Glycemic Durability of Rosiglitazone Metformin or Glyburide Monotherapy

Glycemic Durability of Rosiglitazone, Metformin, or Glyburide Monotherapy Steven E. Kahn, M. B. , Ch. B. , Steven M. Haffner, M. D. , Mark A. Heise, Ph. D. , William H. Herman, M. D. , M. P. H. , Rury R. Holman, F. R. C. P. , Nigel P. Jones, M. A. , Barbara G. Kravitz, M. S. , John M. Lachin, Sc. D. , M. Colleen O'Neill, B. Sc. , Bernard Zinman, M. D. , F. R. C. P. C. , Giancarlo Viberti, M. D. , F. R. C. P. , for the ADOPT Study Group Study Overview v v v This double-blind, randomized, controlled trial evaluated rosiglitazone, metformin, and glyburide as an initial treatment in patients with type 2 diabetes Rosiglitazone reduced the risk of treatment failure (the primary outcome) by 32% as compared with metformin and by 63% as compared with glyburide The potential risks, benefits, and costs of these medications should all be considered to help inform the choice of pharmacotherapy for patients with type 2 diabetes Kahn SE, et al. (ADOPT Study Group) N Engl J Med 2006; 355: 2427 -43
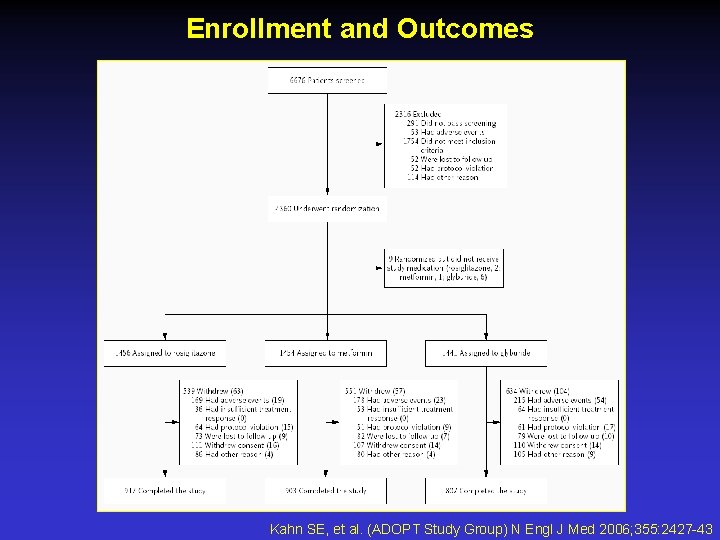
Enrollment and Outcomes Kahn SE, et al. (ADOPT Study Group) N Engl J Med 2006; 355: 2427 -43
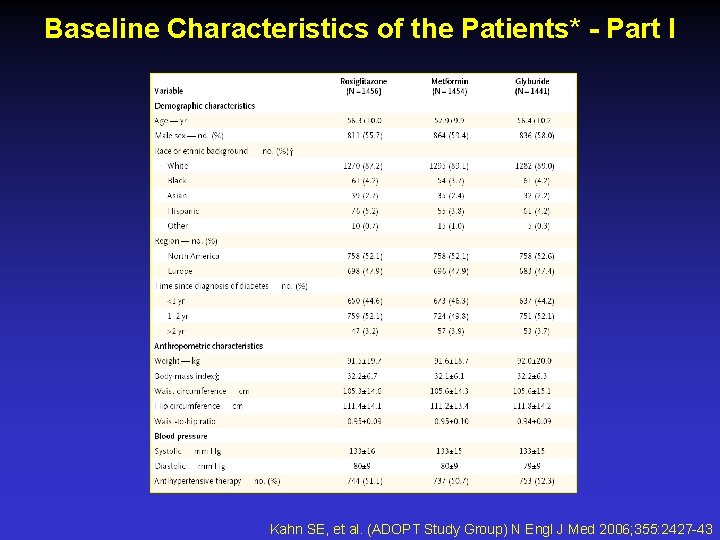
Baseline Characteristics of the Patients* - Part I Kahn SE, et al. (ADOPT Study Group) N Engl J Med 2006; 355: 2427 -43
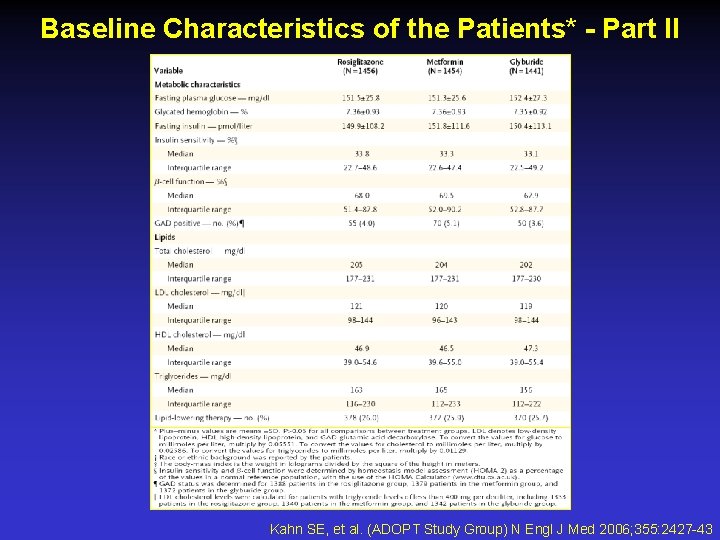
Baseline Characteristics of the Patients* - Part II Kahn SE, et al. (ADOPT Study Group) N Engl J Med 2006; 355: 2427 -43
