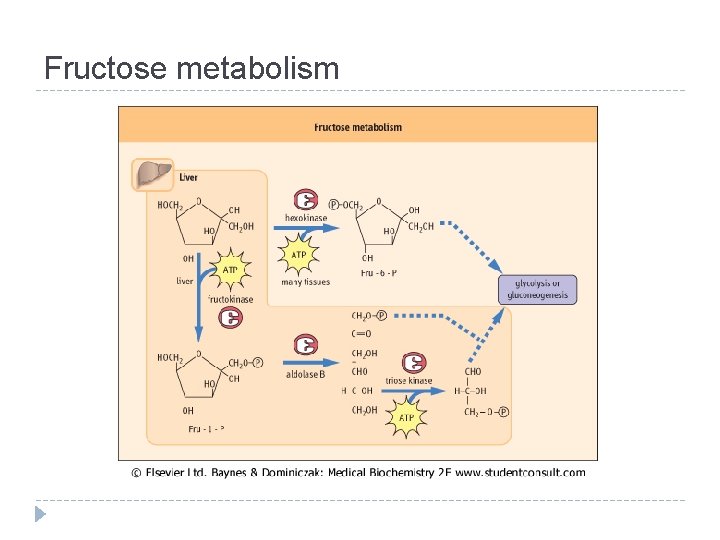
Fructose metabolism
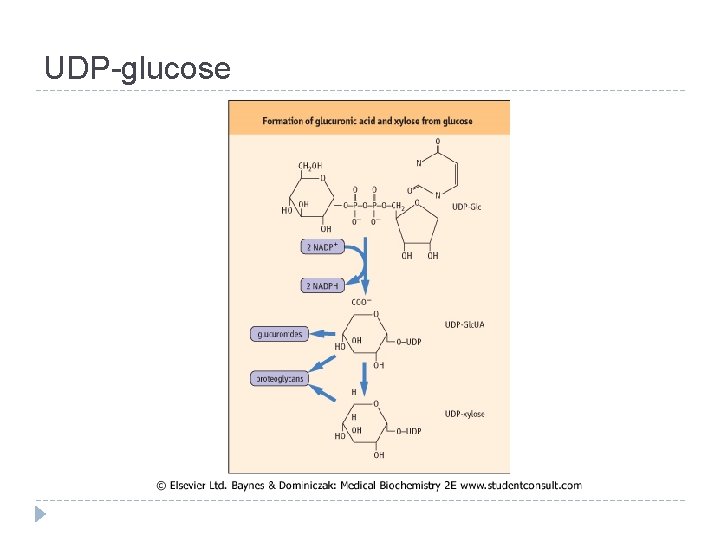
UDP-glucose
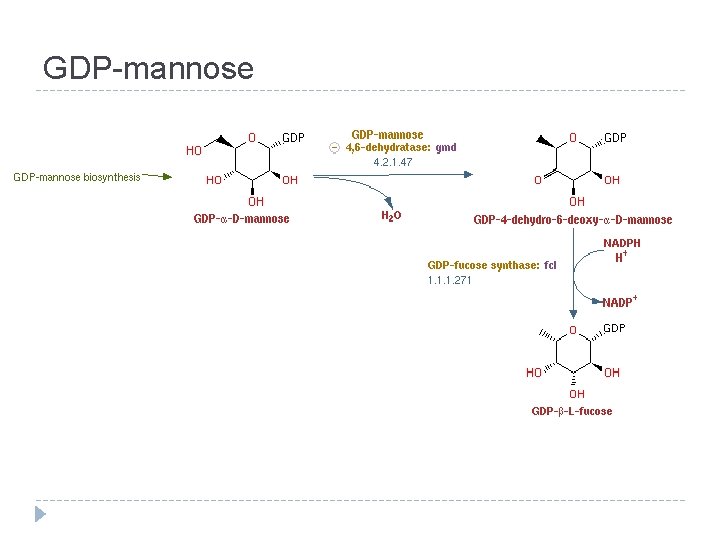
GDP-mannose
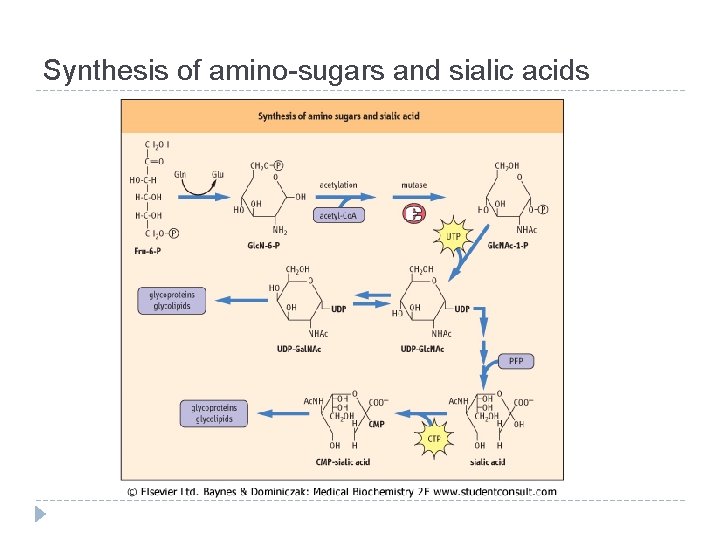
Synthesis of amino-sugars and sialic acids
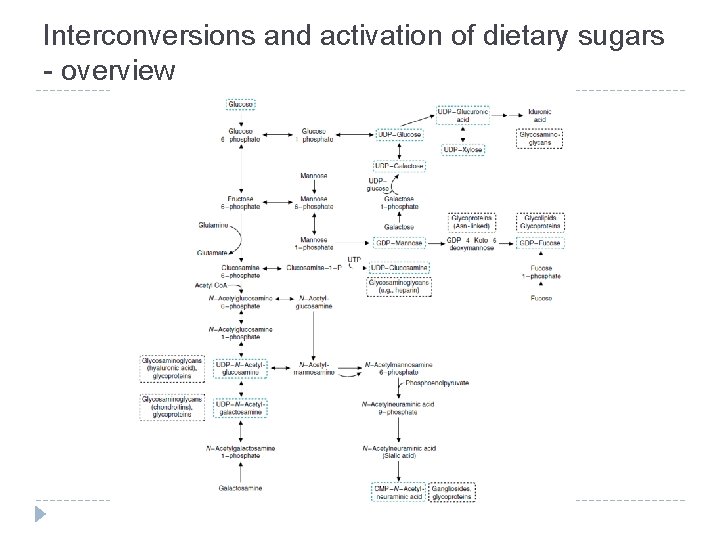
Interconversions and activation of dietary sugars - overview
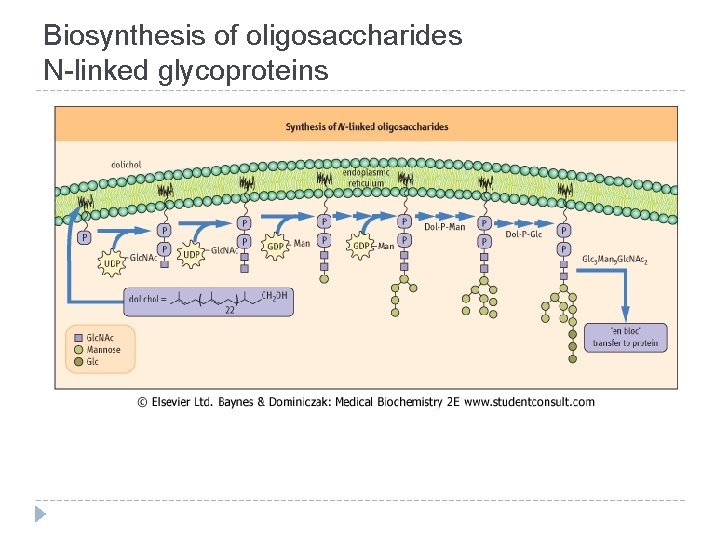
Biosynthesis of oligosaccharides N-linked glycoproteins
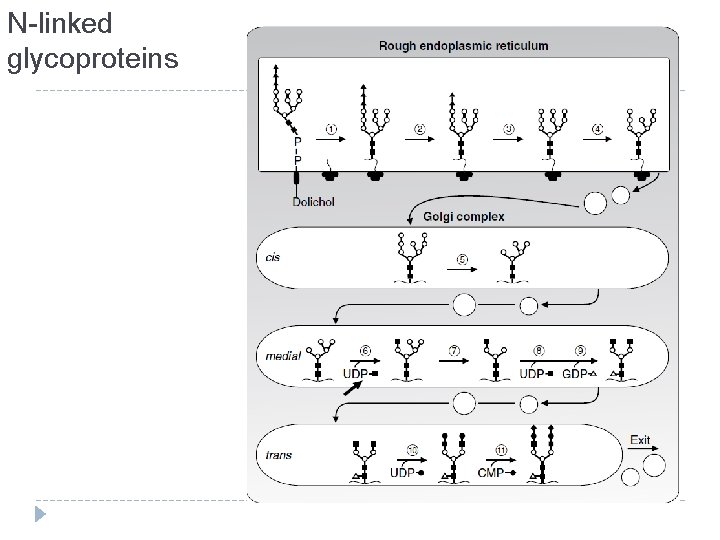
