GLI EVENTI CRITICI DELLA MITOSI SONO PRINCIPALMENTE RIFERIBILI
GLI EVENTI CRITICI DELLA MITOSI SONO PRINCIPALMENTE RIFERIBILI ALLE DINAMICHE DEL DNA E DEI MICROTUBULI

I centrioli
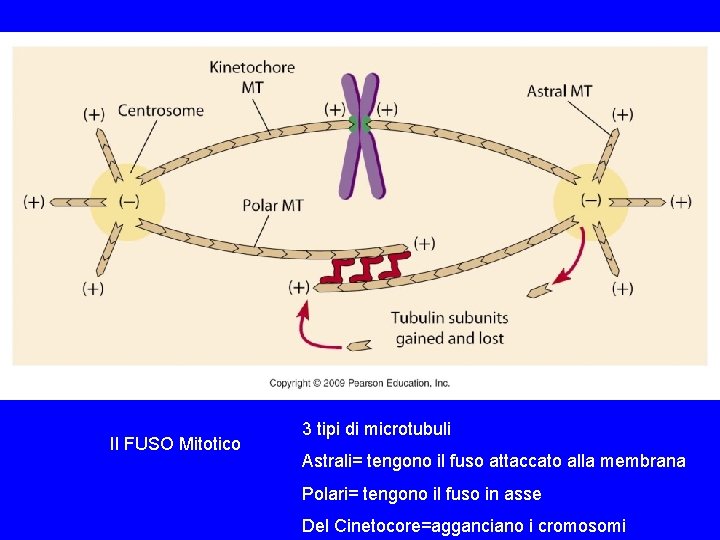
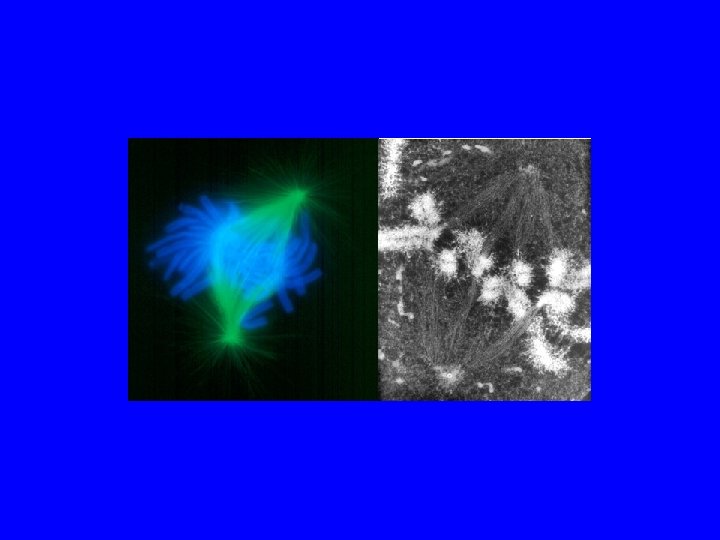
Il FUSO Mitotico 3 tipi di microtubuli Astrali= tengono il fuso attaccato alla membrana Polari= tengono il fuso in asse Del Cinetocore=agganciano i cromosomi
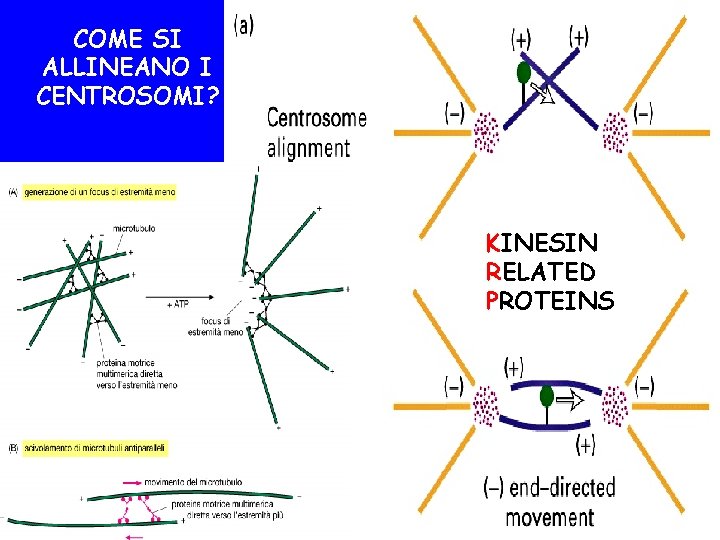
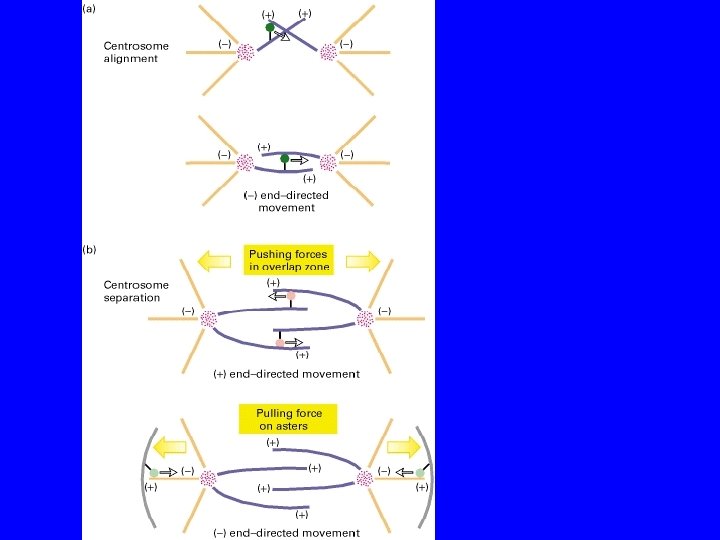
COME SI ALLINEANO I CENTROSOMI? KINESIN RELATED PROTEINS
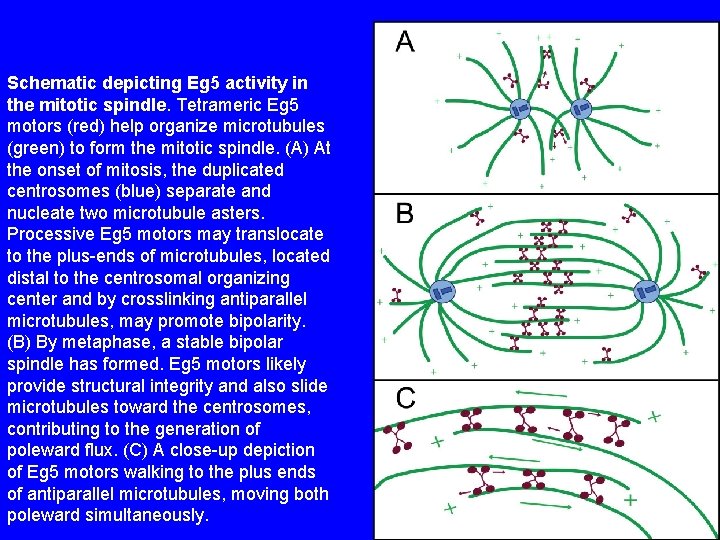
Schematic depicting Eg 5 activity in the mitotic spindle. Tetrameric Eg 5 motors (red) help organize microtubules (green) to form