GLP1 receptor agonists Esteban Jdar Gimeno Dept of

GLP-1 receptor agonists Esteban Jódar Gimeno Dept. of Endocrinology & Clinical Nutrition. University Hospitals Quirón Salud Madrid. Professor of Endocrinology. Health Sciences School. Universidad Europea de Madrid, Spain

Contents • • Pharmacokinetics Size Structure Clinical ‘head-to-head’ comparisons

Anti-diabetic treatment options for T 2 DM Human insulin Thiazolidinediones Metformin 1950 s Sulphonylureas 1960 s 1970 s 1980 s 1990 s DPP-4 inhibitors 2000 s 2010 s GLP-1 receptor SGLT-2 agonists inhibitors DPP-4, dipeptidyl peptidase-4; GLP-1, glucagon-like peptide-1; SGLT 2, sodium glucose co-transporter-2; T 2 DM, type 2 diabetes mellitus
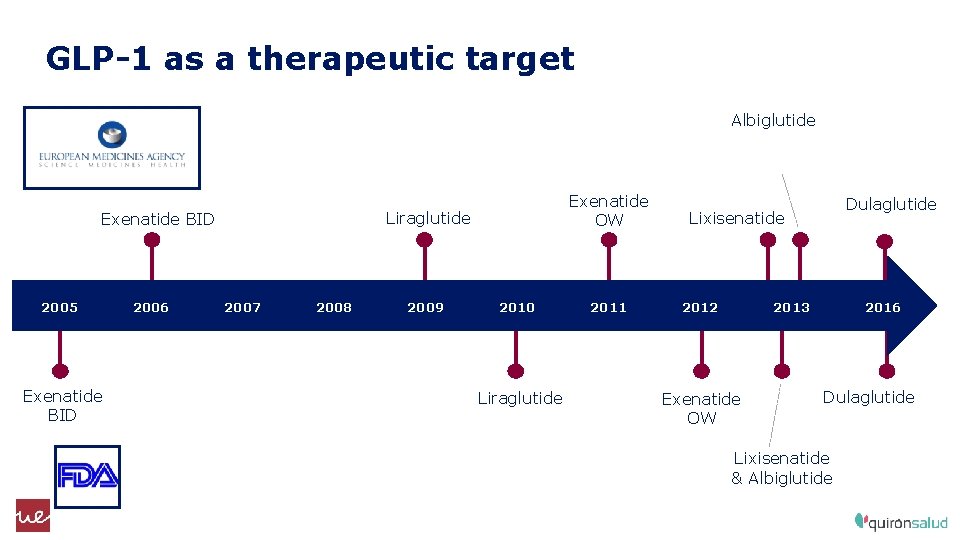
GLP-1 as a therapeutic target Albiglutide Liraglutide Exenatide BID 2005 Exenatide BID 2006 Exenatide OW 2007 2008 2009 2010 Liraglutide 2011 Dulaglutide Lixisenatide 2012 2013 Exenatide OW 2016 Dulaglutide Lixisenatide & Albiglutide
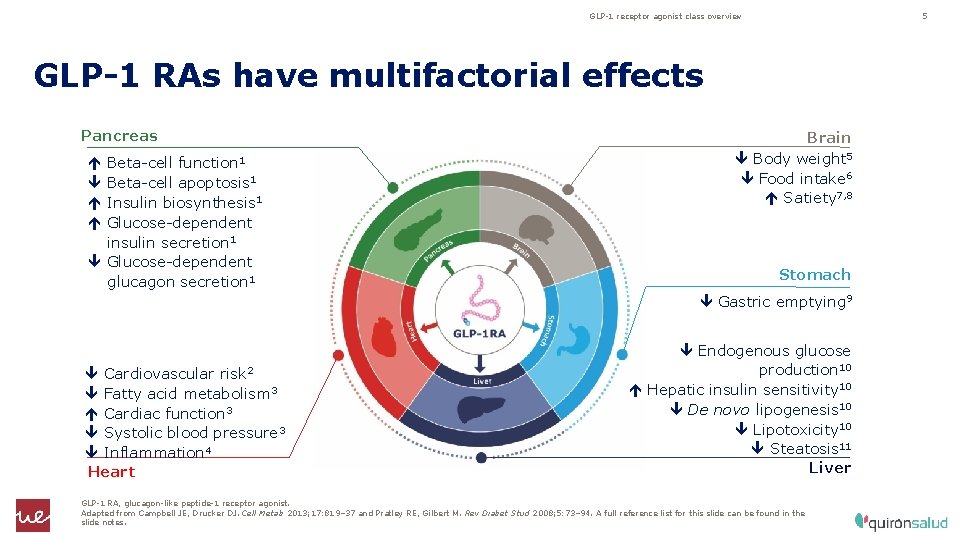
GLP-1 receptor agonist class overview 5 GLP-1 RAs have multifactorial effects Pancreas Beta-cell function 1 Beta-cell apoptosis 1 Insulin biosynthesis 1 Glucose-dependent insulin secretion 1 Glucose-dependent glucagon secretion 1 Brain Body weight 5 Food intake 6 Satiety 7, 8 Stomach Gastric emptying 9 Cardiovascular risk 2 Fatty acid metabolism 3 Cardiac function 3 Systolic blood pressure 3 Inflammation 4 Heart Endogenous glucose production 10 Hepatic insulin sensitivity 10 De novo lipogenesis 10 Lipotoxicity 10 Steatosis 11 Liver GLP-1 RA, glucagon-like peptide-1 receptor agonist. Adapted from Campbell JE, Drucker DJ. Cell Metab 2013; 17: 819– 37 and Pratley RE, Gilbert M. Rev Diabet Stud 2008; 5: 73– 94. A full reference list for this slide can be found in the slide notes.

GLP-1 RAs • • • Exenatide (BID and QW) Lixisenatide Liraglutide Albiglutide Dulaglutide Semaglutide* *Semaglutide is under development and not approved BID, twice daily; GLP-1 RA, glucagon-like peptide-1 receptor agonist; QW, once weekly

GLPRAs therapies: Are all the same?
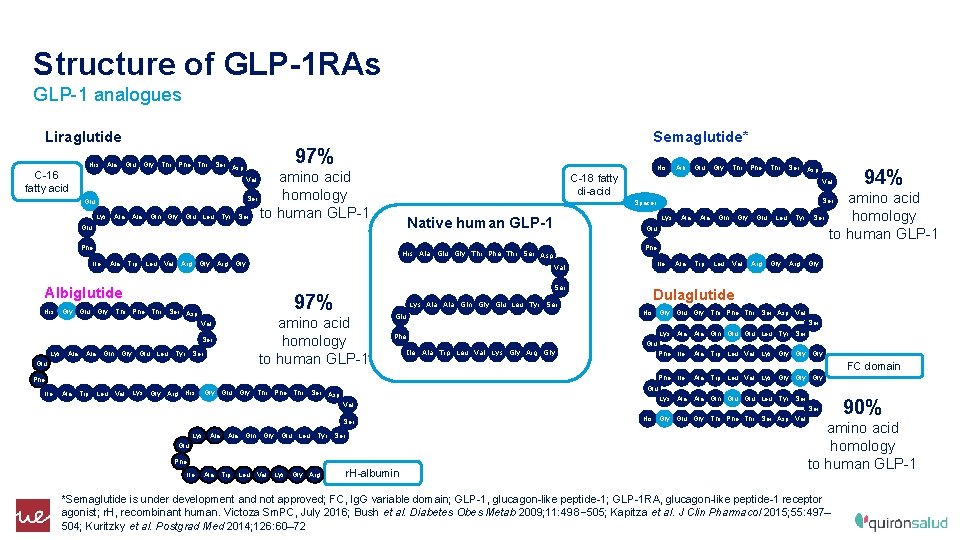
Structure of GLP-1 RAs GLP-1 analogues Liraglutide His Ala Glu Gly Thr Phe Thr 97% Ser Asp C-16 fatty acid Semaglutide* His amino acid homology Ser to human GLP-1 Ser Glu Lys Ala Gln Gly Glu Leu Tyr Trp Leu Arg Gly His Gly Glu Gly Thr Phe Thr Ser Asp Ser Ala Gln Gly Ser Glu Leu Tyr Lys Ala Gln Gly Glu Leu Tyr Ser amino acid homology to human GLP-1 Val Lys Ser 97% Glu Ala Trp Leu Val Lys Gly Arg His Gly Glu Gly Thr Phe Thr Ala Gln Gly Trp Leu Val Lys Gly Arg Asp Ile 94% amino acid homology to human GLP-1 Gly Glu Leu Tyr Ser Ile Ala Trp Leu Val Arg Gly Dulaglutide Ser Glu Ala Trp Leu Val Lys Gly Arg Gly Phe Ile Ala Trp Leu Val Lys Gly Gly Gly FC domain Glu Ser Phe Ala Gln Lys Ala Gln Glu Leu Tyr Ser Glu Ile Ala Phe Ser Asp Glu Leu Tyr Ala His Gly Glu Gly Thr Phe Thr Ser Asp Val Ser Ala Ser Glu Val Lys Thr Ser Phe Ile Phe Val Albiglutide Thr Glu His Ala Glu Gly Thr Phe Thr Ser Asp Arg Gly Val Lys Native human GLP-1 Phe Ala Glu Spacer Glu Ile Aib C-18 fatty di-acid Val r. H-albumin Lys Ala Gln Glu Leu Tyr Ser His Gly Glu Gly Thr Phe Thr Ser Asp Val 90% amino acid homology to human GLP-1 *Semaglutide is under development and not approved; FC, Ig. G variable domain; GLP-1, glucagon-like peptide-1; GLP-1 RA, glucagon-like peptide-1 receptor agonist; r. H, recombinant human. Victoza Sm. PC, July 2016; Bush et al. Diabetes Obes Metab 2009; 11: 498– 505; Kapitza et al. J Clin Pharmacol 2015; 55: 497– 504; Kuritzky et al. Postgrad Med 2014; 126: 60– 72
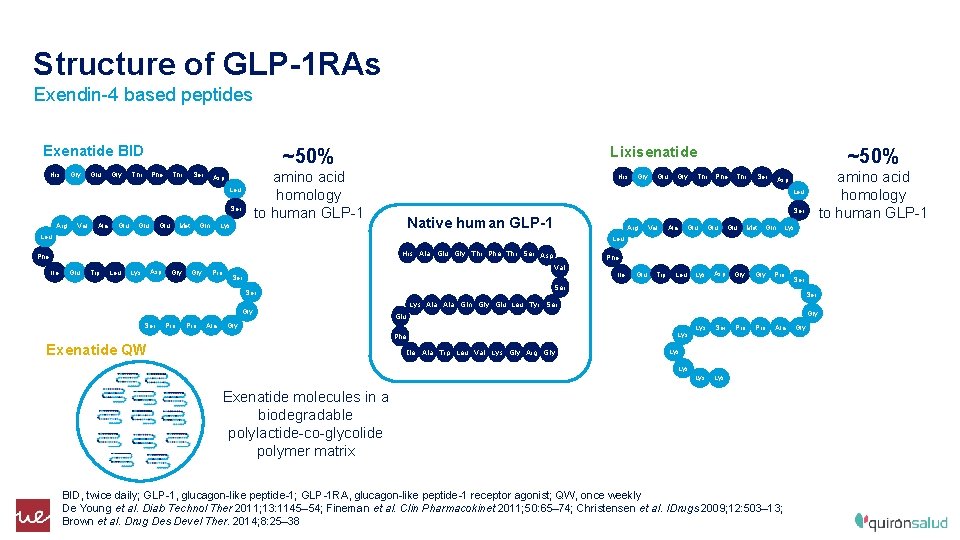
Structure of GLP-1 RAs Exendin-4 based peptides Exenatide BID His Gly Glu Gly Thr Phe Thr Ser amino acid homology to human GLP-1 Asp Leu Ser Arg Val Ala Glu Lixisenatide ~50% Glu Met His Gly Thr Phe Thr Ser amino acid homology to human GLP-1 Asp Ser Native human GLP-1 Arg Leu Val Ala Glu Glu Met Gln Lys Leu His Ala Glu Gly Thr Phe Thr Ser Asp Phe Ile Glu Leu Lys Gln Gly ~50% Glu Trp Leu Asp Lys Gly Pro Ser Gly Pro Ala Ile Glu Trp Leu Asp Gly Pro Ser Lys Ala Gln Gly Glu Leu Tyr Ser Gly Glu Gly Lys Phe Exenatide QW Lys Ser Ser Phe Val Ile Ala Trp Leu Val Lys Gly Arg Gly Lys Ser Lys Pro Ala Lys Exenatide molecules in a biodegradable polylactide-co-glycolide polymer matrix BID, twice daily; GLP-1, glucagon-like peptide-1; GLP-1 RA, glucagon-like peptide-1 receptor agonist; QW, once weekly De Young et al. Diab Technol Ther 2011; 13: 1145– 54; Fineman et al. Clin Pharmacokinet 2011; 50: 65– 74; Christensen et al. IDrugs 2009; 12: 503– 13; Brown et al. Drug Des Devel Ther. 2014; 8: 25– 38 Gly