N-linked glycoproteins
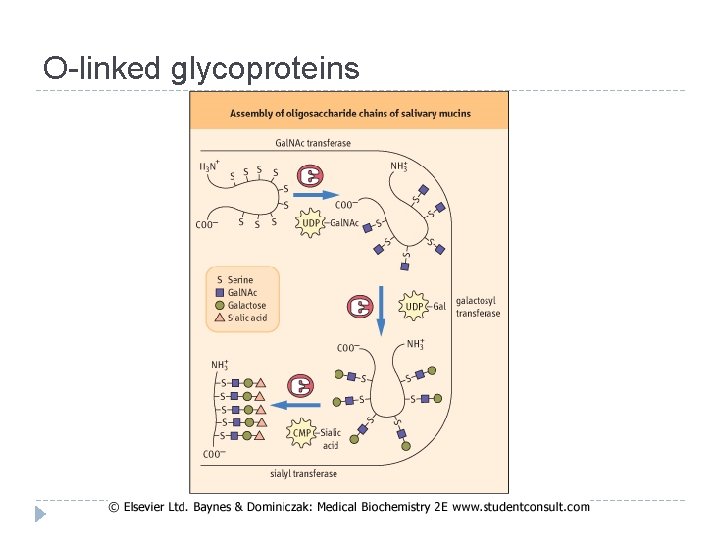
O-linked glycoproteins
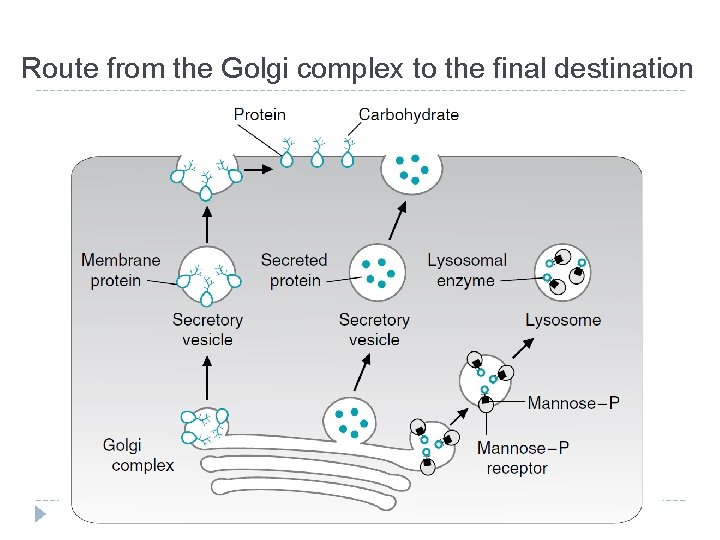
Route from the Golgi complex to the final destination

Functions of the oligosaccharide chains of glycoproteins � N-linked oligosaccharides have an important role in protein folding � High-mannose oligosaccharides target some proteins to specific sites in the cell � The oligosaccharide chains of glycoproteins increase the solubility