GLUCONEOGENESIS Definition Definition the biosynthesis of glucose from

-- GLUCONEOGENESIS -- Definition: Definition the biosynthesis of glucose from simpler molecules, primarily pyruvate and its precursors.

The gluconeogenesis pathway is similar to the reverse of glycolysis but differs at critical sites. control of these opposing pathways is reciprocal so that physiological conditions favoring one disfavor the other and vice versa. General principles of metabolic control -- a) pathways are not simple reversals of each other and b) under reciprocal control

Why do we produce glucose? a) Need to maintain glucose levels in a narrow range in blood. b) Some tissue-- brain, erythrocytes, and muscles in exertion use glucose at a rapid rate and sometimes require glucose in addition to dietary glucose. c) The brain uses mostly glucose and erythrocytes can use only glucose as a source of energy.

Where is glucose synthesized? The liver comes to rescue. The liver is the major location for gluconeogenesis.

What are the sources of precursors? pyruvate- major precursor lactate–from muscle, forms pyruvate some amino acid carbon skeletons- from diet or breakdown of muscle protein during starvation- most important is alanine TCA cycle intermediates propionate from breakdown of fatty acids and amino acids. glycerol from certain lipids.

Cost: The production of glucose is energy expensive. Input: 2 pyruvate + 4 ATP + 2 GTP + 2 NADH Output: glucose + 4 ADP + 2 GDP + 2 NAD++ 6 Pi
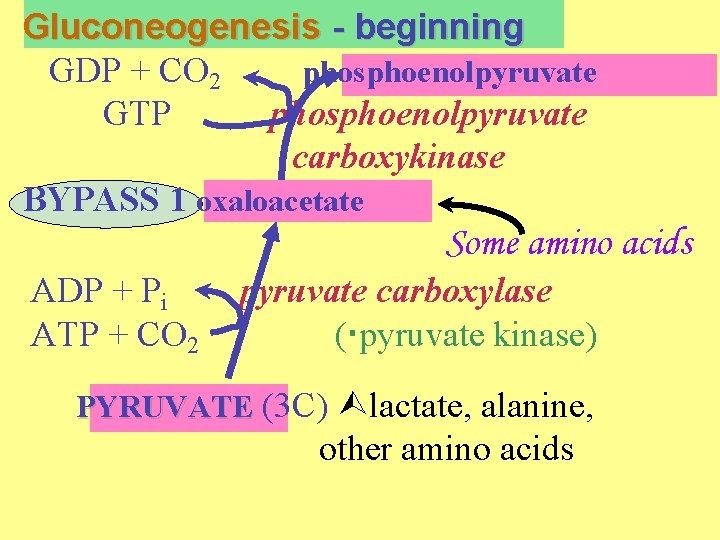
Gluconeogenesis - beginning GDP + CO 2 phosphoenolpyruvate GTP phosphoenolpyruvate carboxykinase BYPASS 1 oxaloacetate Some amino acids ADP + Pi ATP + CO 2 pyruvate carboxylase ( pyruvate kinase) PYRUVATE (3 C) lactate, alanine, other amino acids
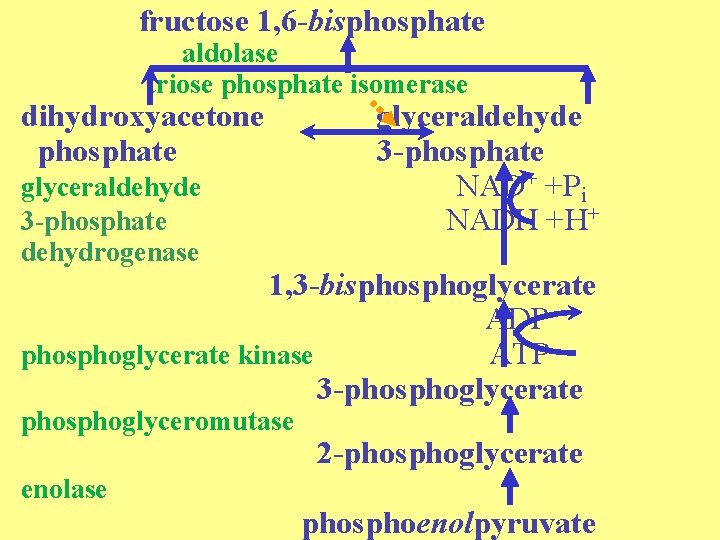
fructose 1, 6 -bisphosphate aldolase triose phosphate isomerase dihydroxyacetone phosphate glyceraldehyde 3 -phosphate dehydrogenase glyceraldehyde 3 -phosphate NAD+ +Pi NADH +H+ 1, 3 -bisphoglycerate ADP phosphoglycerate kinase ATP 3 -phosphoglycerate phosphoglyceromutase enolase 2 -phosphoglycerate phosphoenolpyruvate
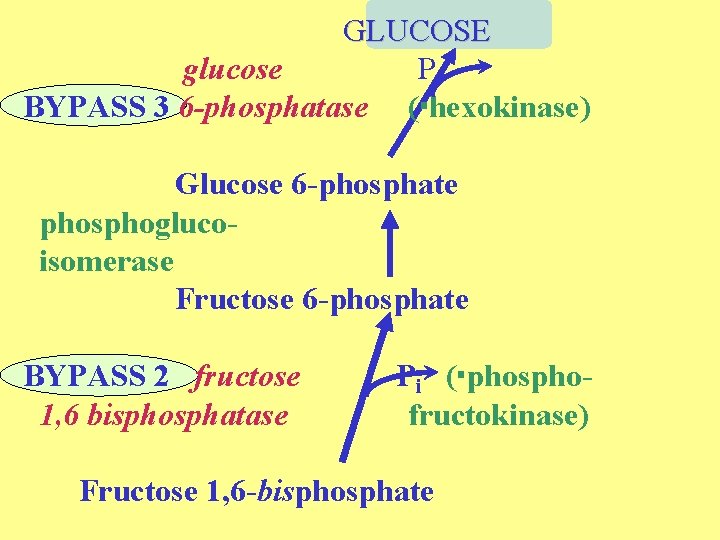
GLUCOSE glucose Pi BYPASS 3 6 -phosphatase ( hexokinase) Glucose 6 -phosphate phosphoglucoisomerase Fructose 6 -phosphate BYPASS 2 fructose 1, 6 bisphosphatase Pi ( phosphofructokinase) Fructose 1, 6 -bisphosphate