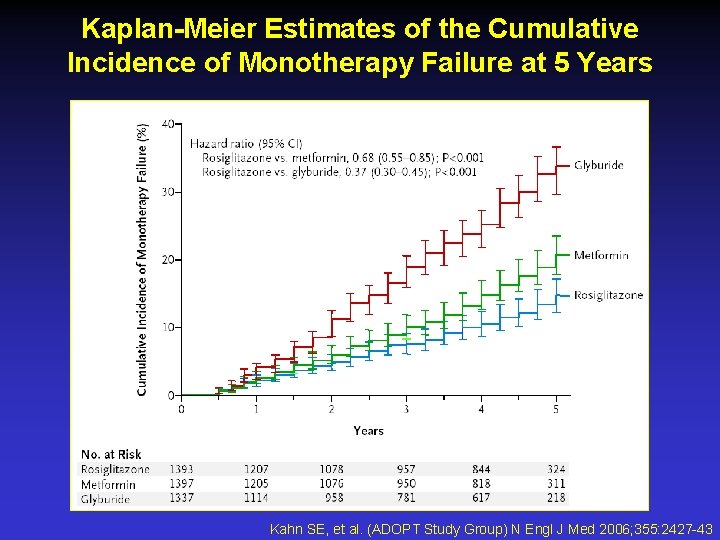
Kaplan-Meier Estimates of the Cumulative Incidence of Monotherapy Failure at 5 Years Kahn SE, et al. (ADOPT Study Group) N Engl J Med 2006; 355: 2427 -43
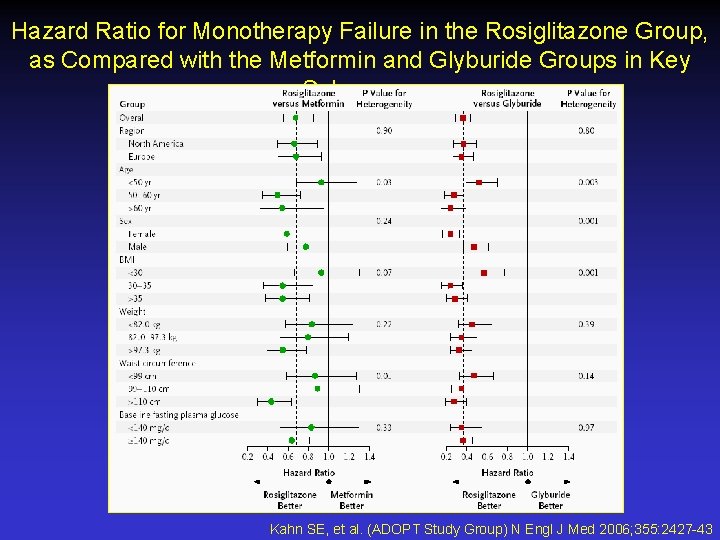
Hazard Ratio for Monotherapy Failure in the Rosiglitazone Group, as Compared with the Metformin and Glyburide Groups in Key Subgroups Kahn SE, et al. (ADOPT Study Group) N Engl J Med 2006; 355: 2427 -43
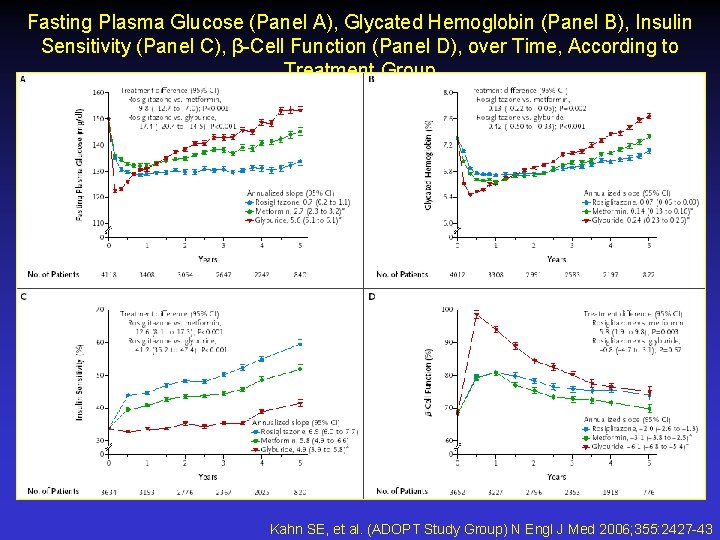
Fasting Plasma Glucose (Panel A), Glycated Hemoglobin (Panel B), Insulin Sensitivity (Panel C), β-Cell Function (Panel D), over Time, According to Treatment Group Kahn SE, et al. (ADOPT Study Group) N Engl J Med 2006; 355: 2427 -43
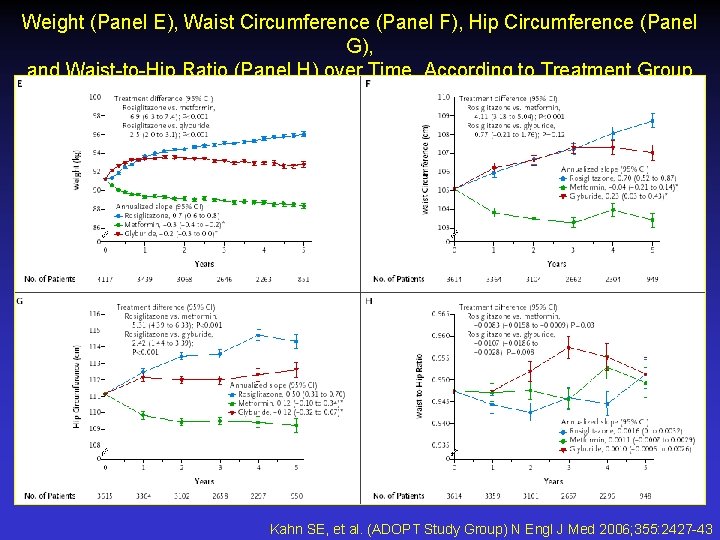