the mitotic spindle. (A) At the onset of mitosis, the duplicated centrosomes (blue) separate and nucleate two microtubule asters. Processive Eg 5 motors may translocate to the plus-ends of microtubules, located distal to the centrosomal organizing center and by crosslinking antiparallel microtubules, may promote bipolarity. (B) By metaphase, a stable bipolar spindle has formed. Eg 5 motors likely provide structural integrity and also slide microtubules toward the centrosomes, contributing to the generation of poleward flux. (C) A close-up depiction of Eg 5 motors walking to the plus ends of antiparallel microtubules, moving both poleward simultaneously.
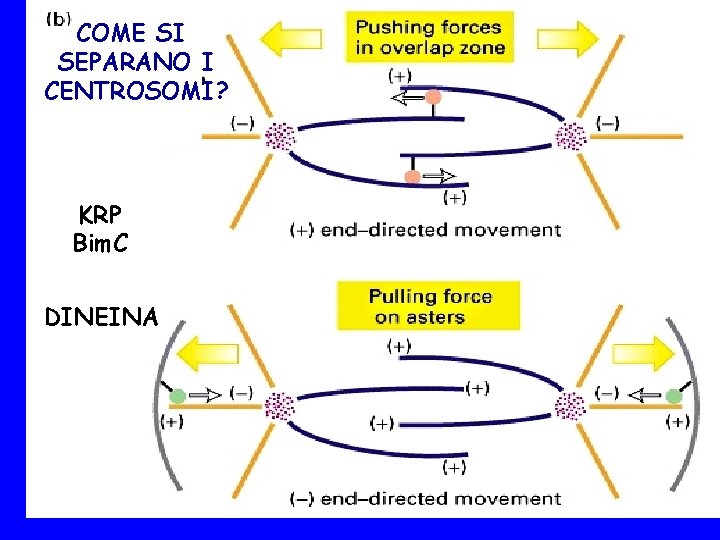
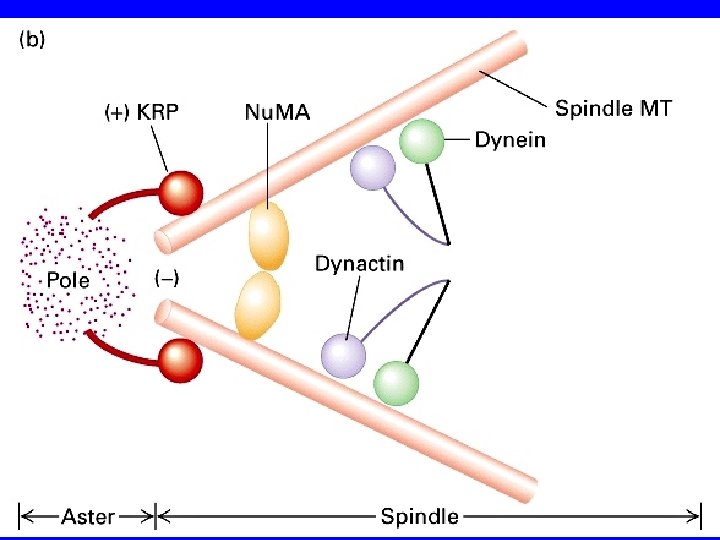
