GLYCOLYSIS General features of Glycolysis 1 Anaerobic degradation

GLYCOLYSIS

General features of Glycolysis 1. Anaerobic degradation of hexose sugar 2. Conversion of a 6 -carbon molecule (glucose, fructose) to a 3 -carbon molecule ( dihydroxyacetone phosphate, glyceraldehyde 3 phosphate; pyruvate 3. One 6 -carbon molecule will give two 3 carbon molecules 4. All the intermediates are phosphorylated; -vely charged at p. H 7 5. Pi bonded by either an ester or anhydride bond 6. 2 phases: activation phase and energy production phase www. lowcarbluxury. com/newsletter/lclnewsvol 03 -no 0
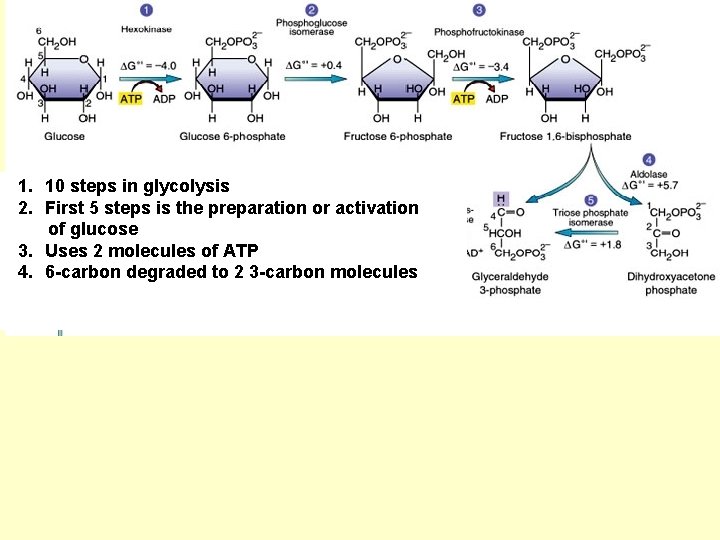
1. 10 steps in glycolysis 2. First 5 steps is the preparation or activation of glucose 3. Uses 2 molecules of ATP 4. 6 -carbon degraded to 2 3 -carbon molecules
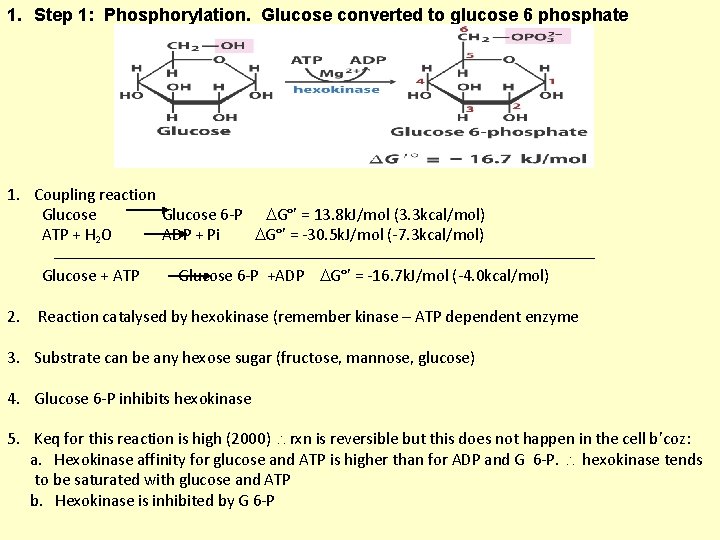