Pyruvate major precursor for gluconeogenesis.

Lactate is the primary source for pyruvate. -- In muscle, lactate is produced in great quantities during exertion. -- This excess lactate cannot be further oxidized in muscle. -- Lactate is released from the muscles to the blood and travels to the liver for conversion to pyruvate and, ultimately to glucose.

In glycolysis, there are three irreversible kinase reactions at control points involving: hexokinase, phosphofructokinase, and pyruvate kinase

In gluconeogenesis, these reactions must be forced the other way. The control points are the same for gluconeogenesis and for glycolysis. Four unique enzymes are used to bypass these irreversible steps. The rest of the steps use the same enzymes as glycolysis.

Bypass number 1. Pyruvate to phosphoenolpyruvate. This bypasses pyruvate kinase. Complex scheme.

a) pyruvate to oxaloacetate Enzyme = pyruvate carboxylase located inside mitochondria. Only this enzyme of the gluconeogenesis pathway is mitochondrial.

Reaction: pyruvate + CO 2 + ATP + H 2 O oxaloacetate + ADP + Pi -- Carboxylations involving CO 2 almost always use the vitamin biotin as a cofactor.

-- Subreactions: Enz-biotin + ATP + CO 2 + H 2 O Enz-carboxybiotin + ADP + Pi Enz-carboxybiotin + pyruvate Enz-biotin + oxaloacetate

-- pyruvate carboxylase - acetyl-Co. A is positive modulator absolutely required for activity higher acetyl-Co. A indicates that adequate carbon levels available for TCA cycle to provide energy glucose can be synthesized and exported from liver. oxaloacetate important in the citric acid cycle, which is more mitochondrial.

For gluconeogenesis, oxaloacetate must leave the mitochondria because all the rest of the gluconeogenesis enzymes are in the cytosol. mitochondrial membranes are nearly impermeable to oxaloacetate. So how does it get out?
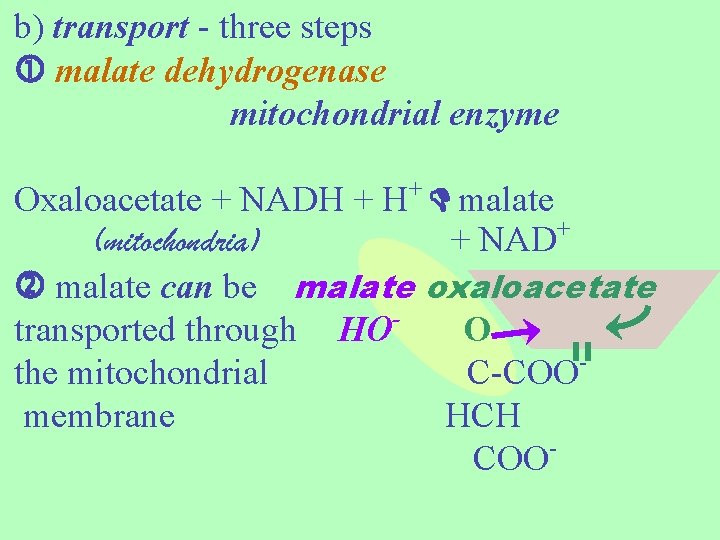
b) transport - three steps malate dehydrogenase mitochondrial enzyme Oxaloacetate + NADH + H+ malate (mitochondria) + NAD+ malate can be malate oxaloacetate transported through HO O the mitochondrial C-COOmembrane HCH COO

malate dehydrogenase -- cytosolic enzyme + malate + NAD oxaloacetate + (cytosolic) NADH + H+

c) The GTP-dependent decarboxylation of oxaloacetate Enzyme = PEP carboxykinase cytosolic enzyme, as all others oxaloacetate + GTP phosphoenolpyruvate + CO 2 + GDP uses GTP, not ATP. CO 2 added is lost in this step. NET so far: pyruvate + ATP + GTP PEP + ADP + GDP + Pi