COME SI SEPARANO I CENTROSOMI? KRP Bim. C DINEINA
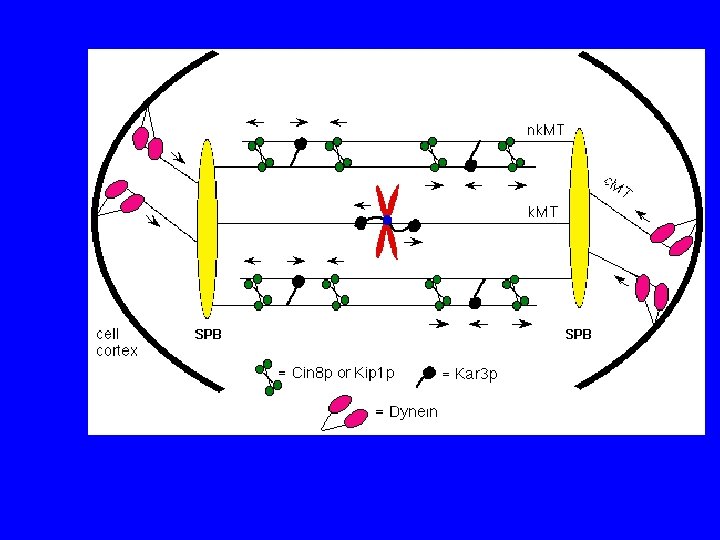
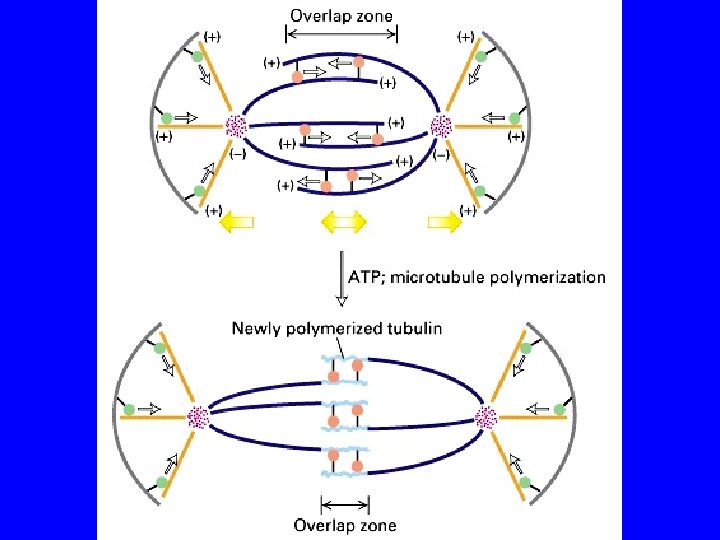
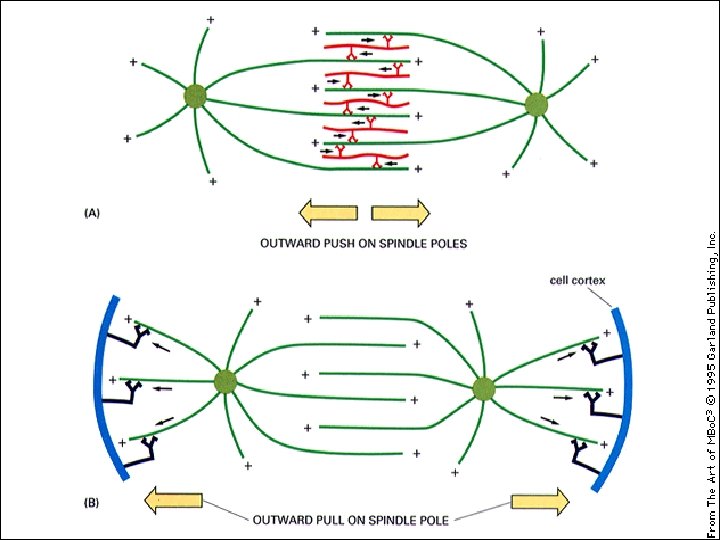

I motori proteici
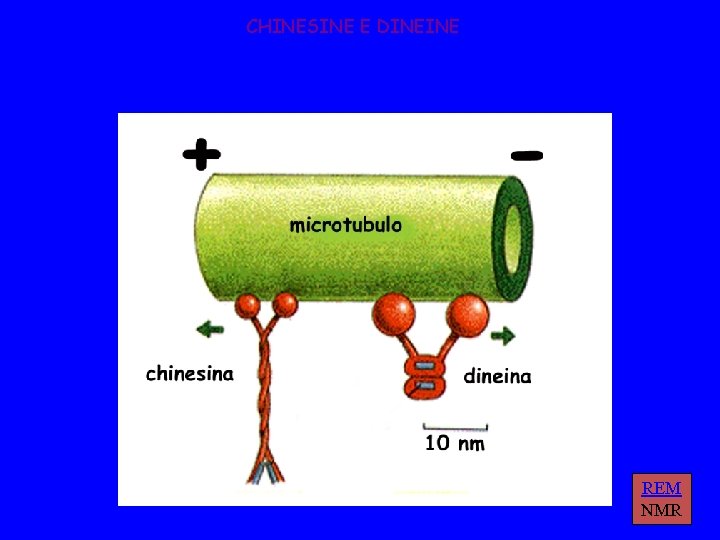
CHINESINE E DINEINE REM NMR

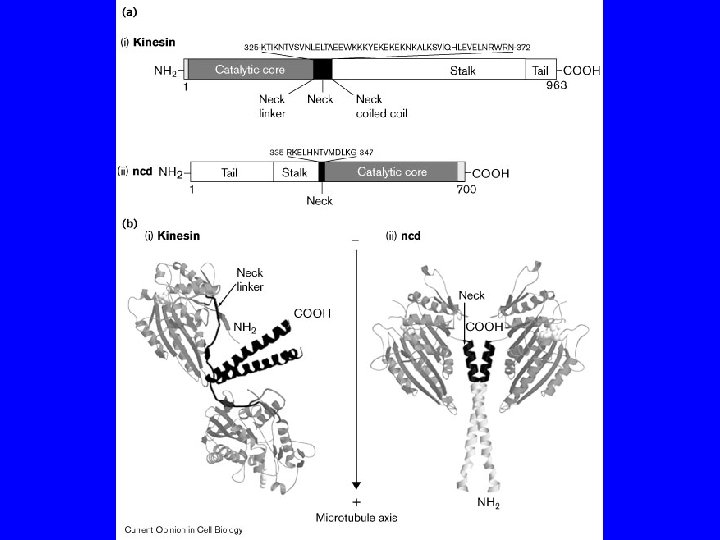
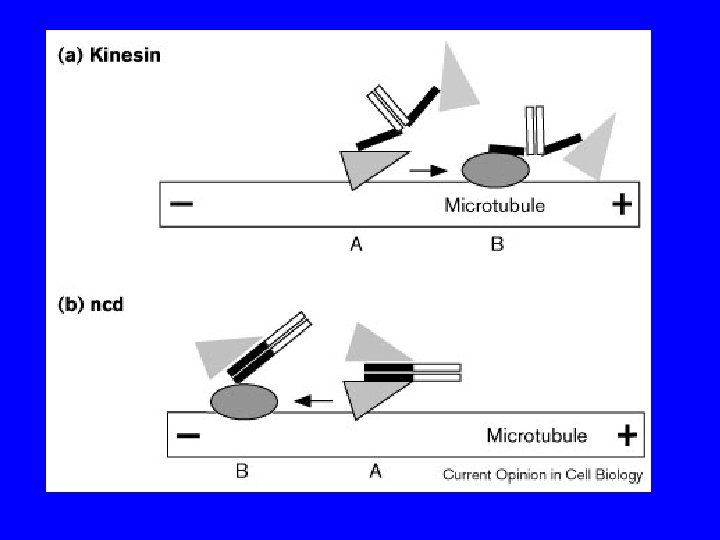
PROPRIETA’ DELLE CHINESINE passo = 8 nm forza = 6 p. N 3 tipi: N - motore N-terminale M - motore centrale C - motore C terminale 2 classi: >>> estremità + >>> estremità - chinesine citosoliche KIF 1 A, KIF 1 B chinesine del fuso CENP-E, ncd, Bim. C
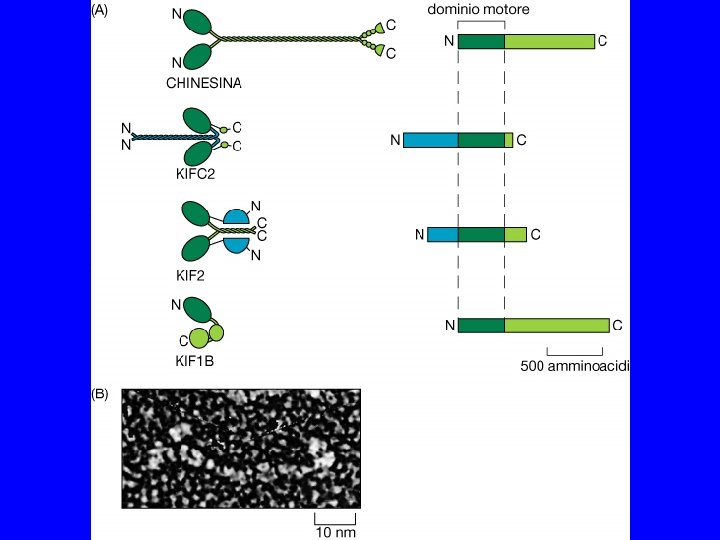

PROPRIETA’ DELLE DINEINE peso molecolare elevato funzionano insieme a MBP 2 classi: dineine citosoliche dineine dell’assonema
