Global Med Technologies Inc Medical Technologies for the

Global. Med Technologies Inc. Medical Technologies for the Future Generation A Company dedicated to the Treatments and Cure of Catastrophic Diseases

Who is Global. Med Tech, (GMT) • GMT is a Bio Medical Technology Company. • GMT has developed a treatment modality which effectively inactivates the HIV virus. • GMT has Government approval to conduct Controlled Clinical Trials on 100 subjects in South America. • GMT has patented it’s process and products worldwide. • GMT Manufactures 5 Main Components: • PCI-1 Hemoirradiator • The GMT - Electronic Master Control Card • GMT Proprietary Software • Patient Intelli - Flash drive Software • The GMT – Cuvette, ( Irradiation Chamber )

PCI THE PROCESS OF HEMOIRRADIATION. • PCI IS THE PROCESS OF EXPOSING A CERTAIN QUANTITY OF BLOOD TO ULTRAVIOLET IRRADIATION VIA A STERILE CLOSED CIRCUIT, CALLED A CUVETTE OR IRRADIATION CHAMBER. • THE BLOOD NEVER COMES IN CONTACT WITH THE AMBIENT ENVIRONMENT.

FUNCTION • AS IN ANY FORM OF THERAPY, THE SUCCESS OF HEMOIRRADIATION IS DEPENDANT ON THE DOSAGE ADMINISTERED. • AND • THE CURATIVE PROPERTIES IN VIVO IS DERIVED FROM THE ACTIVATION AND STIMULATORY EFFECT UPON CELLS AND MOLECULES WHICH COMPOSE HUMAN BLOOD.

NIELS FINSEN • FATHER OF PHOTOTHERAPY: • Won Nobel Prize in Physiology & Medicine in 1903 “In recognition of his contribution to the treatment of diseases, especially Lupus Vulgaris, with concentrated light radiation, whereby he has opened a new avenue for medical science“ • Dr. Finsen’s contribution to medicine is unparallel. He treated 300 patients with Lupus Vulgaris and demonstrated, 100% success rates. Today, we have nothing in our arsenal of medications that can produce such results.
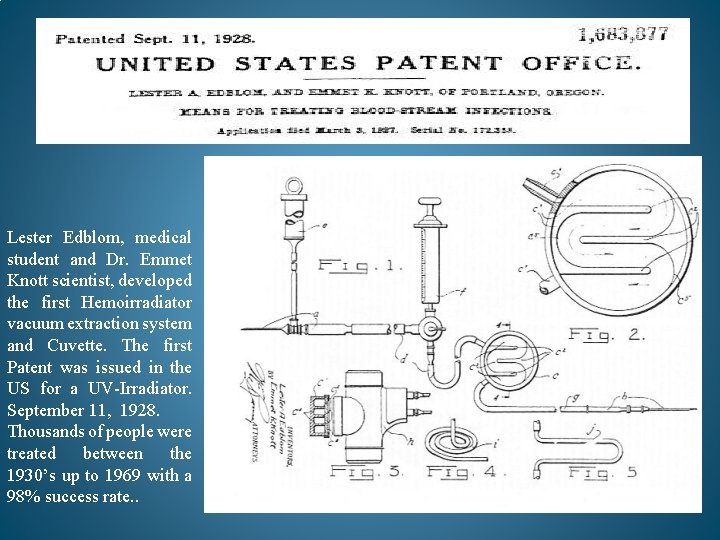
Lester Edblom, medical student and Dr. Emmet Knott scientist, developed the first Hemoirradiator vacuum extraction system and Cuvette. The first Patent was issued in the US for a UV-Irradiator. September 11, 1928. Thousands of people were treated between the 1930’s up to 1969 with a 98% success rate. .

