Global Medical Communications 1 Insight into a Career

Global Medical Communications 1

Insight into a Career in Scientific Writing: Career Opportunities in Global Medical Communications Keri Poi Eli Lilly and Company
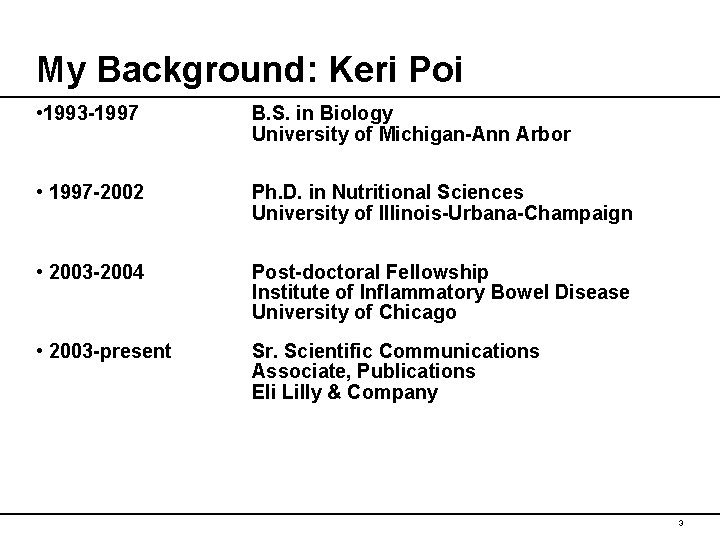
My Background: Keri Poi • 1993 -1997 B. S. in Biology University of Michigan-Ann Arbor • 1997 -2002 Ph. D. in Nutritional Sciences University of Illinois-Urbana-Champaign • 2003 -2004 Post-doctoral Fellowship Institute of Inflammatory Bowel Disease University of Chicago • 2003 -present Sr. Scientific Communications Associate, Publications Eli Lilly & Company 3

Goals for Presentation What is Scientific Communications? What is Scientific Communications at Lilly? Introduction to Eli Lilly and Company 4
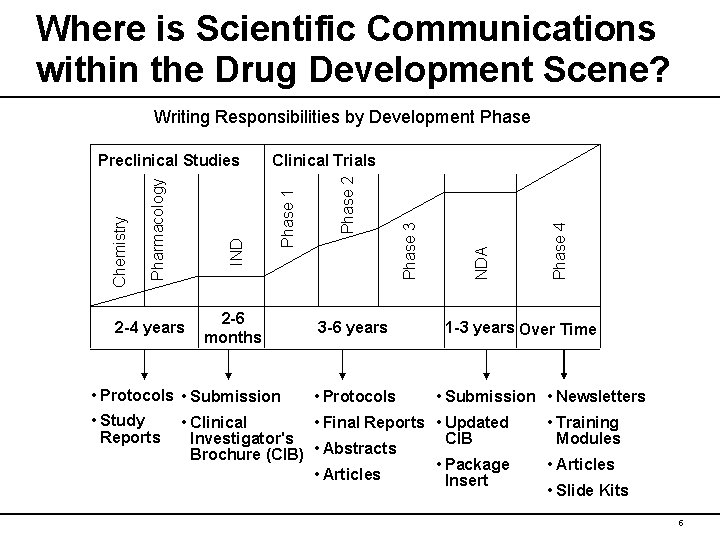
Where is Scientific Communications within the Drug Development Scene? Writing Responsibilities by Development Phase 2 -4 years 2 -6 months 3 -6 years Phase 4 NDA Phase 3 Phase 2 Clinical Trials Phase 1 IND Pharmacology Chemistry Preclinical Studies 1 -3 years Over Time • Protocols • Submission • Newsletters • Study • Clinical • Final Reports • Updated • Training Reports Investigator's CIB Modules • Abstracts Brochure (CIB) • Package • Articles Insert • Slide Kits 5

