Glycolysis is a primary source of substrates that

Glycolysis is a primary source of substrates that feed into the Citric Acid Cycle inside mitochondria! Sept 30 What is anabolism and catabolism? What makes ATP and NADH useful as metabolic intermediates? How do carbohydrates, fatty acids and (not by choice) amino acids become oxidized to provide energy (ATP) to a cell? • Review of Glycolysis: substrates, products and enzymes • How do you make glycolysis go forward considering the Delta G values? • Why regulate enzyme pathways? • How do cells make new glucose? “gluconeogenesis” • What are the main energy signals in a cell? • What glycolytic enzymes are allosterically regulated by metabolites? WHY?

Localization of metabolism to specific eukaryotic organelles/locations. Catabolic Pathways • Glycolysis (glucose pyruvate): cytosol • Citric Acid Cycle (Pyruvate acetate CO 2): mitochondria • Beta-oxidation (fatty acids acetate): mitochondria • Oxidative Phosphorylation (NADH/FADH 2 oxygen) mitochondria Anabolic Pathways • Photosynthesis: chloroplast • Gluconeogenesis: cytosol
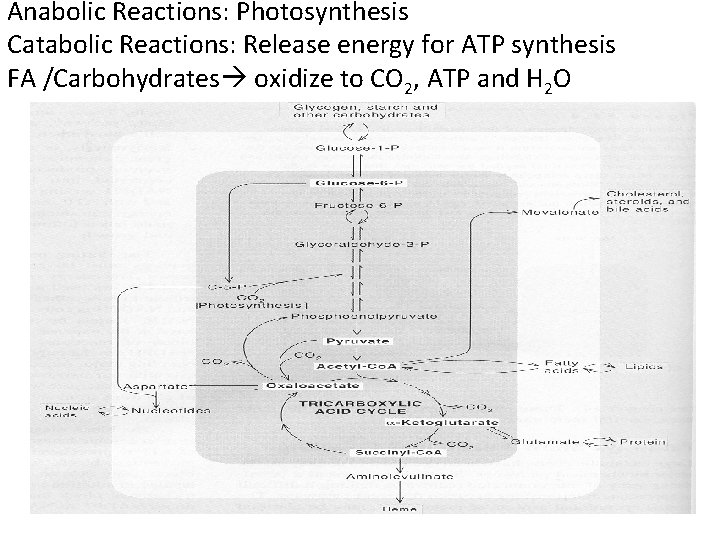
Anabolic Reactions: Photosynthesis Catabolic Reactions: Release energy for ATP synthesis FA /Carbohydrates oxidize to CO 2, ATP and H 2 O