High cost = two energy rich phosphates so a total of four high energy bonds are already utilized here per glucose to be synthesized. Then uses glycolytic enzymes in steps to fructose-1, 6 -bisphosphate

Bypass number 2. Fructose-1, 6 bisphosphate to fructose-6 -phosphate Enzyme = fructose-1, 6 -bisphosphatase Reaction: fructose-1, 6 -bisphosphate + H 2 O fructose-6 -P + Pi bypasses phosphofructokinase a simple hydrolysis. highly exergonic, irreversible enzyme is highly regulated F 6 P isomerizes to glucose-6 -phosphate via phosphoglucoisomerase

Bypass number 3. Glucose-6 -phosphate to glucose. Enzyme = glucose-6 -phosphatase Reaction: glucose-6 -phosphate + H 2 O glucose + Pi bypasses hexokinase highly exergonic, irreversible not present in muscle

******************* * Total Energy Cost = 6 high energy bonds used per glucose synthesized. four more than produced in glycolysis. These four are needed to convert pyruvate to PEP. *******************

CONTROL: -- gluconeogenesis serves as an alternative source of glucose when supplies are low and is largely controlled by diet. -- high carbohydrate in meal reduce gluconeogenesis and fasting increases. -- key enzymes targeted. -- gluconeogenesis and glycolysis are controlled in reciprocal fashion.

REGULATION OF GLUCONEOGENESIS: Key enzymes: 1. PYRUVATE CARBOXYLASE. activated by acetyl-Co. A (required) vs. pyruvate kinase, inhibited by acetyl Co. A. high levels of acetyl-Co. A signals that enough carbon substrate available for citric acid cycle. pyruvate kinase is also inhibited by ATP and the liver form by alanine.

2. FRUCTOSE 1, 6 BISPHOSPHATASE Strongly inhibited by AMP, low energy Strongly inhibited by fructose-2, 6 -bisphosphate, high glucose Recall that the reciprocal enzyme, phosphofructokinase, in glycolysis, is strongly activated by AMP and fructose-2, 6 -bisphosphate.

Fructose-2, 6 -bisphosphate is the most important regulator of glycolysis and gluconeogenesis through its reciprocal effects on fructose 1, 6 -bisphosphatase and phosphofructokinase.

3. PHOSPHOFRUCTOKINASE-2/ FRUCTOSE BISPHOSPHATASE-2 Bifunctional enzyme fructose-6 -phosphate + ATP fructose-2, 6 -bisphosphate fructose-6 -phosphate + Pi Enzyme highly regulated to control levels of F 2, 6 -P
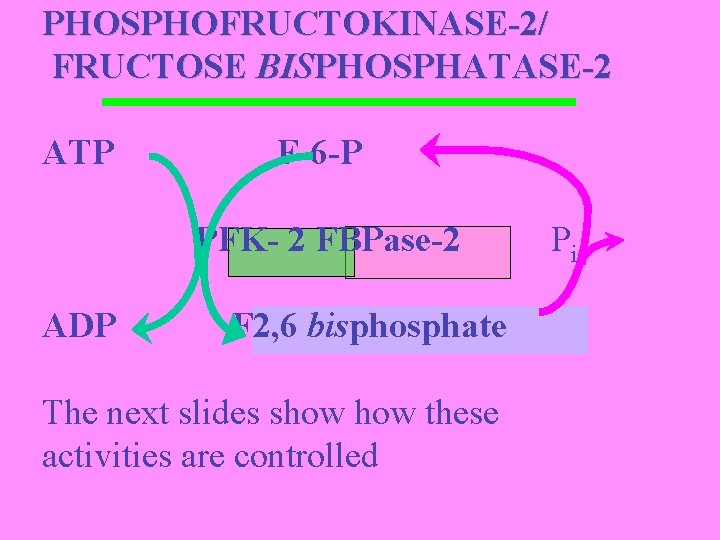
PHOSPHOFRUCTOKINASE-2/ FRUCTOSE BISPHOSPHATASE-2 ATP F-6 -P PFK- 2 FBPase-2 ADP F 2, 6 bisphosphate The next slides show these activities are controlled Pi
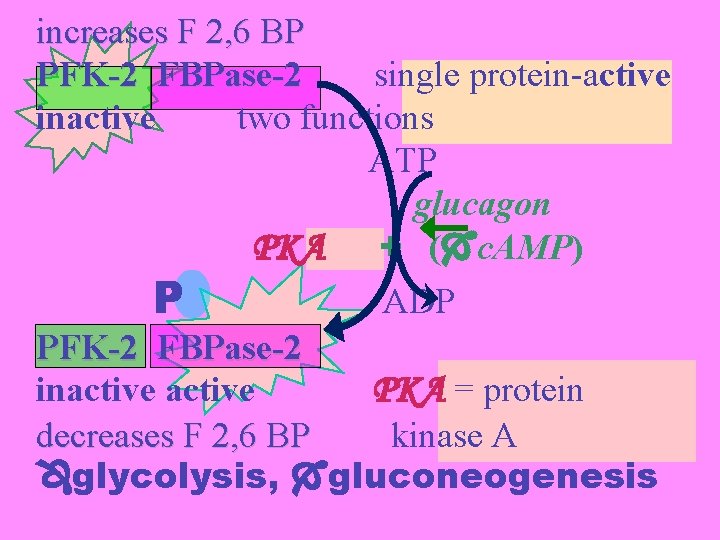
increases F 2, 6 BP PFK-2 FBPase-2 single protein-active inactive two functions ATP glucagon PKA + ( c. AMP) P ADP PFK-2 FBPase-2 inactive PKA = protein decreases F 2, 6 BP kinase A glycolysis, gluconeogenesis