1. Step 1: Phosphorylation. Glucose converted to glucose 6 phosphate 1. Coupling reaction Glucose 6 -P G°’ = 13. 8 k. J/mol (3. 3 kcal/mol) ATP + H 2 O ADP + Pi G°’ = -30. 5 k. J/mol (-7. 3 kcal/mol) Glucose + ATP 2. Glucose 6 -P +ADP G°’ = -16. 7 k. J/mol (-4. 0 kcal/mol) Reaction catalysed by hexokinase (remember kinase – ATP dependent enzyme 3. Substrate can be any hexose sugar (fructose, mannose, glucose) 4. Glucose 6 -P inhibits hexokinase 5. Keq for this reaction is high (2000) rxn is reversible but this does not happen in the cell b’coz: a. Hexokinase affinity for glucose and ATP is higher than for ADP and G 6 -P. hexokinase tends to be saturated with glucose and ATP b. Hexokinase is inhibited by G 6 -P

Hexokinase is an allosteric enzyme: Activator: ADP Inhibitor: ATP and Glucose 6 -Phosphate
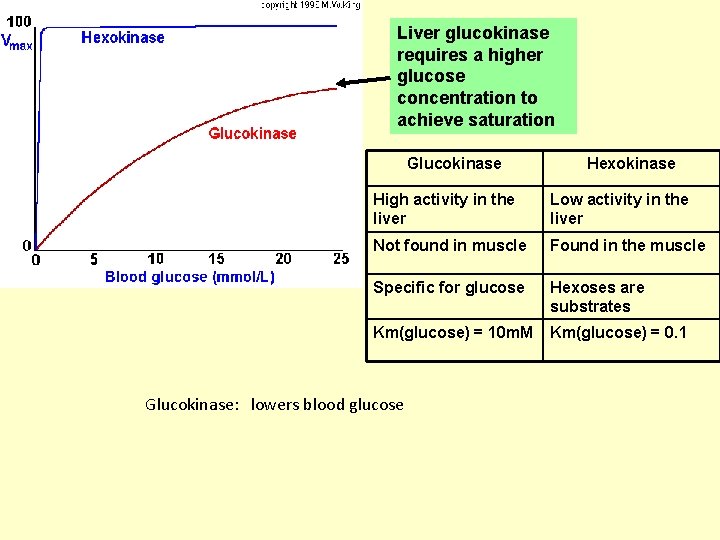
Liver glucokinase requires a higher glucose concentration to achieve saturation Glucokinase Hexokinase High activity in the liver Low activity in the liver Not found in muscle Found in the muscle Specific for glucose Hexoses are substrates Km(glucose) = 10 m. M Km(glucose) = 0. 1 Glucokinase: lowers blood glucose