How does glycolysis allow us to break down glucose (oxidize it) to release energy that directly or indirectly is used for ATP synthesis? • Glycolysis: Glyco-”sweet” Lysis-”break up” • 10 enzymes in pathway The Sweet Valentines Break-Up • Enzyme names typically describe reaction • Net Reaction: • 1 Glucose+2 ATP + 4 ADP+2 NAD+ 2 pyruvate + 2 ADP + 4 ATP + 2 NADH • If oxygen is present mitochondria or bacteria can convert • one NADH into 2 or 3 ATP molecules. • If oxygen absent, NADH MUST BE converted to NAD+ for recycling into glycolysis! No NAD+ No pyruvate and no ATP • Anaerobic Metabolism: (Anoxia-Prokaryotes vs. Hypoxia. Eukaryotes)

Overview of glycolysis: three main phases. One Preparatory Phase • Phase One (Preparatory Phase): Prepare glucose so it is unstable and easy to cleave into smaller parts! – Make it chemically unstable – Use two ATP to create unstable shape Two Pay-Off Phases • Phase Two: Oxidize and regenerate four ATP! – Substrate level phosphorylation – No reactions transfer to ATP • Phase Three: Create pyruvate as final step from unstable PEP! • Use fermentation enzymes to regenerate NAD+ in times
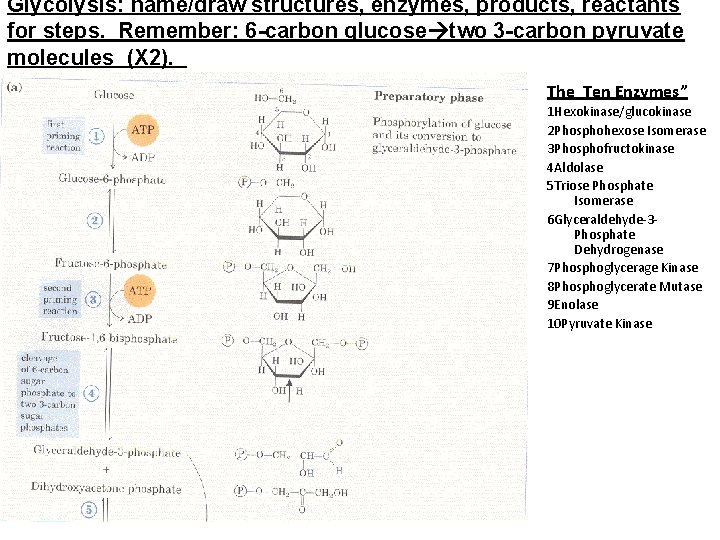
Glycolysis: name/draw structures, enzymes, products, reactants for steps. Remember: 6 -carbon glucose two 3 -carbon pyruvate molecules (X 2). The Ten Enzymes” 1 Hexokinase/glucokinase 2 Phosphohexose Isomerase 3 Phosphofructokinase 4 Aldolase 5 Triose Phosphate Isomerase 6 Glyceraldehyde-3 Phosphate Dehydrogenase 7 Phosphoglycerage Kinase 8 Phosphoglycerate Mutase 9 Enolase 10 Pyruvate Kinase
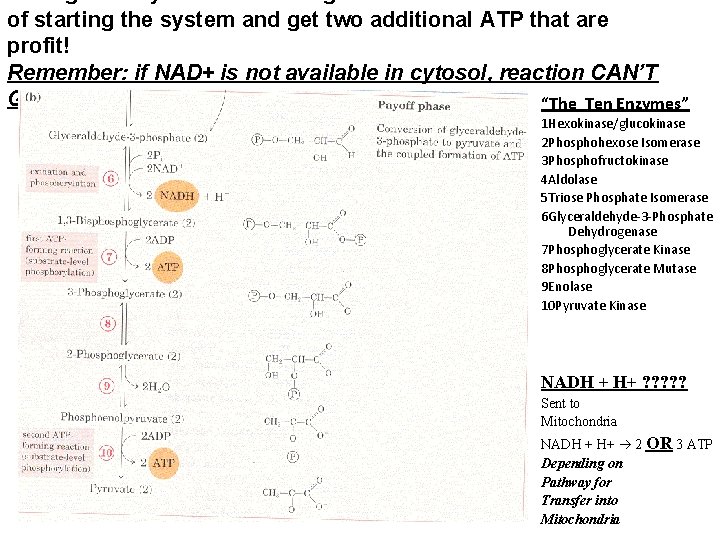