and stability of proteins � Both N- and O-linked glycan structures are involved in recognition processes
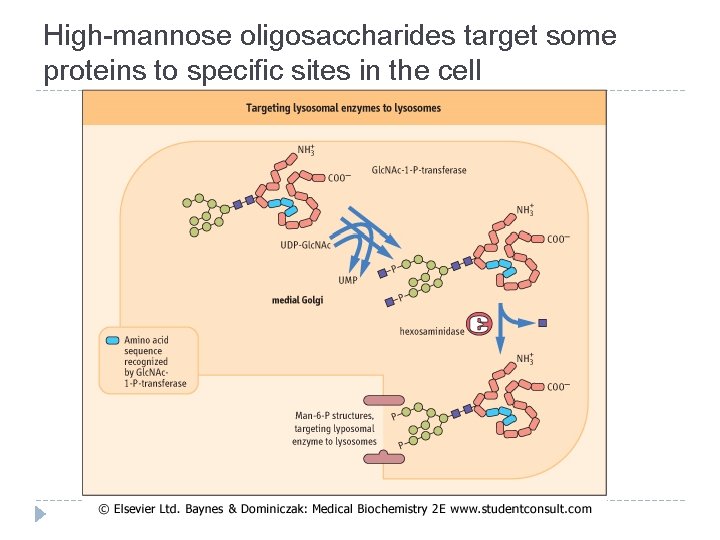
High-mannose oligosaccharides target some proteins to specific sites in the cell
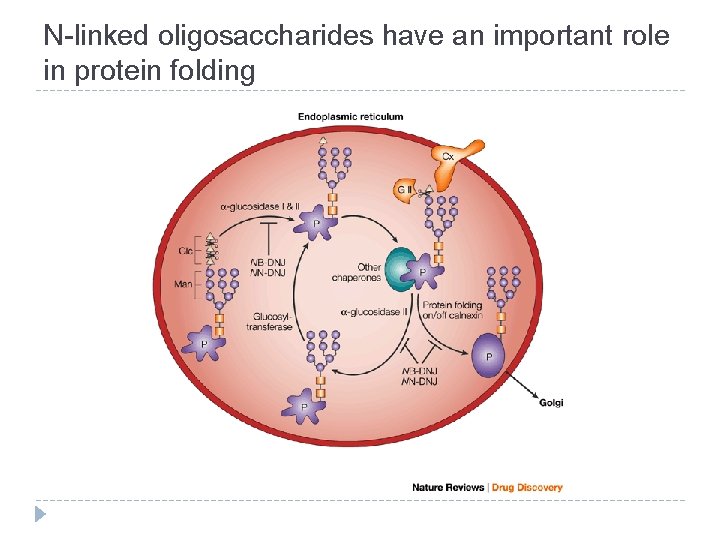
N-linked oligosaccharides have an important role in protein folding
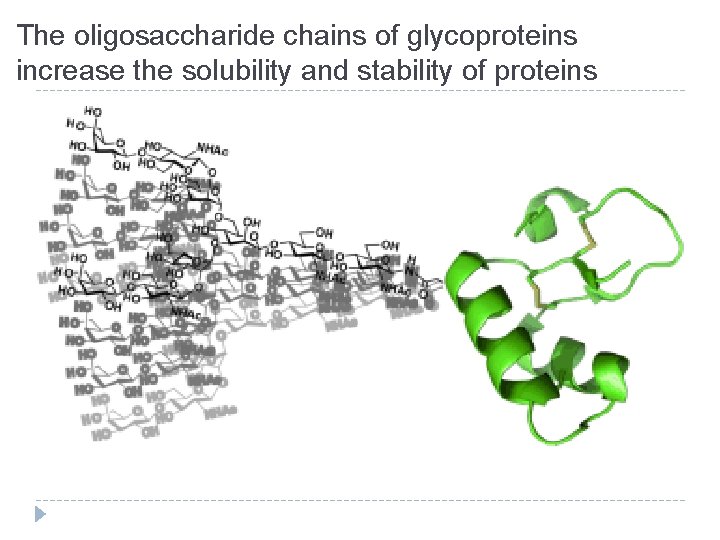
The oligosaccharide chains of glycoproteins increase the solubility and stability of proteins