Emmet Knott Irradiator.
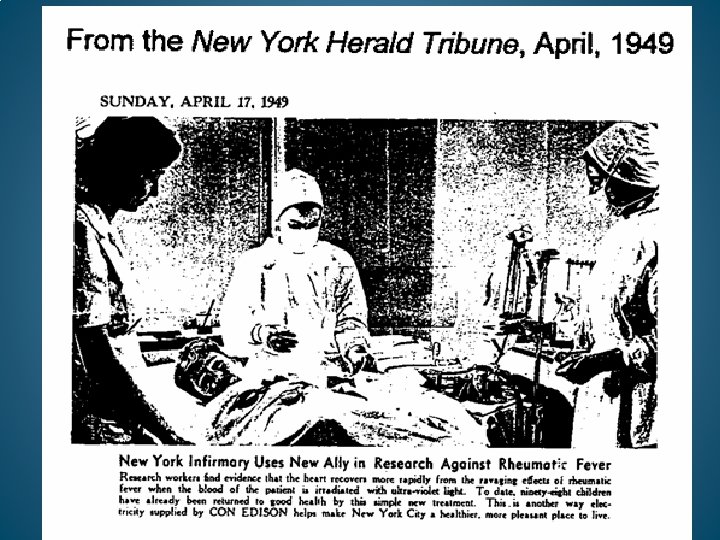
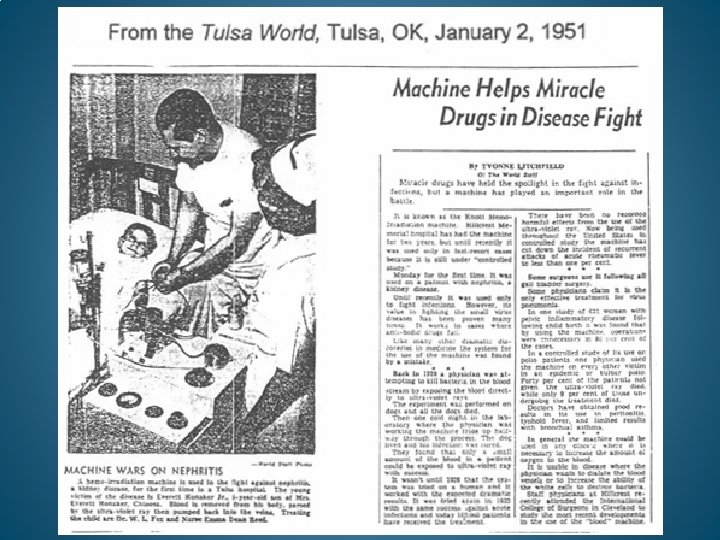

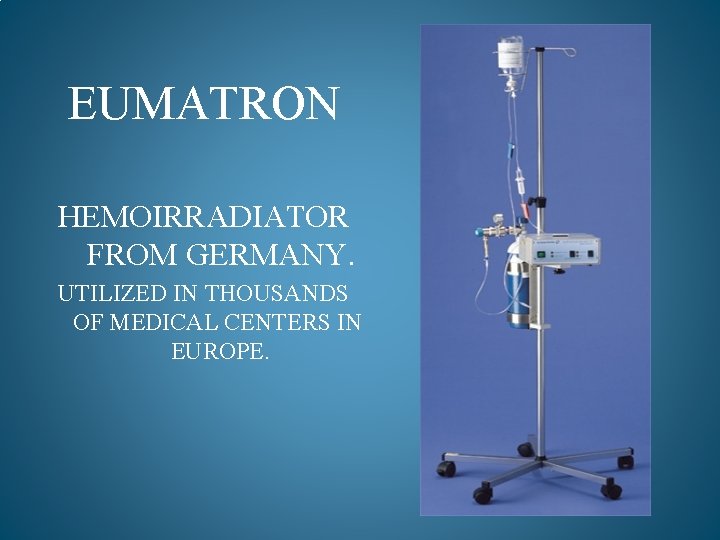
EUMATRON HEMOIRRADIATOR FROM GERMANY. UTILIZED IN THOUSANDS OF MEDICAL CENTERS IN EUROPE.