Pharmacokinetics of GLP-1 RAs GLP-1 RA, glucagon-like peptide-1 receptor agonist
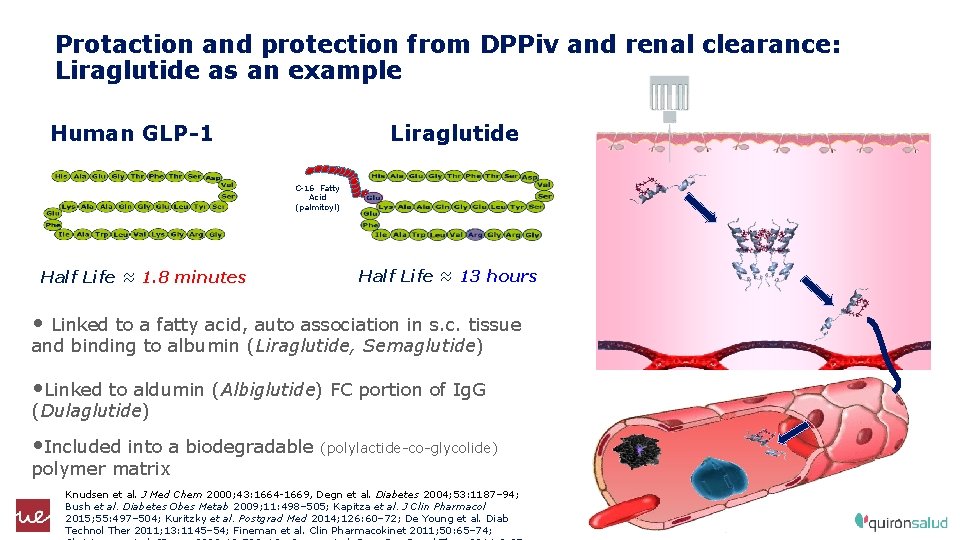
15: 18 Protaction and protection from DPPiv and renal clearance: Liraglutide as an example Human GLP-1 Liraglutide C-16 Fatty Acid (palmitoyl) Half Life ≈ 1. 8 minutes Half Life ≈ 13 hours • Linked to a fatty acid, auto association in s. c. tissue and binding to albumin (Liraglutide, Semaglutide) • Linked to aldumin (Albiglutide) FC portion of Ig. G (Dulaglutide) • Included into a biodegradable (polylactide-co-glycolide) polymer matrix Knudsen et al. J Med Chem 2000; 43: 1664 -1669, Degn et al. Diabetes 2004; 53: 1187– 94; Bush et al. Diabetes Obes Metab 2009; 11: 498– 505; Kapitza et al. J Clin Pharmacol 2015; 55: 497– 504; Kuritzky et al. Postgrad Med 2014; 126: 60– 72; De Young et al. Diab Technol Ther 2011; 13: 1145– 54; Fineman et al. Clin Pharmacokinet 2011; 50: 65– 74; 11
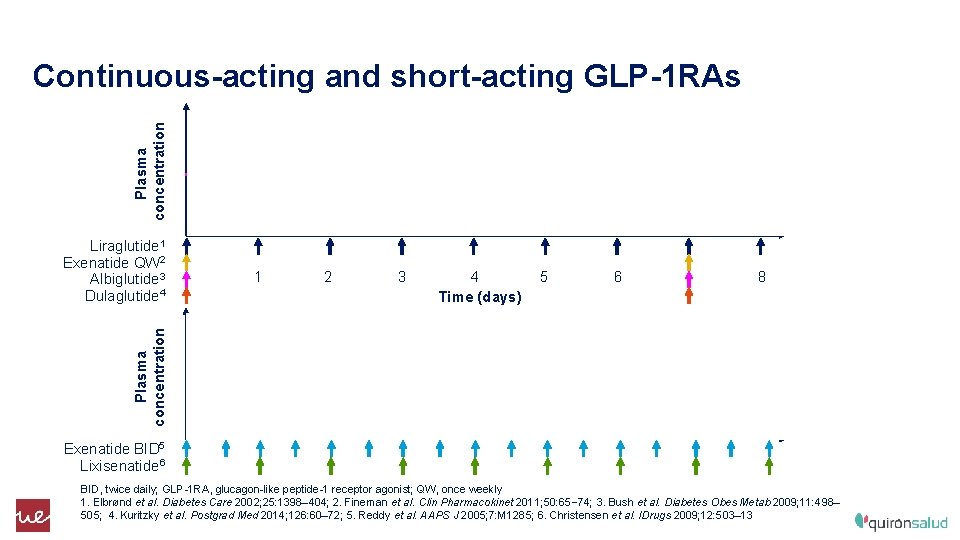
Plasma concentration Continuous-acting and short-acting GLP-1 RAs 1 2 3 4 5 Time (days) 6 8 Plasma concentration Liraglutide 1 Exenatide QW 2 Albiglutide 3 Dulaglutide 4 Exenatide BID 5 Lixisenatide 6 BID, twice daily; GLP-1 RA, glucagon-like peptide-1 receptor agonist; QW, once weekly 1. Elbrønd et al. Diabetes Care 2002; 25: 1398– 404; 2. Fineman et al. Clin Pharmacokinet 2011; 50: 65– 74; 3. Bush et al. Diabetes Obes Metab 2009; 11: 498– 505; 4. Kuritzky et al. Postgrad Med 2014; 126: 60– 72; 5. Reddy et al. AAPS J 2005; 7: M 1285; 6. Christensen et al. IDrugs 2009; 12: 503– 13

Is the pharmacokinetic profile important for efficacy?
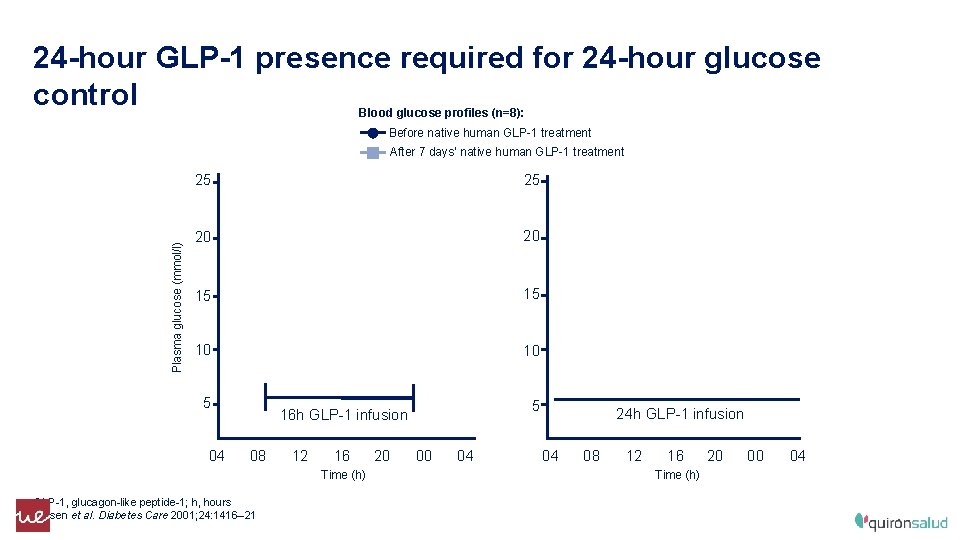
24 -hour GLP-1 presence required for 24 -hour glucose control Blood glucose profiles (n=8): Before native human GLP-1 treatment Plasma glucose (mmol/l) After 7 days’ native human GLP-1 treatment 25 25 20 20 15 15 10 10 5 04 5 16 h GLP-1 infusion 08 12 16 Time (h) GLP-1, glucagon-like peptide-1; h, hours Larsen et al. Diabetes Care 2001; 24: 1416– 21 20 00 04 24 h GLP-1 infusion 04 08 12 16 Time (h) 20 00 04
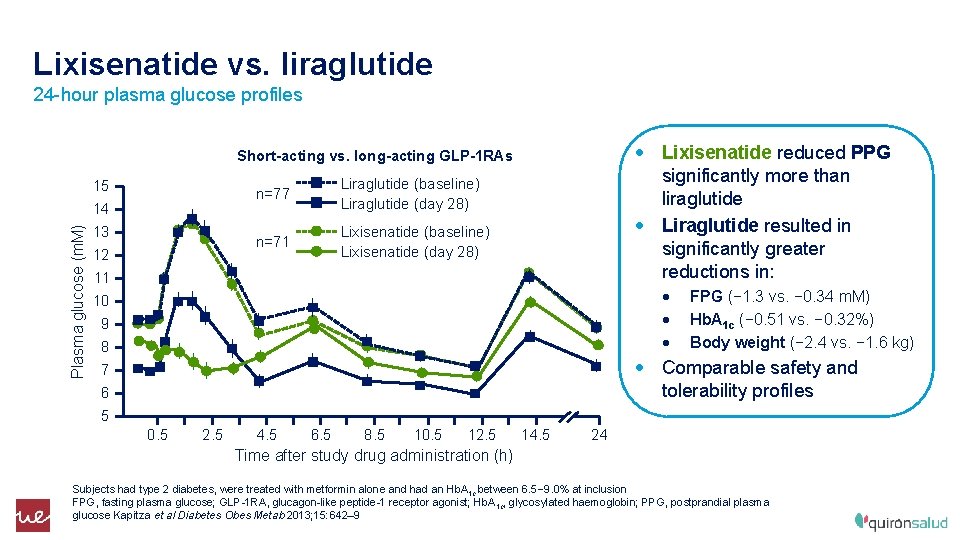
Lixisenatide vs. liraglutide 24 -hour plasma glucose profiles • Lixisenatide reduced PPG significantly more than liraglutide • Liraglutide resulted in significantly greater reductions in: Short-acting vs. long-acting GLP-1 RAs 15 Plasma glucose (m. M) 14 13 12 n=77 Liraglutide (baseline) Liraglutide (day 28) n=71 Lixisenatide (baseline) Lixisenatide (day 28) 11 • • • 10 9 8 FPG (− 1. 3 vs. − 0. 34 m. M) Hb. A 1 c (− 0. 51 vs. − 0. 32%) Body weight (− 2. 4 vs. − 1. 6 kg) • Comparable safety and tolerability profiles 7 6 5 0. 5 2. 5 4. 5 6. 5 8. 5 10. 5 12. 5 14. 5 24 Time after study drug administration (h) Subjects had type 2 diabetes, were treated with metformin alone and had an Hb. A 1 c between 6. 5– 9. 0% at inclusion FPG, fasting plasma glucose; GLP-1 RA, glucagon-like peptide-1 receptor agonist; Hb. A 1 c, glycosylated haemoglobin; PPG, postprandial plasma glucose Kapitza et al Diabetes Obes Metab 2013; 15: 642– 9

Does size matter?