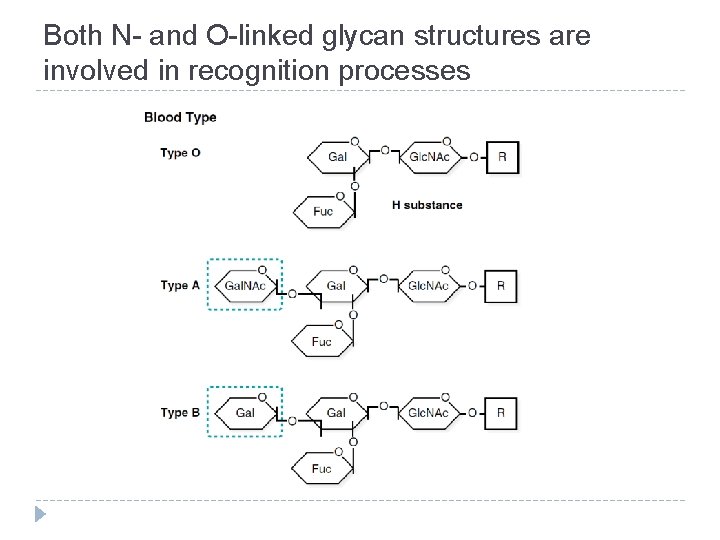
Both N- and O-linked glycan structures are involved in recognition processes
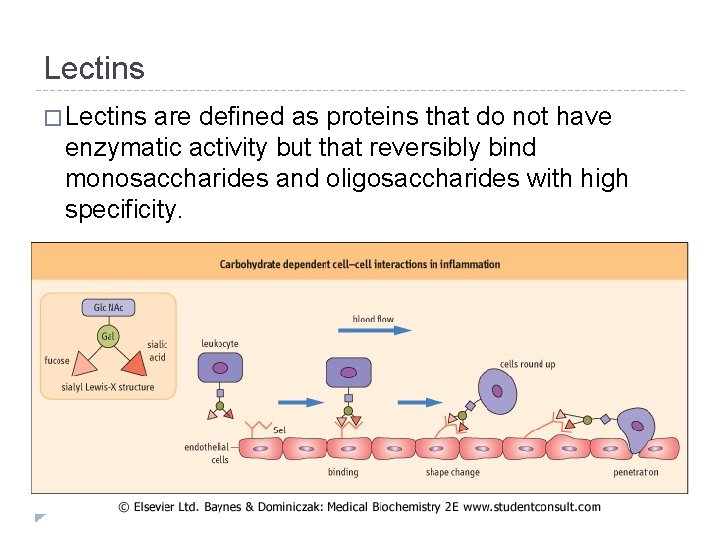
Lectins � Lectins are defined as proteins that do not have enzymatic activity but that reversibly bind monosaccharides and oligosaccharides with high specificity.

Mucins - characterization � Complex glycoproteins synthetized in epithelial cells � Components of mucus secretions covering epithelial cells in gastrointestinal, urogenital, tracheobronchial, ocular and auditory systems of all vertebrates (but they can be found in all eukaryotes) � Very rich in carbohydrates (50 -90% of mucin mass is composed by sugars) and saccharides are linked to protein via O-glycosidic bond � O-glycans are linked to serine/threonine in specific domain called tandem repeat � Some mucins can also contain N-glycosidic oligosaccharides, but they are bound only in cysteine-rich domain � Marked MUC 1 ….