Scientific Communications at Lilly Mission: We combine scientific knowledge and communications expertise to plan, generate and deliver relevant medical communications that are meaningful to customers worldwide, advance the quality and transparency of Lilly research, and enhance patient care. 6
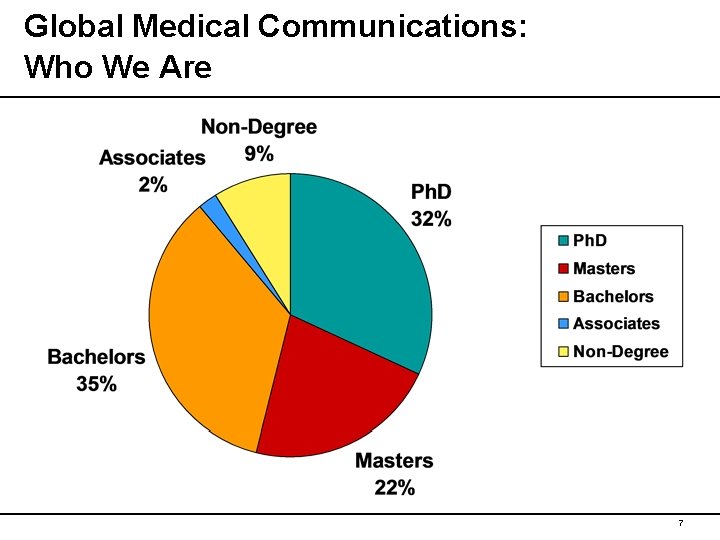
Global Medical Communications: Who We Are 7
Global Medical Communications: Job Titles Publications Coordinator Publications Assistant Team Leader Scientific Communications Associate Manager Editing Assistant Medical Information Associate 8

Current Global Medical Communications Organization The GMC organization focuses on 3 areas of scientific communications. . . • Regulatory – Global Scientific Information and Communications – Regulatory – primary audience: regulatory agencies (e. g. , FDA) • Commercialization – Global Scientific Information and Communications – Commercialization – primary audience: scientific and medical community • Medical Information – Global Medical Information – primary audience: healthcare providers 9

Global Scientific Communication and Information – Regulatory (GSIC-R) Regulatory staff members: • Design, write, and review clinical registration and scientific documents • Lead collaborative writing teams, which includes physicians, statisticians, and clinical researchers • Manage timelines and resources for complex clinical documents • Ensure the quality of scientific documents and compliance with regulatory guidelines • Develop deep expertise in medical science in the pharmaceutical industry • Facilitate the registration of new drugs in other countries 10

Global Scientific Communication and Information – Regulatory (GSIC-R) Primary Deliverables • Protocols • Investigator's Brochures • Clinical Study Reports (CSRs) • Global Registration Documents • Contribution to • • • Statistical Analysis Plans Briefing Documents Safety Updates Preparation for Regulatory Meetings Regulatory Responses 11
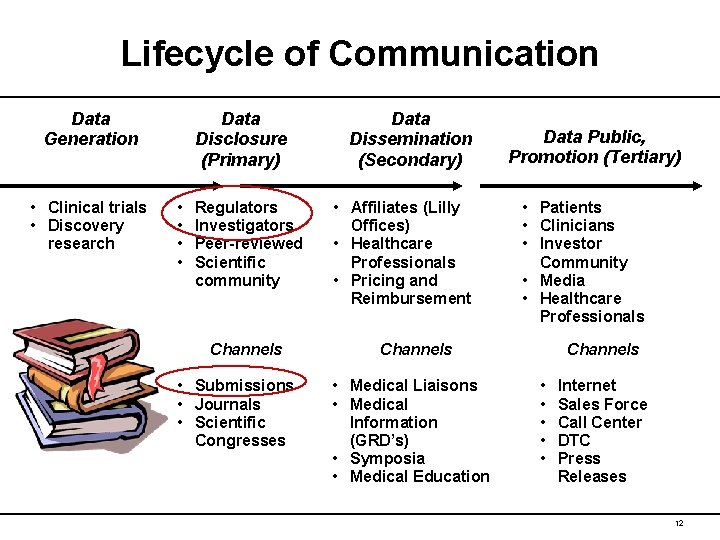
Lifecycle of Communication Data Generation • Clinical trials • Discovery research Data Disclosure (Primary) • • Regulators Investigators Peer-reviewed Scientific community Channels • Submissions • Journals • Scientific Congresses Data Dissemination (Secondary) • Affiliates (Lilly Offices) • Healthcare Professionals • Pricing and Reimbursement Data Public, Promotion (Tertiary) • Patients • Clinicians • Investor Community • Media • Healthcare Professionals Channels • Medical Liaisons • Medical Information (GRD’s) • Symposia • Medical Education Channels • • • Internet Sales Force Call Center DTC Press Releases 12