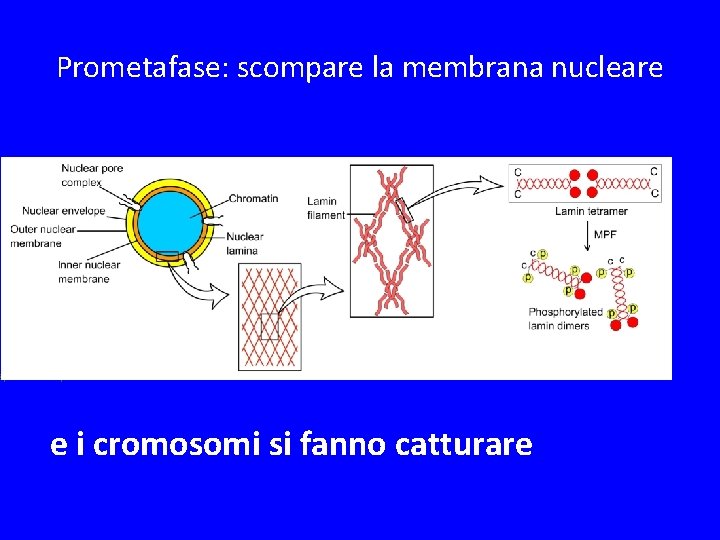
Prometafase: scompare la membrana nucleare Spindle MTs capture chromosomes e i cromosomi si fanno catturare
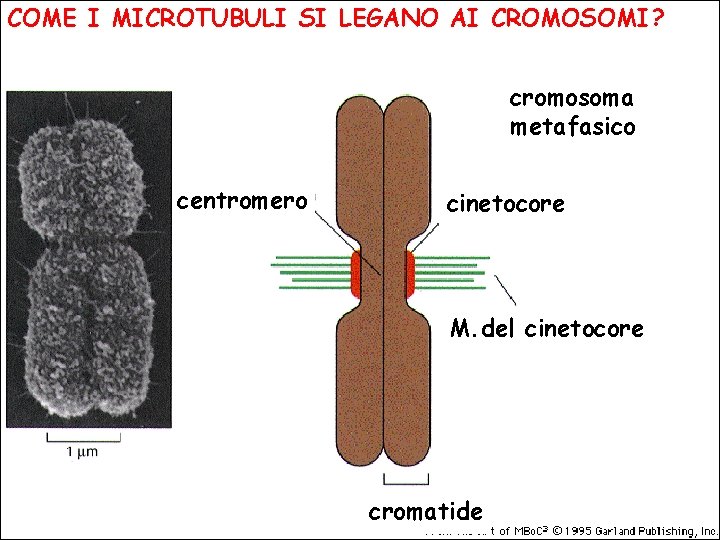
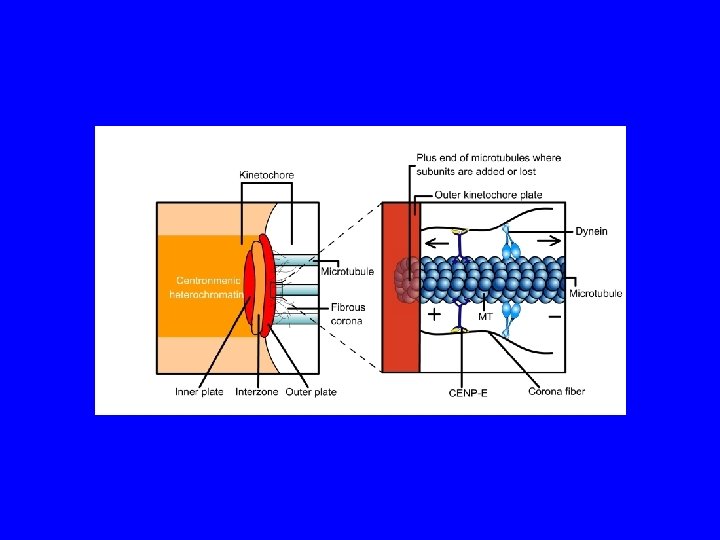
COME I MICROTUBULI SI LEGANO AI CROMOSOMI? cromosoma metafasico centromero cinetocore M. del cinetocore cromatide
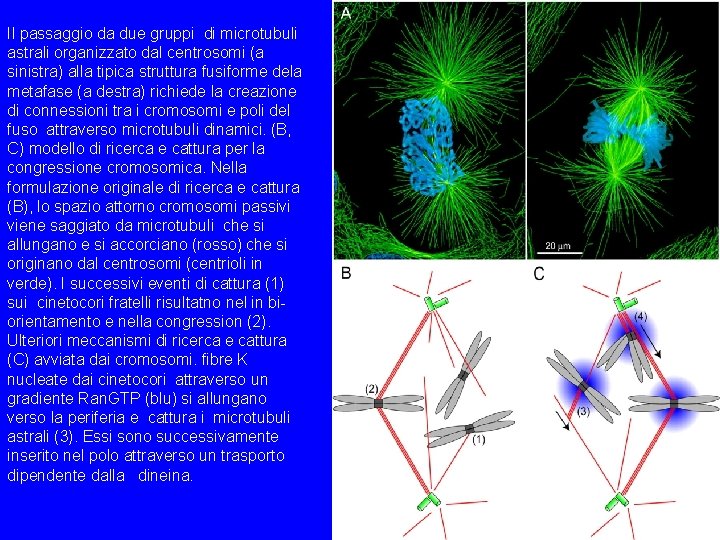
Il passaggio da due gruppi di microtubuli astrali organizzato dal centrosomi (a sinistra) alla tipica struttura fusiforme dela metafase (a destra) richiede la creazione di connessioni tra i cromosomi e poli del fuso attraverso microtubuli dinamici. (B, C) modello di ricerca e cattura per la congressione