The differing molecular masses of the GLP-1 RA class Molecular mass (k. Da) 0 10 Liraglutide 1 20 C-16 30 His Ala Glu Gly Thr Phe Thr Ser Asp Val Ser Glu Lys Ala Gln Gly Glu Leu Tyr Ser Glu (3. 8 k. Da) Phe Ile Ala Trp Leu Val Arg Gly 40 50 His Gly Glu Gly Thr Phe Thr Ser Asp Val Ser Lys Ala Gln Glu Leu Tyr Ser Glu Phe Ile Ala Trp Leu Val Lys Gly Gly Gly Glu Lys Ala Gln Glu Leu Tyr Ser His Gly Glu Gly Thr Phe Thr Ser Asp Val 60 70 Dulaglutide 2 (59. 7 k. Da) His. Gly. Glu. Gly Thr. Phe. Thr. Ser. Asp Leu Exenatide 2 (4. 2 k. Da) Ser Arg Val Ala Glu. Met. Gln. Lys Leu Phe Ile Glu. Trp. Leu. Lys. Asp. Gly. Pro. Ser Gly Ser. Pro Ala Gly His Gly Glu. Gly Thr. Phe. Thr. Ser. Asp Val Ser Lys Ala Gln Gly Glu. Leu. Tyr Ser Glu His. Gly. Glu. Gly Thr. Phe. Thr. Ser. Asp Leu Phe Ile Ala. Trp. Leu. Val Lys Gly. Arg. His Gly Glu. Gly Thr. Phe. Thr. Ser. Asp Val Ser Arg Val Ala Glu. Met. Gln. Lys Leu Lixisenatide 2 Ser Phe Ile Glu. Trp. Leu. Lys. Asp. Gly. Pro. Ser (4. 9 k. Da) Ser Gly Lys Pro Ala Gly Lys Ala Gln Gly Glu. Leu. Tyr Ser Glu Phe Ile Ala. Trp. Leu. Val Lys Gly. Arg Lys Lys Semaglutide 2* (4. 1 k. Da) 80 C-18 fatty di-acid His Aib Glu. Gly Thr. Phe. Thr. Ser. Asp Val Spacer Ser Lys Ala Gln Gly Glu. Leu Tyr Ser Glu Phe Ile Ala Trp. Leu Val Arg Gly *Semaglutide is under development and not approved C-16, 16 -carbon fatty acid; GLP-1 RA, glucagon-like peptide-1 receptor agonist; r. H, recombinant human 1. Malm-Erjefält et al. Drug Metab Dispos 2010; 38: 1944– 53; 2. Dhruv et al. JCD 2016; 2: 18– 25 r. H-albumin Albiglutide 2 (73. 0 k. Da) 90 100
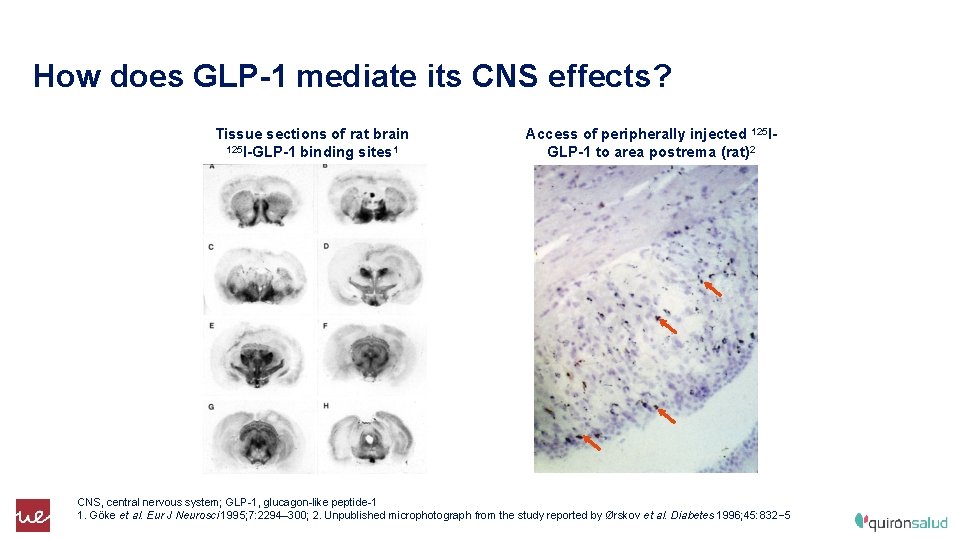
How does GLP-1 mediate its CNS effects? Tissue sections of rat brain 125 I-GLP-1 binding sites 1 Access of peripherally injected 125 IGLP-1 to area postrema (rat)2 CNS, central nervous system; GLP-1, glucagon-like peptide-1 1. Göke et al. Eur J Neurosci 1995; 7: 2294– 300; 2. Unpublished microphotograph from the study reported by Ørskov et al. Diabetes 1996; 45: 832– 5
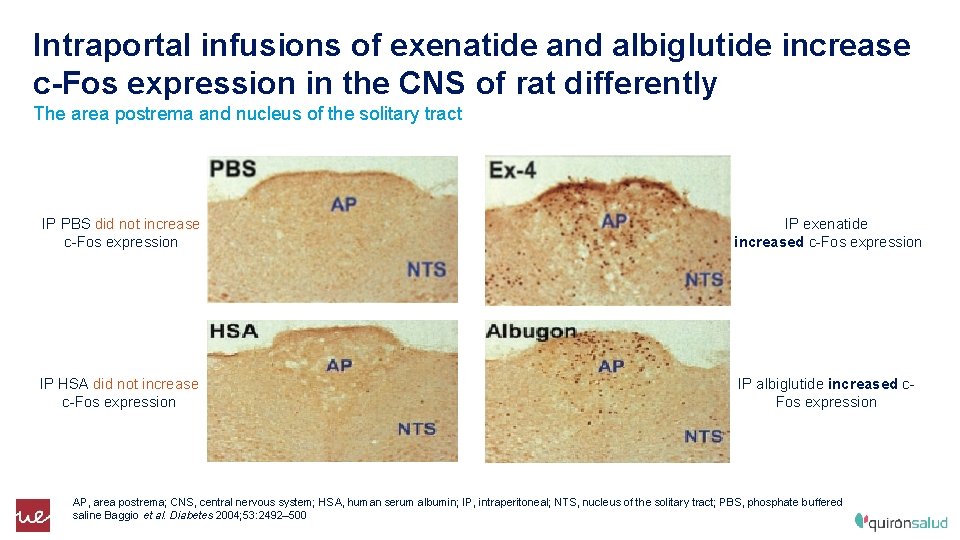
Intraportal infusions of exenatide and albiglutide increase c-Fos expression in the CNS of rat differently The area postrema and nucleus of the solitary tract IP PBS did not increase c-Fos expression IP exenatide increased c-Fos expression IP HSA did not increase c-Fos expression IP albiglutide increased c. Fos expression AP, area postrema; CNS, central nervous system; HSA, human serum albumin; IP, intraperitoneal; NTS, nucleus of the solitary tract; PBS, phosphate buffered saline Baggio et al. Diabetes 2004; 53: 2492– 500

Is structure (exendin-4 vs. human analogue) important?

Immunogenicity of the available GLP-1 RAs EMA specifications GLP-1 analogue 80 Exendin-4 based peptide 70 57 44 EMA Sm. PC VICTOZA 1 • Antibody formation has not been associated with change in Hb. A 1 c • • EMA Sm. PC TRULICITY 2 EMA Sm. PC EPERZAN 3 • Antibody formation has not been associated with change in Hb. A 1 c • Patients who develop antibodies tend to have more injection-site reactions • 40 EMA Sm. PC LYXUMIA 4 • Antibody status is not predictive of Hb. A 1 c reduction • Patients who develop antibodies tend to have more injection-site reactions 20 Exenatide QW Exenatide BID 4 Lixisenatide 1. 6 Albiglutide • Dulaglutide 9 0 • • Antibody formation has not been associated with change in Hb. A 1 c 60 Liraglutide Proportion of patients with antibodies (%) 100 EMA Sm. PC BYETTA 5 • In most patients antibody titres diminish over time and remain low • Patients who develop antibodies tend to have more injection-site reactions • No cross-reactivity with GLP-1 or glucagon • EMA Sm. PC BYDUREON 6 • Hb. A 1 c was comparable to that observed in patients without antibody titres • Patients who develop antibodies tend to have more injection-site reactions • No cross-reactivity with GLP-1 or glucagon BID, twice daily; EMA, European Medicines Agency; GLP-1, glucagon-like peptide-1; GLP-1 RA, glucagon-like peptide-1 receptor agonist; Hb. A 1 c, glycosylated haemoglobin; QW, once weekly; Sm. PC, summary of product characteristics 1. Victoza Sm. PC, July 2016; 2. Trulicity Sm. PC, March 2016; 3. Eperzan Sm. PC, April 2016; 4. Lyxumia Sm. PC, May 2016; 5. Byetta Sm. PC, January 2016; 6. Bydureon Sm. PC, March 2016

