GLOBAL BIOGEOCHEMICAL CYCLING NITROGEN OXYGEN CARBON SULFUR Daniel

GLOBAL BIOGEOCHEMICAL CYCLING: NITROGEN, OXYGEN, CARBON, SULFUR Daniel J. Jacob Harvard University http: //www-as. harvard. edu/chemistry/trop

THE EARTH: ASSEMBLAGE OF ATOMS OF THE 92 NATURAL ELEMENTS • Most abundant elements: iron (core), silicon (mantle), hydrogen (oceans), nitrogen, oxygen, carbon, sulfur… • The elemental compostion of the Earth has remained essentially unchanged over its 4. 5 Gyr history – Extraterrestrial inputs (e. g. , from meteorites, cometary material) have been relatively unimportant – Escape to space has been restricted by gravity • Biogeochemical cycling of these elements between the different reservoirs of the Earth system determines the composition of the Earth’s atmosphere and the evolution of life
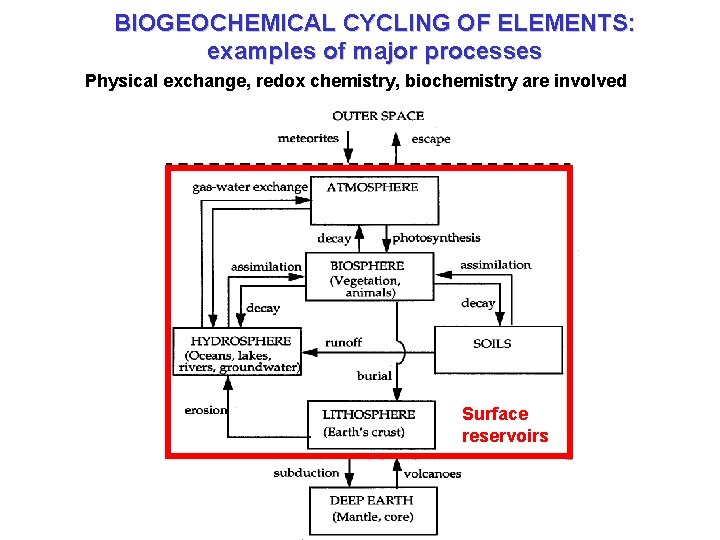
BIOGEOCHEMICAL CYCLING OF ELEMENTS: examples of major processes Physical exchange, redox chemistry, biochemistry are involved Surface reservoirs
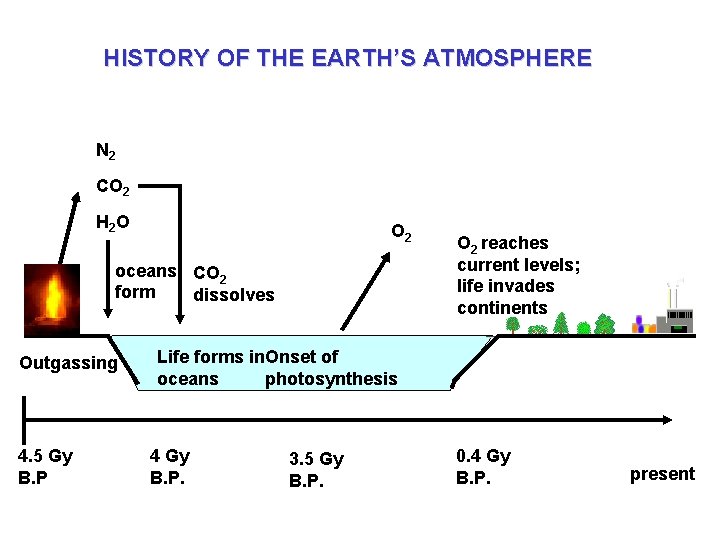
