GLT 111 GENERAL LABORATORY TECHNIQUES BY EMMAUNEL IFEANYI

GLT 111 GENERAL LABORATORY TECHNIQUES BY EMMAUNEL IFEANYI NWOKEDI DEPARTMENT OF PURE AND APPLIED SCIENCES

TABLE OF CONTENT 1: General laboratory safety procedures and rules 2: Laboratory hazards and risks 3: Radiation exposure 4: Laboratory glassware 5: Common laboratory glassware and apparatus 6: Calibration 7: Electronic balance 8: Microscope 9: Microtome 10: Construction of Ammeter and Voltmeter from Galvanometer 11: Microbial contaminants 12: Chemical and acid burns 13: Classification of fire 14: Solution preparation 15: Heating apparatus 16: Temperature measuring devices

General laboratory safety procedures and rules A laboratory is a facility that provides controlled conditions in which scientific or technological research, experiments, and measurement maybe performed. Laboratories can be found in schools, universities, industries, etc. Laboratory techniques refers to the sum of procedures used on natural sciences such as chemistry, biology, and physics in order to conduct an experiment. Laboratory safety rules and emergency procedure that must be adhered to by students before any laboratory session includes; v Emergency Response v Electrical safety v Personal and General laboratory safety v Mechanical safety v Laser safety v Chemical safety v Additional Safety Guidelines

Laboratory hazards and risks Only with proper guidance and a trained eye can injury and illness be avoided in the laboratory. Understanding the required occupational safety and recognizing workplace hazards will help to identify and minimize many of the common safety and health hazards associated with running a research laboratory. Types Of Hazards Most hazards encountered fall into three main categories; 1. Chemical Hazard: the use of chemicals in the laboratory is inevitable, and the potential for harm or injury could be significant if they are misused or mishandled. 2. Biological Hazards: these encompass microbes, recombinant organisms, viral vectors, and biological agents introduced into experimental animals. 3. Physical Hazards: these are electrical safety hazards, ergonomic hazards associated with manual material handling and equipment use, handling sharps and basic housekeeping issues.

Radiation exposure Radiation is the energy in the form of particles or waves. Radiation is emitted naturally sunlight and is also made by man for use in X-ray, cancer treatment, and for nuclear facilities and weapons. Basic terms in radiation includes; Ionizing radiation, Non-ionizing radiation, Radioactivity, Radioactive material, Radioactive contamination. Common Types of Radiation 1. Gamma rays. 2. Betas. 4. Neutrons. 5. X-rays. 3. Alphas. Common Units used in measuring radiation 1. Roentgen (R). 2. Rad (radiation absorbed dose). 3. Rem (roentgen equivalent man). 4. Curie (Ci). 5. Gray (Gy). 7. Becquerel (Bq). 6. Sievert (Sv).

Laboratory glassware refers to a variety of equipment, traditionally made of glass, used for scientific experiments and other work in science, especially in chemistry and biology laboratories. There are different types of glass, e. g. Borosilicate glass, Quartz glass, Darkened brown or Amber(Actinic) glass, Heavy-wall glass. Laboratory glassware production; this involves Heating reagents or samples, Negative pressure and vacuums, Hermetic sealing. Cleaning Disposal Transparency
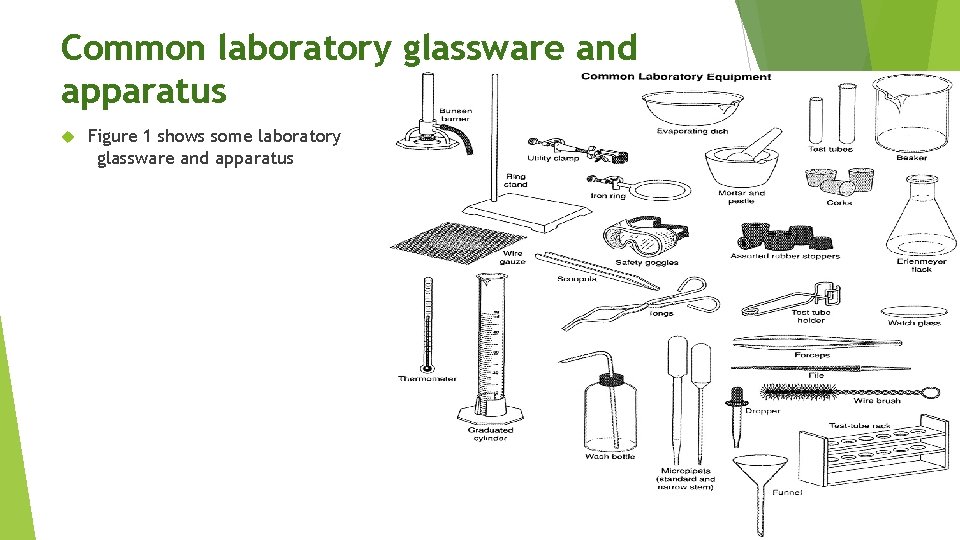
Common laboratory glassware and apparatus Figure 1 shows some laboratory glassware and apparatus

Calibration of glassware is commonly done using a liquid of known, specific density, and an analytical balance. The procedure is to determine the mass of liquid the glassware will hold, and to divide this mass of liquid by the density of the liquid, obtaining the corresponding volume of liquid. The procedure above can be used for both pipette and burette. it is important to note that the weight of substances are not taken in the laboratory rather their mass is determined.