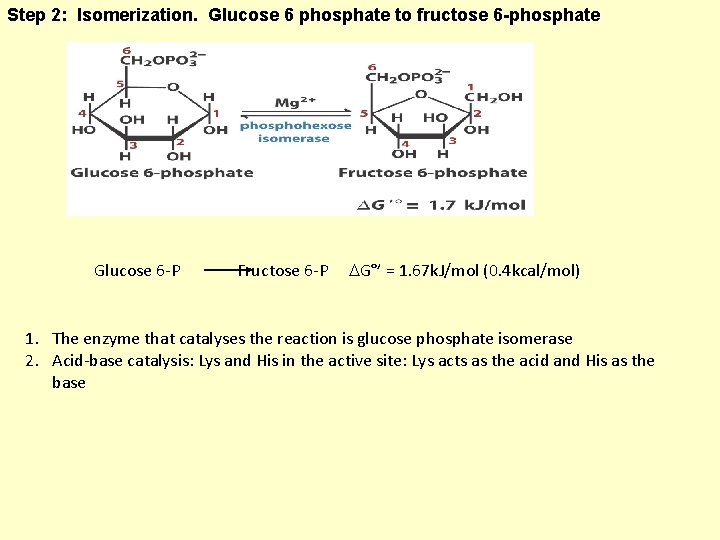
Step 2: Isomerization. Glucose 6 phosphate to fructose 6 -phosphate Glucose 6 -P Fructose 6 -P G°’ = 1. 67 k. J/mol (0. 4 kcal/mol) 1. The enzyme that catalyses the reaction is glucose phosphate isomerase 2. Acid-base catalysis: Lys and His in the active site: Lys acts as the acid and His as the base
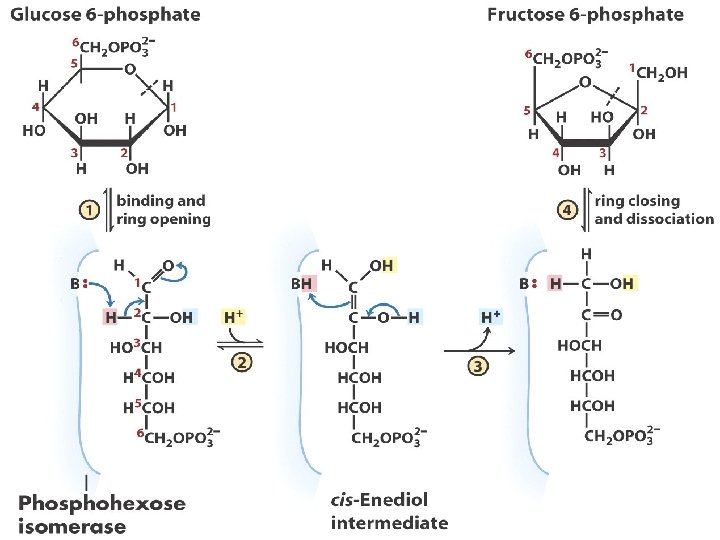
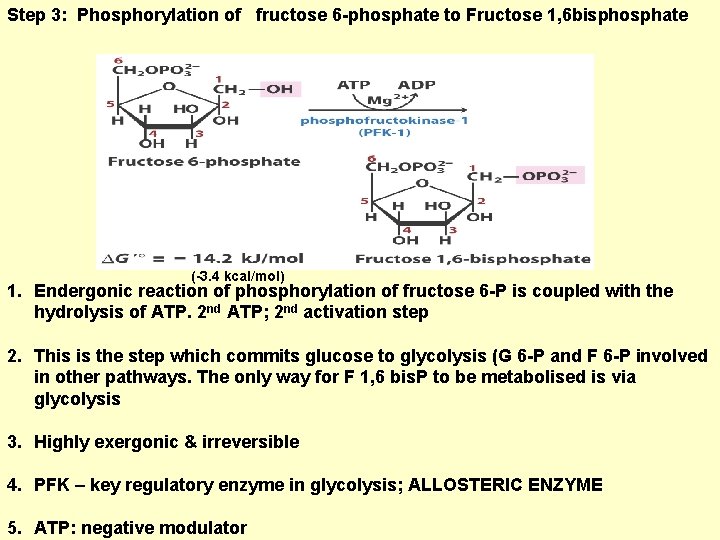
Step 3: Phosphorylation of fructose 6 -phosphate to Fructose 1, 6 bisphosphate (-3. 4 kcal/mol) 1. Endergonic reaction of phosphorylation of fructose 6 -P is coupled with the hydrolysis of ATP. 2 nd ATP; 2 nd activation step 2. This is the step which commits glucose to glycolysis (G 6 -P and F 6 -P involved in other pathways. The only way for F 1, 6 bis. P to be metabolised is via glycolysis 3. Highly exergonic & irreversible 4. PFK – key regulatory enzyme in glycolysis; ALLOSTERIC ENZYME 5. ATP: negative modulator