HISTORY OF THE EARTH’S ATMOSPHERE N 2 CO 2 H 2 O O 2 oceans CO 2 form dissolves Outgassing 4. 5 Gy B. P O 2 reaches current levels; life invades continents Life forms in. Onset of oceans photosynthesis 4 Gy B. P. 3. 5 Gy B. P. 0. 4 Gy B. P. present
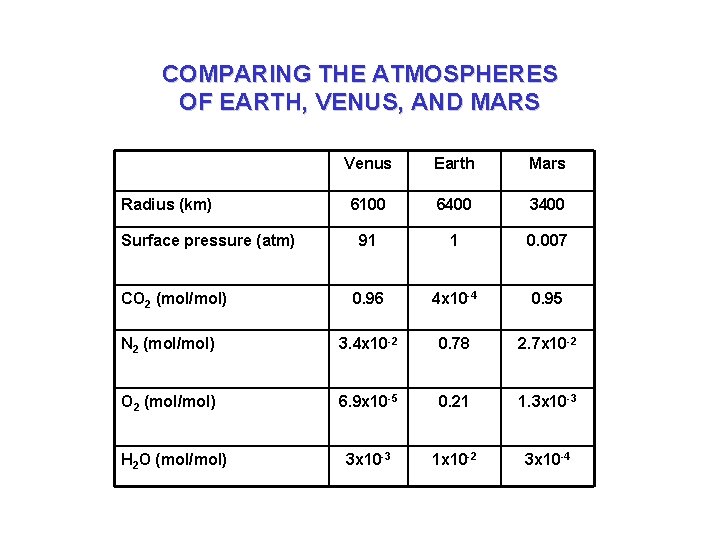
COMPARING THE ATMOSPHERES OF EARTH, VENUS, AND MARS Venus Earth Mars 6100 6400 3400 91 1 0. 007 0. 96 4 x 10 -4 0. 95 N 2 (mol/mol) 3. 4 x 10 -2 0. 78 2. 7 x 10 -2 O 2 (mol/mol) 6. 9 x 10 -5 0. 21 1. 3 x 10 -3 1 x 10 -2 3 x 10 -4 Radius (km) Surface pressure (atm) CO 2 (mol/mol) H 2 O (mol/mol)
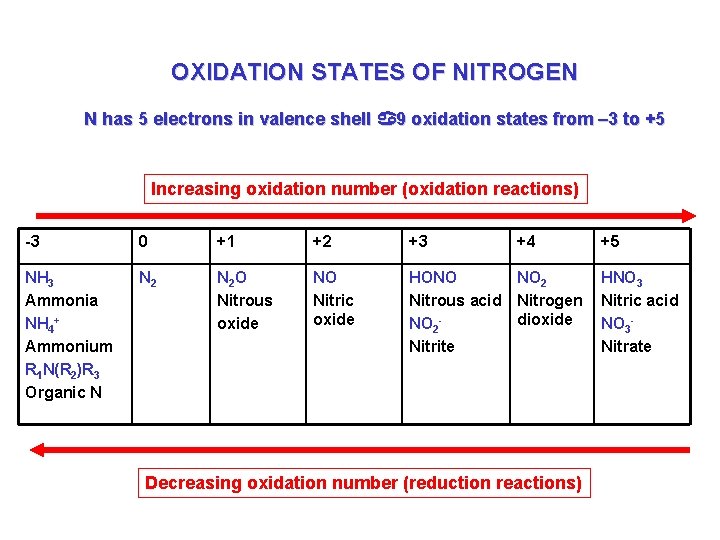
OXIDATION STATES OF NITROGEN N has 5 electrons in valence shell a 9 oxidation states from – 3 to +5 Increasing oxidation number (oxidation reactions) -3 0 +1 +2 +3 +4 NH 3 Ammonia NH 4+ Ammonium R 1 N(R 2)R 3 Organic N N 2 O Nitrous oxide NO Nitric oxide HONO NO 2 Nitrous acid Nitrogen dioxide NO 2 Nitrite Decreasing oxidation number (reduction reactions) +5 HNO 3 Nitric acid NO 3 Nitrate
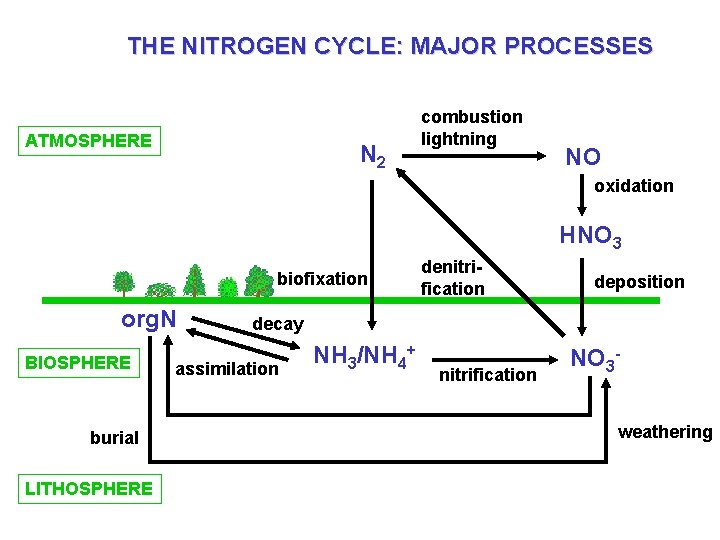