Global Scientific Communication and Information – Commercialization (GSIC-C) Commercialization staff members: • Actively participate in planning and implementing data dissemination strategy • Plan, write, and manage manuscripts, abstracts and posters • Monitor scientific literature to build therapeutic area expertise and for competitive intelligence • Based on knowledge of clinical trial data, develop novel publication ideas and plan additional analyses in collaboration with physicians and statisticians • Review and provide input to press releases and other “lay press” documents related to publications and clinical data to ensure accuracy 13
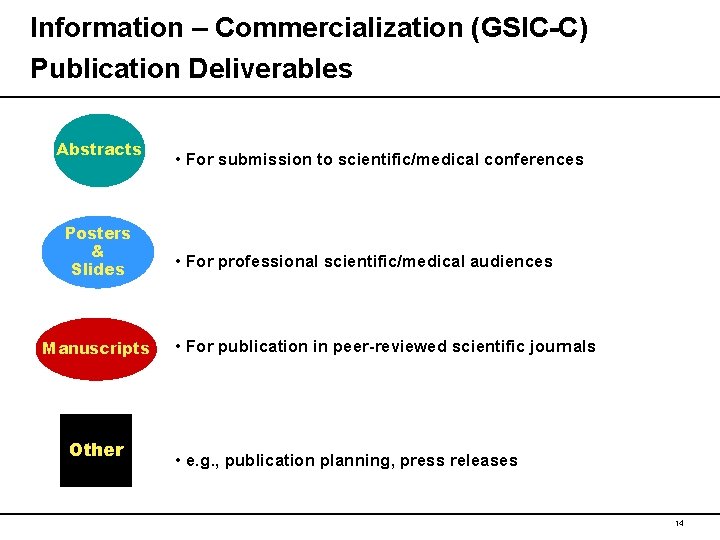
Information – Commercialization (GSIC-C) Publication Deliverables Abstracts Posters & Slides Manuscripts Other • For submission to scientific/medical conferences • For professional scientific/medical audiences • For publication in peer-reviewed scientific journals • e. g. , publication planning, press releases 14

Principles of Medical Research • Lilly discloses publicly all medical research results that are significant to patients, health care providers, or payers — whether favorable or unfavorable to a Lilly product — in an accurate, objective, and balanced manner, in order for our customers to make more informed decisions about our products. • Publications writers at Lilly are not “ghost authors, ” nor do they condone “guest authorship”! 15