Weight (Panel E), Waist Circumference (Panel F), Hip Circumference (Panel G), and Waist-to-Hip Ratio (Panel H) over Time, According to Treatment Group Kahn SE, et al. (ADOPT Study Group) N Engl J Med 2006; 355: 2427 -43
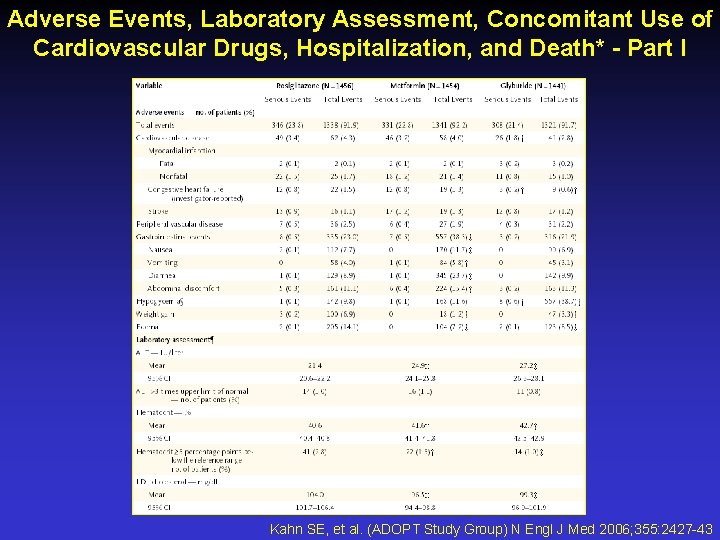
Adverse Events, Laboratory Assessment, Concomitant Use of Cardiovascular Drugs, Hospitalization, and Death* - Part I Kahn SE, et al. (ADOPT Study Group) N Engl J Med 2006; 355: 2427 -43
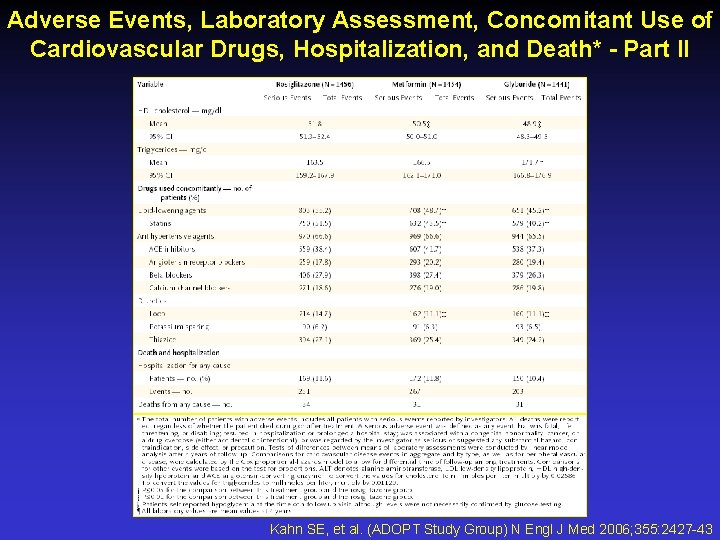
Adverse Events, Laboratory Assessment, Concomitant Use of Cardiovascular Drugs, Hospitalization, and Death* - Part II Kahn SE, et al. (ADOPT Study Group) N Engl J Med 2006; 355: 2427 -43
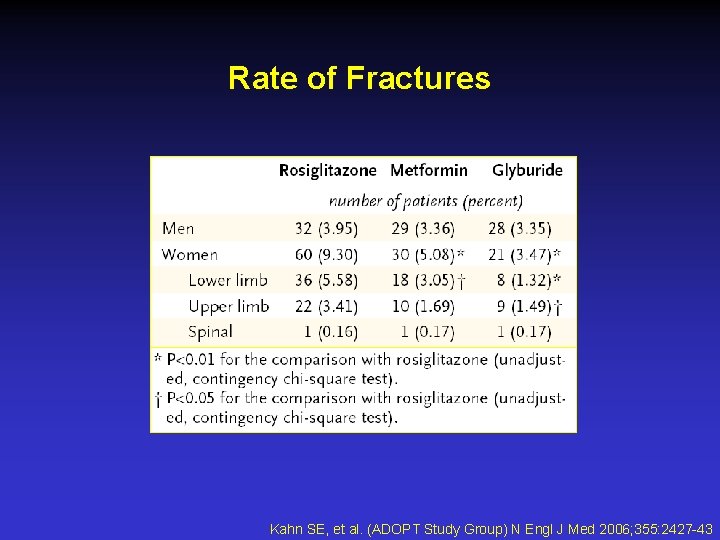
Rate of Fractures Kahn SE, et al. (ADOPT Study Group) N Engl J Med 2006; 355: 2427 -43
- Slides: 11