THE NITROGEN CYCLE: MAJOR PROCESSES ATMOSPHERE N 2 combustion lightning NO oxidation HNO 3 biofixation org. N BIOSPHERE burial LITHOSPHERE denitrification deposition decay assimilation NH 3/NH 4+ nitrification NO 3 weathering
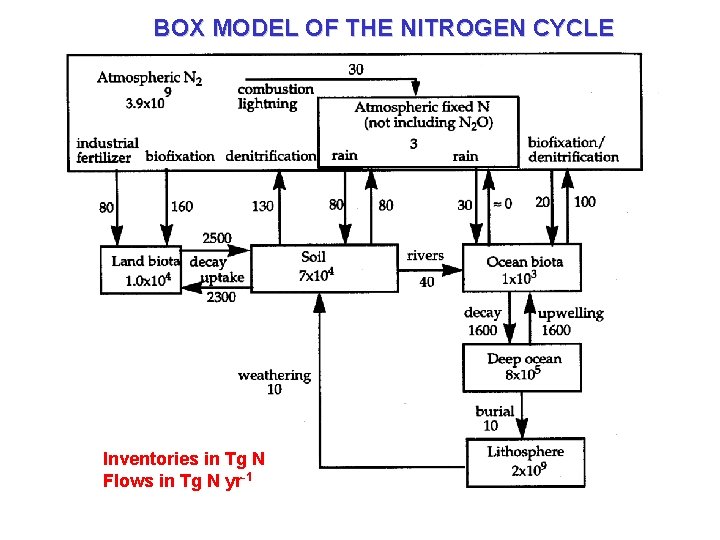
BOX MODEL OF THE NITROGEN CYCLE Inventories in Tg N Flows in Tg N yr-1
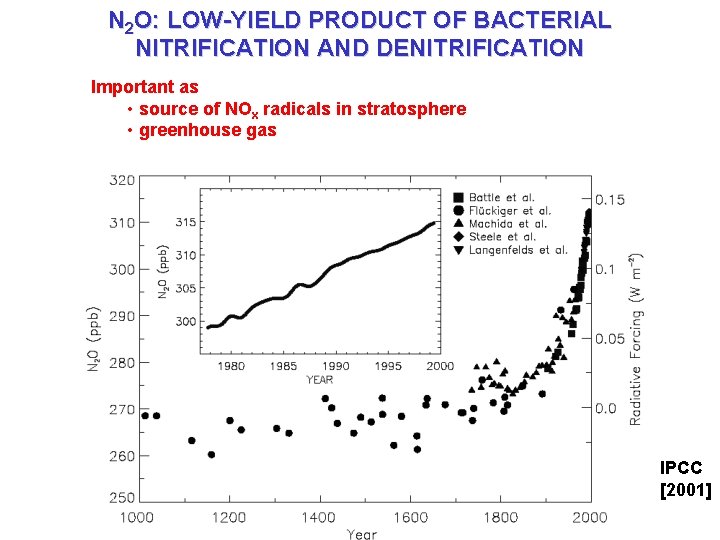
N 2 O: LOW-YIELD PRODUCT OF BACTERIAL NITRIFICATION AND DENITRIFICATION Important as • source of NOx radicals in stratosphere • greenhouse gas IPCC [2001]
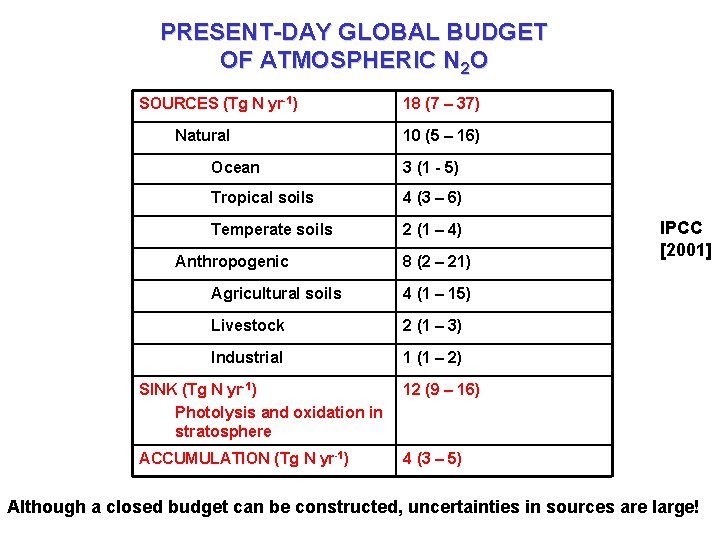
PRESENT-DAY GLOBAL BUDGET OF ATMOSPHERIC N 2 O SOURCES (Tg N yr-1) Natural 18 (7 – 37) 10 (5 – 16) Ocean 3 (1 - 5) Tropical soils 4 (3 – 6) Temperate soils 2 (1 – 4) Anthropogenic 8 (2 – 21) Agricultural soils 4 (1 – 15) Livestock 2 (1 – 3) Industrial 1 (1 – 2) SINK (Tg N yr-1) Photolysis and oxidation in stratosphere 12 (9 – 16) ACCUMULATION (Tg N yr-1) 4 (3 – 5) IPCC [2001] Although a closed budget can be constructed, uncertainties in sources are large!