PTX 4 PHOTONIC PUMP USA. NOT-FDA APPROVED UTILIZED IN VARIOUS ALTERNATIVE CENTERS IN THE US
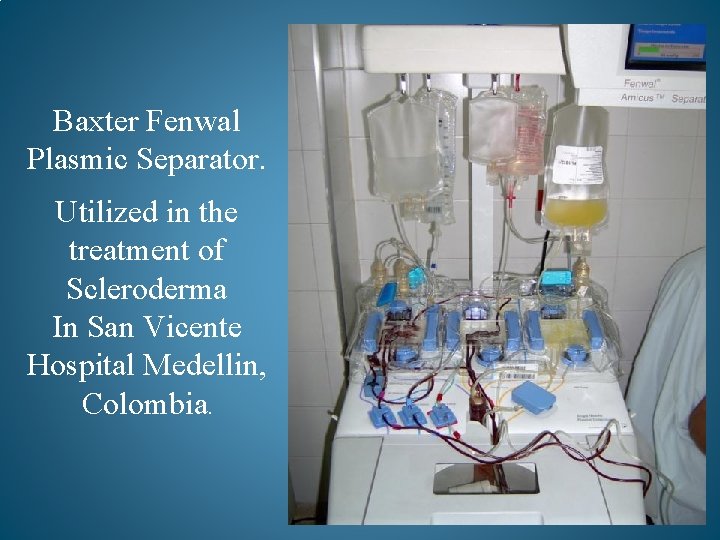
Baxter Fenwal Plasmic Separator. Utilized in the treatment of Scleroderma In San Vicente Hospital Medellin, Colombia.
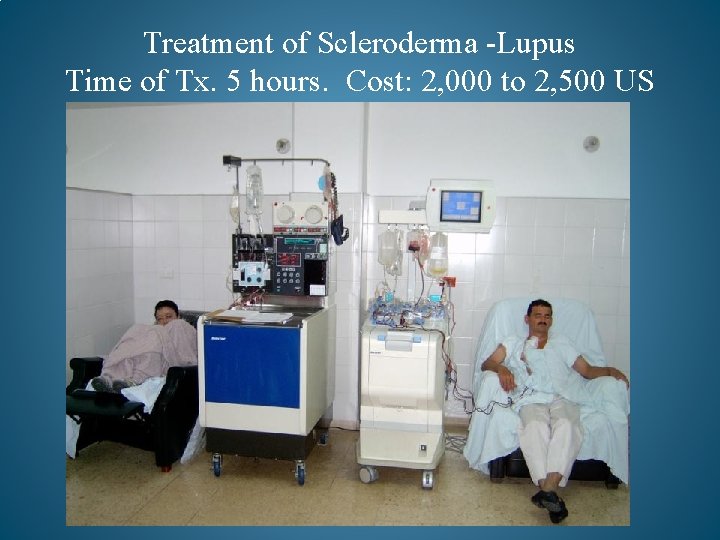
Treatment of Scleroderma -Lupus Time of Tx. 5 hours. Cost: 2, 000 to 2, 500 US

THERAKOS JOHNSON & JOHNSON Ultraviolet A Irradiator developed by Dr. Elderson and utilized in clinical trials on AIDS Related ARC patients at Columbia University in 1990. Four subjects were registered as SERO Negative after 5 to 19 Sessions.
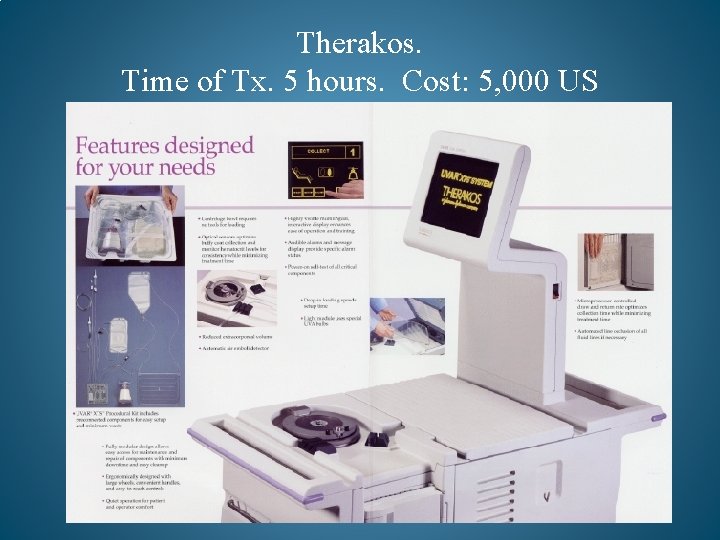
Therakos. Time of Tx. 5 hours. Cost: 5, 000 US