Clinical GLP-1 RA head-to-head comparisons Pharmacodynamics, efficacy and/or safety DURATION-1 DURATION-6 HARMONY-7 Exenatide QW vs. exenatide BID 1 Exenatide QW vs. liraglutide 2 Albiglutide vs. liraglutide 3 LEAD-6 Nauck et al. AWARD-1 Liraglutide vs. exenatide BID 4 Semaglutide* vs. liraglutide 5 Dulaglutide vs. exenatide BID 6 Rosenstock et al. Get. Goal-X AWARD-6 Albiglutide vs. exenatide BID 7 Lixisenatide vs. exenatide BID 8 Dulaglutide vs. liraglutide 9 DURATION-5 Kapitza et al. Lira–Lixi Exenatide QW vs. exenatide BID 10 Lixisenatide vs. liraglutide 11 Liraglutide vs. lixisenatide 12 SUSTAIN-3 Semaglutide* vs. exenatide QW 13 *Semaglutide is under development and not approved; BID, twice daily; GLP-1 RA, glucagon-like peptide-1 receptor agonist; QW, once weekly 1. Drucker et al. Lancet 2008; 372: 1240– 50; 2. Buse et al. Lancet 2013; 381: 117– 24; 3. Pratley et al. Lancet Diabetes Endocrinol 2014; 2: 289– 97; 4. Buse et al. Lancet 2009; 374: 39– 47; 5. Nauck et al. Diabetes Care 2016; 39: 231– 41; 6. Wysham et al. Diabetes Care 2014; 37: 2159– 67; 7. Rosenstock et al. Diabetes Care 2009; 32: 1880– 6; 8. Rosenstock et al. Diabetes Care 2013; 36: 2945– 51; 9. Dungan et al. Lancet 2014; 384: 1349– 57; 10. Blevins et al. J Clin Endocrinol Metab 2011; 96: 1301– 10; 11. Kapitza et al. Diabetes Obes Metab 2013; 15: 642– 9; 12. Nauck et al. Diabetes Care 2016; 39: 1501– 9; 13. https: //clinicaltrials. gov/ct 2/show/NCT 01885208
Clinical ‘head-to-head’ comparisons of different GLP-1 RAs (efficacy and safety) Short-acting / Long-acting Exendin-4 -based / GLP-1 -based GLP-1 RA X vs. GLP-1 RA Y Small / Large GLP-1, glucagon-like peptide-1; GLP-1 RA, glucagon-like peptide-1 receptor agonist
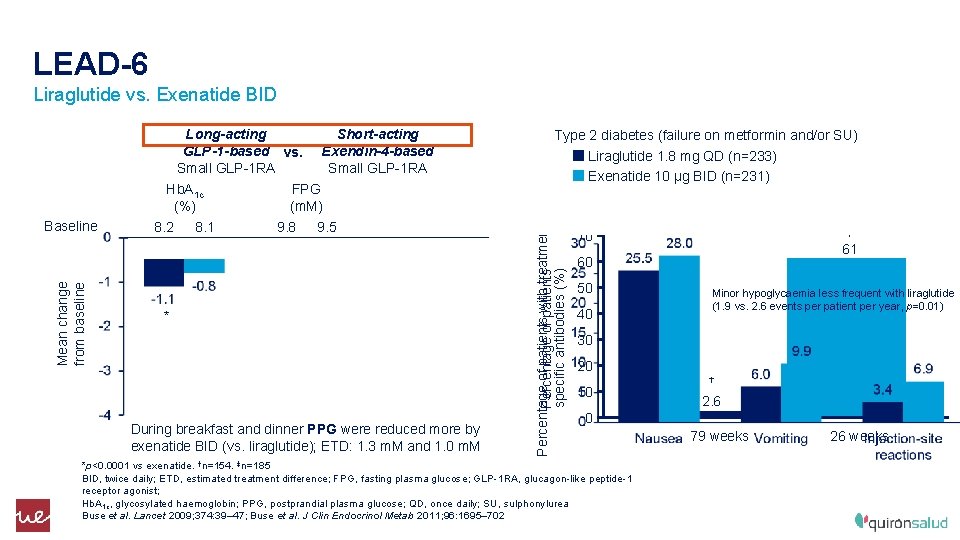
LEAD-6 Liraglutide vs. Exenatide BID Mean change from baseline Baseline Hb. A 1 c (%) 8. 2 8. 1 Short-acting Exendin-4 -based Small GLP-1 RA FPG (m. M) 9. 8 9. 5 Body weight (kg) 93. 0 * * During breakfast and dinner PPG were reduced more by exenatide BID (vs. liraglutide); ETD: 1. 3 m. M and 1. 0 m. M Type 2 diabetes (failure on metformin and/or SU) Liraglutide 1. 8 mg QD (n=233) Exenatide 10 μg BID (n=231) Percentage of patients with treatment. Percentage of patients specific antibodies (%) Long-acting GLP-1 -based vs. Small GLP-1 RA ‡ 70 61 60 50 40 Minor hypoglycaemia less frequent with liraglutide (1. 9 vs. 2. 6 events per patient per year, p=0. 01) 30 20 10 † 2. 6 0 *p<0. 0001 vs exenatide. †n=154. ‡n=185 BID, twice daily; ETD, estimated treatment difference; FPG, fasting plasma glucose; GLP-1 RA, glucagon-like peptide-1 receptor agonist; Hb. A 1 c, glycosylated haemoglobin; PPG, postprandial plasma glucose; QD, once daily; SU, sulphonylurea Buse et al. Lancet 2009; 374: 39– 47; Buse et al. J Clin Endocrinol Metab 2011; 96: 1695– 702 79 weeks 26 weeks
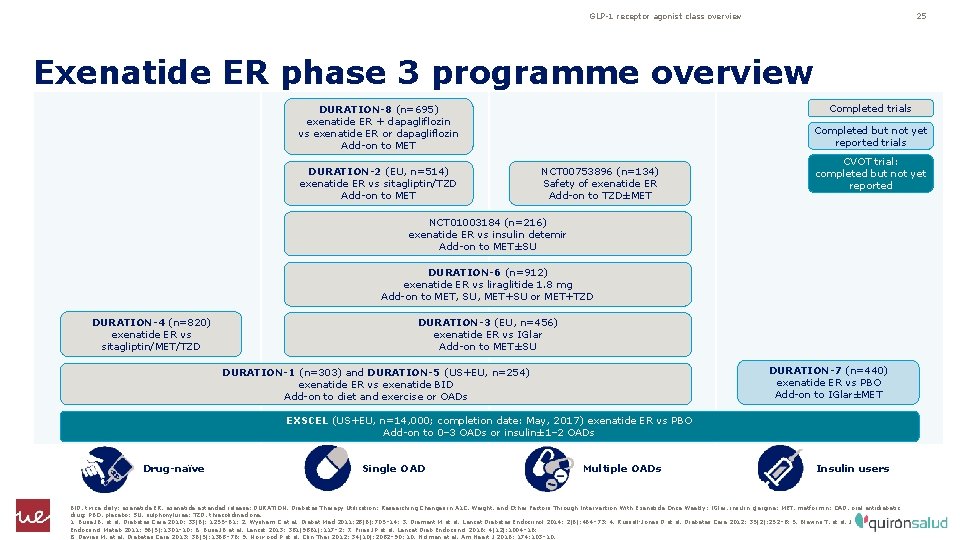
GLP-1 receptor agonist class overview 25 Exenatide ER phase 3 programme overview Completed trials DURATION-8 (n=695) exenatide ER + dapagliflozin vs exenatide ER or dapagliflozin Add-on to MET DURATION-2 (EU, n=514) exenatide ER vs sitagliptin/TZD Add-on to MET Completed but not yet reported trials NCT 00753896 (n=134) Safety of exenatide ER Add-on to TZD±MET CVOT trial: completed but not yet reported NCT 01003184 (n=216) exenatide ER vs insulin detemir Add-on to MET±SU DURATION-6 (n=912) exenatide ER vs liraglitide 1. 8 mg Add-on to MET, SU, MET+SU or MET+TZD DURATION-4 (n=820) exenatide ER vs sitagliptin/MET/TZD DURATION-3 (EU, n=456) exenatide ER vs IGlar Add-on to MET±SU DURATION-7 (n=440) exenatide ER vs PBO Add-on to IGlar±MET DURATION-1 (n=303) and DURATION-5 (US+EU, n=254) exenatide ER vs exenatide BID Add-on to diet and exercise or OADs EXSCEL (US+EU, n=14, 000; completion date: May, 2017) exenatide ER vs PBO Add-on to 0– 3 OADs or insulin± 1– 2 OADs Drug-naïve Single OAD Multiple OADs Insulin users BID, twice daily; exenatide ER, exenatide extended release; DURATION, Diabetes Therapy Utilization: Researching Changes in A 1 C, Weight, and Other Factors Through Intervention With Exenatide Once Weekly; IGlar, insulin glargine; MET, metformin; OAD, oral antidiabetic drug; PBO, placebo; SU, sulphonylurea; TZD, thiazolidinedione. 1. Buse JB, et al. Diabetes Care 2010; 33(6): 1255– 61; 2. Wysham C et al. Diabet Med 2011; 28(6): 705– 14; 3. Diamant M et al. Lancet Diabetes Endocrinol 2014; 2(6): 464– 73; 4. Russell-Jones D et al. Diabetes Care 2012; 35(2): 252– 8; 5. Blevins T, et al. J Clin Endocrinol Metab 2011; 96(5): 1301– 10; 6. Buse JB et al. Lancet 2013; 381(9861): 117– 2; 7. Frias JP et al. Lancet Diab Endocrinol 2016; 4(12): 1004– 16; 8. Davies M, et al. Diabetes Care 2013; 36(5): 1368– 76; 9. Norwood P et al. Clin Ther 2012; 34(10): 2082– 90; 10. Holman et al. Am Heart J 2016; 174: 103– 10.
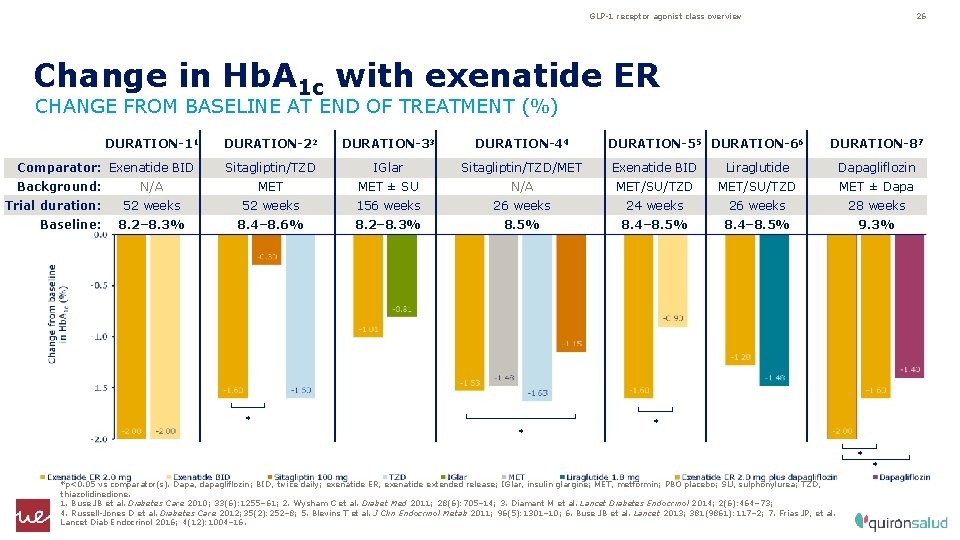
GLP-1 receptor agonist class overview 26 Change in Hb. A 1 c with exenatide ER CHANGE FROM BASELINE AT END OF TREATMENT (%) DURATION-11 DURATION-22 DURATION-33 DURATION-44 Comparator: Exenatide BID Sitagliptin/TZD IGlar Sitagliptin/TZD/MET Exenatide BID Liraglutide Dapagliflozin N/A MET ± SU N/A MET/SU/TZD MET ± Dapa 52 weeks 156 weeks 24 weeks 26 weeks 28 weeks 8. 2– 8. 3% 8. 4– 8. 6% 8. 2– 8. 3% 8. 5% 8. 4– 8. 5% 9. 3% Background: Trial duration: Baseline: * * DURATION-55 DURATION-66 DURATION-87 * *p<0. 05 vs comparator(s). Dapa, dapagliflozin; BID, twice daily; exenatide ER, exenatide extended release; IGlar, insulin glargine; MET, metformin; PBO placebo; SU, sulphonylurea; TZD, thiazolidinedione. 1. Buse JB et al. Diabetes Care 2010; 33(6): 1255– 61; 2. Wysham C et al. Diabet Med 2011; 28(6): 705– 14; 3. Diamant M et al. Lancet Diabetes Endocrinol 2014; 2(6): 464– 73; 4. Russell-Jones D et al. Diabetes Care 2012; 35(2): 252– 8; 5. Blevins T et al. J Clin Endocrinol Metab 2011; 96(5): 1301– 10; 6. Buse JB et al. Lancet 2013; 381(9861): 117– 2; 7. Frias JP, et al. Lancet Diab Endocrinol 2016; 4(12): 1004– 16.