During the Payoff Phase we regenerate 2 ATP to cover the cost of starting the system and get two additional ATP that are profit! Remember: if NAD+ is not available in cytosol, reaction CAN’T GO FORWARD! “The Ten Enzymes” 1 Hexokinase/glucokinase 2 Phosphohexose Isomerase 3 Phosphofructokinase 4 Aldolase 5 Triose Phosphate Isomerase 6 Glyceraldehyde-3 -Phosphate Dehydrogenase 7 Phosphoglycerate Kinase 8 Phosphoglycerate Mutase 9 Enolase 10 Pyruvate Kinase NADH + H+ ? ? ? Sent to Mitochondria NADH + H+ 2 OR 3 ATP Depending on Pathway for Transfer into Mitochondria
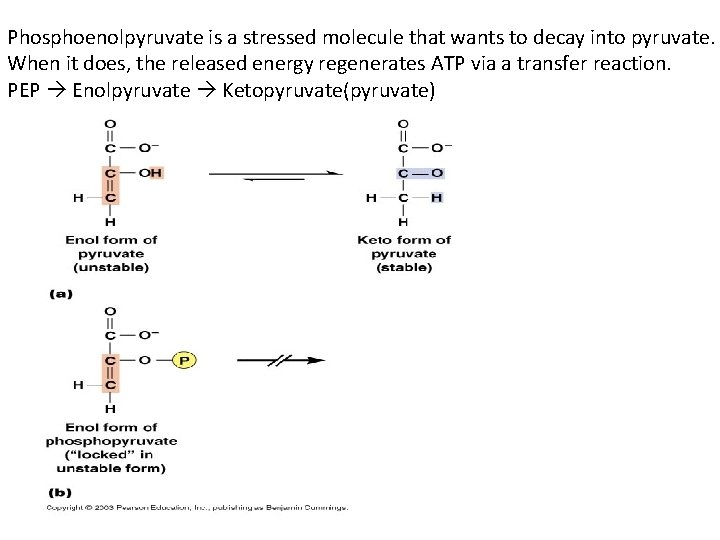
Phosphoenolpyruvate is a stressed molecule that wants to decay into pyruvate. When it does, the released energy regenerates ATP via a transfer reaction. PEP Enolpyruvate Ketopyruvate(pyruvate)
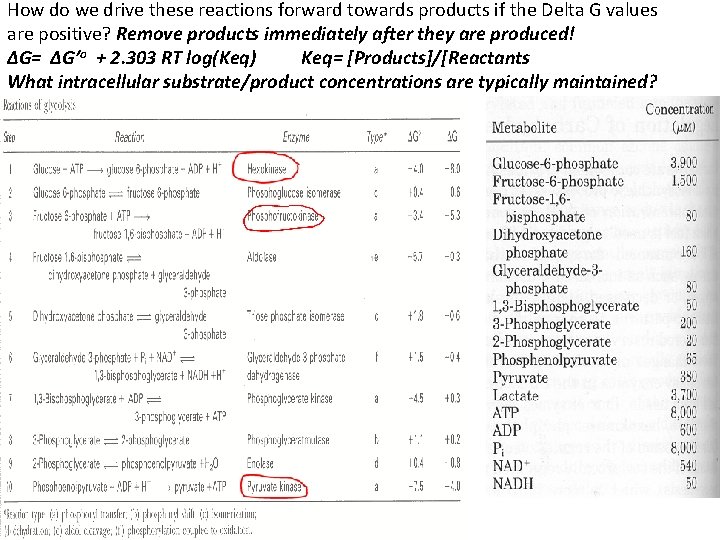
How do we drive these reactions forward towards products if the Delta G values are positive? Remove products immediately after they are produced! ∆G= ∆G’o + 2. 303 RT log(Keq) Keq= [Products]/[Reactants What intracellular substrate/product concentrations are typically maintained?
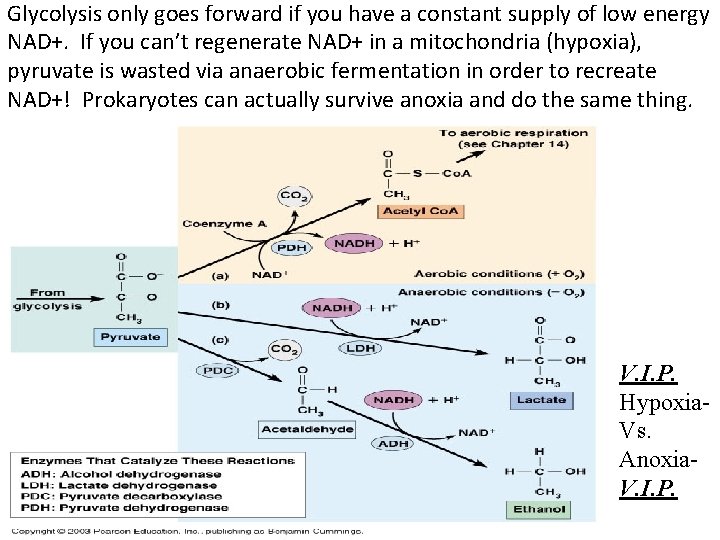
Glycolysis only goes forward if you have a constant supply of low energy NAD+. If you can’t regenerate NAD+ in a mitochondria (hypoxia), pyruvate is wasted via anaerobic fermentation in order to recreate NAD+! Prokaryotes can actually survive anoxia and do the same thing. V. I. P. Hypoxia. Vs. Anoxia. V. I. P.

