Global Harmonisation Task Force SG 2 Information exchange

Global Harmonisation Task Force SG 2: Information exchange Dr. Ekkehard Stösslein Federal Institute for Drugs and Medical Devices Tel. : +49 -228 -207 5384; Fax: +49 -228 -207 -5300 e. stoesslein@bfarm. de Riyadh, November 29/30, 2005 Dr. Ekkehard Stösslein
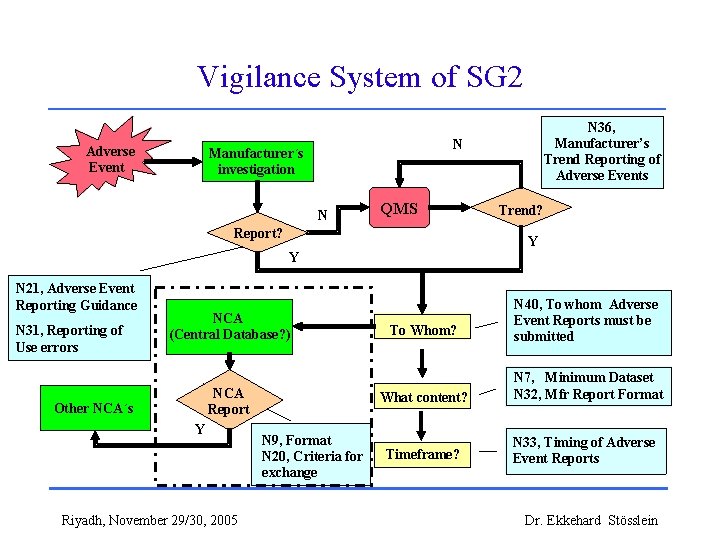
Vigilance System of SG 2 Adverse Event N Manufacturer´s Manufacturer investigation N Report? QMS N 31, Reporting of Use errors Other National Other NCA´s Regulators National NCA Regulator (Central Database? ) Vigilance NCA Report? Report Y Riyadh, November 29/30, 2005 N 9, Format N 20, Criteria for exchange Trend? Y Y N 21, Adverse Event Reporting Guidance N 36, Manufacturer’s Trend Reporting of Adverse Events To To Whom? N 40, To whom Adverse Event Reports must be submitted What content? N 7, Minimum Dataset N 32, Mfr Report Format When? Timeframe? N 33, Timing of Adverse Event Reports Dr. Ekkehard Stösslein

GHTF-Vigilance Report Exchange (1) SG 2 N 20 R 7: Competent Authority Reporting Criteria for the Exchange seriousness should be linked with many of the following criteria • • • unexpectedness of incident/event population vulnerable (e. g paediatric, unconscious patients) preventability (useful recommendations) public concern / outrage (radioactivity, infections ) benefit/risk ratio, state of the art, alternatives lack of scientific date (especially long term effects) repeated device problems that re-surfaced class I recalls or equivalent (in EU acc. to EN 46001) written notifications by one NCA to the public Riyadh, November 29/30, 2005 Dr. Ekkehard Stösslein

GHTF-Vigilance Report Exchange (2) • 2 levels of information exchange • highly sensitive and/or confidential (e. g. on ongoing investigations) • selected public (non-confidential) information • active exchange by email via the NCAR secretariat in ENGLISH LANGUAGE • NCAR secretariat is responsible for checking the completness of the report and maintaining the distribution list Riyadh, November 29/30, 2005 Dr. Ekkehard Stösslein

Principle Rules for Exchange • contact mfr before sending out a report to other NCA´s • use NCA reporting form SG 2 N 9 latest version • if investigation is completed and NO action is required the report should not be exchanged actively • if investigation is not completed but actions are contemplated a report should be sent Riyadh, November 29/30, 2005 Dr. Ekkehard Stösslein

Exchange (1) • started in January 1999 with a pilot pahse • about 100 reports have been exchanged between 1999 and December 2000 (DE , UK, US, AU, NO, CH ) • regular exchange since January 2001 • about 618 reports (up to Mai 31 st, 2004) have been exchanged • most NCA declare it to be a useful vehicle to exchange information about vigilance • example: DE-2004 -09 -06 -150. doc DE-2004 -10 -28 -189. doc • requirements on how NCAs outside GHTF could participate on that exchange (SG 2 N 38) Riyadh, November 29/30, 2005 Dr. Ekkehard Stösslein