Glucagon: hormone released when glucose levels are low. signal to elevate blood glucose levels increases intracellular levels of c. AMP in liver and elsewhere c. AMP activates protein kinase A = c. AMP-dependent protein kinase stimulating gluconeogenesis and glycogenolysis

Consequences A. glucose & AMP PFK-2 F 2, 6 bis. P PFK-1 glycolysis Also F 1, 6 BPase gluconeogenesis B. glucose & ATP c. AMP FBPase-2 F 2, 6 bis. P F 1, 6 BPase gluconeogenesis Also PFK-1 glycolysis

Summary of Gluconeogenesis purpose- alternative source of glucose rather than dietary carbohydrates or glycogen breakdown primary precursors are pyruvate, lactate, glycerol, part of fatty acids and certain amino acids (glucogenic) 3 essentially irreversible steps of glycolysis are bypassed

regulated via pyruvate carboxylase, fructose 1, 6 bisphosphatase, and phosphofructokinase-2/fructose bisphosphatase-2 ENotice glucose cannot be made from acetyl Co. A

GLYCOGEN METABOLISM Glycogen: a highly branched polymer of glucose. Chains have glycosidic links α 1 4. Branches are linked α 1 6.

Glucose stored in polymeric form as glycogen mostly in the liver and skeletal muscle. Glucose can be rapidly delivered to the blood stream when needed upon degradation of glycogen. = glycogenolysis Enough glucose and energy triggers synthesis of glycogen. = glycogenesis

Glycogenesis = GLYCOGEN BIOSYNTHESIS =============== JGlucose is transported into the liver cell by a specific glucose transporter and immediately phosphorylated. =============== -- Most of the glucose in a cell is in the form of glucose-6 -phosphate.

1 - Conversion of glucose-6 -phosphate to glucose-1 -phosphate Enzyme = phosphoglucomutase α-D-glucose-6α-D- glucose-1 phosphate reversible reaction allows G 1 P conversion to G 6 P in glycogenolysis mechanism involves phosphorylated enzyme intermediate and glucose-1, 6 bisphosphate bound intermediate similar to phosphoglycerate mutase
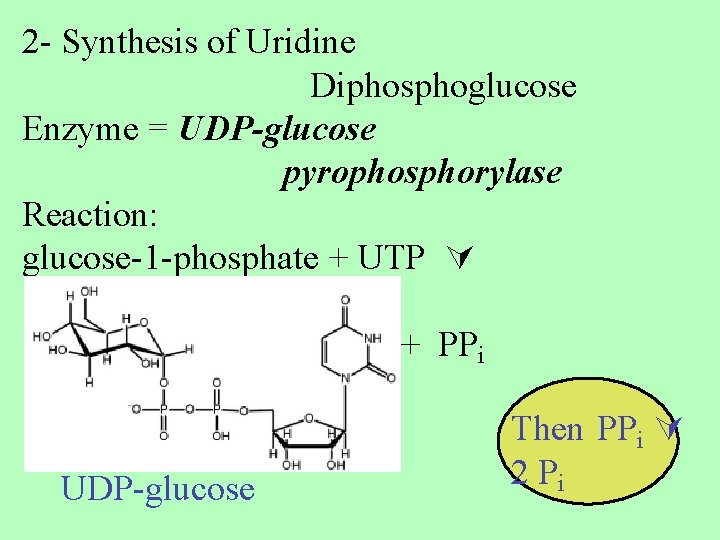
2 - Synthesis of Uridine Diphosphoglucose Enzyme = UDP-glucose pyrophosphorylase Reaction: glucose-1 -phosphate + UTP + PPi UDP-glucose Then PPi 2 Pi