What are the ten glycolytic enzymes? Could you match these to their reactions? • 1) Hexokinase/glucokinase • 2) Phosphohexose Isomerase • 3) Phosphofructokinase • 4) Aldolase • 5) Triose Phosphate Isomerase • 6) Glyceraldehyde-3 -Phosphate Dehydrogenase • 7) Phosphoglycerage Kinase • 8) Phosphoglycerate Mutase • 9) Enolase • 10) Pyruvate Kinase Fermentation: Anerobic-prokaryotic cells /Hypoxic-Most eukaryotic cells – Lactate Dehydrogenase – Pyruvate. Decarboxylase and Alcohol Dehydrogenase
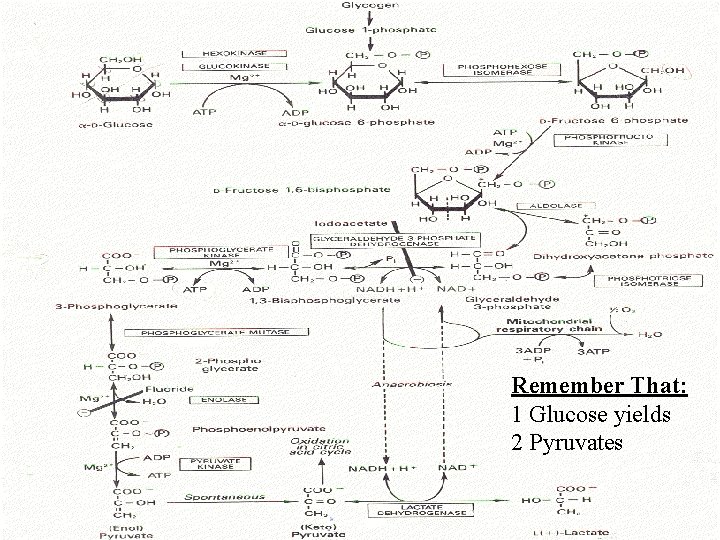
Remember That: 1 Glucose yields 2 Pyruvates

What happens when glucose is not available to a cell? Gluconeogenesis: making glucose from pyruvate or amino acids that can be converted to pyruvate! • Think Low. Carb Diet! Req: 4 new enzymes! • What are the Committed Steps to gluco. NEOgenesis? How does gluconeogenesis differ from glycolysis? • How do we prevent futile cycles?

Why does a cell have to tightly regulate both glycolysis and gluconeogenesis? • Futile Cycles: occur when two enzyme pathways are going in opposite directions at the same time. • Implications: – Excess energy production? – Inadequate energy production? • What pathways should predominate in a cell? – During Starvation: Glucagon – During Well fed state: Insulin – Please remember that cell clusters on different locations in the body work as organs, whose metabolic activities may appear to

How does a cell control the rate of glucose use/synthesis? • Enzyme production/destruction: – Called “induction” or “suppression”– Problem: it takes 30 minutes to several hours to create enzymes! • Allosteric Regulation modifies the rate of preexisting enzymes. • Enzymes are bound by a molecule (allosteric regulator) at a regulatory binding site such that the activity of the enzyme either speeds up or slows down. • Regulatory molecules are typically products or reactants in the pathway affected by the enzyme. • Covalent Modification: use a kinase to add a phosphate or a phosphatase to remove a

Do the presence of the following metabolites suggest a well fed or low energy state to a cell? • If these metabolites appear in the cytosol, should the cell speed up or slow down glycolysis or gluconeogenesis? • High G-6 -P: _____ • High ATP: ______ • High Acetyl-Co. A: _____ • High F-1, 6 -BP: _______ • High ADP: _____ • High AMP: _____ • Specific allosteric binding sites for the above metabolites are found on the “Committed Step Enzymes” of glycolysis and gluconeogenesis.