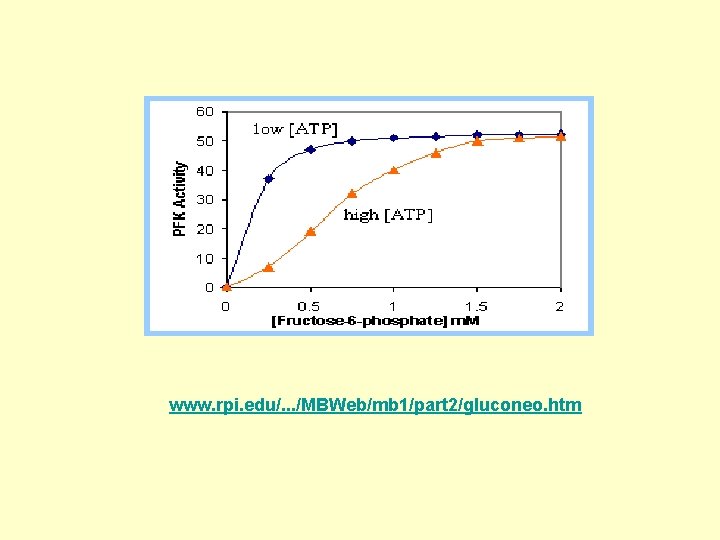
www. rpi. edu/. . . /MBWeb/mb 1/part 2/gluconeo. htm
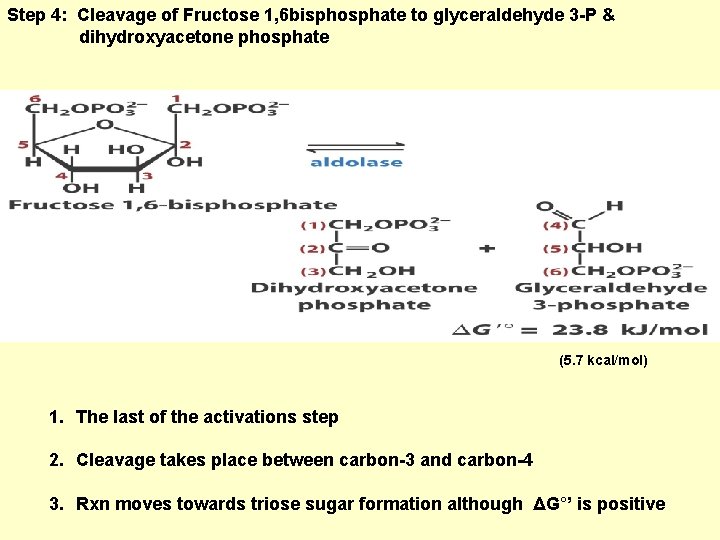
Step 4: Cleavage of Fructose 1, 6 bisphosphate to glyceraldehyde 3 -P & dihydroxyacetone phosphate (5. 7 kcal/mol) 1. The last of the activations step 2. Cleavage takes place between carbon-3 and carbon-4 3. Rxn moves towards triose sugar formation although ΔG°’ is positive
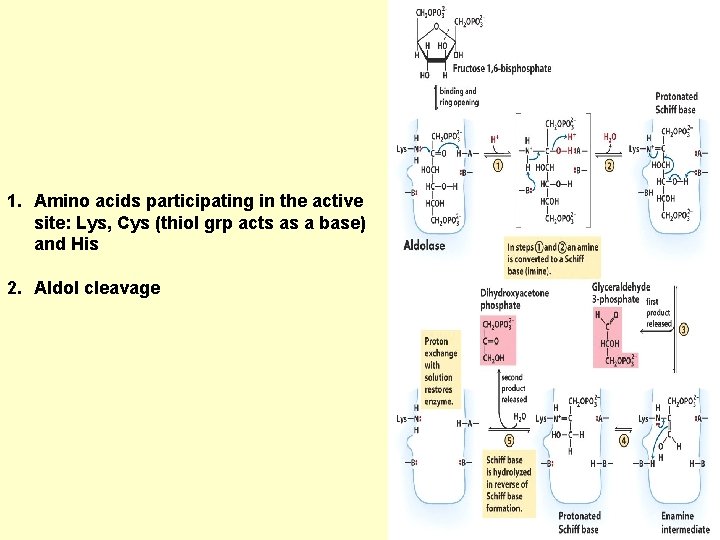
1. Amino acids participating in the active site: Lys, Cys (thiol grp acts as a base) and His 2. Aldol cleavage
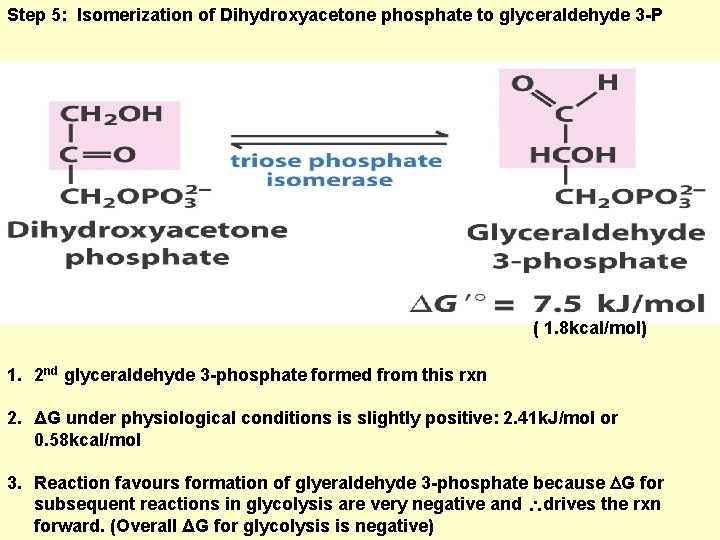
Step 5: Isomerization of Dihydroxyacetone phosphate to glyceraldehyde 3 -P ( 1. 8 kcal/mol) 1. 2 nd glyceraldehyde 3 -phosphate formed from this rxn 2. ΔG under physiological conditions is slightly positive: 2. 41 k. J/mol or 0. 58 kcal/mol 3. Reaction favours formation of glyeraldehyde 3 -phosphate because G for subsequent reactions in glycolysis are very negative and drives the rxn forward. (Overall ΔG for glycolysis is negative)