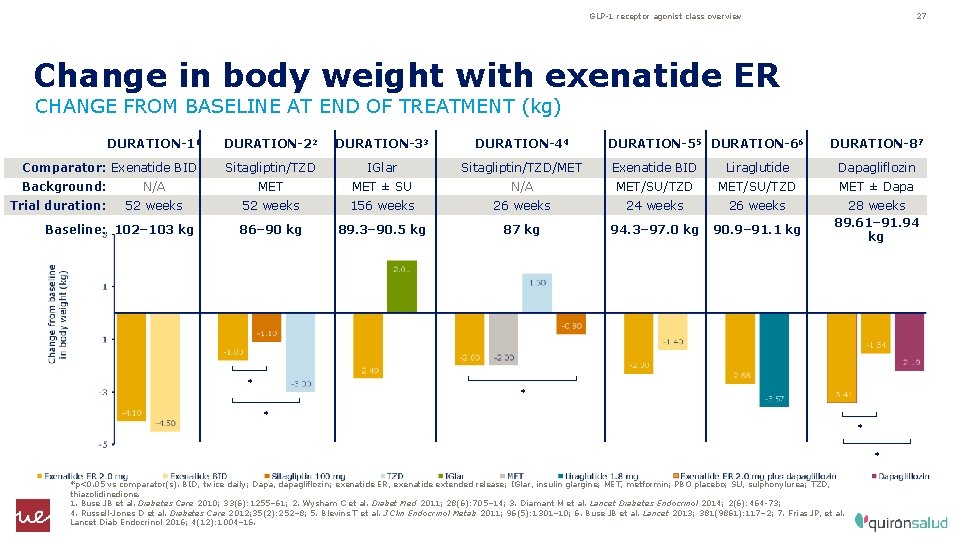
GLP-1 receptor agonist class overview 27 Change in body weight with exenatide ER CHANGE FROM BASELINE AT END OF TREATMENT (kg) DURATION-11 DURATION-22 DURATION-33 DURATION-44 Comparator: Exenatide BID Sitagliptin/TZD IGlar Sitagliptin/TZD/MET Exenatide BID Liraglutide Dapagliflozin N/A MET ± SU N/A MET/SU/TZD MET ± Dapa 52 weeks 156 weeks 24 weeks 26 weeks 86– 90 kg 89. 3– 90. 5 kg 87 kg 94. 3– 97. 0 kg 90. 9– 91. 1 kg 28 weeks 89. 61– 91. 94 kg Background: Trial duration: Baseline: 102– 103 kg * DURATION-55 DURATION-66 DURATION-87 * * *p<0. 05 vs comparator(s). BID, twice daily; Dapa, dapagliflozin; exenatide ER, exenatide extended release; IGlar, insulin glargine; MET, metformin; PBO placebo; SU, sulphonylurea; TZD, thiazolidinedione. 1. Buse JB et al. Diabetes Care 2010; 33(6): 1255– 61; 2. Wysham C et al. Diabet Med 2011; 28(6): 705– 14; 3. Diamant M et al. Lancet Diabetes Endocrinol 2014; 2(6): 464 -73; 4. Russell-Jones D et al. Diabetes Care 2012; 35(2): 252– 8; 5. Blevins T et al. J Clin Endocrinol Metab 2011; 96(5): 1301– 10; 6. Buse JB et al. Lancet 2013; 381(9861): 117– 2; 7. Frias JP, et al. Lancet Diab Endocrinol 2016; 4(12): 1004– 16.
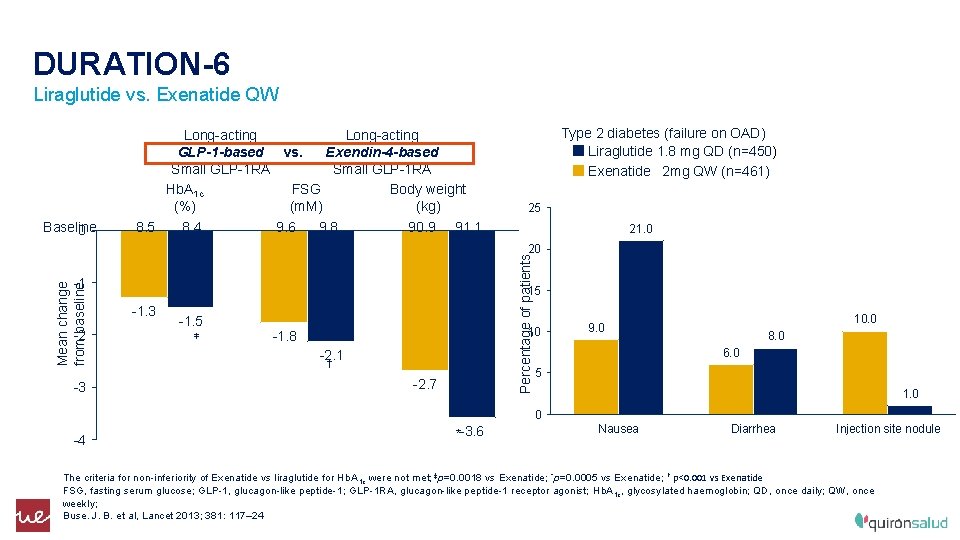
DURATION-6 Liraglutide vs. Exenatide QW Long-acting GLP-1 -based vs. Exendin-4 -based Small GLP-1 RA Hb. A 1 c FSG Body weight (%) (m. M) (kg) Type 2 diabetes (failure on OAD) Liraglutide 1. 8 mg QD (n=450) Exenatide 2 mg QW (n=461) 25 Baseline 8. 5 8. 4 9. 6 9. 8 90. 9 91. 1 0 21. 0 Percentage of patients 20 Mean change from baseline -1 -2 -3 15 -1. 3 -1. 5 ‡ 10 -1. 8 -2. 1 † -2. 7 10. 0 9. 0 8. 0 6. 0 5 1. 0 0 -4 *-3. 6 Nausea Diarrhea Injection site nodule The criteria for non-inferiority of Exenatide vs liraglutide for Hb. A 1 c were not met; ‡p=0. 0018 vs Exenatide; *p=0. 0005 vs Exenatide; † p<0. 001 vs Exenatide FSG, fasting serum glucose; GLP-1, glucagon-like peptide-1; GLP-1 RA, glucagon-like peptide-1 receptor agonist; Hb. A 1 c, glycosylated haemoglobin; QD, once daily; QW, once weekly; Buse. J. B. et al, Lancet 2013; 381: 117– 24
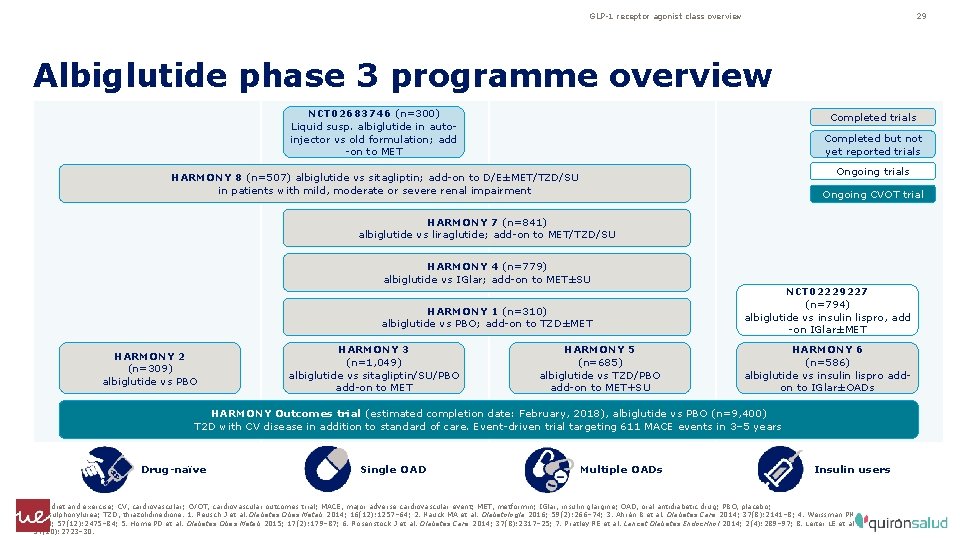
GLP-1 receptor agonist class overview 29 Albiglutide phase 3 programme overview NCT 02683746 (n=300) Liquid susp. albiglutide in autoinjector vs old formulation; add -on to MET Completed trials Completed but not yet reported trials Ongoing trials HARMONY 8 (n=507) albiglutide vs sitagliptin; add-on to D/E±MET/TZD/SU in patients with mild, moderate or severe renal impairment Ongoing CVOT trial HARMONY 7 (n=841) albiglutide vs liraglutide; add-on to MET/TZD/SU HARMONY 4 (n=779) albiglutide vs IGlar; add-on to MET±SU HARMONY 1 (n=310) albiglutide vs PBO; add-on to TZD±MET HARMONY 2 (n=309) albiglutide vs PBO HARMONY 3 (n=1, 049) albiglutide vs sitagliptin/SU/PBO add-on to MET HARMONY 5 (n=685) albiglutide vs TZD/PBO add-on to MET+SU NCT 02229227 (n=794) albiglutide vs insulin lispro, add -on IGlar±MET HARMONY 6 (n=586) albiglutide vs insulin lispro addon to IGlar±OADs HARMONY Outcomes trial (estimated completion date: February, 2018), albiglutide vs PBO (n=9, 400) T 2 D with CV disease in addition to standard of care. Event-driven trial targeting 611 MACE events in 3– 5 years Drug-naïve Single OAD Multiple OADs Insulin users D/E, diet and exercise; CV, cardiovascular; CVOT, cardiovascular outcomes trial; MACE, major adverse cardiovascular event; MET, metformin; IGlar, insulin glargine; OAD, oral antidiabetic drug; PBO, placebo; SU, sulphonylurea; TZD, thiazolidinedione. 1. Reusch J et al. Diabetes Obes Metab 2014; 16(12): 1257– 64; 2. Nauck MA et al. Diabetologia 2016; 59(2): 266– 74; 3. Ahrén B et al. Diabetes Care 2014; 37(8): 2141– 8; 4. Weissman PN et al. Diabetologia 2014; 57(12): 2475– 84; 5. Home PD et al. Diabetes Obes Metab 2015; 17(2): 179– 87; 6. Rosenstock J et al. Diabetes Care 2014; 37(8): 2317– 25; 7. Pratley RE et al. Lancet Diabetes Endocrinol 2014; 2(4): 289– 97; 8. Leiter LE et al. Diabetes Care 2014; 37(10): 2723– 30.
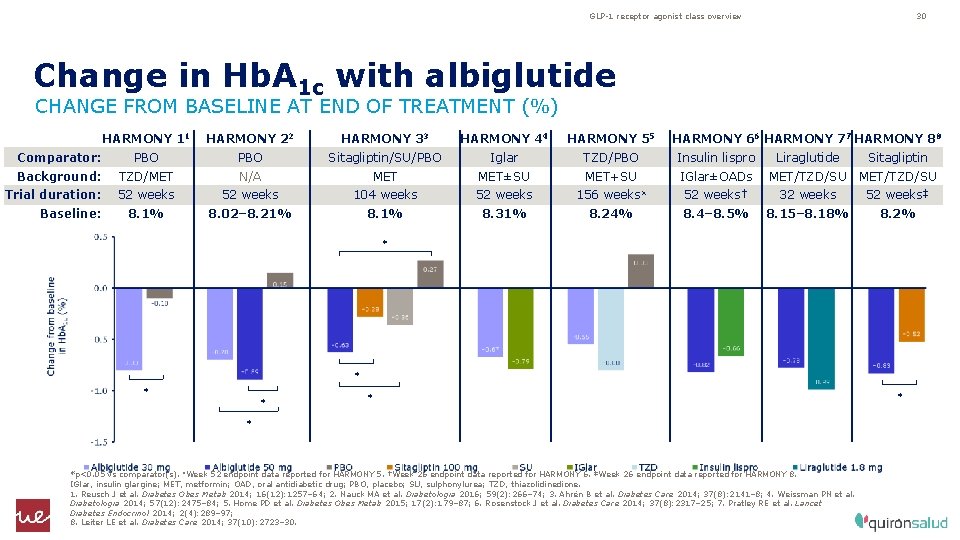
GLP-1 receptor agonist class overview 30 Change in Hb. A 1 c with albiglutide CHANGE FROM BASELINE AT END OF TREATMENT (%) HARMONY 44 HARMONY 11 HARMONY 22 HARMONY 33 Comparator: PBO Sitagliptin/SU/PBO Iglar TZD/PBO Insulin lispro Liraglutide Sitagliptin Background: TZD/MET N/A MET±SU MET+SU IGlar±OADs MET/TZD/SU 52 weeks† 32 weeks 52 weeks‡ 8. 4– 8. 5% 8. 15– 8. 18% 8. 2% Trial duration: Baseline: HARMONY 55 52 weeks 104 weeks 52 weeks 156 weeks¤ 8. 1% 8. 02– 8. 21% 8. 31% 8. 24% HARMONY 66 HARMONY 77 HARMONY 88 * * * *p<0. 05 vs comparator(s). ¤Week 52 endpoint data reported for HARMONY 5. †Week 26 endpoint data reported for HARMONY 6. ‡Week 26 endpoint data reported for HARMONY 8. IGlar, insulin glargine; MET, metformin; OAD, oral antidiabetic drug; PBO, placebo; SU, sulphonylurea; TZD, thiazolidinedione. 1. Reusch J et al. Diabetes Obes Metab 2014; 16(12): 1257– 64; 2. Nauck MA et al. Diabetologia 2016; 59(2): 266– 74; 3. Ahrén B et al. Diabetes Care 2014; 37(8): 2141– 8; 4. Weissman PN et al. Diabetologia 2014; 57(12): 2475– 84; 5. Home PD et al. Diabetes Obes Metab 2015; 17(2): 179– 87; 6. Rosenstock J et al. Diabetes Care 2014; 37(8): 2317– 25; 7. Pratley RE et al. Lancet Diabetes Endocrinol 2014; 2(4): 289– 97; 8. Leiter LE et al. Diabetes Care 2014; 37(10): 2723– 30. *
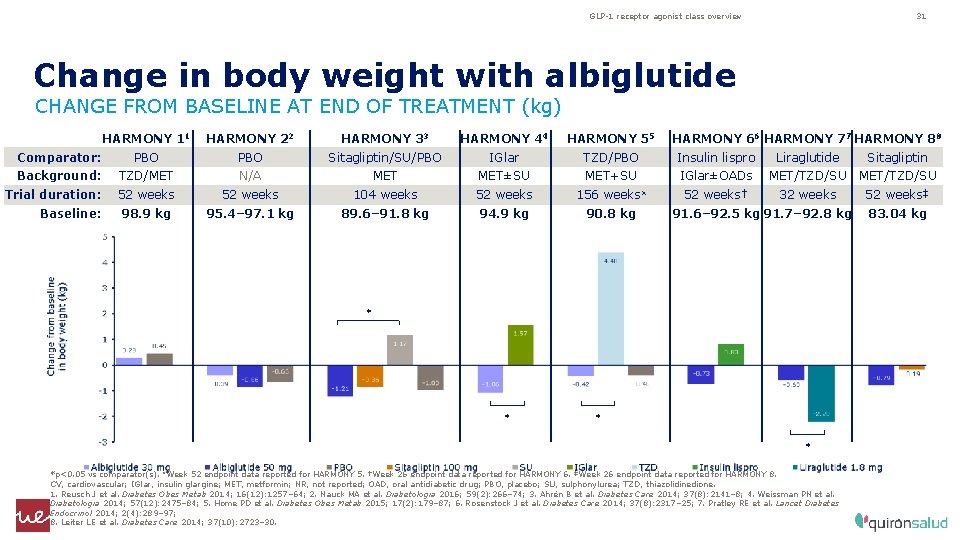