Global. Med Technologies: • • • PCI-1 Hemoirradiator GMT- Cuvette (Irradiation Chamber) Electronic Master Control Card Electronic UV Detector GMT Proprietary Software Patient Inteli - Flash drive Software

PCI-1 GLOBALMED Time of Tx. 25 minutes Cost: $100. 00 )

GMT-PCI-1 Bar Code Scanner Automatic Peristalsis Pump Inteliflash-Card UVC Irradiation Bulb LCD UV protection Lid Keyboard

GMT-Cuvette Utilized during Treatments. Developed and Manufactured by GMT. Patent Registered Worldwide.

GMT -Cuvette (Irradiation Chamber) Prototype Blood Banks Human

GMT -Cuvette (Irradiation Chamber) External Body Quartz Glass Top O-Ring 1 O-Ring 2
GMT –Kit
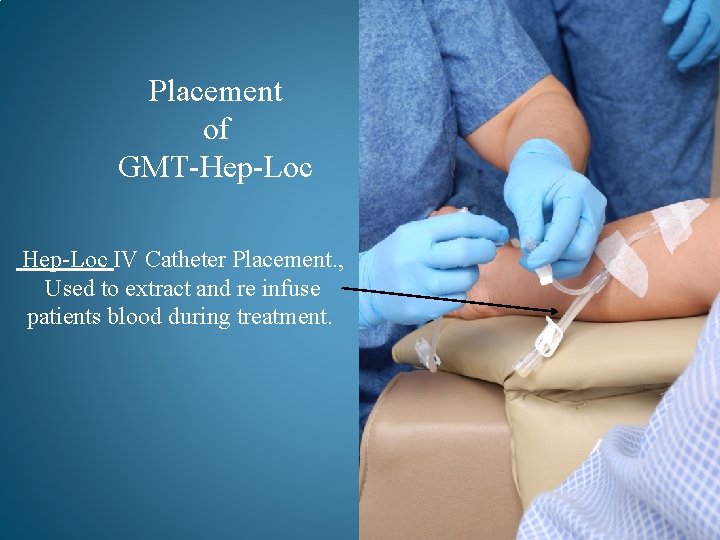
Placement of GMT-Hep-Loc IV Catheter Placement. , Used to extract and re infuse patients blood during treatment.
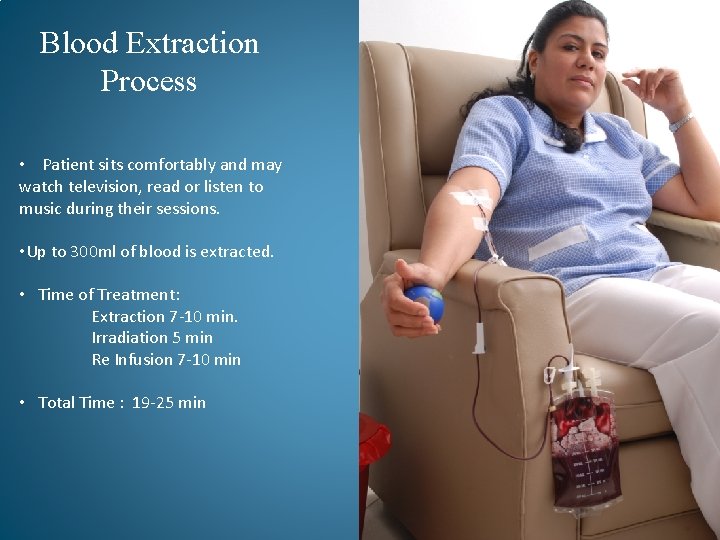
Blood Extraction Process • Patient sits comfortably and may watch television, read or listen to music during their sessions. • Up to 300 ml of blood is extracted. • Time of Treatment: Extraction 7 -10 min. Irradiation 5 min Re Infusion 7 -10 min • Total Time : 19 -25 min
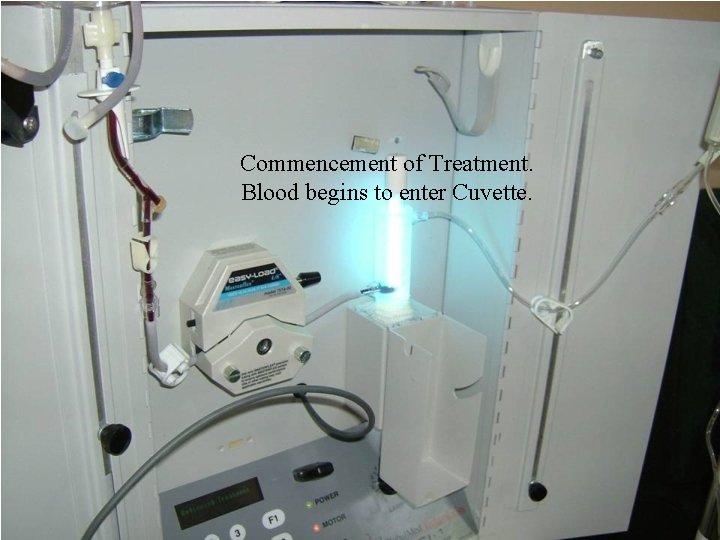
Commencement of Treatment. Blood begins to enter Cuvette.