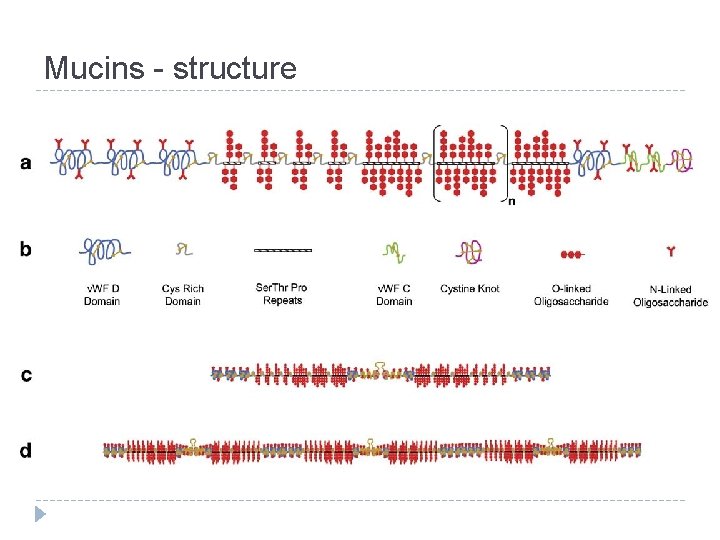
Mucins - structure

Mucins - types � Membrane-tethered with TR (membrane mucins) – e. g. MUC 1, MUC 4 � Secreted, cysteine-poor with TR (gel forming cysteinpoor mucins) - e. g. MUC 7 � Secreted, � Mucins cysteine-rich with TR - MUC 2, MUC 5 AC without TR
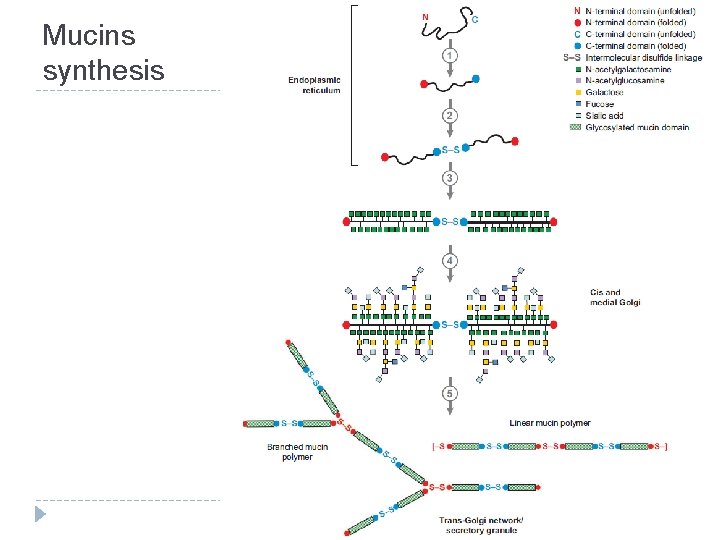
Mucins synthesis
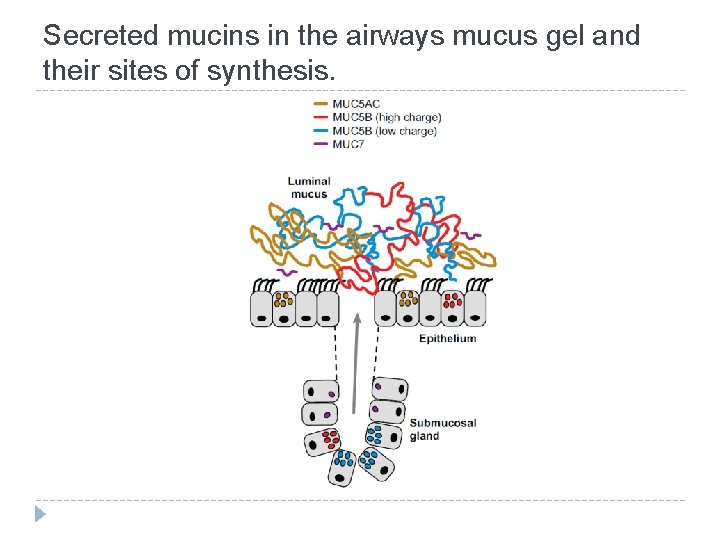
Secreted mucins in the airways mucus gel and their sites of synthesis.