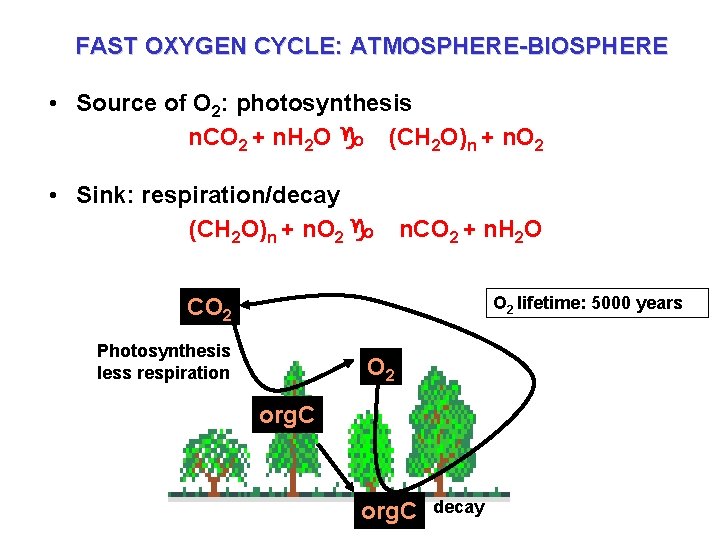
FAST OXYGEN CYCLE: ATMOSPHERE-BIOSPHERE • Source of O 2: photosynthesis n. CO 2 + n. H 2 O g (CH 2 O)n + n. O 2 • Sink: respiration/decay (CH 2 O)n + n. O 2 g n. CO 2 + n. H 2 O O 2 lifetime: 5000 years CO 2 Photosynthesis less respiration O 2 org. C litter org. C decay
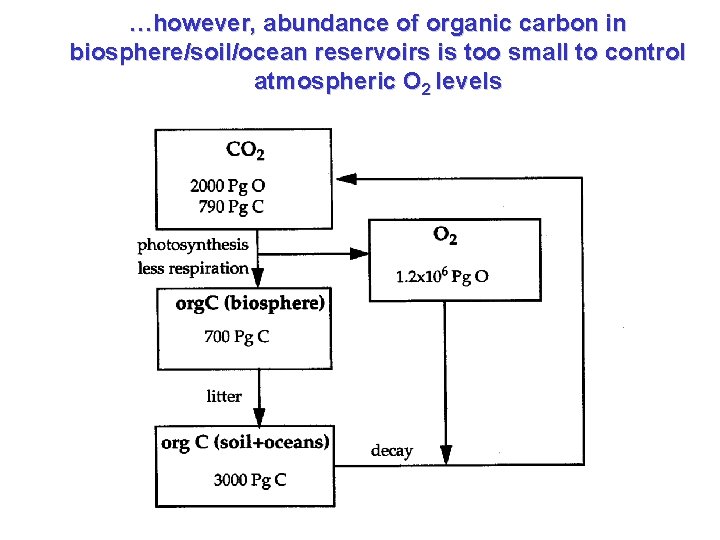
…however, abundance of organic carbon in biosphere/soil/ocean reservoirs is too small to control atmospheric O 2 levels
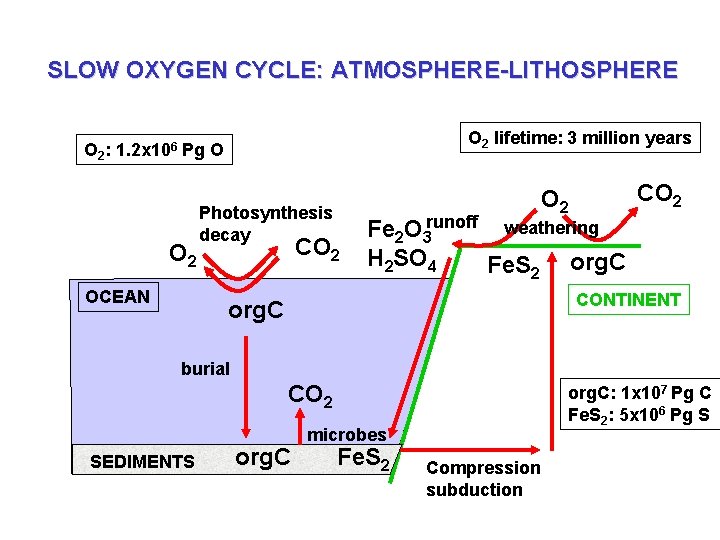
SLOW OXYGEN CYCLE: ATMOSPHERE-LITHOSPHERE O 2 lifetime: 3 million years O 2: 1. 2 x 106 Pg O O 2 OCEAN Photosynthesis decay CO 2 Fe 2 O 3 runoff weathering H 2 SO 4 Fe. S 2 org. C CONTINENT org. C Uplift burial CO 2 SEDIMENTS CO 2 org. C: 1 x 107 Pg C Fe. S 2: 5 x 106 Pg S microbes Fe. S 2 Compression subduction
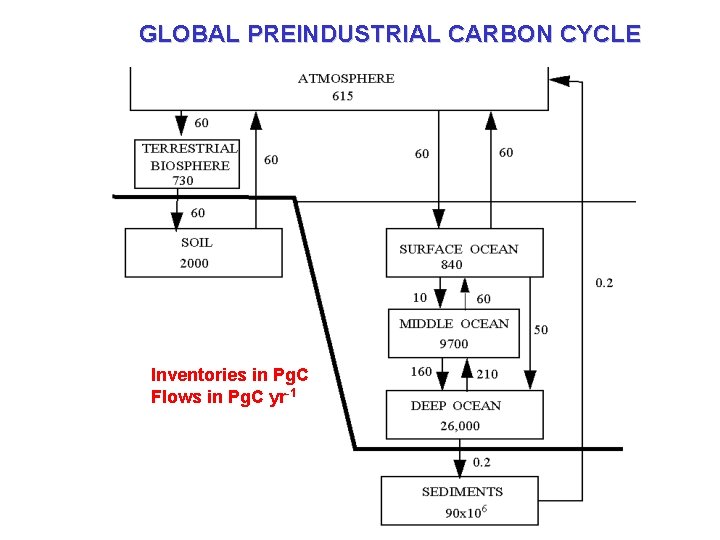
GLOBAL PREINDUSTRIAL CARBON CYCLE Inventories in Pg. C Flows in Pg. C yr-1
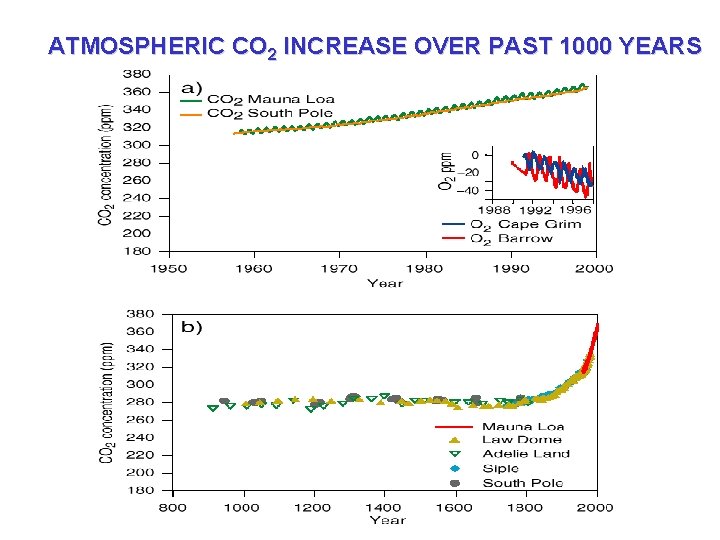
ATMOSPHERIC CO 2 INCREASE OVER PAST 1000 YEARS
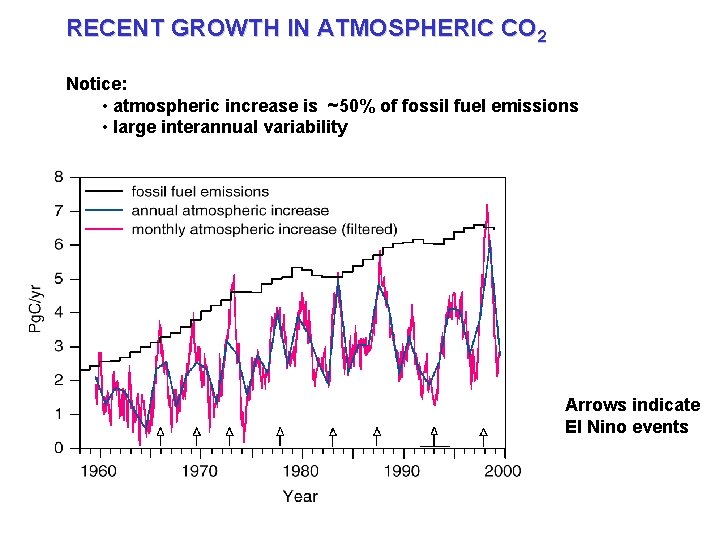
RECENT GROWTH IN ATMOSPHERIC CO 2 Notice: • atmospheric increase is ~50% of fossil fuel emissions • large interannual variability Arrows indicate El Nino events
