Global Medical Device Nomenclature GMDN Mark Wasmuth CEO

Global Medical Device Nomenclature (GMDN) Mark Wasmuth – CEO, GMDN Agency
Why the need for Medical Device names?

Consistent naming is important Regulators need to: • Identify the products on their national market • Speed up Pre-market approval • Information on imports and exports • Identify trends in new equipment use • Identify individual and ‘systematic’ failure types Healthcare Providers need to: • Identify the products they use • Manage their inventory more efficiently • Identify the most effective devices Medical Devices are traded internationally we need a global system for naming

Why we need the GMDN Existing (national) nomenclatures: • Too vague with poor definitions • Too rigid structure – difficult to amend • Can’t keep up with volume of innovation • Too many duplicate descriptions • Usually Single language • Uncontrolled – no updating method for users • Unsustainable funding models • Not suitable for international harmonisation The GMDN is a global harmonizing standard

What are the benefits of using the GMDN • Used by 90 national Medical Device Regulators • Has over 7000 Manufacturers registered worldwide • Translated into 24 languages • 24, 000 product descriptions with detailed definitions • Controlled distribution and updating • Up-to-date with latest medical technology • Support for transition from other nomenclatures • Funded by Manufacturers who benefit from reduced costs of compliance The GMDN is available now

Who uses the GMDN • US FDA require GMDN in their national regulation of Unique Device Identification (UDI Rule) • European Commission uses the GMDN for their current market surveillance database (EUDAMED) and are evaluating use for their new regulation (MDR & IVDR) • The WHO use the GMDN in their publications • Widely supported by device Trade Associations (GMTA, DITTA, Advamed, Med. Tech Europe, etc) in promoting global harmonisation & reducing costs of compliance The GMDN is widely used by many countries today