the commiting step Phosphoryl transfer -- UTP is the energy equivalent of ATP -- energy is used to activate glucose -- two phosphates from UTP are lost as PPi is broken down by an enzyme PPi 2 Pi driving the reaction to the right
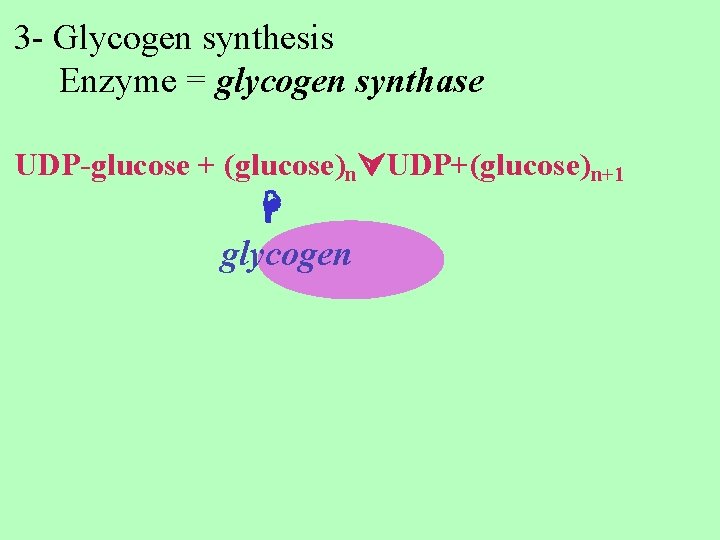
3 - Glycogen synthesis Enzyme = glycogen synthase UDP-glucose + (glucose)n UDP+(glucose)n+1 glycogen
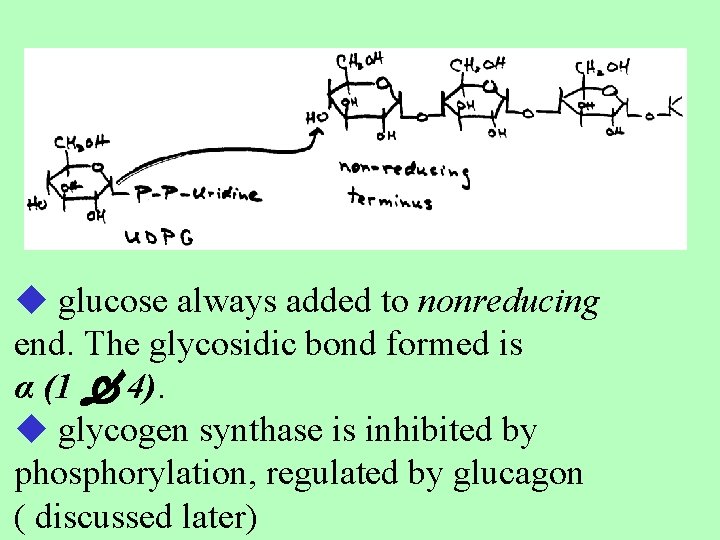
glucose always added to nonreducing end. The glycosidic bond formed is α (1 4). glycogen synthase is inhibited by phosphorylation, regulated by glucagon ( discussed later)

4 - Branching Enzyme = branching enzyme introduces branching by transferring a teminal fragment of 6 -7 residues from a growing chain to a 6 -position farther back in a chain. makes a branch with an α (1 6) link creating two ends to add glucose. branching accelerates the rate of glucose release during degradation.
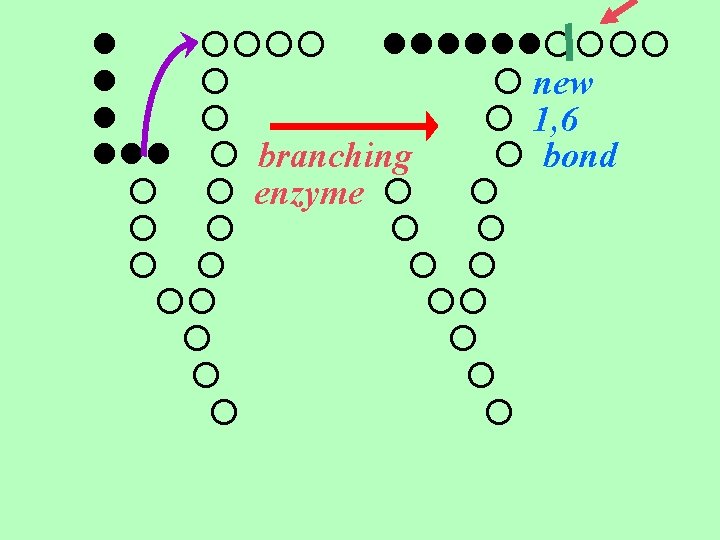
new 1, 6 branching bond enzyme