![GLOBAL CO 2 BUDGET (Pg C yr-1) IPCC [2001] GLOBAL CO 2 BUDGET (Pg C yr-1) IPCC [2001]](http://slidetodoc.com/presentation_image_h2/5c5f59851ebbfb1e2b5a67ae206e1ade/image-17.jpg)
GLOBAL CO 2 BUDGET (Pg C yr-1) IPCC [2001]
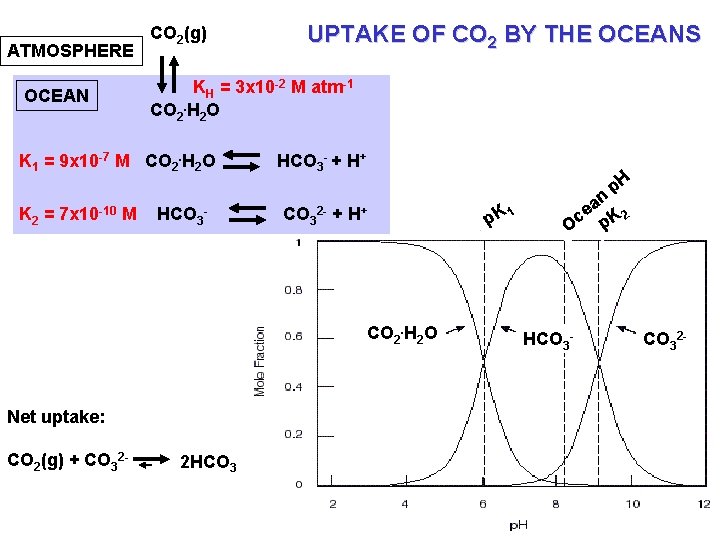
ATMOSPHERE CO 2(g) KH = 3 x 10 -2 M atm-1 CO 2. H 2 O OCEAN K 1 = 9 x 10 -7 M CO 2. H 2 O K 2 = 7 x 10 -10 UPTAKE OF CO 2 BY THE OCEANS M HCO 3 - HCO 3 - + H+ CO 3 + 2 - p. H H+ CO 2. H 2 O Net uptake: CO 2(g) + CO 32 - 2 HCO 3 - p K 1 an e c p. K 2 O HCO 3 - CO 32 -

EQUILIBRIUM PARTITIONING OF CO 2 BETWEEN ATMOSPHERE AND GLOBAL OCEAN Equilibrium for present-day ocean: varies roughly as [H+] [ only 3% of total inorganic carbon is in the atmosphere But CO 2(g) k [ [H+] k [ F k … positive feedback to increasing CO 2 Pose problem differently: how does a CO 2 addition d. N partition between the atmosphere and ocean at equilibrium? varies roughly as [H+]2 [ 28% of added CO 2 remains in atmosphere!
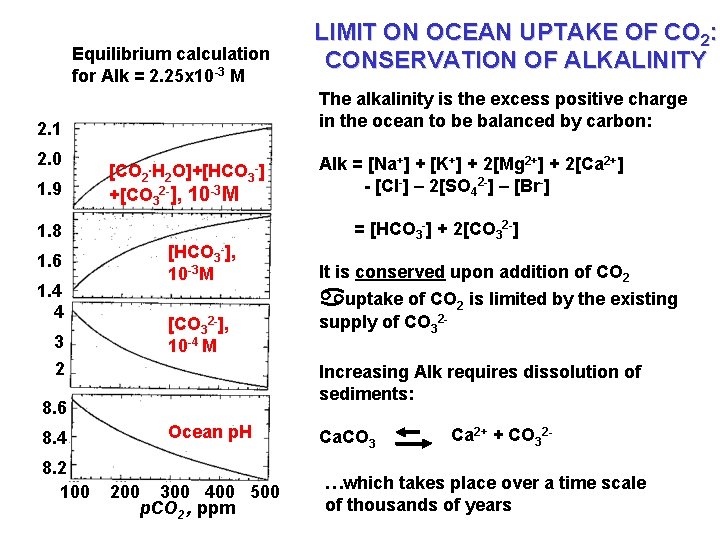
Equilibrium calculation for Alk = 2. 25 x 10 -3 M The alkalinity is the excess positive charge in the ocean to be balanced by carbon: 2. 1 2. 0 1. 9 [CO 2. H 2 O]+[HCO 3 -] +[CO 32 -], 10 -3 M 1. 8 1. 6 1. 4 4 3 [HCO 3 -], 10 -3 M [CO 32 -], 10 -4 M = [HCO 3 -] + 2[CO 32 -] It is conserved upon addition of CO 2 supply of CO 32 - Increasing Alk requires dissolution of sediments: 8. 