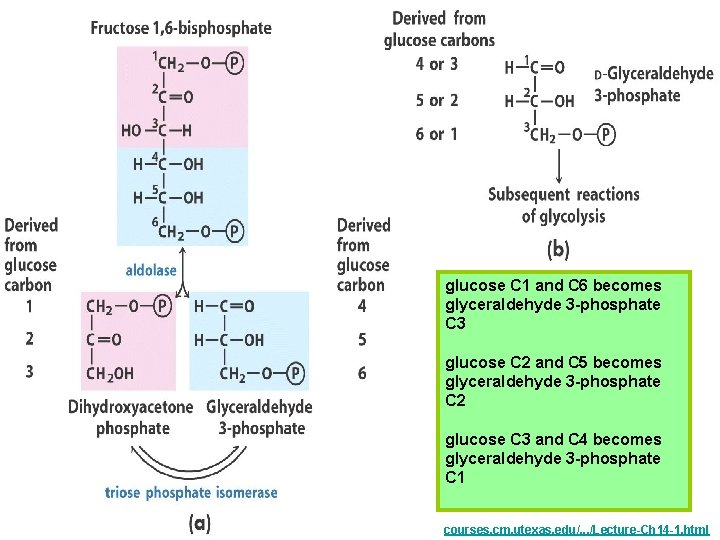
glucose C 1 and C 6 becomes glyceraldehyde 3 -phosphate C 3 glucose C 2 and C 5 becomes glyceraldehyde 3 -phosphate C 2 glucose C 3 and C 4 becomes glyceraldehyde 3 -phosphate C 1 courses. cm. utexas. edu/. . . /Lecture-Ch 14 -1. html
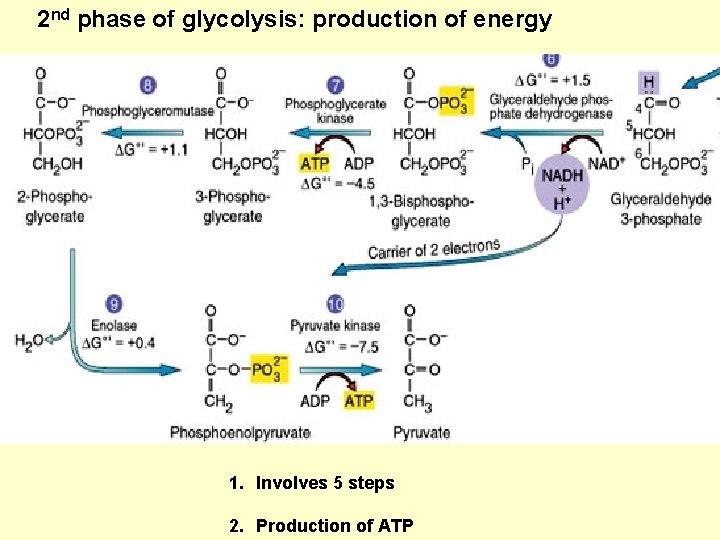
2 nd phase of glycolysis: production of energy 1. Involves 5 steps 2. Production of ATP
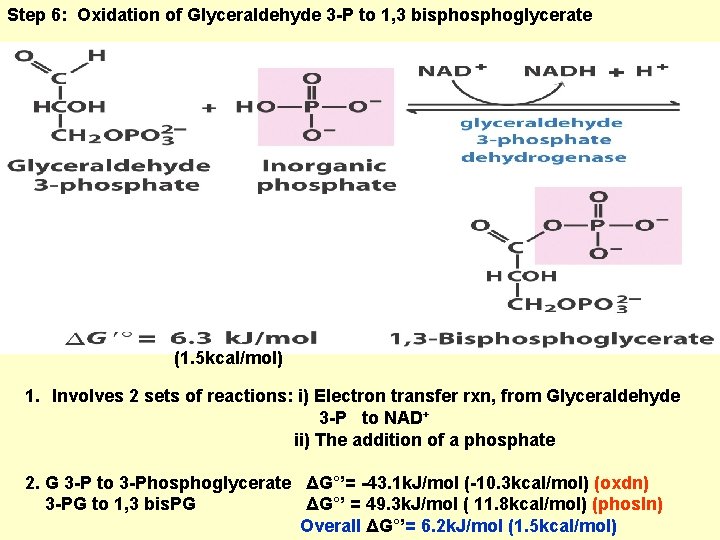
Step 6: Oxidation of Glyceraldehyde 3 -P to 1, 3 bisphoglycerate (1. 5 kcal/mol) 1. Involves 2 sets of reactions: i) Electron transfer rxn, from Glyceraldehyde 3 -P to NAD + ii) The addition of a phosphate 2. G 3 -P to 3 -Phosphoglycerate ΔG°’= -43. 1 k. J/mol (-10. 3 kcal/mol) (oxdn) 3 -PG to 1, 3 bis. PG ΔG°’ = 49. 3 k. J/mol ( 11. 8 kcal/mol) (phosln) Overall ΔG°’= 6. 2 k. J/mol (1. 5 kcal/mol)
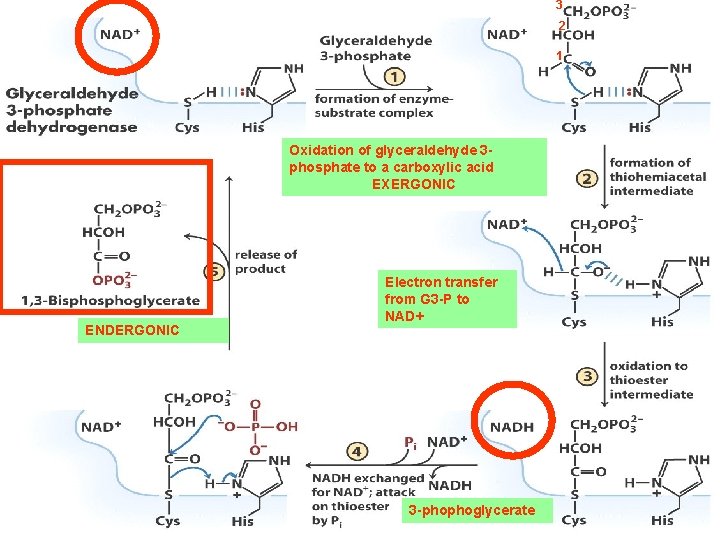