cromosomica. Nella formulazione originale di ricerca e cattura (B), lo spazio attorno cromosomi passivi viene saggiato da microtubuli che si allungano e si accorciano (rosso) che si originano dal centrosomi (centrioli in verde). I successivi eventi di cattura (1) sui cinetocori fratelli risultatno nel in biorientamento e nella congression (2). Ulteriori meccanismi di ricerca e cattura (C) avviata dai cromosomi. fibre K nucleate dai cinetocori attraverso un gradiente Ran. GTP (blu) si allungano verso la periferia e cattura i microtubuli astrali (3). Essi sono successivamente inserito nel polo attraverso un trasporto dipendente dalla dineina.
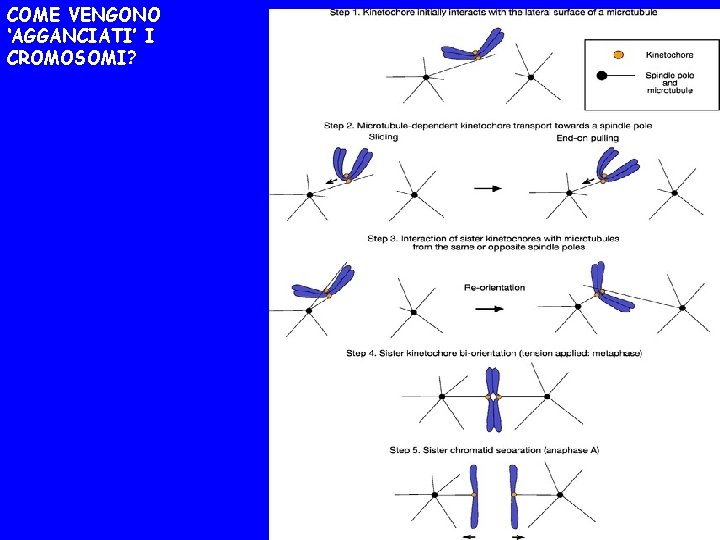
COME VENGONO ‘AGGANCIATI’ I CROMOSOMI?