-- GLYCOGENOLYSIS-DEGRADATION OF GLYCOGEN 1. Release of glucose-1 -phosphate Enzyme = glycogen phosphorylase nonreducing + Pi ends glucose-1 - + phosphate
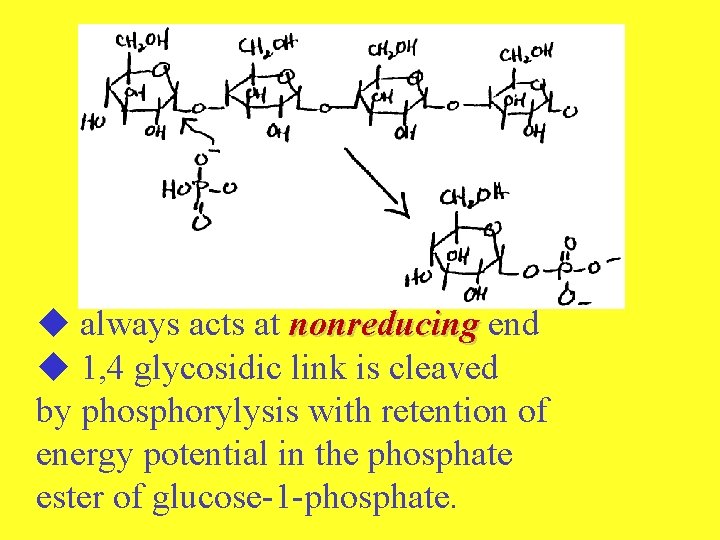
always acts at nonreducing end 1, 4 glycosidic link is cleaved by phosphorylysis with retention of energy potential in the phosphate ester of glucose-1 -phosphate.

stops at fourth glucose from a 1, 6 branch point contrast with enzymes acting on starch and glycogen in the gut, which yield sugars, not sugar phosphates, as products. activated by phosphorylation, regulated by glucagon and epinephrine
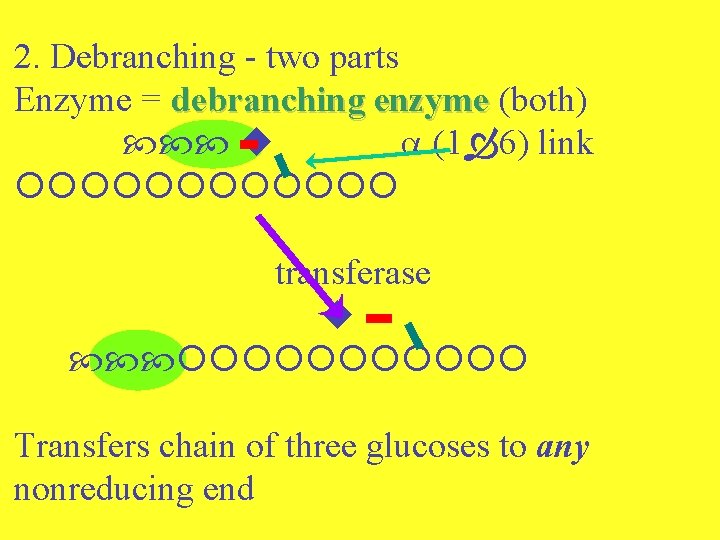
2. Debranching - two parts Enzyme = debranching enzyme (both) (1 6) link transferase Transfers chain of three glucoses to any nonreducing end
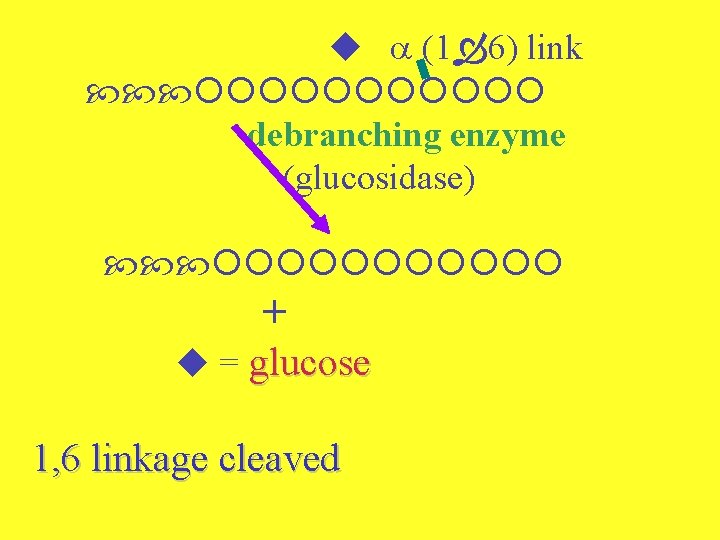
(1 6) link debranching enzyme (glucosidase) + = glucose 1, 6 linkage cleaved

glycogen phosphorylase or phosphorylase for short glucose-1 -phosphate one at a time as previously shown -- phosphoglucomutase then yields glucose-6 -phosphate, which can be dephosphorylated or enter glycolysis.