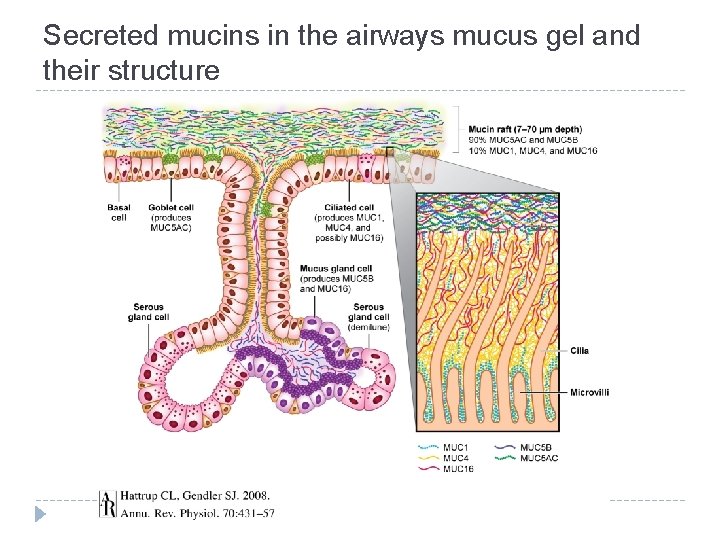
Secreted mucins in the airways mucus gel and their structure

Pathological glycosylation and glycation

I-CELL DISEASE � I-cell disease results from an enzyme deficiency so that lysosomal enzymes do not aquire the targeting signal, mannose 6 -phosphate. � Fibroblasts in this disease have dense inclusion bodies (I-cells) and are deficient in many lysosomal enzymes. � The lysosomes become engorged with indigestible substrates, leading to death in infancy.
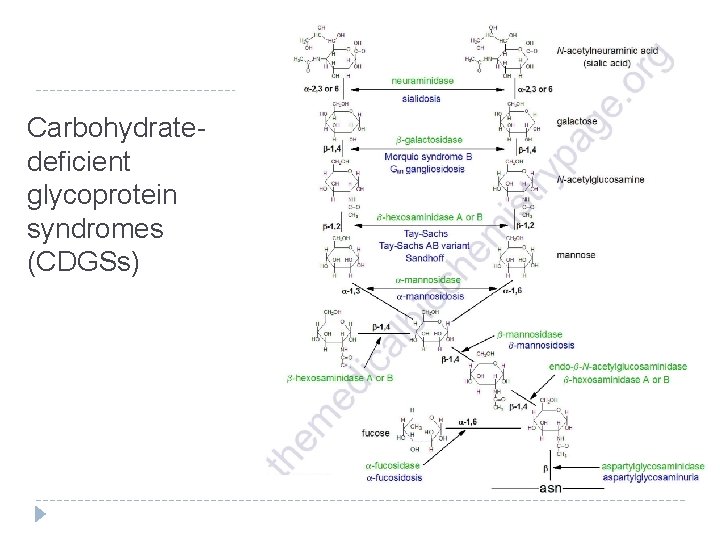
Carbohydratedeficient glycoprotein syndromes (CDGSs)

Carbohydrate-deficient glycoprotein syndromes (CDGSs) Disease Enzyme Deficiency Symptoms/Comments progressive mental retardation, delayed speech and motor development, coarse facial features Aspartylglucosaminuria aspartylglucosaminidase (N-aspartyl-β-glucosaminidase) β-Mannosidosis β-mannosidase primarily neurological defects, speech impairment α-Mannosidosis α-mannosidase mental retardation, dystosis multiplex, hepatosplenomegaly, hearing loss, delayed speech GM 1 Gangliosidosis β-galactosidase also identified as a glycosphingolipid storage disease or