GLP-1 receptor agonist class overview 31 Change in body weight with albiglutide CHANGE FROM BASELINE AT END OF TREATMENT (kg) HARMONY 11 HARMONY 22 HARMONY 33 HARMONY 44 HARMONY 55 Comparator: PBO Sitagliptin/SU/PBO IGlar TZD/PBO Insulin lispro Liraglutide Sitagliptin Background: TZD/MET N/A MET±SU MET+SU IGlar±OADs MET/TZD/SU 52 weeks† 32 weeks 52 weeks‡ Trial duration: 52 weeks 104 weeks 52 weeks 156 weeks¤ Baseline: 98. 9 kg 95. 4– 97. 1 kg 89. 6– 91. 8 kg 94. 9 kg 90. 8 kg HARMONY 66 HARMONY 77 HARMONY 88 91. 6– 92. 5 kg 91. 7– 92. 8 kg * * *p<0. 05 vs comparator(s). ¤Week 52 endpoint data reported for HARMONY 5. †Week 26 endpoint data reported for HARMONY 6. ‡Week 26 endpoint data reported for HARMONY 8. CV, cardiovascular; IGlar, insulin glargine; MET, metformin; NR, not reported; OAD, oral antidiabetic drug; PBO, placebo; SU, sulphonylurea; TZD, thiazolidinedione. 1. Reusch J et al. Diabetes Obes Metab 2014; 16(12): 1257– 64; 2. Nauck MA et al. Diabetologia 2016; 59(2): 266– 74; 3. Ahrén B et al. Diabetes Care 2014; 37(8): 2141– 8; 4. Weissman PN et al. Diabetologia 2014; 57(12): 2475– 84; 5. Home PD et al. Diabetes Obes Metab 2015; 17(2): 179– 87; 6. Rosenstock J et al. Diabetes Care 2014; 37(8): 2317– 25; 7. Pratley RE et al. Lancet Diabetes Endocrinol 2014; 2(4): 289– 97; 8. Leiter LE et al. Diabetes Care 2014; 37(10): 2723– 30. 83. 04 kg
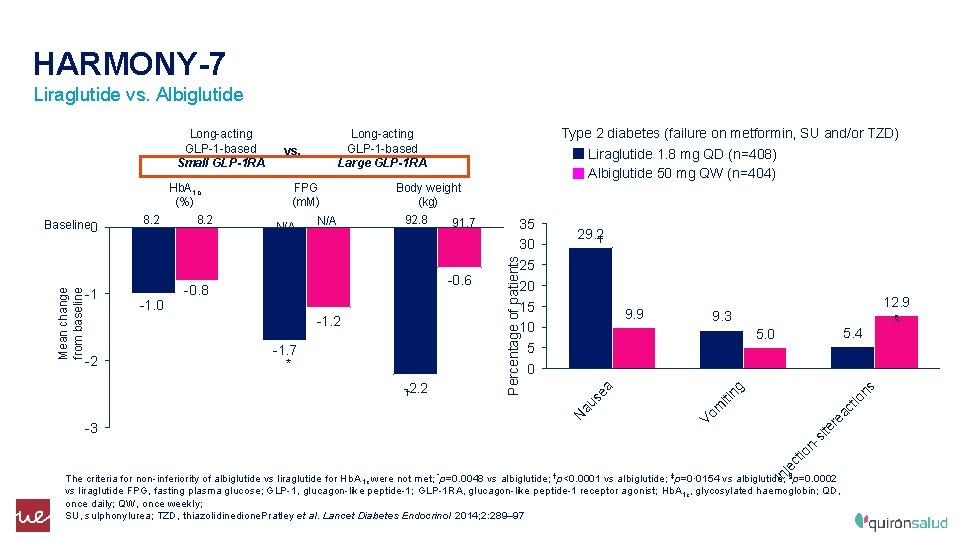
HARMONY-7 Liraglutide vs. Albiglutide 5. 4 5. 0 ns je ct io n- -3 N au -2. 2 † ‡ § tio * 9. 3 ac -1. 7 12. 9 re -2 9. 9 te -1. 2 29. 2† si -0. 6 -0. 8 35 30 25 20 15 10 5 0 ng N/A iti N/A Body weight (kg) 92. 8 91. 7 Vo m -1. 0 FPG (m. M) a -1 vs. se Mean change from baseline Baseline 0 Hb. A 1 c (%) 8. 2 Type 2 diabetes (failure on metformin, SU and/or TZD) Liraglutide 1. 8 mg QD (n=408) Albiglutide 50 mg QW (n=404) Long-acting GLP-1 -based Large GLP-1 RA Percentage of patients Long-acting GLP-1 -based Small GLP-1 RA In The criteria for non-inferiority of albiglutide vs liraglutide for Hb. A 1 c were not met; *p=0. 0048 vs albiglutide; †p<0. 0001 vs albiglutide; ‡p=0· 0154 vs albiglutide; §p=0. 0002 vs liraglutide FPG, fasting plasma glucose; GLP-1, glucagon-like peptide-1; GLP-1 RA, glucagon-like peptide-1 receptor agonist; Hb. A 1 c, glycosylated haemoglobin; QD, once daily; QW, once weekly; SU, sulphonylurea; TZD, thiazolidinedione. Pratley et al. Lancet Diabetes Endocrinol 2014; 2: 289– 97
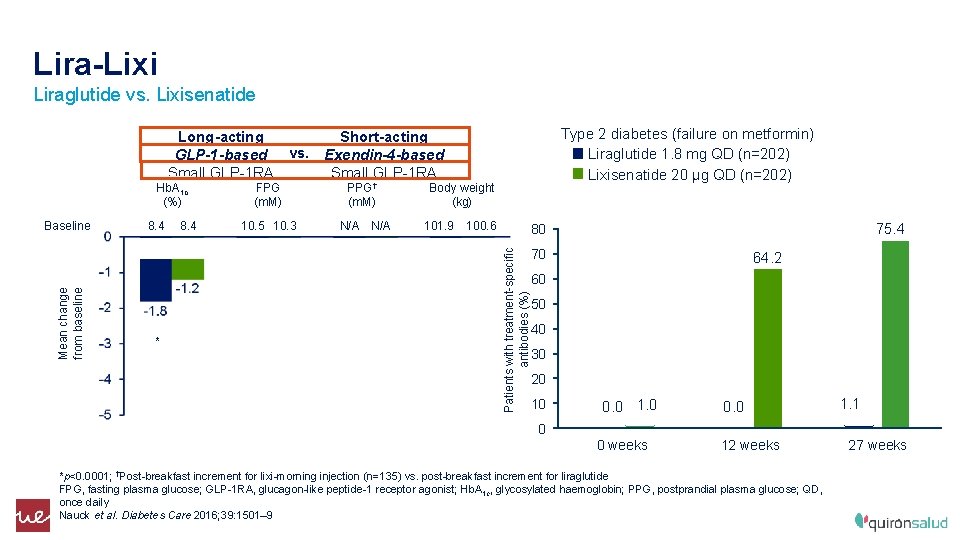
Lira-Lixi Liraglutide vs. Lixisenatide Type 2 diabetes (failure on metformin) Liraglutide 1. 8 mg QD (n=202) Lixisenatide 20 µg QD (n=202) Long-acting Short-acting GLP-1 -based vs. Exendin-4 -based Small GLP-1 RA Mean change from baseline Baseline 8. 4 FPG (m. M) PPG† (m. M) 10. 5 10. 3 N/A * * Body weight (kg) 101. 9 100. 6 75. 4 80 Patients with treatment-specific antibodies (%) Hb. A 1 c (%) 70 64. 2 60 50 40 30 20 10 0. 0 1. 1 0 0 weeks 12 weeks *p<0. 0001; †Post-breakfast increment for lixi-morning injection (n=135) vs. post-breakfast increment for liraglutide FPG, fasting plasma glucose; GLP-1 RA, glucagon-like peptide-1 receptor agonist; Hb. A 1 c, glycosylated haemoglobin; PPG, postprandial plasma glucose; QD, once daily Nauck et al. Diabetes Care 2016; 39: 1501– 9 27 weeks
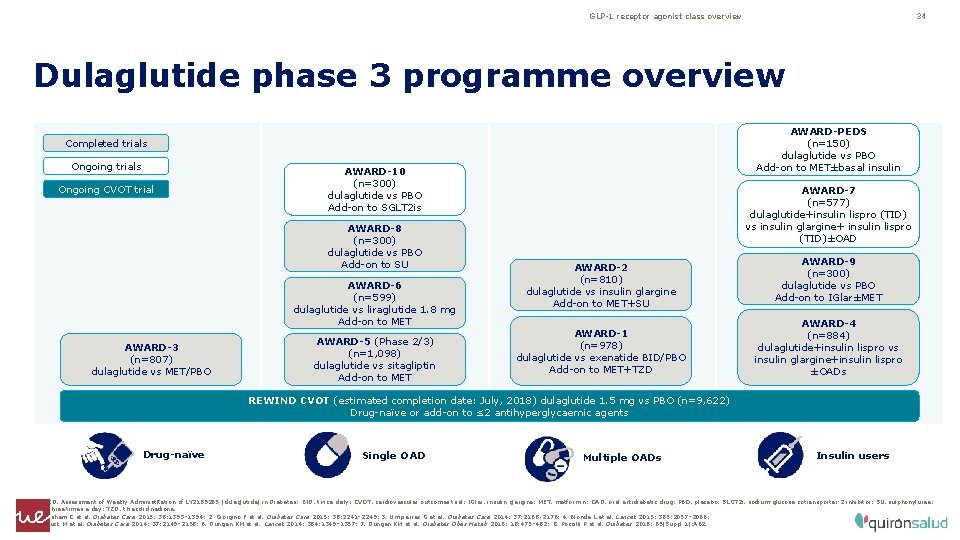
GLP-1 receptor agonist class overview 34 Dulaglutide phase 3 programme overview AWARD-PEDS (n=150) dulaglutide vs PBO Add-on to MET±basal insulin Completed trials Ongoing CVOT trial AWARD-10 (n=300) dulaglutide vs PBO Add-on to SGLT 2 is AWARD-8 (n=300) dulaglutide vs PBO Add-on to SU AWARD-6 (n=599) dulaglutide vs liraglutide 1. 8 mg Add-on to MET AWARD-3 (n=807) dulaglutide vs MET/PBO AWARD-5 (Phase 2/3) (n=1, 098) dulaglutide vs sitagliptin Add-on to MET AWARD-7 (n=577) dulaglutide+insulin lispro (TID) vs insulin glargine+ insulin lispro (TID)±OAD AWARD-2 (n=810) dulaglutide vs insulin glargine Add-on to MET+SU AWARD-1 (n=978) dulaglutide vs exenatide BID/PBO Add-on to MET+TZD AWARD-9 (n=300) dulaglutide vs PBO Add-on to IGlar±MET AWARD-4 (n=884) dulaglutide+insulin lispro vs insulin glargine+insulin lispro ±OADs REWIND CVOT (estimated completion date: July, 2018) dulaglutide 1. 5 mg vs PBO (n=9, 622) Drug-naïve or add-on to ≤ 2 antihyperglycaemic agents Drug-naïve Single OAD Multiple OADs Insulin users AWARD, Assessment of Weekly Administ. Ration of LY 2189265 (dulaglutide) in Diabetes; BID, twice daily; CVOT, cardiovascular outcomes trial; IGlar, insulin glargine; MET, metformin; OAD, oral antidiabetic drug; PBO, placebo; SLGT 2 i, sodium-glucose cotransporter-2 inhibitor; SU, sulphonylurea; TID, three times a day; TZD, thiazolidinedione. 1. Wysham C et al. Diabetes Care 2015; 38: 1393– 1394; 2. Giorgino F et al. Diabetes Care 2015; 38: 2241– 2249; 3. Umpierrez G et al. Diabetes Care 2014; 37: 2168 ‑ 2176; 4. Blonde L et al. Lancet 2015; 385: 2057– 2066; 5. Nauck M et al. Diabetes Care 2014; 37: 2149– 2158; 6. Dungan KM et al. Lancet 2014; 384: 1349– 1357; 7. Dungan KM et al. Diabetes Obes Metab 2016; 18: 475– 482; 8. Pozzilli P et al. Diabetes 2016; 65(Suppl 1): A 62.
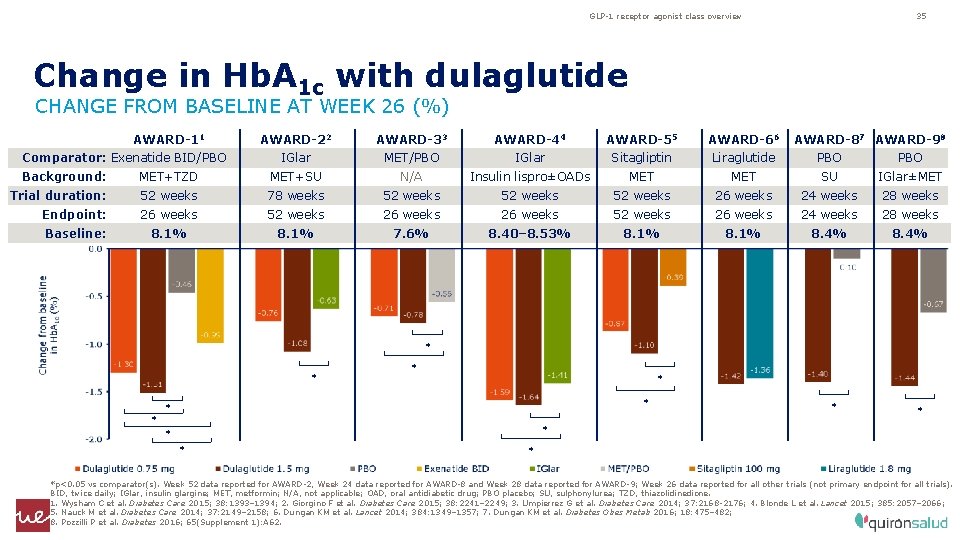
GLP-1 receptor agonist class overview 35 Change in Hb. A 1 c with dulaglutide CHANGE FROM BASELINE AT WEEK 26 (%) AWARD-11 Comparator: Exenatide BID/PBO AWARD-22 AWARD-33 AWARD-44 AWARD-55 AWARD-66 AWARD-87 AWARD-98 IGlar MET/PBO IGlar Sitagliptin Liraglutide PBO Background: MET+TZD MET+SU N/A Insulin lispro±OADs MET SU IGlar±MET Trial duration: 52 weeks 78 weeks 52 weeks 26 weeks 24 weeks 28 weeks Endpoint: 26 weeks 52 weeks 26 weeks 24 weeks 28 weeks Baseline: 8. 1% 7. 6% 8. 40– 8. 53% 8. 1% 8. 4% * * * *p<0. 05 vs comparator(s). Week 52 data reported for AWARD-2, Week 24 data reported for AWARD-8 and Week 28 data reported for AWARD-9; Week 26 data reported for all other trials (not primary endpoint for all trials). BID, twice daily; IGlar, insulin glargine; MET, metformin; N/A, not applicable; OAD, oral antidiabetic drug; PBO placebo; SU, sulphonylurea; TZD, thiazolidinedione. 1. Wysham C et al. Diabetes Care 2015; 38: 1393– 1394; 2. Giorgino F et al. Diabetes Care 2015; 38: 2241– 2249; 3. Umpierrez G et al. Diabetes Care 2014; 37: 2168‑ 2176; 4. Blonde L et al. Lancet 2015; 385: 2057– 2066; 5. Nauck M et al. Diabetes Care 2014; 37: 2149– 2158; 6. Dungan KM et al. Lancet 2014; 384: 1349– 1357; 7. Dungan KM et al. Diabetes Obes Metab 2016; 18: 475– 482; 8. Pozzilli P et al. Diabetes 2016; 65(Supplement 1): A 62.