Metabolic Regulation of Mammalian Glycogen Levels -- Glycogen reserves are the most immediately available large source of metabolic energy for mammals. -- Storage and utilization are under dietary and hormonal control.

Primary hormones = -- epinephrine (adrenaline) = “fight-or-flight” -- glucagon -- insulin Primary enzyme targets in glycogen metabolism= glycogen phosphorylase and glycogen synthase. The actions of the hormones are indirect.

Example- hormones and diet Dinner at 9: 00 pm -- steak, mashed potatoes, sherbert for dessert, wine Sleep immediately, sleep late During sleep: amino acids, CH 2 O high blood glucose levels higher insulin glycogenesis

Wake late for class adrenaline rush run to class glycogen glucose lactate epinephrine (= adrenaline) glycogenolysis

==== HORMONES ===== Glucagon - low glucose levels -- A polypeptide hormone produced in α-cells of the islets of Langerhan of the pancreas. -- Acts primarily on liver cells. -- Receptors on surface of liver cells. -- Stimulates glycogen breakdown & inhibits glycogenesis. -- Glucagon also blocks glycolysis & stimulates gluconeogenesis.

Epinephrine - low glucose levels -- Acts primarily on skeletal muscle. -- Receptors on surface of cells. -- Stimulates glycogen breakdown & inhibits glycogenesis. Glucagon and epinephrine both stimulate intracellular pathway via increasing levels of c. AMP.

Insulin -- High levels of glucose induce release of insulin from β-cells of islets of Langerhan in the pancreas. -- Insulin is polypeptide hormone. -- Detected by receptors at surface of muscle cells. -- Increases glycogenesis in muscle. -- Intracellular signal pathway involves complex sequential phosphorylations and dephosphorylations.
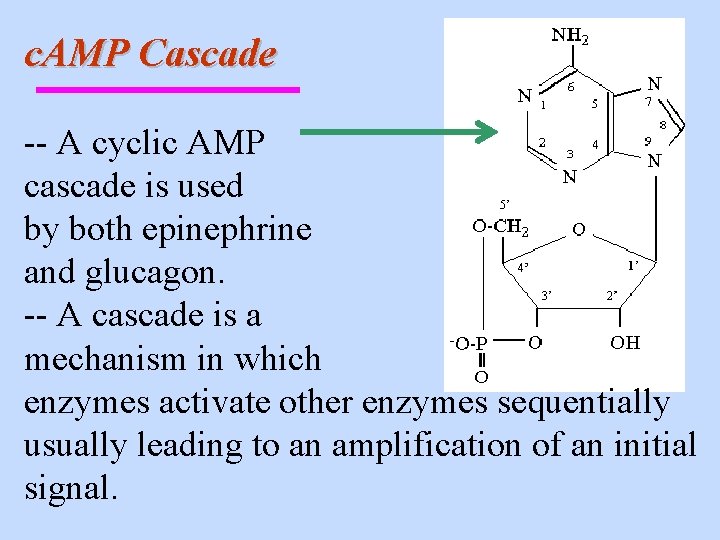
c. AMP Cascade -- A cyclic AMP cascade is used by both epinephrine and glucagon. -- A cascade is a mechanism in which enzymes activate other enzymes sequentially usually leading to an amplification of an initial signal.
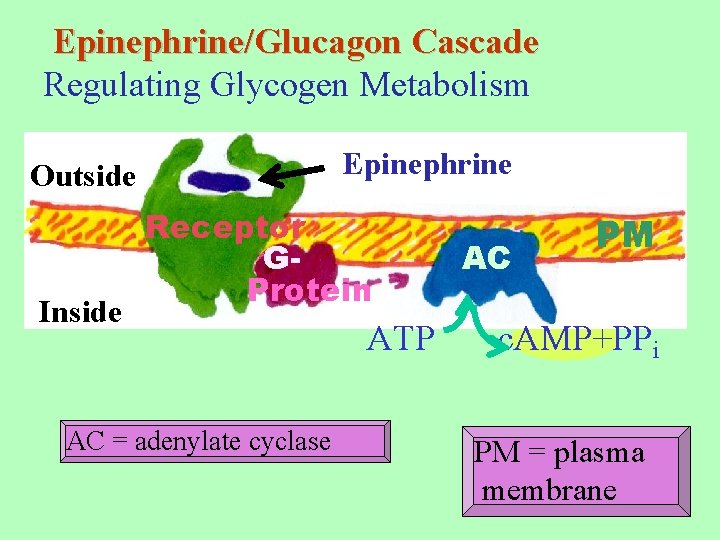
Epinephrine/Glucagon Cascade Regulating Glycogen Metabolism Epinephrine Outside Inside Receptor GProtein AC = adenylate cyclase ATP AC PM c. AMP+PPi PM = plasma membrane
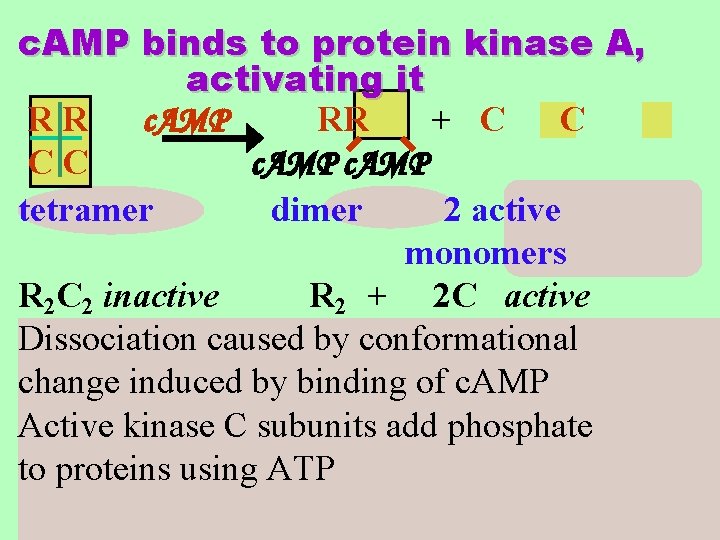
c. AMP binds to protein kinase A, activating it RR c. AMP RR + C C CC c. AMP tetramer dimer 2 active monomers R 2 C 2 inactive R 2 + 2 C active Dissociation caused by conformational change induced by binding of c. AMP Active kinase C subunits add phosphate to proteins using ATP
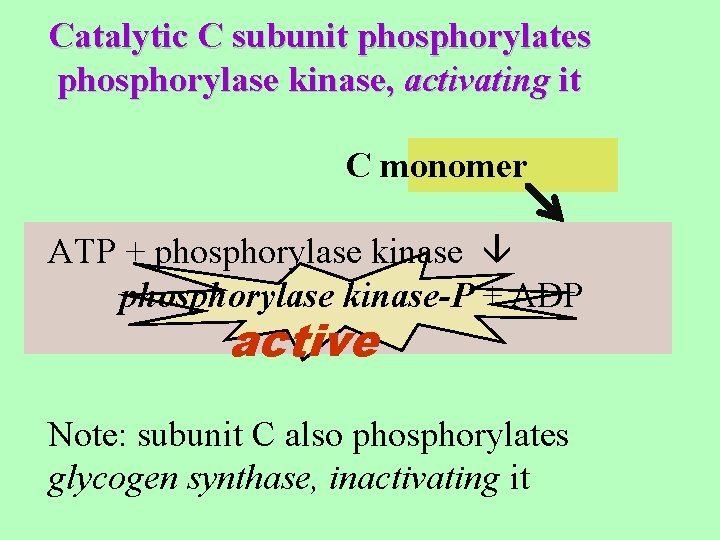
Catalytic C subunit phosphorylates phosphorylase kinase, activating it C monomer ATP + phosphorylase kinase-P + ADP active Note: subunit C also phosphorylates glycogen synthase, inactivating it