lysosomal storage disease β-hexosaminidases A and B also identified as a glycosphingolipid storage disease or lysosomal storage disease neuraminidase (sialidase) myoclonus, congenital ascites, hepatosplenomegaly, coarse facial features, delayed mental and motor development Sandhoff disease Sialidosis (also identified as Mucolipidosis I) Fucosidosis α-fucosidase progressive motor and mental deterioration, growth retardation, coarse facial features, recurrent sinus and pulmonary infections
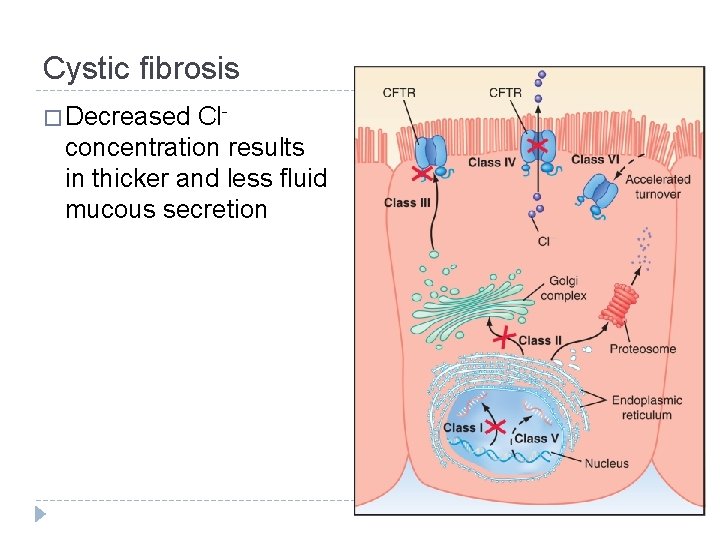
Cystic fibrosis � Decreased Clconcentration results in thicker and less fluid mucous secretion
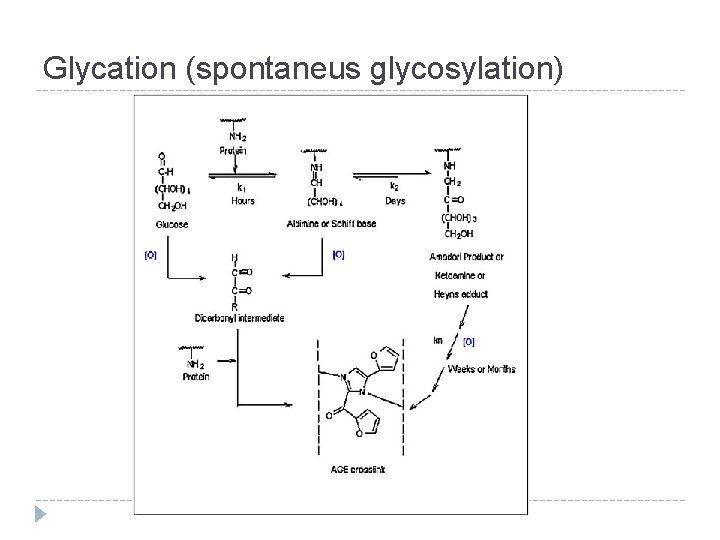
Glycation (spontaneus glycosylation)

Glycation (spontaneus glycosylation) Extracellular proteins Intracellular proteins Long-lived proteins Collagen Type I, III and IV, cartilage, elastin, myelin, proteoglycan Lens crystallins, Neurofilaments Short-lived proteins Plasma proteins such as Apo B, LDL, albumin, immunoglobulins Hemoglobin, enzymes, lysosomes, ribonuclease and several membrane proteins
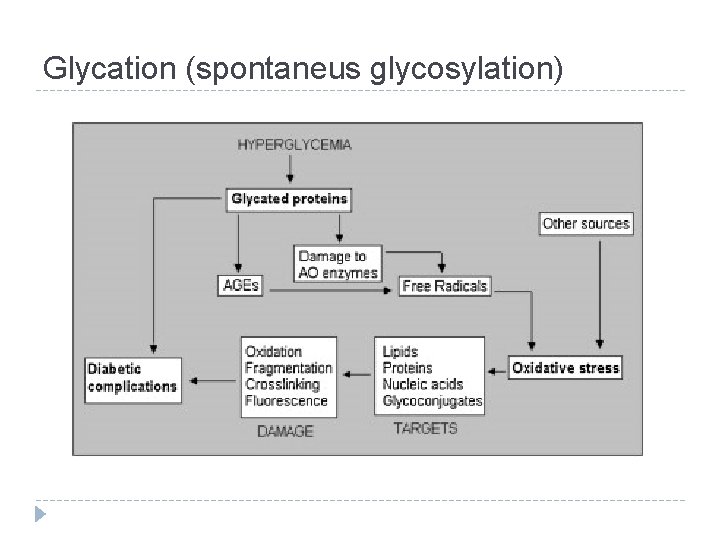
Glycation (spontaneus glycosylation)
- Slides: 35