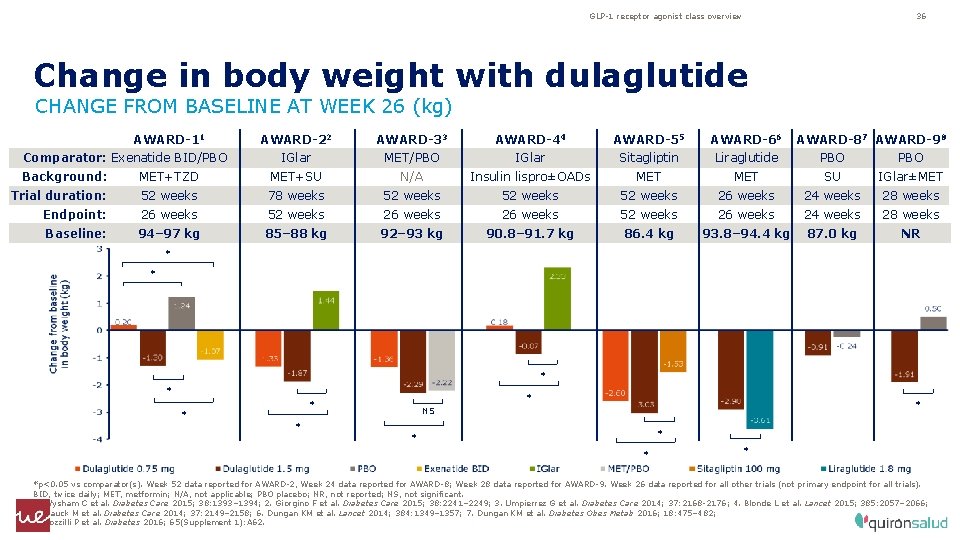
GLP-1 receptor agonist class overview 36 Change in body weight with dulaglutide CHANGE FROM BASELINE AT WEEK 26 (kg) AWARD-11 Comparator: Exenatide BID/PBO AWARD-22 AWARD-33 AWARD-44 AWARD-55 AWARD-66 AWARD-87 AWARD-98 IGlar MET/PBO IGlar Sitagliptin Liraglutide PBO Background: MET+TZD MET+SU N/A Insulin lispro±OADs MET SU IGlar±MET Trial duration: 52 weeks 78 weeks 52 weeks 26 weeks 24 weeks 28 weeks Endpoint: 26 weeks 52 weeks 26 weeks 24 weeks 28 weeks Baseline: 94– 97 kg 85– 88 kg 92– 93 kg 90. 8– 91. 7 kg 86. 4 kg 93. 8– 94. 4 kg 87. 0 kg NR * * * * * NS * * *p<0. 05 vs comparator(s). Week 52 data reported for AWARD-2, Week 24 data reported for AWARD-8; Week 28 data reported for AWARD-9. Week 26 data reported for all other trials (not primary endpoint for all trials). BID, twice daily; MET, metformin; N/A, not applicable; PBO placebo; NR, not reported; NS, not significant. 1. Wysham C et al. Diabetes Care 2015; 38: 1393– 1394; 2. Giorgino F et al. Diabetes Care 2015; 38: 2241– 2249; 3. Umpierrez G et al. Diabetes Care 2014; 37: 2168‑ 2176; 4. Blonde L et al. Lancet 2015; 385: 2057– 2066; 5. Nauck M et al. Diabetes Care 2014; 37: 2149– 2158; 6. Dungan KM et al. Lancet 2014; 384: 1349– 1357; 7. Dungan KM et al. Diabetes Obes Metab 2016; 18: 475– 482; 8. Pozzilli P et al. Diabetes 2016; 65(Supplement 1): A 62.
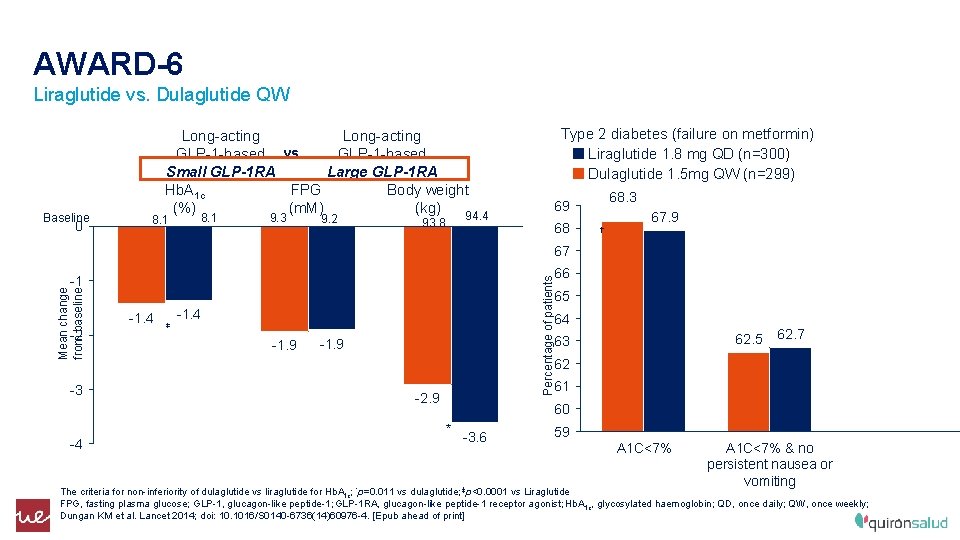
AWARD-6 Liraglutide vs. Dulaglutide QW Baseline 0 Long-acting GLP-1 -based vs. GLP-1 -based Small GLP-1 RA Large GLP-1 RA Hb. A 1 c FPG Body weight (%) (m. M) (kg) 94. 4 8. 1 9. 3 9. 2 93. 8 Type 2 diabetes (failure on metformin) Liraglutide 1. 8 mg QD (n=300) Dulaglutide 1. 5 mg QW (n=299) 68. 3 69 68 67. 9 † 67 66 -2 -3 Percentage of patients Mean change from baseline -1 65 -1. 4 ‡ -1. 4 64 -1. 9 62. 7 62 61 -2. 9 60 * -4 62. 5 63 -1. 9 -3. 6 59 A 1 C<7% & no persistent nausea or vomiting The criteria for non-inferiority of dulaglutide vs liraglutide for Hb. A 1 c; *p=0. 011 vs dulaglutide; ‡p<0. 0001 vs Liraglutide FPG, fasting plasma glucose; GLP-1, glucagon-like peptide-1; GLP-1 RA, glucagon-like peptide-1 receptor agonist; Hb. A 1 c, glycosylated haemoglobin; QD, once daily; QW, once weekly; Dungan KM et al. Lancet 2014; doi: 10. 1016/S 0140 -6736(14)60976 -4. [Epub ahead of print]
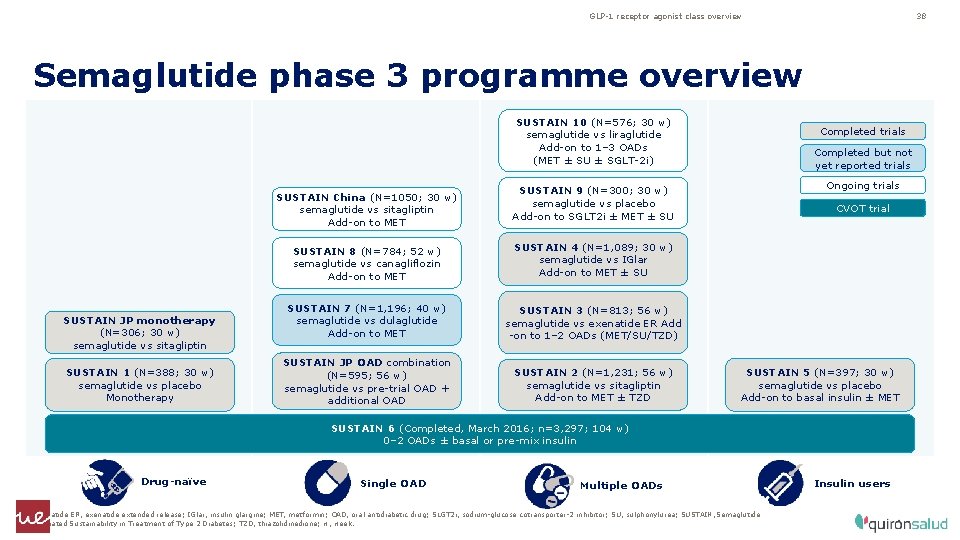
GLP-1 receptor agonist class overview 38 Semaglutide phase 3 programme overview SUSTAIN 10 (N=576; 30 w) semaglutide vs liraglutide Add-on to 1– 3 OADs (MET ± SU ± SGLT-2 i) SUSTAIN China (N=1050; 30 w) semaglutide vs sitagliptin Add-on to MET SUSTAIN JP monotherapy (N=306; 30 w) semaglutide vs sitagliptin SUSTAIN 1 (N=388; 30 w) semaglutide vs placebo Monotherapy Completed trials Completed but not yet reported trials Ongoing trials SUSTAIN 9 (N=300; 30 w) semaglutide vs placebo Add-on to SGLT 2 i ± MET ± SU SUSTAIN 8 (N=784; 52 w) semaglutide vs canagliflozin Add-on to MET SUSTAIN 4 (N=1, 089; 30 w) semaglutide vs IGlar Add-on to MET ± SU SUSTAIN 7 (N=1, 196; 40 w) semaglutide vs dulaglutide Add-on to MET SUSTAIN 3 (N=813; 56 w) semaglutide vs exenatide ER Add -on to 1– 2 OADs (MET/SU/TZD) SUSTAIN JP OAD combination (N=595; 56 w) semaglutide vs pre-trial OAD + additional OAD SUSTAIN 2 (N=1, 231; 56 w) semaglutide vs sitagliptin Add-on to MET ± TZD CVOT trial SUSTAIN 5 (N=397; 30 w) semaglutide vs placebo Add-on to basal insulin ± MET SUSTAIN 6 (Completed, March 2016; n=3, 297; 104 w) 0– 2 OADs ± basal or pre-mix insulin Drug-naïve Single OAD Multiple OADs Exenatide ER, exenatide extended release; IGlar, insulin glargine; MET, metformin; OAD, oral antidiabetic drug; SLGT 2 i, sodium-glucose cotransporter-2 inhibitor; SU, sulphonylurea; SUSTAIN, Semaglutide Unabated Sustainability in Treatment of Type 2 Diabetes; TZD, thiazolidinedione; w, week. Insulin users
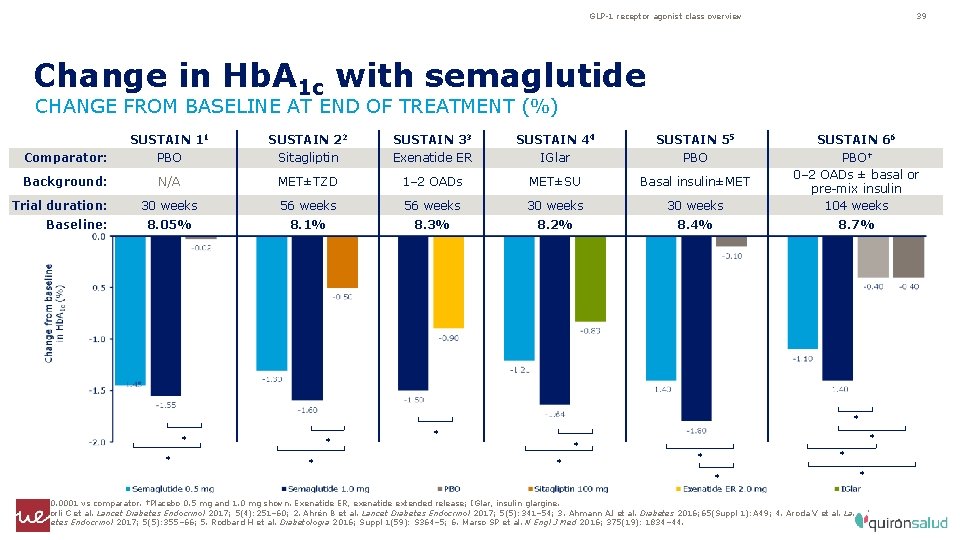
GLP-1 receptor agonist class overview 39 Change in Hb. A 1 c with semaglutide CHANGE FROM BASELINE AT END OF TREATMENT (%) SUSTAIN 11 SUSTAIN 22 SUSTAIN 33 SUSTAIN 44 SUSTAIN 55 SUSTAIN 66 Comparator: PBO Sitagliptin Exenatide ER IGlar PBO Background: N/A MET±TZD 1– 2 OADs MET±SU Basal insulin±MET 30 weeks 56 weeks 30 weeks PBO† 0– 2 OADs ± basal or pre-mix insulin 104 weeks 8. 05% 8. 1% 8. 3% 8. 2% 8. 4% 8. 7% Trial duration: Baseline: * * * *p<0. 0001 vs comparator. †Placebo 0. 5 mg and 1. 0 mg shown. Exenatide ER, exenatide extended release; IGlar, insulin glargine. 1. Sorli C et al. Lancet Diabetes Endocrinol 2017; 5(4): 251– 60; 2. Ahrén B et al. Lancet Diabetes Endocrinol 2017; 5(5): 341– 54; 3. Ahmann AJ et al. Diabetes 2016; 65(Suppl 1): A 49; 4. Aroda V et al. Lancet Diabetes Endocrinol 2017; 5(5): 355– 66; 5. Rodbard H et al. Diabetologia 2016; Suppl 1(59): S 364– 5; 6. Marso SP et al. N Engl J Med 2016; 375(19): 1834– 44.
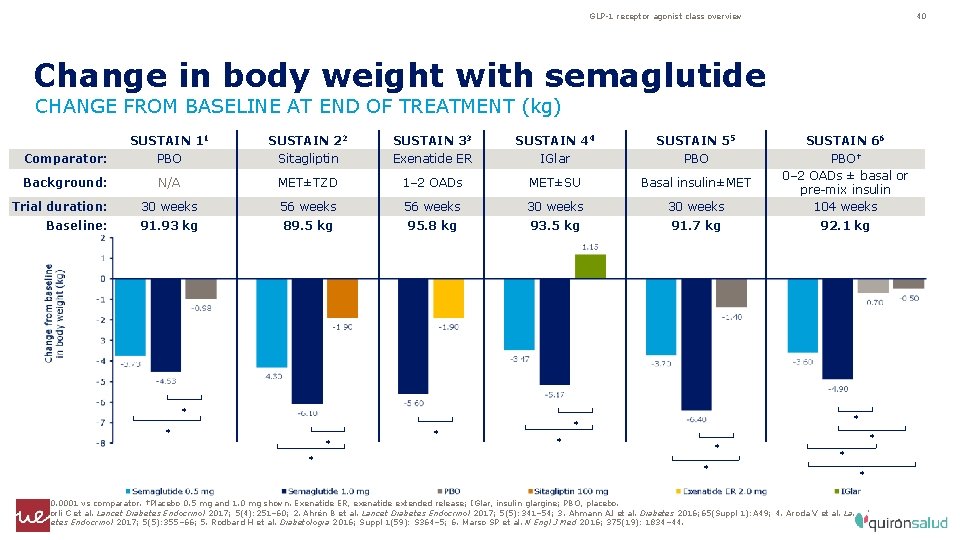
GLP-1 receptor agonist class overview 40 Change in body weight with semaglutide CHANGE FROM BASELINE AT END OF TREATMENT (kg) SUSTAIN 11 SUSTAIN 22 SUSTAIN 33 SUSTAIN 44 SUSTAIN 55 SUSTAIN 66 Comparator: PBO Sitagliptin Exenatide ER IGlar PBO Background: N/A MET±TZD 1– 2 OADs MET±SU Basal insulin±MET Trial duration: 30 weeks 56 weeks 30 weeks PBO† 0– 2 OADs ± basal or pre-mix insulin 104 weeks Baseline: 91. 93 kg 89. 5 kg 95. 8 kg 93. 5 kg 91. 7 kg 92. 1 kg * * * *p<0. 0001 vs comparator. †Placebo 0. 5 mg and 1. 0 mg shown. Exenatide ER, exenatide extended release; IGlar, insulin glargine; PBO, placebo. 1. Sorli C et al. Lancet Diabetes Endocrinol 2017; 5(4): 251– 60; 2. Ahrén B et al. Lancet Diabetes Endocrinol 2017; 5(5): 341– 54; 3. Ahmann AJ et al. Diabetes 2016; 65(Suppl 1): A 49; 4. Aroda V et al. Lancet Diabetes Endocrinol 2017; 5(5): 355– 66; 5. Rodbard H et al. Diabetologia 2016; Suppl 1(59): S 364– 5; 6. Marso SP et al. N Engl J Med 2016; 375(19): 1834– 44.