Active phosphorylase kinase transfers phosphate to glycogen phosphorylase, activating this enzyme. ATP + phosphorylase b ADP + phosphorylase a- P Glycogen phosphorylase a (active form) then degrades glycogen: glycogen + Pi glucose-1 -P+(glucose)n-1

How is Glycogenolysis Inhibited? -- when need for glucose is over, glycogenolysis is inhibited. -- a phosphoprotein phosphatase hydrolyzes the protein phosphates to reconvert: reconvert phosphorylase a phosphorylase b (inactive) and phosphorylase kinase (active) phosphorylase kinase (inactive)

How is Glycogenesis Activated? -- Complex process stimulated by insulin. -- Insulin indirectly activates a phosphoprotein phosphatase: glycogen synthase(D) glycogen synthase(I) phosphorylated dephosphorylated less active dependent on G-6 -P active independent of G-6 -P

-- dephosphorylation is the major pathway for stimulation of glycogenesis in liver and resting muscles. -- Another way: In active muscle, there may still be high glucose-6 -phosphate. The less active phosphorylated glycogen synthase can be activated by high levels of glucose-6 -phosphate. -- bypasses hormonal regulation

How is Glycogenesis Inhibited? -- epinephrine and glucagon inhibit glycogen synthesis. a) protein kinase A subunit C phosphorylates glycogen synthase, synthase decreasing its activity. b) also phosphorylase kinase can phosphorylate glycogen synthase, inactivating it. [It’s called synthase phosphorylase kinase because of its dual function. ]

SIMPLISTIC SUMMARY: -- Epinephrine and glucagon stimulate glycogenolysis and inhibit glycogenesis via a c. AMP and a phosphorylation cascade. releases glucose -- Glycogenesis is stimulated by insulin in a pathway ending in the dephosphorylation of glycogen synthase. -- Glycogenolysis is also inhibited via dephosphorylation. takes up glucose

Glycogen Storage Diseases: -- A family of serious, although not necessarily fatal, diseases caused by mutations in the enzymes involving in glycogen storage and breakdown.

- Slides: 72