ICMJE “Uniform Requirements” All three of the following criteria must be satisfied to warrant authorship: • – Making substantial contributions to conception and design, or acquisition of data, or analysis and interpretation of data. – Drafting the article or revising it critically for important intellectual content. – Giving final approval of the version to be published. • Authors get credit for and are responsible for the publication’s contents. • If a writer does not meet authorship criteria, then his or her name and contributions are listed in the “Acknowledgments. ” 16
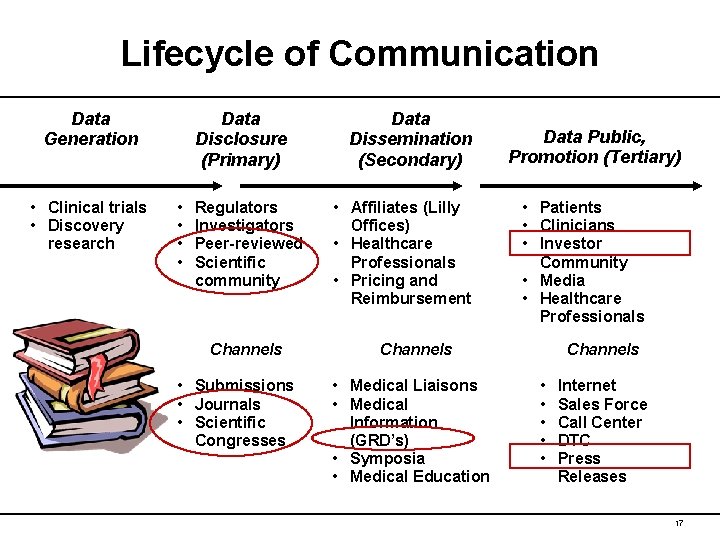
Lifecycle of Communication Data Generation • Clinical trials • Discovery research Data Disclosure (Primary) • • Regulators Investigators Peer-reviewed Scientific community Channels • Submissions • Journals • Scientific Congresses Data Dissemination (Secondary) • Affiliates (Lilly Offices) • Healthcare Professionals • Pricing and Reimbursement Data Public, Promotion (Tertiary) • Patients • Clinicians • Investor Community • Media • Healthcare Professionals Channels • Medical Liaisons • Medical Information (GRD’s) • Symposia • Medical Education Channels • • • Internet Sales Force Call Center DTC Press Releases 17

Global Medical Information (GMI) GMI staff members: • Plan, write, and edit Global Response Documents (medical letters) and unique responses to health care professionals’ questions • Interface with scientific opinion leaders • Participate in planning submission strategies and publication strategies, improving clinical document processes 18
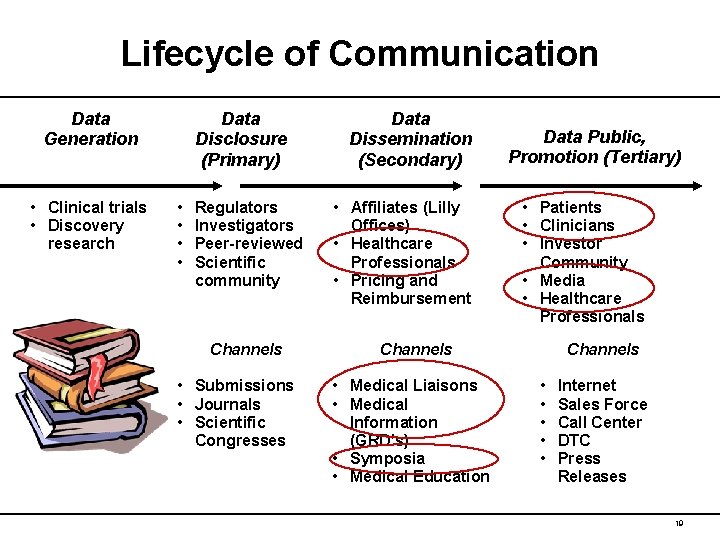
Lifecycle of Communication Data Generation • Clinical trials • Discovery research Data Disclosure (Primary) • • Regulators Investigators Peer-reviewed Scientific community Channels • Submissions • Journals • Scientific Congresses Data Dissemination (Secondary) • Affiliates (Lilly Offices) • Healthcare Professionals • Pricing and Reimbursement Data Public, Promotion (Tertiary) • Patients • Clinicians • Investor Community • Media • Healthcare Professionals Channels • Medical Liaisons • Medical Information (GRD’s) • Symposia • Medical Education Channels • • • Internet Sales Force Call Center DTC Press Releases 19

Lilly GMC Writer Qualifications • Graduate degrees preferred (bachelors degree required) • Demonstrated writing experience or aptitude. Writing exercise is required as part of candidate evaluation process • Excellent analytical, interpersonal, teamwork, project and self management, influence/negotiation, innovation/creativity, and leadership skills • Excellent written and oral communication skills 20

What is my life like as a Scientific Writer at Lilly? • I enjoy writing and had experience writing scientific papers prior to Lilly. • I am able to work in a project team, which means juggling multiple projects to meet deadlines. • I am constantly learning new things and need to pay attention to the details. • I was able to learn a particular therapeutic area (e. g. oncology, diabetes, neuroscience). • I continually use my scientific training by interpreting complex data. • I attend medical conferences that enable me to interact with physicians and learn more about clinical science. • I have had the opportunity to work with the world leaders in the field of Diabetes. • The science advancements that I have contributed, ultimately will help patients. 21