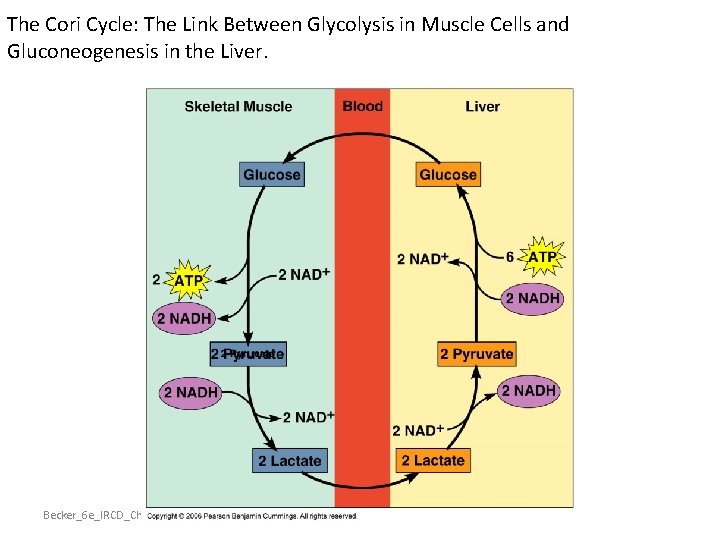
The Cori Cycle: The Link Between Glycolysis in Muscle Cells and Gluconeogenesis in the Liver. Becker_6 e_IRCD_Chapter_9 17
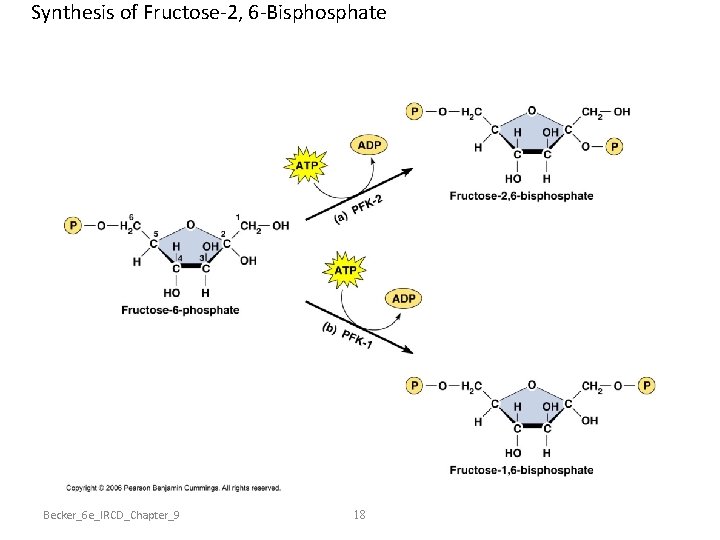
Synthesis of Fructose-2, 6 -Bisphosphate Becker_6 e_IRCD_Chapter_9 18
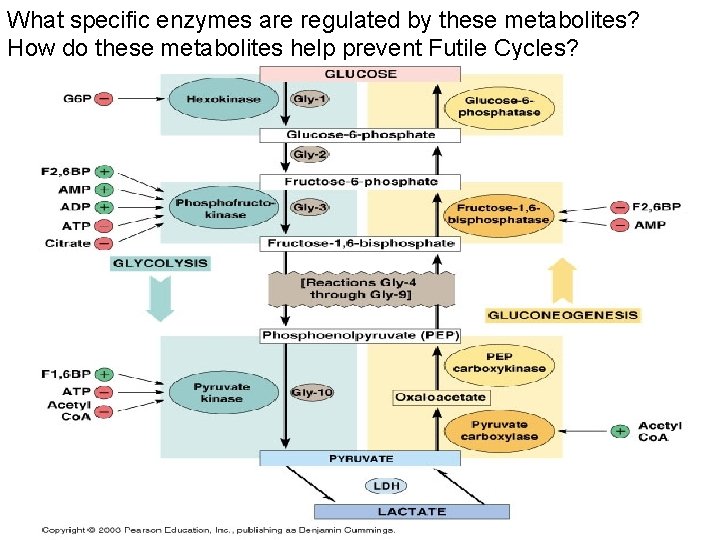
What specific enzymes are regulated by these metabolites? How do these metabolites help prevent Futile Cycles?