6 8. 2 100 Alk = [Na+] + [K+] + 2[Mg 2+] + 2[Ca 2+] - [Cl-] – 2[SO 42 -] – [Br-] auptake of CO 2 is limited by the existing 2 8. 4 LIMIT ON OCEAN UPTAKE OF CO 2: CONSERVATION OF ALKALINITY Ocean p. H 200 300 400 500 p. CO 2 , ppm Ca. CO 3 Ca 2+ + CO 32 - …which takes place over a time scale of thousands of years
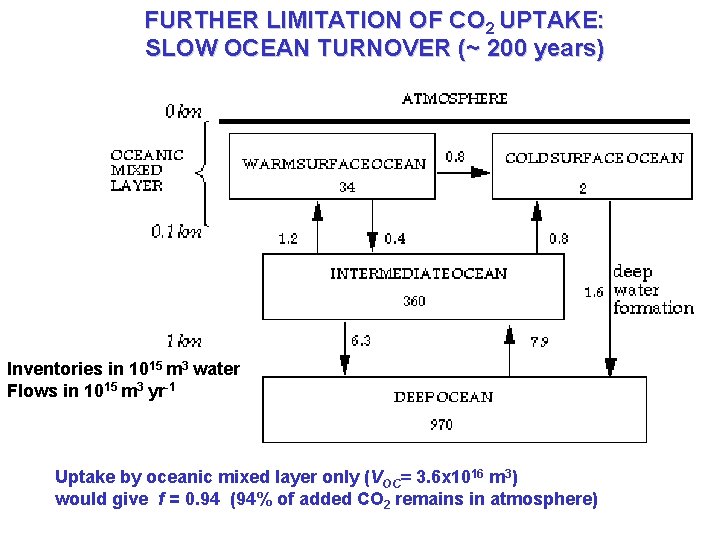
FURTHER LIMITATION OF CO 2 UPTAKE: SLOW OCEAN TURNOVER (~ 200 years) Inventories in 1015 m 3 water Flows in 1015 m 3 yr-1 Uptake by oceanic mixed layer only (VOC= 3. 6 x 1016 m 3) would give f = 0. 94 (94% of added CO 2 remains in atmosphere)
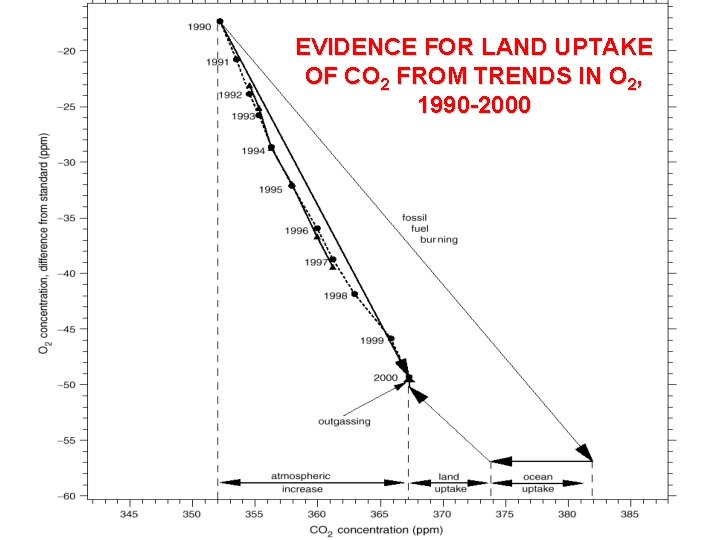
EVIDENCE FOR LAND UPTAKE OF CO 2 FROM TRENDS IN O 2, 1990 -2000
![GLOBAL CO 2 BUDGET (Pg C yr-1) IPCC [2001] GLOBAL CO 2 BUDGET (Pg C yr-1) IPCC [2001]](http://slidetodoc.com/presentation_image_h2/5c5f59851ebbfb1e2b5a67ae206e1ade/image-23.jpg)
GLOBAL CO 2 BUDGET (Pg C yr-1) IPCC [2001]

NET UPTAKE OF CO 2 BY TERRESTRIAL BIOSPHERE (1. 4 Pg C yr-1 in the 1990 s; IPCC [2001]) is a small residual of large atm-bio exchange • Gross primary production (GPP): GPP = CO 2 uptake by photosynthesis = 120 Pg. C yr-1 • Net primary production (NPP): NPP = GPP – “autotrophic” respiration by green