Irradiation Process. The blood is propelled by the Peristaltic Pump via the Irradiation Chamber (Cuvette) where the blood is irradiated with the precise amount of UV Dosage. After irradiation the blood continues to the secondary Blood Bag and from there is returned via a Blood Administration with a 70 um Filter.
Re Infusion Process

Government Approved Clinical Study in the Republic Dominican with 36 Patients living with HIV/AIDS

PROTOCOL ISUV-001 -04 CLASSIFICATION BY AGE GROUP SUBJECTS 18 – 30 31 – 40 41 – 51 TOTAL: 12 16 8 36
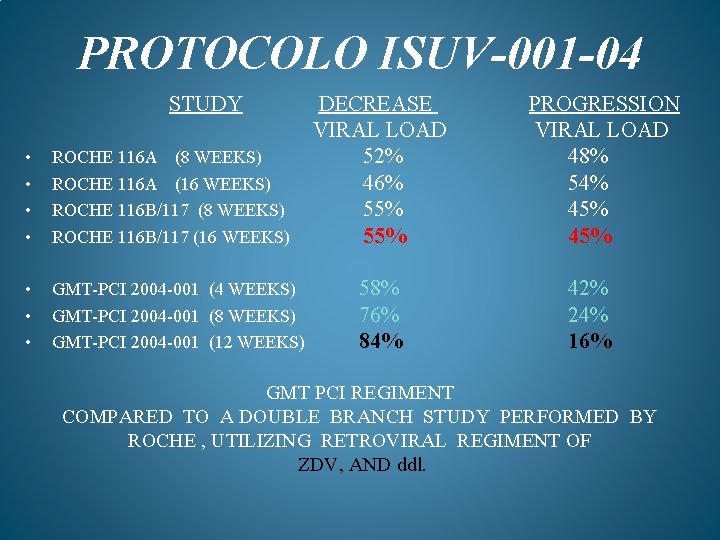
PROTOCOLO ISUV-001 -04 STUDY DECREASE VIRAL LOAD 52% 46% 55% • • ROCHE 116 A (8 WEEKS) ROCHE 116 A (16 WEEKS) ROCHE 116 B/117 (8 WEEKS) ROCHE 116 B/117 (16 WEEKS) • • • GMT-PCI 2004 -001 (4 WEEKS) 58% GMT-PCI 2004 -001 (8 WEEKS) 76% GMT-PCI 2004 -001 (12 WEEKS) 84% PROGRESSION VIRAL LOAD 48% 54% 45% 42% 24% 16% GMT PCI REGIMENT COMPARED TO A DOUBLE BRANCH STUDY PERFORMED BY ROCHE , UTILIZING RETROVIRAL REGIMENT OF ZDV, AND ddl.