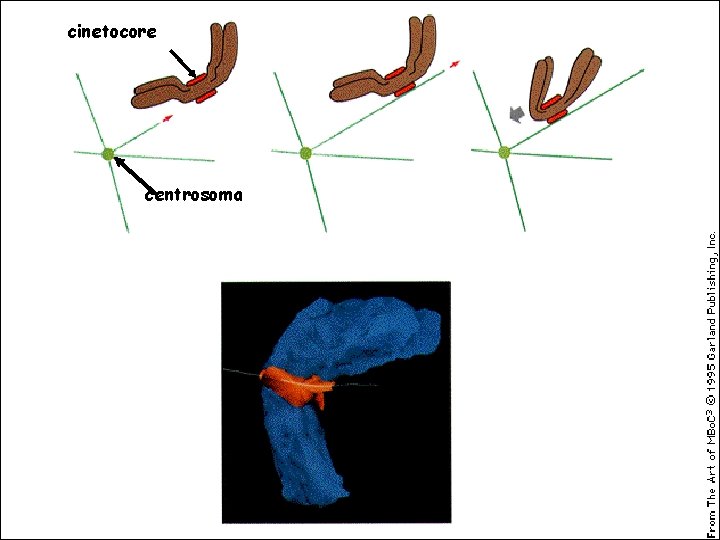
cinetocore centrosoma
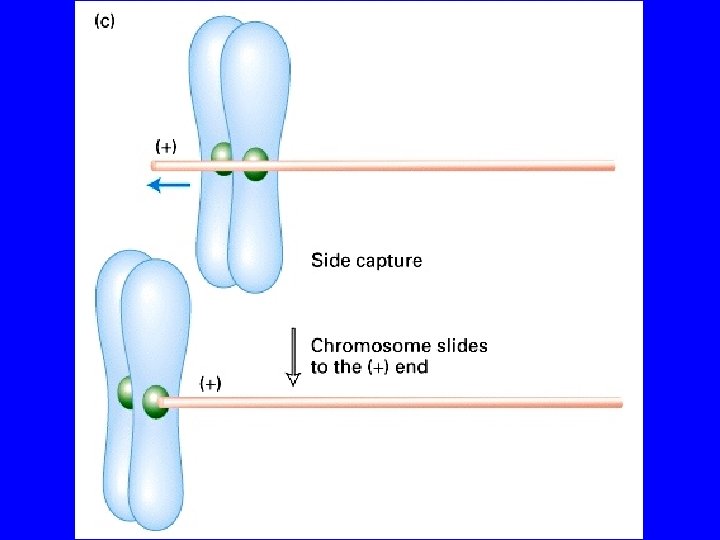

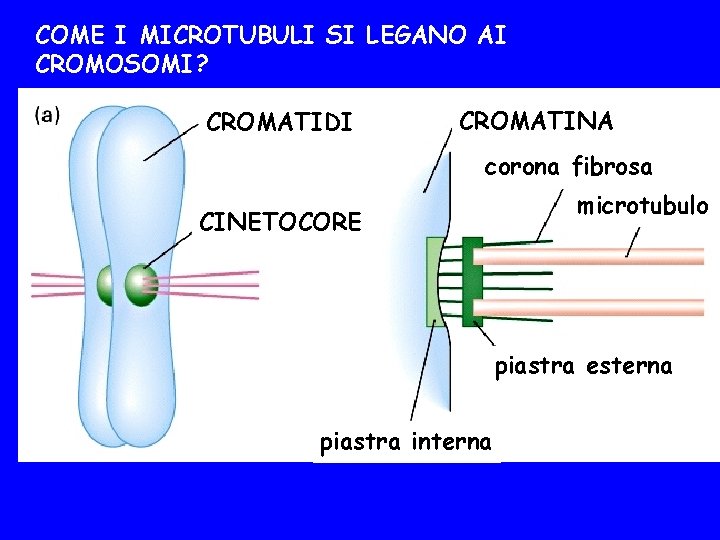
COME I MICROTUBULI SI LEGANO AI CROMOSOMI? CROMATIDI CROMATINA corona fibrosa CINETOCORE microtubulo piastra esterna piastra interna
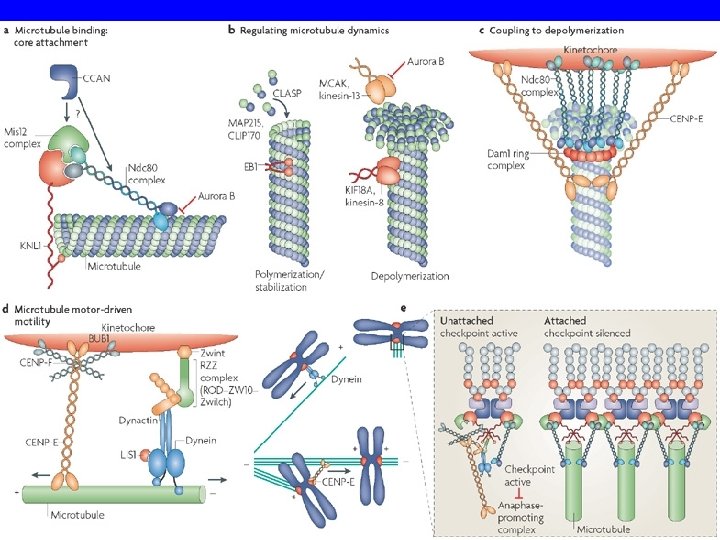

COME SI MUOVONO I CROMOSOMI? • The pushing and pulling forces drive the chromosomes to the metaphase plate • MT behavior during formation of the metaphase plate. Initially, MT from opposite poles are different in length.