3 2 1 Oxidation of glyceraldehyde 3 phosphate to a carboxylic acid EXERGONIC ENDERGONIC Electron transfer from G 3 -P to NAD+ 3 -phophoglycerate
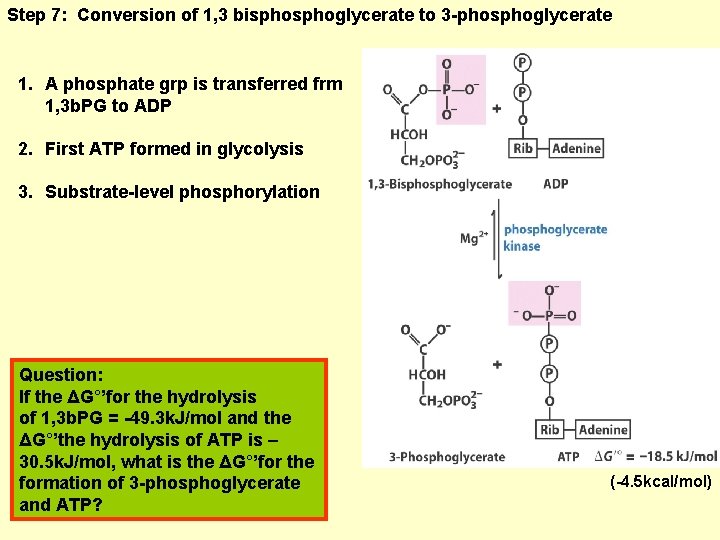
Step 7: Conversion of 1, 3 bisphoglycerate to 3 -phosphoglycerate 1. A phosphate grp is transferred frm 1, 3 b. PG to ADP 2. First ATP formed in glycolysis 3. Substrate-level phosphorylation Question: If the ΔG°’for the hydrolysis of 1, 3 b. PG = -49. 3 k. J/mol and the ΔG°’the hydrolysis of ATP is – 30. 5 k. J/mol, what is the ΔG°’for the formation of 3 -phosphoglycerate and ATP? (-4. 5 kcal/mol)
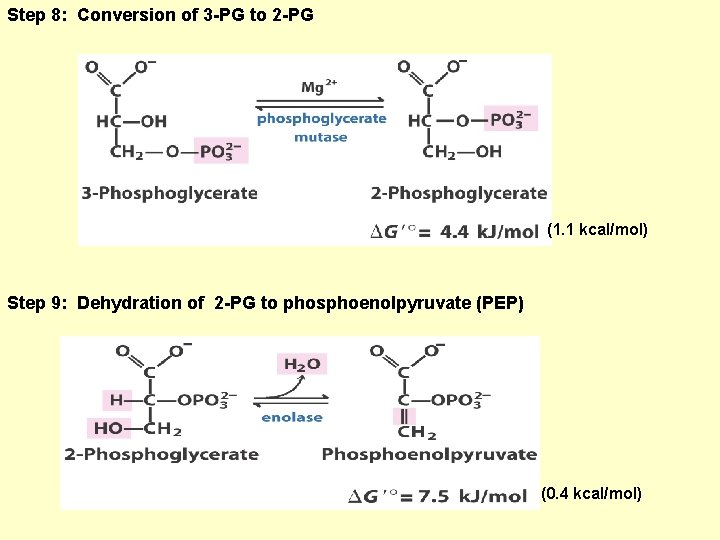
Step 8: Conversion of 3 -PG to 2 -PG (1. 1 kcal/mol) Step 9: Dehydration of 2 -PG to phosphoenolpyruvate (PEP) (0. 4 kcal/mol)
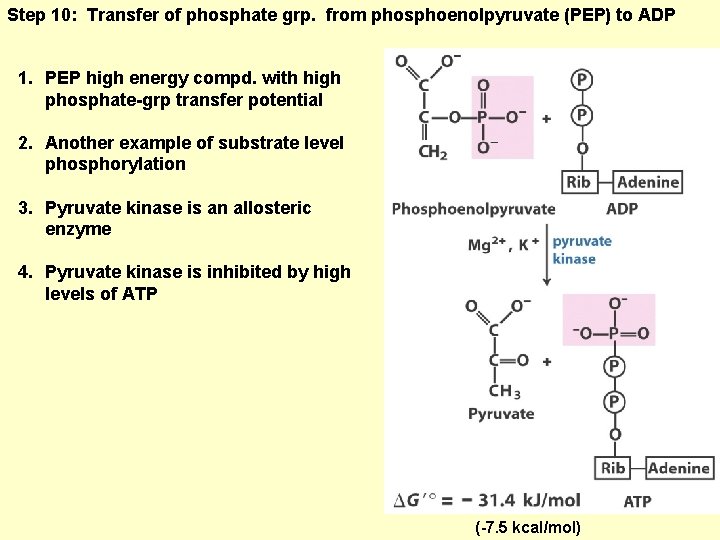
Step 10: Transfer of phosphate grp. from phosphoenolpyruvate (PEP) to ADP 1. PEP high energy compd. with high phosphate-grp transfer potential 2. Another example of substrate level phosphorylation 3. Pyruvate kinase is an allosteric enzyme 4. Pyruvate kinase is inhibited by high levels of ATP (-7. 5 kcal/mol)