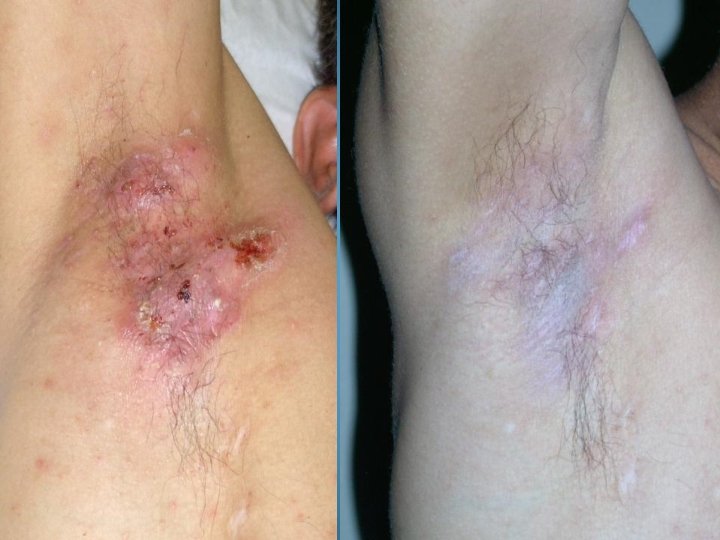
BEFORE AND AFTER PHOTOS. AFTER 90 DAYS OF TREATMENT, 100% OF OPPORTUNISTIC INFECTIONS HAD SUBSIDED. GMT believes it can reduce the cost to keep one HIV/AIDS patient alive by 90% a year. Savings to Medicare and Medicaid would be in the Billions. Current Across the Board yearly cost in US to keep 1 million HIV/AIDS