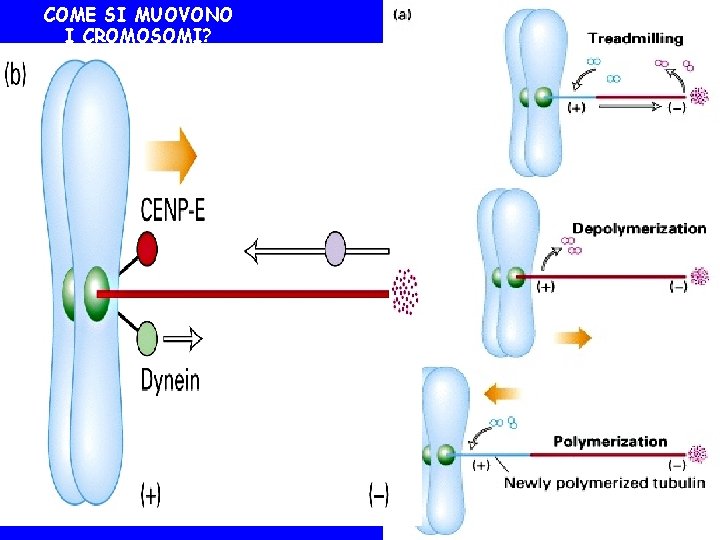
COME SI MUOVONO I CROMOSOMI?
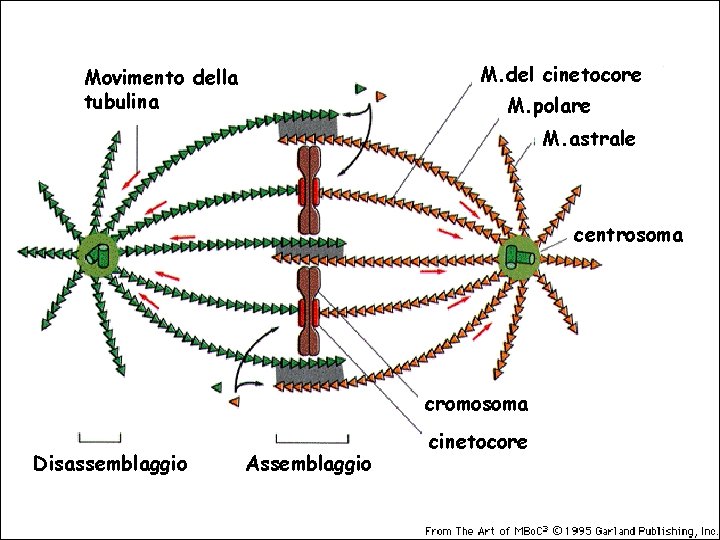
M. del cinetocore Movimento della tubulina M. polare M. astrale centrosoma cromosoma Disassemblaggio Assemblaggio cinetocore
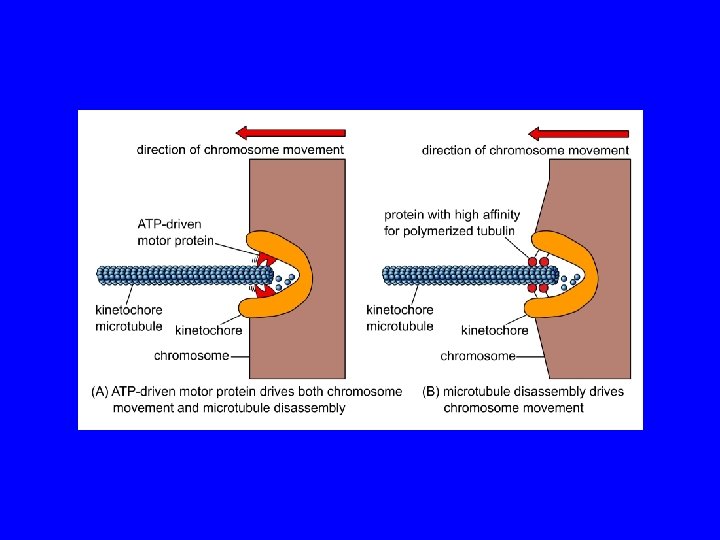
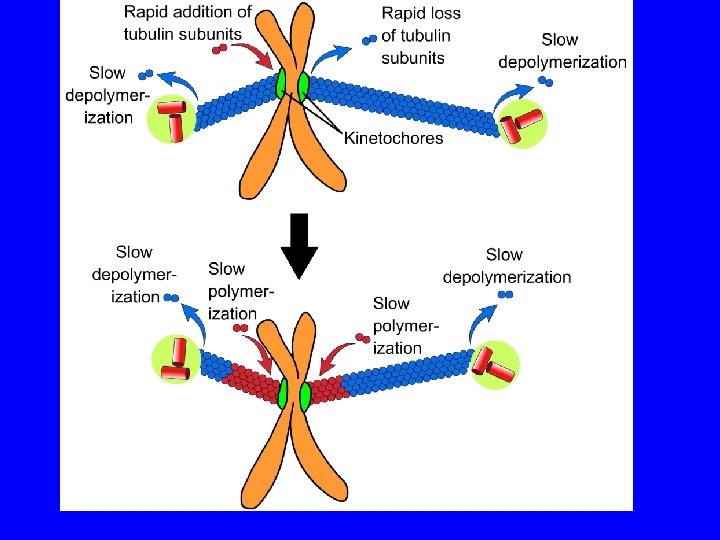
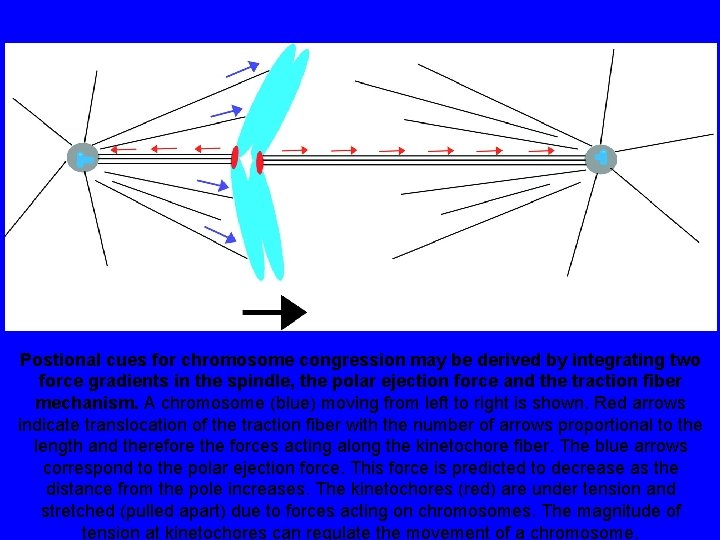
Postional cues for chromosome congression may be derived by integrating two force gradients in the spindle, the polar ejection force and the traction fiber mechanism. A chromosome (blue) moving from left to right is shown. Red arrows indicate translocation of the traction fiber with the number of arrows proportional to the length and therefore the forces acting along the kinetochore fiber. The blue arrows correspond to the polar ejection force. This force is predicted to decrease as the distance from the pole increases. The kinetochores (red) are under tension and stretched (pulled apart) due to forces acting on chromosomes. The magnitude of tension at kinetochores can regulate the movement of a chromosome.