Find out More from Professional Organizations • American Medical Writers Association (AMWA) www. amwa. org • European Medical Writers Association (EMWA) www. emwa. org • Council of Science Editors (CSE) www. cbe. org • Drug Information Association (DIA) www. diahome. org • Society for Technical Communication (STC) www. stc. org • The Board of Editors in the Life Sciences (BELS) www. bels. org • Therapeutic-related organizations (ADA, APA, etc. ) 22
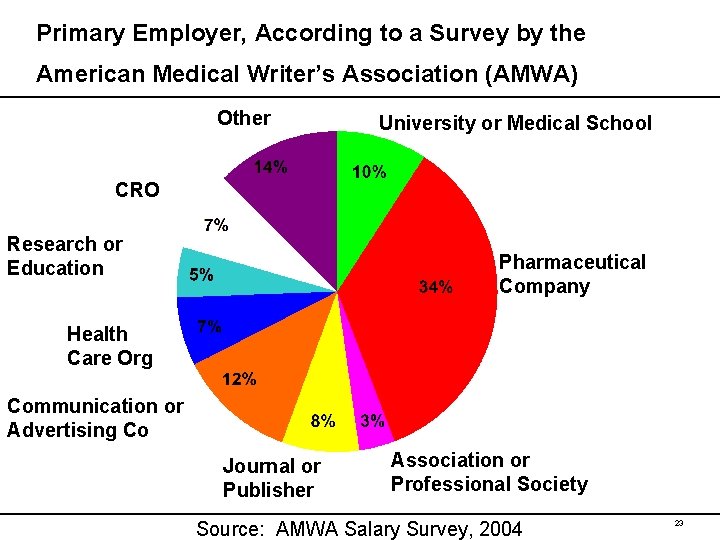
Primary Employer, According to a Survey by the American Medical Writer’s Association (AMWA) Other University or Medical School CRO Research or Education Pharmaceutical Company Health Care Org Communication or Advertising Co Journal or Publisher Association or Professional Society Source: AMWA Salary Survey, 2004 23