plants = 60 Pg. C yr-1 • Net ecosystem production (NEP): NEP = NPP – “heterotrophic” respiration by decomposers = 10 Pg. C yr-1 • Net biome production (NBP) NBP = NEP – fires/erosion/harvesting = 1. 4 Pg. C yr-1 Atmospheric CO 2 observations show that the net uptake is at northern midlatitudes but cannot resolve American vs. Eurasian contributions
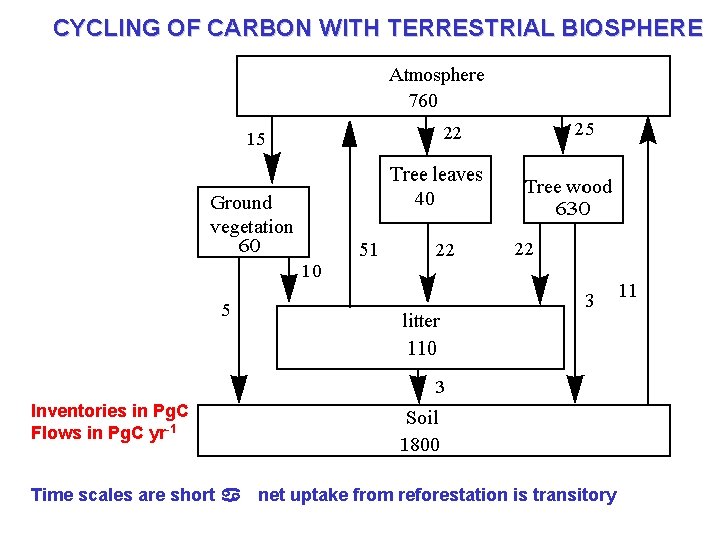
CYCLING OF CARBON WITH TERRESTRIAL BIOSPHERE Inventories in Pg. C Flows in Pg. C yr-1 Time scales are short a net uptake from reforestation is transitory
![PROJECTED FUTURE TRENDS IN CO 2 UPTAKE BY OCEANS AND TERRESTRIAL BIOSPHERE IPCC [2001] PROJECTED FUTURE TRENDS IN CO 2 UPTAKE BY OCEANS AND TERRESTRIAL BIOSPHERE IPCC [2001]](http://slidetodoc.com/presentation_image_h2/5c5f59851ebbfb1e2b5a67ae206e1ade/image-26.jpg)
PROJECTED FUTURE TRENDS IN CO 2 UPTAKE BY OCEANS AND TERRESTRIAL BIOSPHERE IPCC [2001]
![PROJECTIONS OF FUTURE CO 2 CONCENTRATIONS [IPCC, 2001] PROJECTIONS OF FUTURE CO 2 CONCENTRATIONS [IPCC, 2001]](http://slidetodoc.com/presentation_image_h2/5c5f59851ebbfb1e2b5a67ae206e1ade/image-27.jpg)
PROJECTIONS OF FUTURE CO 2 CONCENTRATIONS [IPCC, 2001]