GLP-1 receptor agonist class overview Sustain 7 https: //www. novonordisk. com/bin/get. PDF. 2127298. pdf 41

Summary

GLP-1 receptor agonists are not alike… Pharmacokinetics Structure Size Short-acting Long-acting Exendin-4 based GLP-1 -based Small Large Exenatide BID Exenatide QW Exenatide BID Liraglutide Exenatide BID Albiglutide Lixisenatide Liraglutide Exenatide QW Albiglutide Exenatide QW Dulaglutide Albiglutide Lixisenatide Semaglutide Liraglutide Dulaglutide Lixisenatide Semaglutide Dulaglutide Short-acting GLP-1 RAs retain effect on gastric emptying (and PPG), while longacting GLP-1 RAs seem to have more pronounced effects on FPG and Hb. A 1 c Semaglutide Exendin-based GLP-1 RAs seem to give rise to the formation of antibodies to a higher degree than the GLP-1 based ones; clinical implication uncertain The large GLP-1 RAs may not be able to penetrate into the brain to the same extent as the smaller types; possibly affecting appetite signalling differently *Semaglutide is under development and not approved; BID, twice daily; FPG, fasting plasma glucose; GLP-1 RA, glucagon-like peptide-1 receptor agonist; Hb. A 1 c, glycosylated haemoglobin; PPG, postprandial plasma glucose; QW, once weekly

- Slides: 44