Global Leadership Meeting Craig Picinich Agenda 2 2

Global Leadership Meeting Craig Picinich

Agenda • • • 2 2 CTMS/e. TMF Implementation First Study Live R 1. 1 High Level Scope Next Steps Q&A

Global Leadership Meeting CTMS will transform our clinical trial processes and systems to create a global solution that unquestionably complies with regulatory requirements and enhances our capability to deliver robust and accurate study data. CTMS will eliminate tedious manual effort and focus employees on more interesting work truly valued by our clients. The system will improve quality and productivity for clinical trials and recognized by our existing and future clients as the industry leading CRO partner

Global Leadership Meeting e. TMF Electronic Trial Master File (e. TMF) Creating a regulatory compliant electronic trial master filing system, which provides us with: –Streamlined integrated processes –Instant access to up-to-date trial master file information –And enables us to delight our clients every time
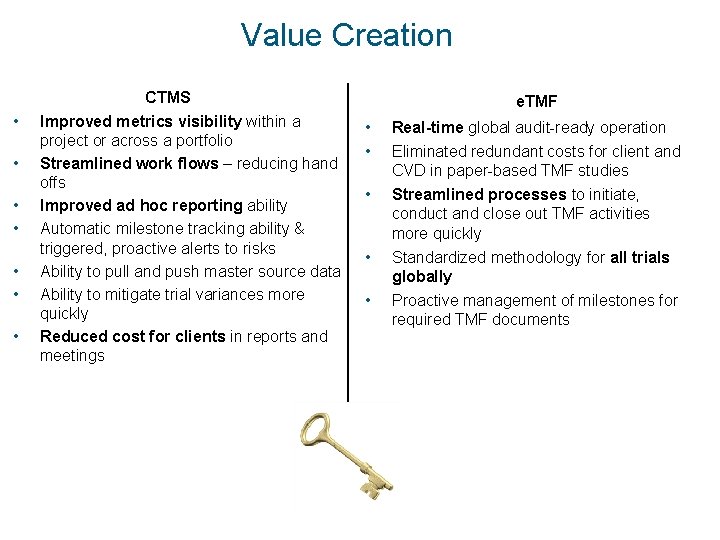
Value Creation • • CTMS Improved metrics visibility within a project or across a portfolio Streamlined work flows – reducing hand offs Improved ad hoc reporting ability Automatic milestone tracking ability & triggered, proactive alerts to risks Ability to pull and push master source data Ability to mitigate trial variances more quickly Reduced cost for clients in reports and meetings e. TMF • • Real-time global audit-ready operation Eliminated redundant costs for client and CVD in paper-based TMF studies • Streamlined processes to initiate, conduct and close out TMF activities more quickly • Standardized methodology for all trials globally Proactive management of milestones for required TMF documents •
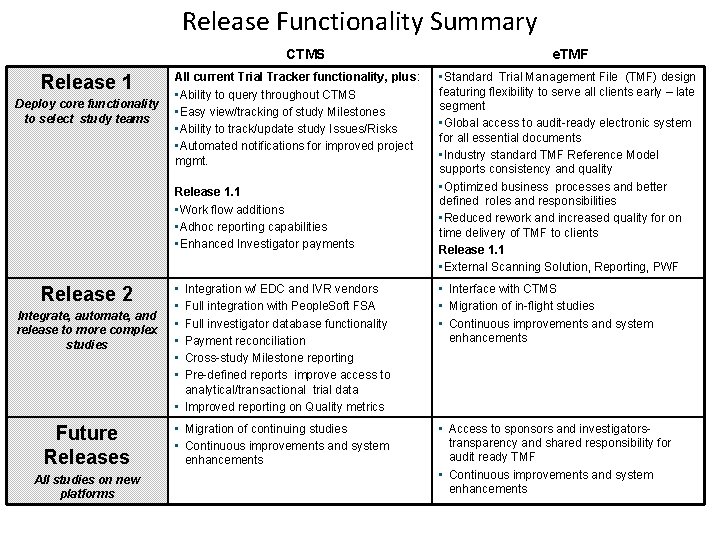
Release Functionality Summary CTMS Release 1 Deploy core functionality to select study teams All current Trial Tracker functionality, plus: • Ability to query throughout CTMS • Easy view/tracking of study Milestones • Ability to track/update study Issues/Risks • Automated notifications for improved project mgmt. Release 1. 1 • Work flow additions • Adhoc reporting capabilities • Enhanced Investigator payments Release 2 Integrate, automate, and release to more complex studies Future Releases All studies on new platforms 6 e. TMF • Standard Trial Management File (TMF) design featuring flexibility to serve all clients early – late segment • Global access to audit-ready electronic system for all essential documents • Industry standard TMF Reference Model supports consistency and quality • Optimized business processes and better defined roles and responsibilities • Reduced rework and increased quality for on time delivery of TMF to clients Release 1. 1 • External Scanning Solution, Reporting, PWF • • • Integration w/ EDC and IVR vendors Full integration with People. Soft FSA Full investigator database functionality Payment reconciliation Cross-study Milestone reporting Pre-defined reports improve access to analytical/transactional trial data • Improved reporting on Quality metrics • Interface with CTMS • Migration of in-flight studies • Continuous improvements and system enhancements • Migration of continuing studies • Continuous improvements and system enhancements • Access to sponsors and investigatorstransparency and shared responsibility for audit ready TMF • Continuous improvements and system enhancements