OXIDATION STATES OF SULFUR S has 6 electrons in valence shell a oxidation states from – 2 to +6 Increasing oxidation number (oxidation reactions) -2 +4 +6 Fe. S 2 Pyrite H 2 S Hydrogen sulfide (CH 3)2 S Dimethylsulfide (DMS) CS 2 Carbon disulfide COS Carbonyl sulfide SO 2 Sulfur dioxide H 2 SO 4 Sulfuric acid SO 42 Sulfate Decreasing oxidation number (reduction reactions)
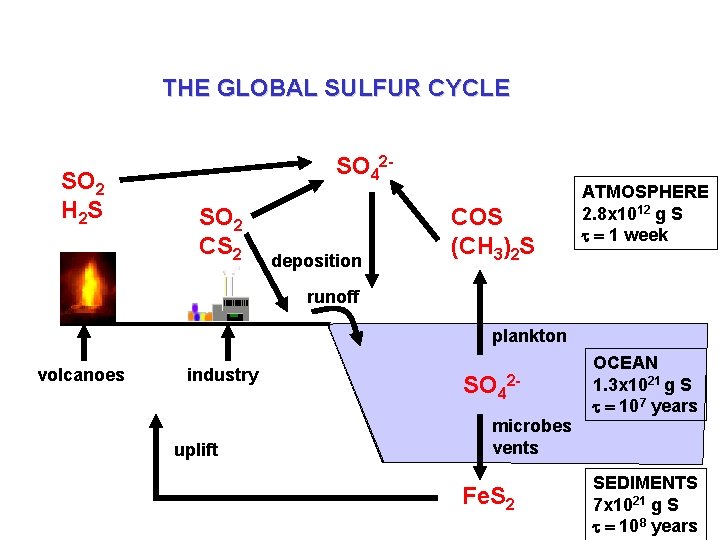
THE GLOBAL SULFUR CYCLE SO 2 H 2 S SO 42 SO 2 CS 2 deposition COS (CH 3)2 S ATMOSPHERE 2. 8 x 1012 g S t = 1 week runoff plankton volcanoes industry uplift SO 42 microbes vents Fe. S 2 OCEAN 1. 3 x 1021 g S t = 107 years SEDIMENTS 7 x 1021 g S t = 108 years
![GLOBAL SULFUR EMISSION TO THE ATMOSPHERE 1990 annual mean Chin et al. [2000] GLOBAL SULFUR EMISSION TO THE ATMOSPHERE 1990 annual mean Chin et al. [2000]](http://slidetodoc.com/presentation_image_h2/5c5f59851ebbfb1e2b5a67ae206e1ade/image-30.jpg)
GLOBAL SULFUR EMISSION TO THE ATMOSPHERE 1990 annual mean Chin et al. [2000]

FURTHER READING • Jacob, D. J. , Introduction to Atmospheric Chemistry, Princeton University Press, 1999. …don’t leave home without it! • Warneck, P. , Chemistry of the Natural Atmosphere, 2 nd ed. , Academic Press, 1999 Great chapters on biogeochemical cycles • Intergovernmental Panel on Climate Change (IPCC), Climate Change 2001: The Scientific Basis, 2001 The latest on the carbon cycle
- Slides: 31