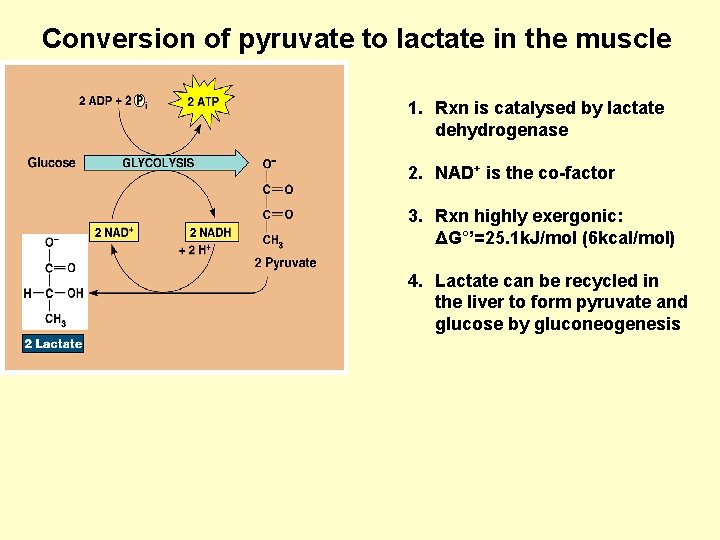
Conversion of pyruvate to lactate in the muscle 1. Rxn is catalysed by lactate dehydrogenase 2. NAD+ is the co-factor 3. Rxn highly exergonic: ΔG°’=25. 1 k. J/mol (6 kcal/mol) 4. Lactate can be recycled in the liver to form pyruvate and glucose by gluconeogenesis

www. biologie. uni-hamburg. de/b-online/e 19/19 d. htm
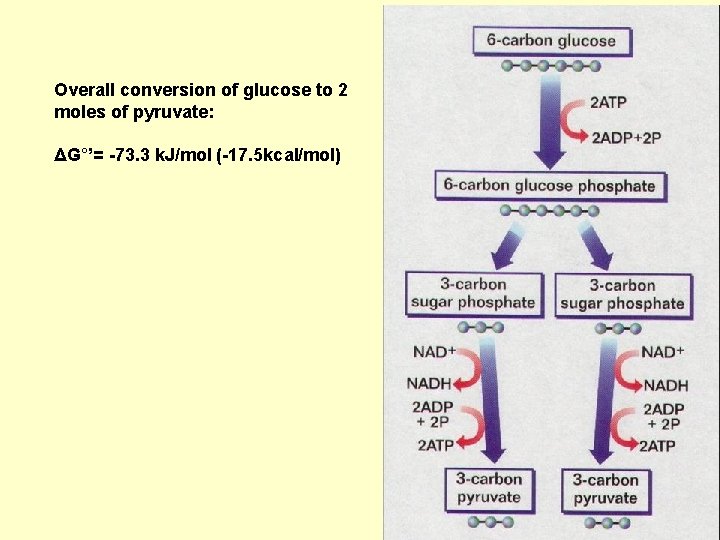
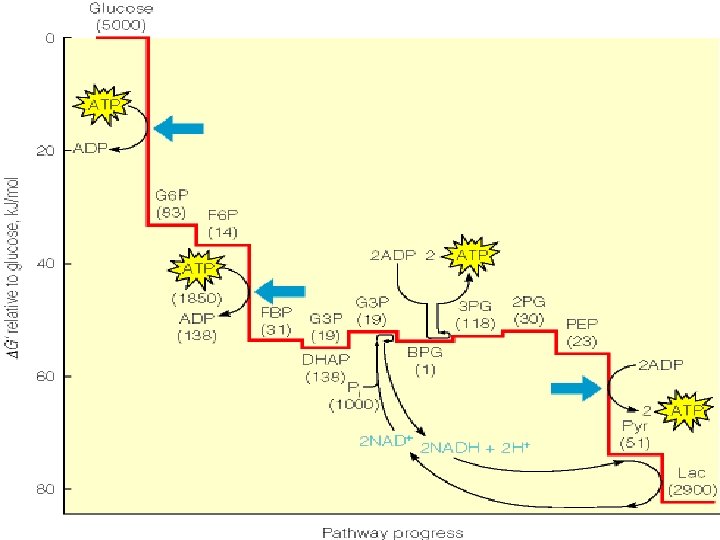
Overall conversion of glucose to 2 moles of pyruvate: ΔG°’= -73. 3 k. J/mol (-17. 5 kcal/mol)
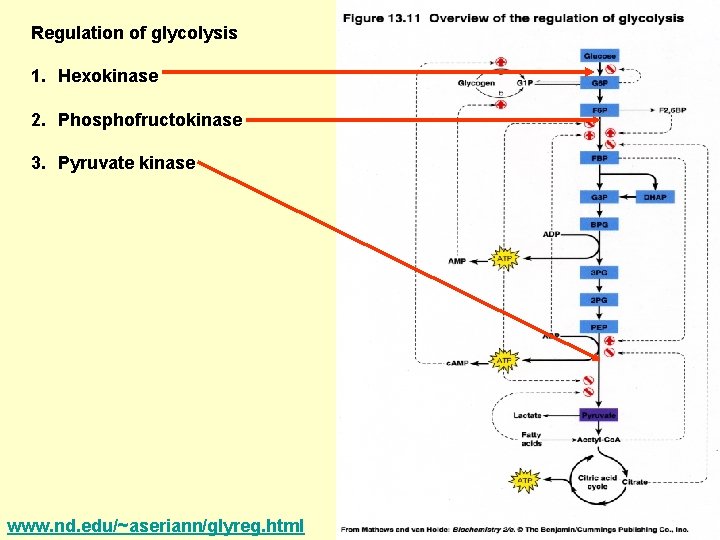
Regulation of glycolysis 1. Hexokinase 2. Phosphofructokinase 3. Pyruvate kinase www. nd. edu/~aseriann/glyreg. html
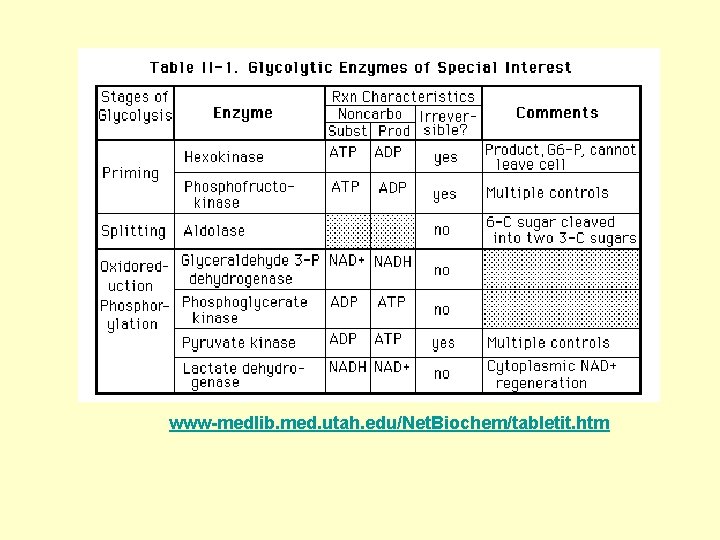
www-medlib. med. utah. edu/Net. Biochem/tabletit. htm
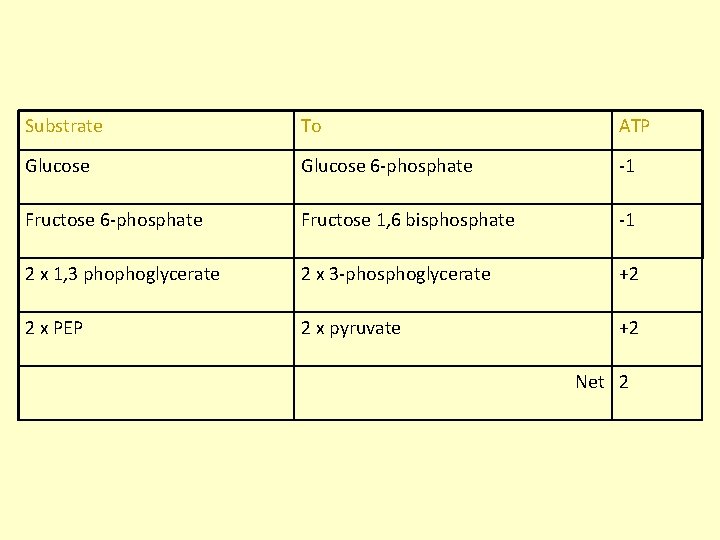