Release 1 Study Selection Diamond 2 evaluated potential studies as candidates to be used in Release 1. 0 Project Study Parameters Duration Short-term Phase I/II # sites ~20 # CDS resources TBD # study participants ~80 patients Other Considerations: ØStudies will take place in only 1 or 2 countries ØFirst Patient Enrolled – projected for October ØSame studies being considered for CTMS and e. TMF 7

Controlled Pilot Objectives • Exercise using the new systems, processes, index and SOPs in support of a clinical study trial with a limited set of users • Actively monitor user experience and system issues to identify challenges prior to the broader release • Validate project specific SOPs for global use • Validate the new Index 8

How e. TMF will Improve CAPA Results 11 CAPA top three categories/findings: Finding Category 1 Essential Documentation – Inadequate or Incomplete How e. TMF Release 1. 0 will solve • • 2 Essential Documentation – Documents Missing With No Notes to File (NTFs) • • • 3 Filing/Archiving—Missing Documents/Mis-filed Documents • • • 9 Compile a “What’s Missing” list to show completeness of TMF instantly Reporting features assist with proactive management of TMF Be able to identify an artifact and whether it is a NTF Show if the NTF is related to another document or if that artifact will never be expected Have specific reporting views to show NTFs by Trial, Country, Site New Business Processes reinforce and verify quality document delivery Capabilities within e. TMF to search on content or documents by name Access the “What’s Missing” list to show completeness of TMF instantly Ability to copy documents from study to study Ability to reclassify (“re-index”) a document (re-file in the correct place)

Covance TMF Index As you know… The TMF Index has been redesigned: • • 10 Standardized the classification of the TMF Based on the TMF Reference Model Harmonized across ECD, Phase 1 -4 Developed and refined by a Global Team

New e. TMF Index approved by CDS leadership based on the TMF Reference Model ØAll “Artifacts” were assessed for inclusion or exclusion in the e. TMF Ø 308 documents were defined and assigned a place in the Index and an “owner” who is responsible for the review and approval of each “final” document to be filed ØFramework to be used as the filing plan for all studies and sponsors to insure consistency and quality Harmonized and applicable across ECD and Phases I - IV 11

SOP Communications Standard Operating Procedures (SOP) published to support: • • Documentation of new business processes along with AD’s and WI’s, which outlines roles and responsibilities The development and approval of the TMF Plan and TMF Index The Set-up, maintenance and archiving of e. TMF and Paper TMF User access to information on the creation of the Regulatory Binder index and Pharmacy File index Why now… • • 12 Right time and right situation New virtual filing and index system New roles and responsibilities Provide guidance on how to operate and use new system