GMDN Term Structure Each GMDN Term consists of 3 parts: • Term Name General-purpose syringe, single-use • Definition “A sterile device consisting of a calibrated barrel (cylinder) with plunger intended to be used for injection/withdrawal of fluids/gas (e. g. , medication) to/from a medical device or the body (i. e. , capable of both)…” • Code 47017
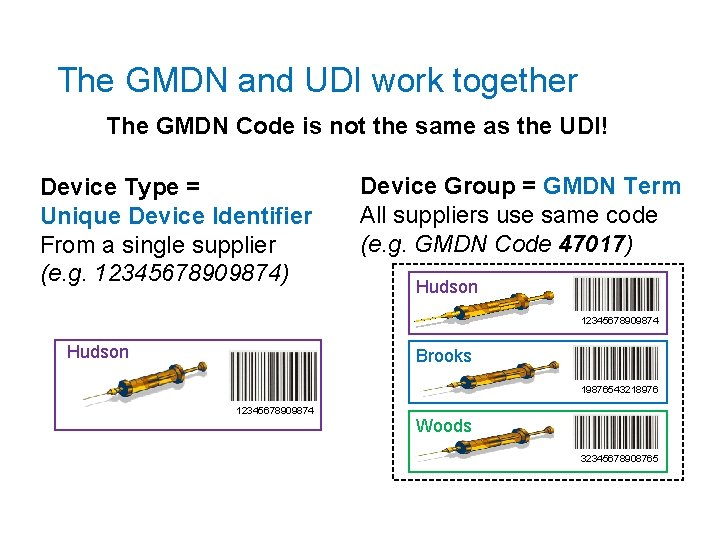
The GMDN and UDI work together The GMDN Code is not the same as the UDI! Device Type = Unique Device Identifier From a single supplier (e. g. 12345678909874) Device Group = GMDN Term All suppliers use same code (e. g. GMDN Code 47017) Hudson 12345678909874 Hudson Brooks 19876543218976 12345678909874 Woods 32345678908765
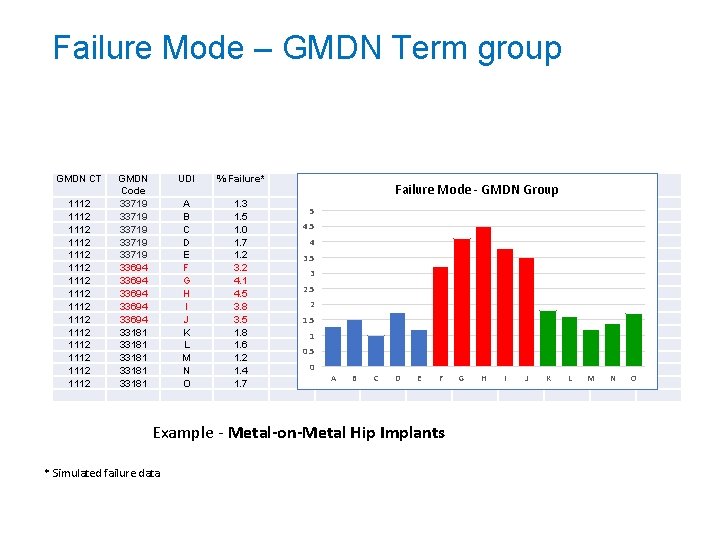
Failure Mode – GMDN Term group GMDN CT 1112 1112 1112 1112 GMDN Code 33719 33719 33694 33694 33181 33181 UDI % Failure* A B C D E F G H I J K L M N O 1. 3 1. 5 1. 0 1. 7 1. 2 3. 2 4. 1 4. 5 3. 8 3. 5 1. 8 1. 6 1. 2 1. 4 1. 7 Failure Mode - GMDN Group 5 4 3. 5 3 2. 5 2 1. 5 1 0. 5 0 A B C D E F Example - Metal-on-Metal Hip Implants * Simulated failure data G H I J K L M N O
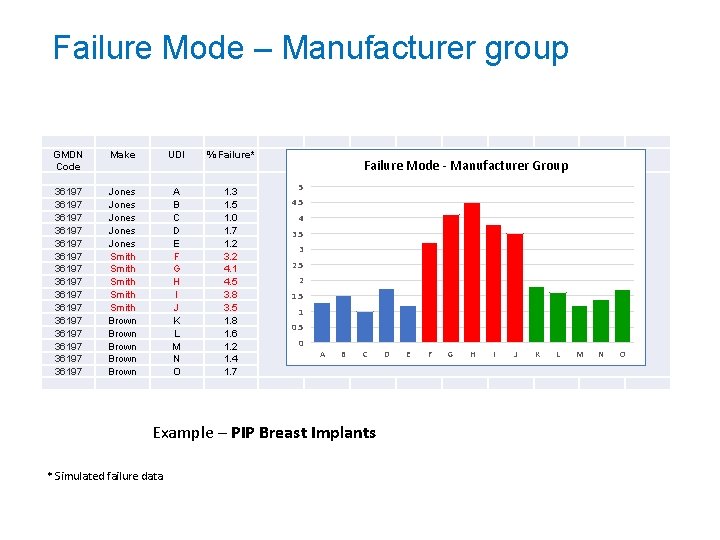
Failure Mode – Manufacturer group GMDN Code 36197 36197 36197 36197 Make UDI % Failure* Jones Jones Smith Smith Brown Brown A B C D E F G H I J K L M N O 1. 3 1. 5 1. 0 1. 7 1. 2 3. 2 4. 1 4. 5 3. 8 3. 5 1. 8 1. 6 1. 2 1. 4 1. 7 Failure Mode - Manufacturer Group 5 4 3. 5 3 2. 5 2 1. 5 1 0. 5 0 A B C Example – PIP Breast Implants * Simulated failure data D E F G H I J K L M N O

Who is the GMDN Agency? • An independent, non-profit, UK Registered Charity • Responsible for maintaining the GMDN • Set-up by Regulators (GHTF / IMDRF / AHWP) • Governance: • Board of Trustees • Policy Advisory Group – guided by: • Regulators • Healthcare Providers • Manufacturers Join us at www. gmdnagency. org
It’s all about sharing information!
- Slides: 12