Electronic balance Types of electronic balance; 1. Electromagnetic balancing type 2. Electrical resistance wire type (load cell type) However, there also hidden pitfalls to these electronic balances in operation and convenience that could result in committing major mistakes or error. Types of Error; 1. Sensitivity error. 4. Repeatability. 2. Linearity error. 5. Eccentric error. Causes and corrective measures for error 1. Gravitational acceleration. Containers. Static electricity. 3. Balance error 2. Temperature. 4. Air flow. 3. 5. Daily inspection of balances; When to inspection fails. What to inspect. What to do if the

Microscope The simple microscope consist of a single convex lens which produces a magnified image with limited magnification. Types of microscope; 1. Compound microscope. 2. Electron microscope. 3. Phase contrast microscope. 4. Interference microscope. 5. Polarizing microscope. 6. Dark-field microscope. Compound microscope; principle of operation and magnification Electron microscope; types of electron microscope Important microscopic terms; Numerical aperture, Cover glass, Field, Focal length, Resolving power, Objective lens, Eye pieces, Condenser, Base, Stage, Disc diaphragm, Iris diaphragm, Body, Nosepiece, Coarse adjustment knob, Fine adjustment knob, Lamp. Illumination of the object; Kohler illumination and Critical illumination. Care and maintenance of a microscope

Microtome is a machine used for cutting thin section from piece of tissue. This machine is used in the study of histology. There are different variety of microtome, but all of them are made up of the following principal components; v A knife held in a firm support v A device for holding the piece of tissue firmly v A mechanism for advancing the piece of tissue across the knife blade or the knife across the tissue. v A mechanism for advancing the piece of tissue across very small and accurately measure distance so that section of suitable thickness can be obtained. v Types of Microtomes includes; 1. Cambridge Rocker microtome. with moving block. with moving knife. 5. Freezing microtome. 2. Sledge microtome 3. Sledge microtome 4. Rotary microtome.
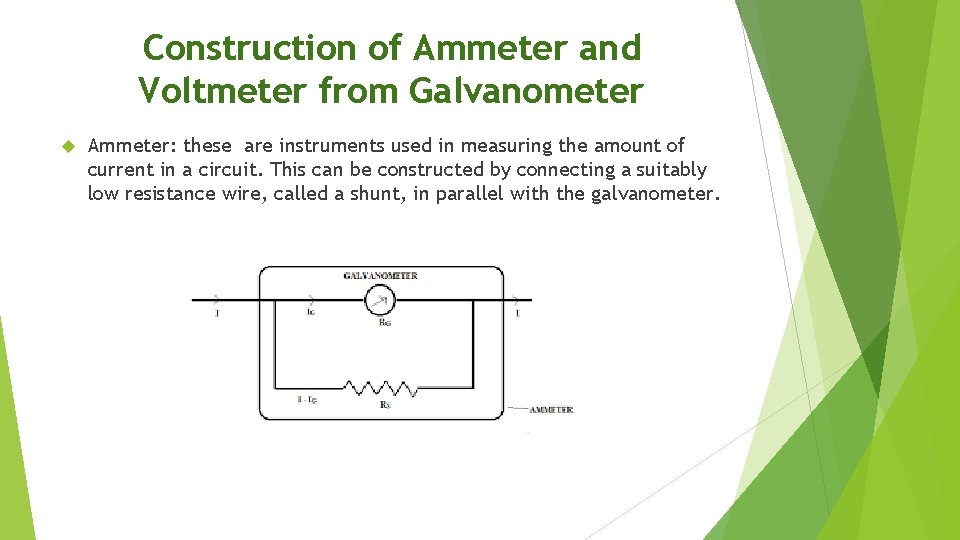
Construction of Ammeter and Voltmeter from Galvanometer Ammeter: these are instruments used in measuring the amount of current in a circuit. This can be constructed by connecting a suitably low resistance wire, called a shunt, in parallel with the galvanometer.

Construction of Ammeter and Voltmeter from Galvanometer CONT Voltmeters are instruments used to measure the potential difference across two points in a circuit. A galvanometer is converted to a voltmeter by connecting a high resistance called a multiplier.

Construction of Ammeter and Voltmeter from Galvanometer CONT Multimeter (Avometer): this is a more sensitive meter which can be used to measure high and low values of circuit

Microbial contaminants Since the development of techniques to obtain microorganisms in pure culture, the susceptibility of such culture to the unwanted growth of other microbes has been recognized, in the medical setting, microbial contamination can be life threatening. Such contamination can be lessened, if not prevented completely. There are two main measures for microbial contamination, they are; First Aid Measures and Accidental Release (spillage/environmental precaution) measures. Handling and Storage: cultures must be opened and used by trained persons in a laboratory of appropriate safety level. Exposure controls/personal protection Toxicology information Disposal

Chemical and acid burns Acid products include toilet cleaners, battery acid, bleach, etc. acid solid and liquid can cause injury, depending on the type, the strength, and the length of time the acid is in contact with the body. Most chemical burns are treated first by rinsing the chemical off your body with a large amount of cool water, while some acid burns are made worse if rinsed with water, they are; carboxylic acid or phenol, sulfuric acid, hydrofluoric acid, and metal compounds. Chemicals that cause acid burns; 1. Acetic acid, glacial(poisonous corrosive) 2. Acetone (flammable) 3. Ammoniumhydroxide (poisonous corrosive) 4. Benedict’s (corrosive) 5. Biuret (poisonous corrosive)

Classification of fire Fire is the most common serious hazard that one faces in a typical laboratory while proper procedure and training can minimize the chances of an accidental fire, laboratory workers should stilled be prepared to deal with a fire emergency should it occur. Most fires that occur will fall into one of the following categories: Class A, Class B, Class C Class D, and Class K. Treatment of burns Most fires that occur The type of treatment a burn victim receives depends on the degree of the burn and how he/she got it. If you judge the burn to be serious, then, it is either imperative that you either take the victim to the hospital, or call an emergency line.

THE END THANK YOU
- Slides: 18