Substrate To ATP Glucose 6 -phosphate -1 Fructose 6 -phosphate Fructose 1, 6 bisphosphate -1 2 x 1, 3 phophoglycerate 2 x 3 -phosphoglycerate +2 2 x PEP 2 x pyruvate +2 Net 2

ATP production and Efficiency of Glycolysis Glucose + 2 ADP + 2 Pi 2 Lactate + 2 ATP G ’ = -184. 5 k. J/mol(-44. 1 kcal/mol) But in glycolysis only 2 ATPs are formed when glucose is oxidised to lactate. To form the ATP molecules would require : 161. 1 k. J/mol(-14. 6 kcal/mol) 2 ADP + 2 Pi 2 ATP ΔG°’= 61. 1 k. J/mol(-14. 6 kcal/mol) % of energy conserved is 61. 1/184. 5 x 100 = 33. 1%
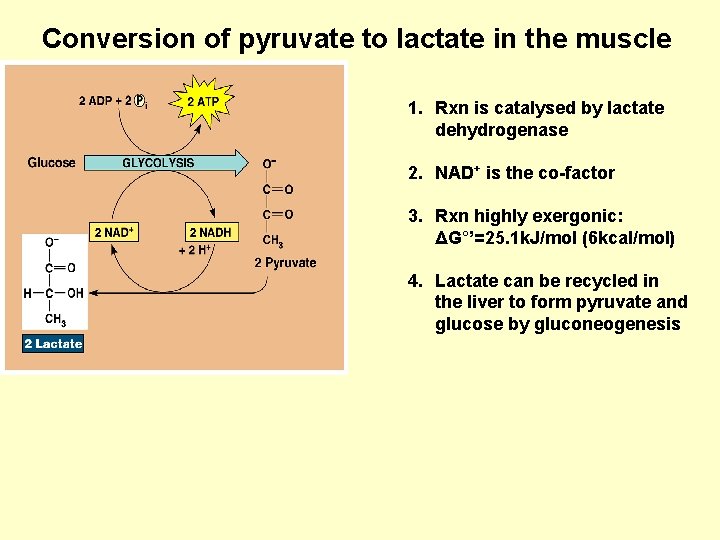
Conversion of pyruvate to lactate in the muscle 1. Rxn is catalysed by lactate dehydrogenase 2. NAD+ is the co-factor 3. Rxn highly exergonic: ΔG°’=25. 1 k. J/mol (6 kcal/mol) 4. Lactate can be recycled in the liver to form pyruvate and glucose by gluconeogenesis
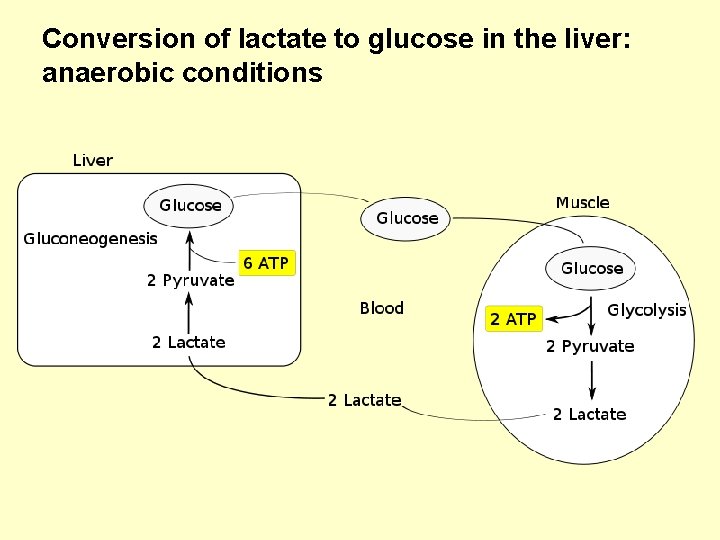
Conversion of lactate to glucose in the liver: anaerobic conditions
- Slides: 31