CTMS/e. TMF – Pilot Training Detail • • • Just-in-time role-based training was conducted to support Release 1. 0 Courses were separated by Function/Responsibility/Capability Course Audience was determined by matching a participants role to their required functional capabilities September – Instructor-led Training Schedule (Princeton, NJ) Date Course Name Audience by Role September 12 e. TMF and CTMS Fundamentals All Roles (Clinical Business Administrators, CPA’s, e. TMF Specialist, PMs, GSS) September 18 e. TMF Set-up Clinical Business Administrators September 19 e. TMF and CTMS for Project Managers September 20 e. TMF for Project Managers September 24 e. TMF for the e. TMF Specialists e. TMF Specialist September 25 e. TMF Fundamentals CRAs September 25 CTMS Fundamentals CRAs September 26 Using CTMS for CRA's CRAs September 27 Using e. TMF for Contributors CRAs, GSS September 27 GSS for PM’s/PC’s GSS Staff and PM/PC Staff 13
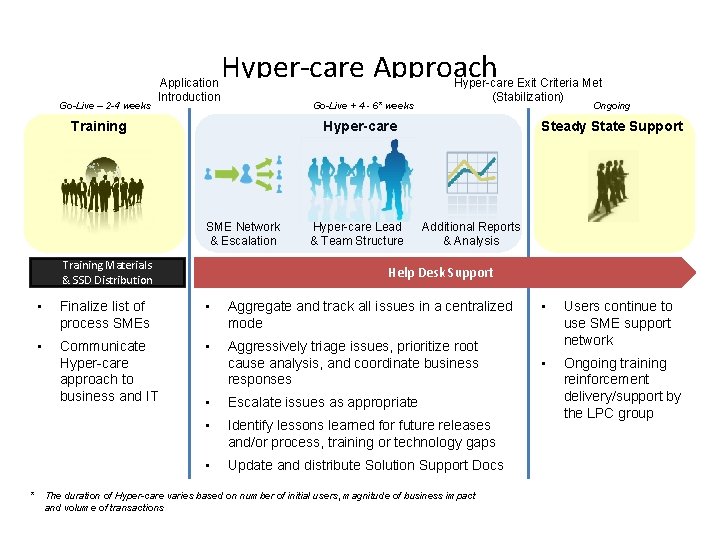
Go-Live – 2 -4 weeks Application Introduction Hyper-care Approach Go-Live + 4 - 6* weeks Training Hyper-care Exit Criteria Met (Stabilization) Ongoing Hyper-care SME Network & Escalation Training Materials & SSD Distribution Hyper-care Lead & Team Structure Steady State Support Additional Reports & Analysis Help Desk Support • Finalize list of process SMEs • Aggregate and track all issues in a centralized mode • Communicate Hyper-care approach to business and IT • Aggressively triage issues, prioritize root cause analysis, and coordinate business responses • Escalate issues as appropriate • Identify lessons learned for future releases and/or process, training or technology gaps • Update and distribute Solution Support Docs * The duration of Hyper-care varies based on number of initial users, magnitude of business impact and volume of transactions 14 • Users continue to use SME support network • Ongoing training reinforcement delivery/support by the LPC group
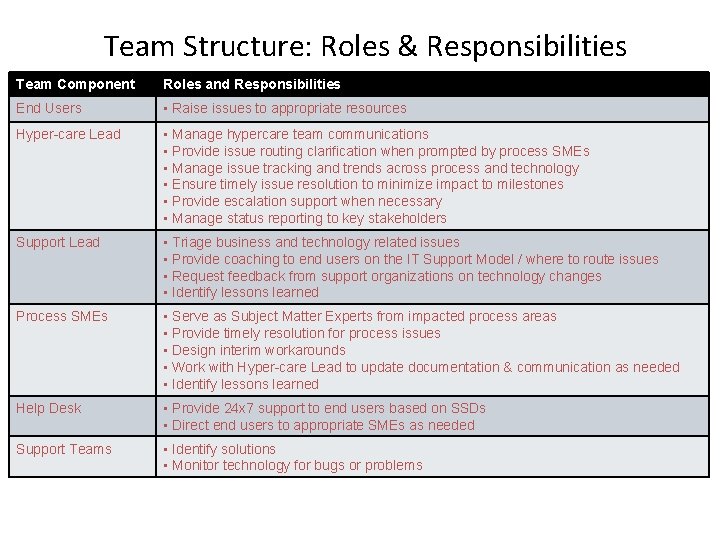
Team Structure: Roles & Responsibilities Team Component Roles and Responsibilities End Users • Raise issues to appropriate resources Hyper-care Lead • Manage hypercare team communications • Provide issue routing clarification when prompted by process SMEs • Manage issue tracking and trends across process and technology • Ensure timely issue resolution to minimize impact to milestones • Provide escalation support when necessary • Manage status reporting to key stakeholders Support Lead • Triage business and technology related issues • Provide coaching to end users on the IT Support Model / where to route issues • Request feedback from support organizations on technology changes • Identify lessons learned Process SMEs • Serve as Subject Matter Experts from impacted process areas • Provide timely resolution for process issues • Design interim workarounds • Work with Hyper-care Lead to update documentation & communication as needed • Identify lessons learned Help Desk • Provide 24 x 7 support to end users based on SSDs • Direct end users to appropriate SMEs as needed Support Teams • Identify solutions • Monitor technology for bugs or problems 15
- Slides: 15