Application Process (1) • Associate Participant: An organisation that participates in the NCAR program that receives only public information (see definition of public information) from other NCAR participants. Associate participants may contribute NCARs that contain either public or confidential information, but are not compelled to do so. An associate participant may not necessarily be a National Competent Authority. • Full Participant: An organisation that participates in the NCAR program that receives both public and confidential information from other NCAR participants. Full participation is open only to National Competent Authorities. Riyadh, November 29/30, 2005 Dr. Ekkehard Stösslein
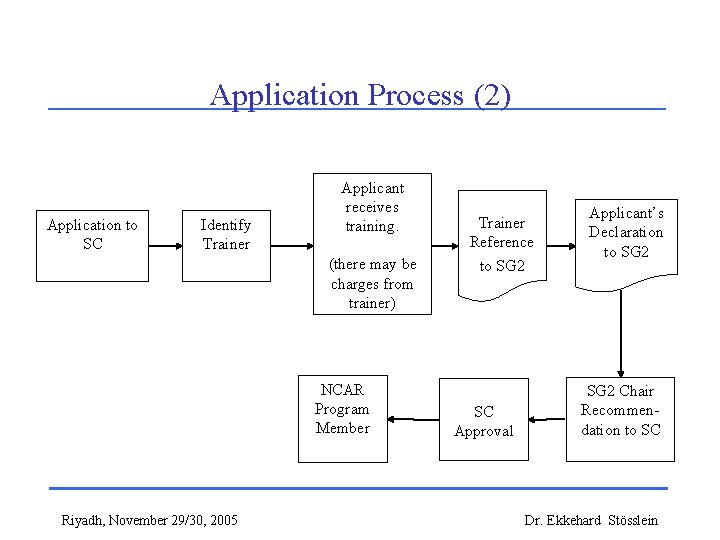
Application Process (2) Application to SC Identify Trainer Applicant receives training. (there may be charges from trainer) NCAR Program Member Riyadh, November 29/30, 2005 Trainer Reference to SG 2 SC Approval Applicant’s Declaration to SG 2 Chair Recommendation to SC Dr. Ekkehard Stösslein
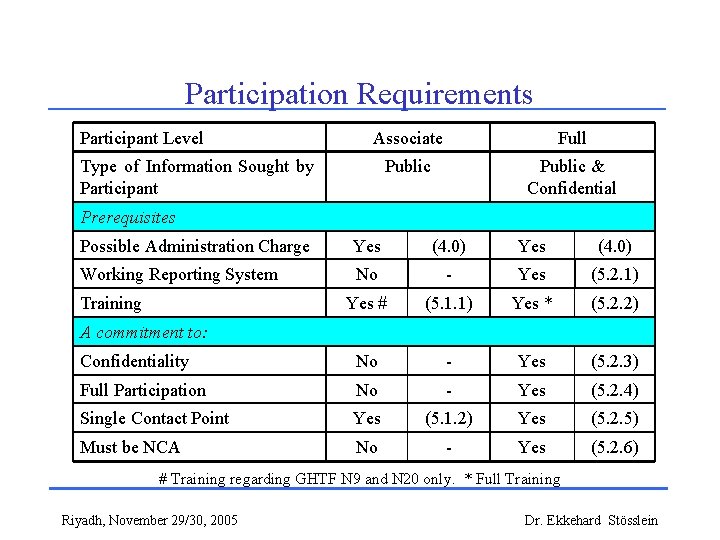
Participation Requirements Participant Level Associate Full Public & Confidential Type of Information Sought by Participant Prerequisites Possible Administration Charge Yes (4. 0) Working Reporting System No - Yes (5. 2. 1) Yes # (5. 1. 1) Yes * (5. 2. 2) Confidentiality No - Yes (5. 2. 3) Full Participation No - Yes (5. 2. 4) Single Contact Point Yes (5. 1. 2) Yes (5. 2. 5) Must be NCA No - Yes (5. 2. 6) Training A commitment to: # Training regarding GHTF N 9 and N 20 only. * Full Training Riyadh, November 29/30, 2005 Dr. Ekkehard Stösslein

For More Information See www. ghtf. org Thanks for Your Attention Riyadh, November 29/30, 2005 Dr. Ekkehard Stösslein
- Slides: 10