patients alive = 200 Billion Estimated cost with GMT Protocol 1 million patients 15 to 20 Billion
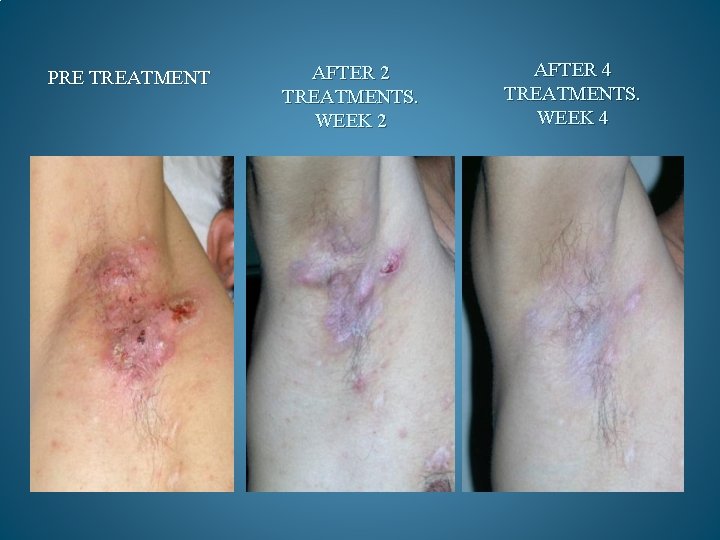
PRE TREATMENT AFTER 2 TREATMENTS. WEEK 2 AFTER 4 TREATMENTS. WEEK 4
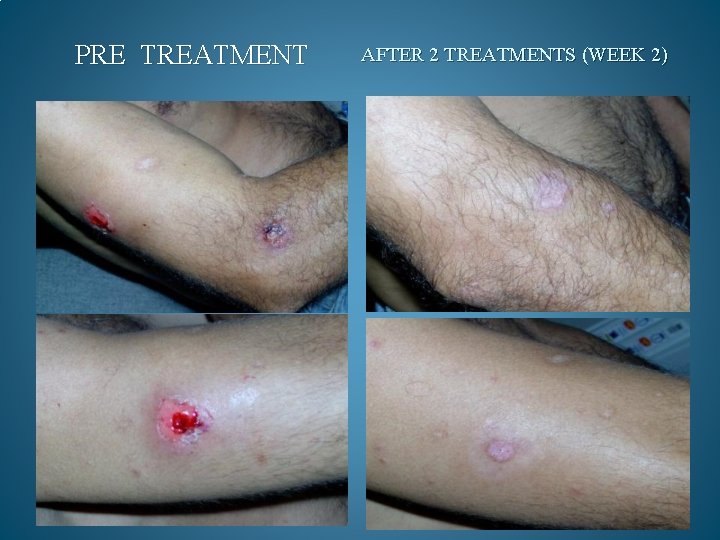
PRE TREATMENT AFTER 2 TREATMENTS (WEEK 2)
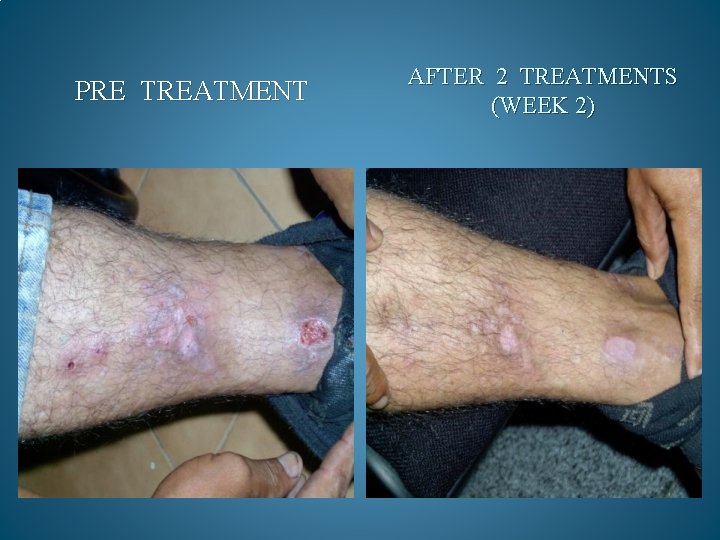
PRE TREATMENT AFTER 2 TREATMENTS (WEEK 2)