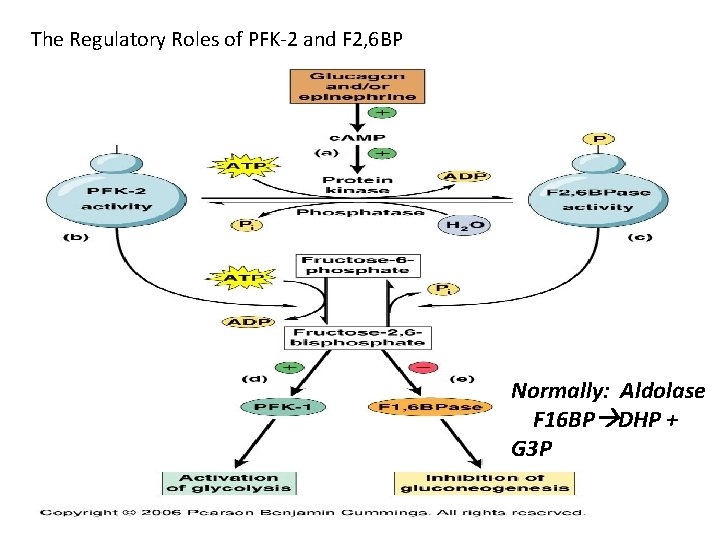
The Regulatory Roles of PFK-2 and F 2, 6 BP Normally: Aldolase F 16 BP DHP + G 3 P Becker_6 e_IRCD_Chapter_9 20
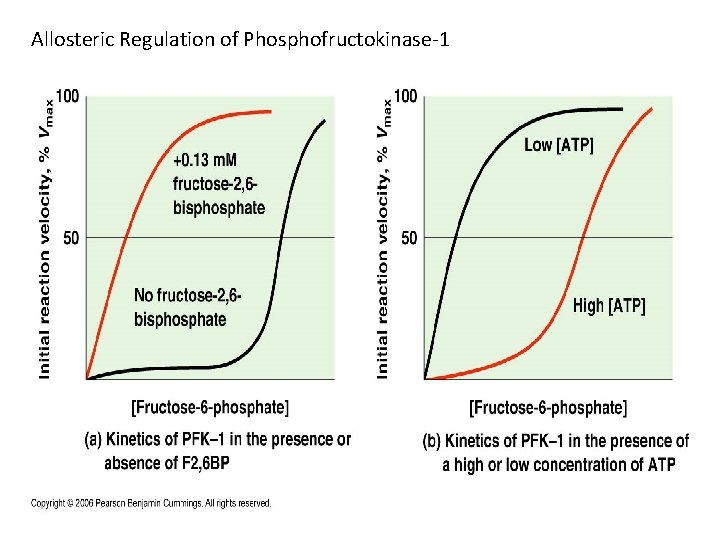
Allosteric Regulation of Phosphofructokinase-1 Becker_6 e_IRCD_Chapter_9 21
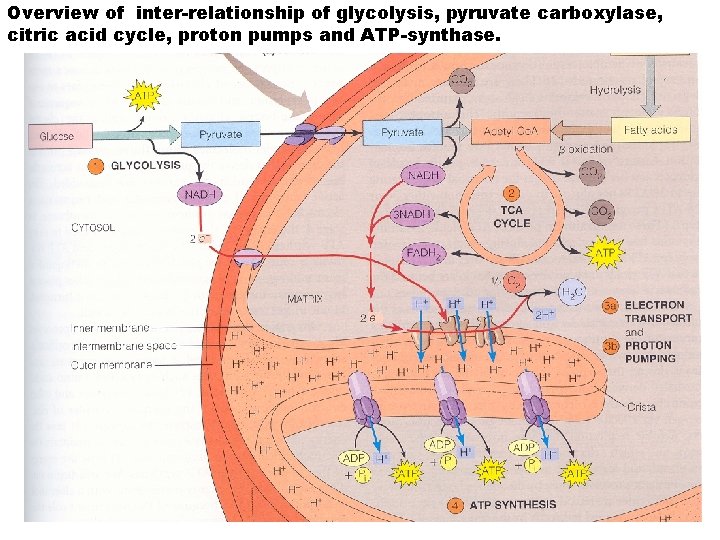
Overview of inter-relationship of glycolysis, pyruvate carboxylase, citric acid cycle, proton pumps and ATP-synthase.