Field of Highest Degree, According to a Survey by the American Medical Writer’s Association (AMWA) • Science (33. 8%) • Communication (4. 3%) • English (14. 4%) • Public Health (3. 1%) • Journalism (5. 3%) • Nursing (2. 4%) • Pharmacy (5. 1%) • Technical Writing (2. 4%) • Medicine (4. 8%) • Other (24. 3%) Source: AMWA Salary Survey, 2004 24
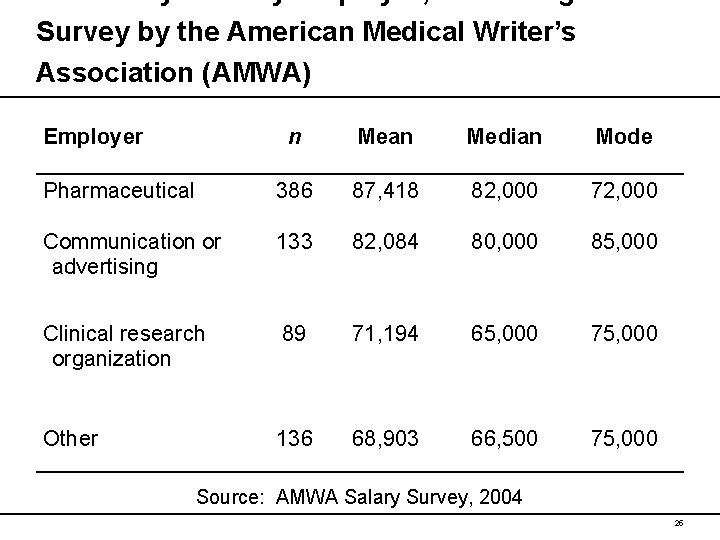
Income by Primary Employer, According to a Survey by the American Medical Writer’s Association (AMWA) Employer n Mean Median Mode Pharmaceutical 386 87, 418 82, 000 72, 000 Communication or advertising 133 82, 084 80, 000 85, 000 Clinical research organization 89 71, 194 65, 000 75, 000 Other 136 68, 903 66, 500 75, 000 Source: AMWA Salary Survey, 2004 25
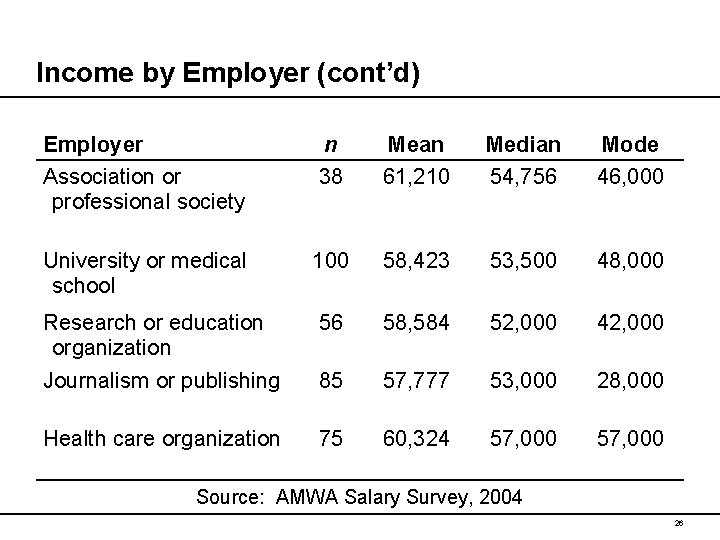
Income by Employer (cont’d) Employer Association or professional society n 38 Mean 61, 210 Median 54, 756 Mode 46, 000 University or medical school 100 58, 423 53, 500 48, 000 Research or education organization 56 58, 584 52, 000 42, 000 Journalism or publishing 85 57, 777 53, 000 28, 000 Health care organization 75 60, 324 57, 000 Source: AMWA Salary Survey, 2004 26
Scientific Communications at Lilly is a Challenging and Rewarding Career Managing Projects Writing K Medicine & Science Teamwork 27

Why Choose Lilly? Developing People • Cross-functional teams and assignments • Opportunities with global impact • Many career path options • Promotion from within • Recognized by Harvard Business Review for Succession Management Process Work-Life Balance • Flexible hours and work options • On-site conveniences • On-site child development centers • Employee volunteer policy • Business casual attire • Global recognitions • Employee clubs and activities 28

Lilly at a Glance • Founded in 1876 in Indianapolis, Indiana • 129 -year history of innovation • $14 B in sales • 43, 000 employees worldwide • Clinical research in more than 60 countries and sell products in more than 143 countries • Award winning culture: -FORTUNE magazine “ 100 Best Companies to Work For” -FORTUNE magazine "Global Most Admired Companies“ - MONEY magazine "America's Best Company Benefits" -Science magazine Best Companies for Scientists - Industry. Week "100 Best-Managed Companies“ -Business Ethics magazine 100 Best Corporate Citizens -FORTUNE magazine "50 Best Companies for Minorities“ -Working Mother magazine “ 100 Best Companies for Working Mothers” www. lilly. com/careers 29

Lilly Mission and Values • Lilly’s Mission: We provide customers “Answers that Matter” through innovative medicines, information, and exceptional customer service to enable people to live longer, healthier, and more active lives. • Integrity –Leadership by example –Honesty • Respect for People –People are our greatest asset. Lilly is committed to communicating early and often about any changes that may occur. • Excellence -We are pursuing only first-in-class/best-in-class products. -History of health care breakthroughs -Industry’s strongest late-stage pipeline Living examples of providing “Answers that Matter. ” 30

Who We’re Looking For • People who embrace the Lilly values • People with a strong desire to contribute to the world of health care • Talented individuals who can help us solve problems and meet our growth goals • People with a proven track record of leadership • Individuals who show flexibility, adaptability, and ability to manage through uncertainty 31
- Slides: 31