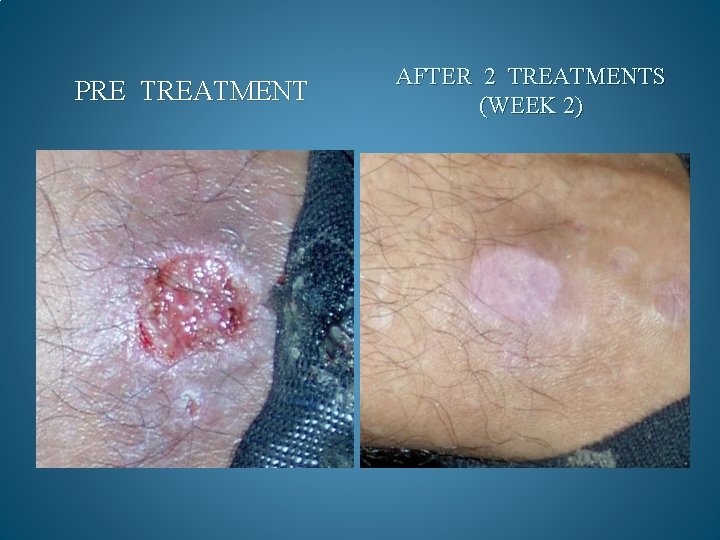
PRE TREATMENT AFTER 2 TREATMENTS (WEEK 2)
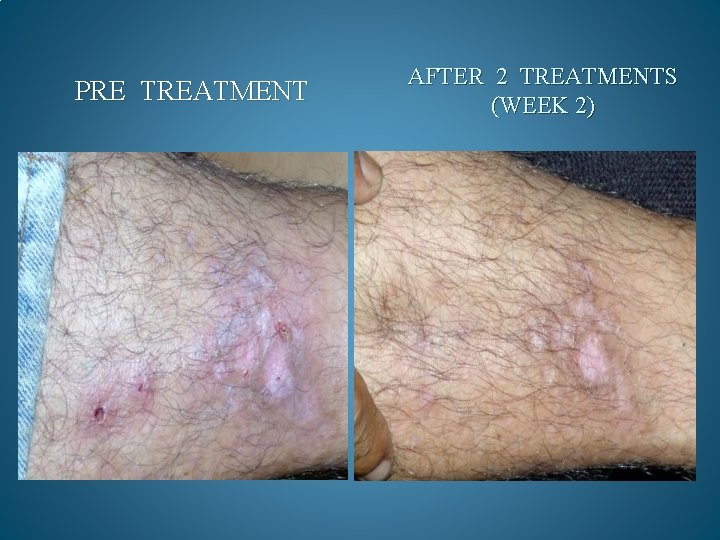
PRE TREATMENT AFTER 2 TREATMENTS (WEEK 2)
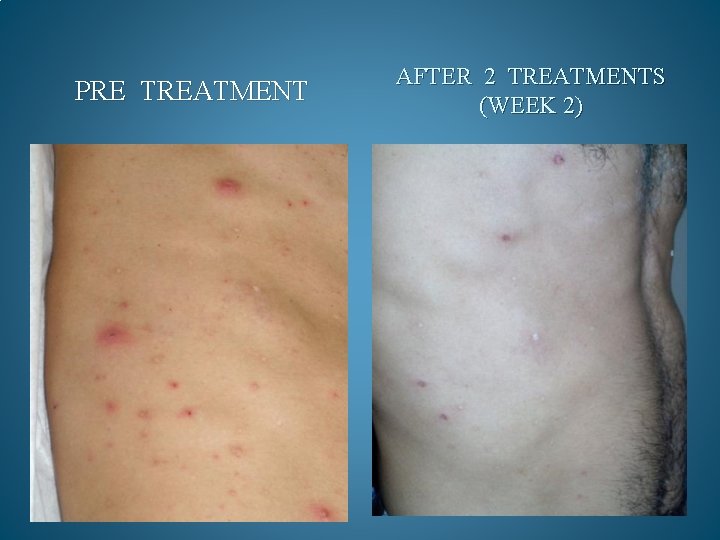
PRE TREATMENT AFTER 2 TREATMENTS (WEEK 2)
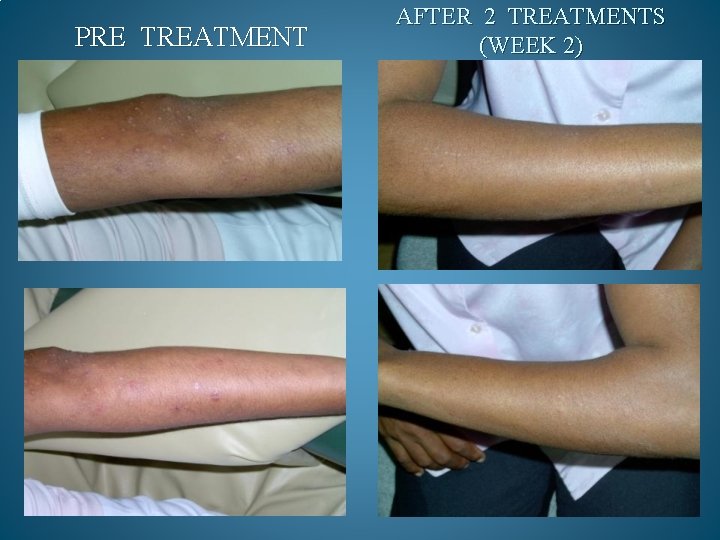
PRE TREATMENT AFTER 2 TREATMENTS (WEEK 2)
- Slides: 40