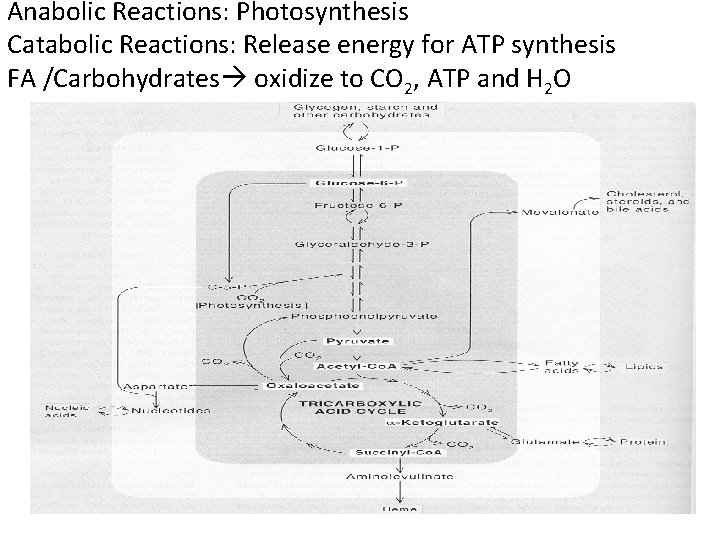
Anabolic Reactions: Photosynthesis Catabolic Reactions: Release energy for ATP synthesis FA /Carbohydrates oxidize to CO 2, ATP and H 2 O

Essay Questions and how to answer them: Read the test question, consider its parts and answer all parts of the question for full credit. Discuss the Fluid Mosaic Model of a plasma membrane with respect to the character and relative mobility of the phospholids and proteins found in it. Also discuss the effect that glycosylation has on membrane lipid/protein mobility. (5 pts; Discuss at least three items/characteristics with at least 20 -30 words) • The Fluid Mosaic Model suggests that the polar heads (i. e. choline) of membrane phospholipids face the hydrophyllic environment on the intracellular and extracellular sides of the plasma membrane, but the hydrophobic fatty acid tails (hydrocarbons) face each other. 1) Each PL is free to move on its transverse plane (laterally) but cannot easily flip between sides due to hydrophobic interactions of the polar heads with the non-polar interior tails unless the enzyme “flipase” makes this possible. 2) Proteins can generally freely diffuse laterally and are fixed to one plane or the other by the amino and carboxyl-terminal ends of the protein, by the hydrophyllic amino acids that face the outsides, and the hydrophobic charges on R -groups of a. a. facing the FA tails of the hydrophobic center of the bilayer. 3) Glycosylations (very hydrophyllic sugar units) on select lipids and proteins create additional limits to flipping between the intra and extracellular sides of a membrane. 4) Ironically, even though it is called a “fluid mosaic” because lipids and proteins “can” move about on their respective transverse planes, some proteins are linked to ankyrin, spectin or actin on the intracellualr side, which potentially localizes them to one “part” of one “side” of a membrane (they loose the ability to diffuse freely). • Adding a simple sketch/diagram would also help make your point for this essay.
- Slides: 24