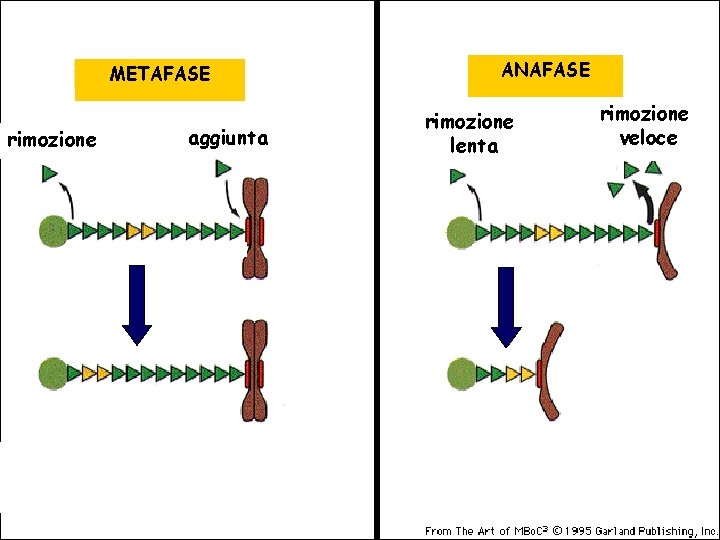
METAFASE rimozione aggiunta ANAFASE rimozione lenta rimozione veloce
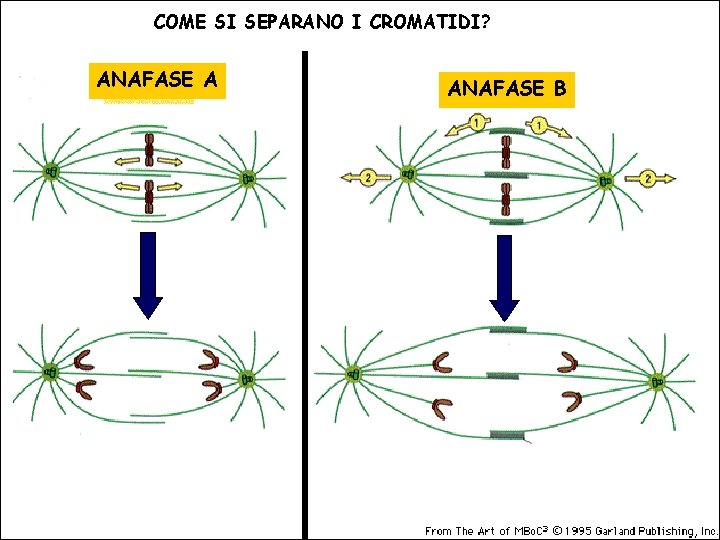
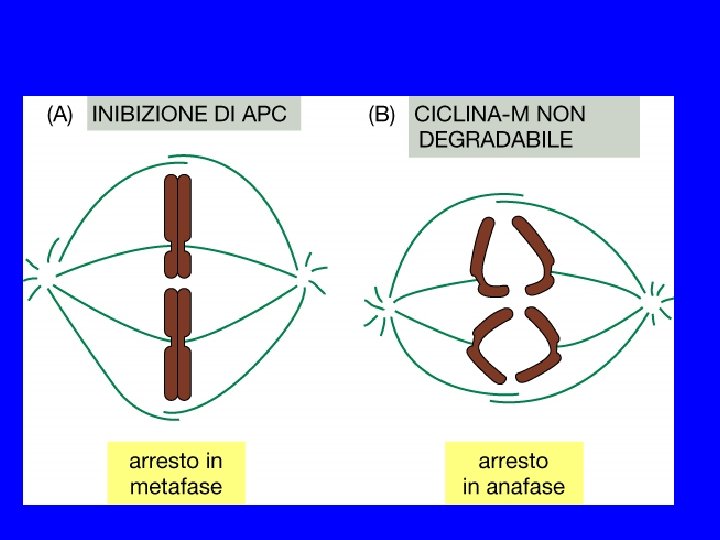
COME SI SEPARANO I CROMATIDI? ANAFASE A ANAFASE B
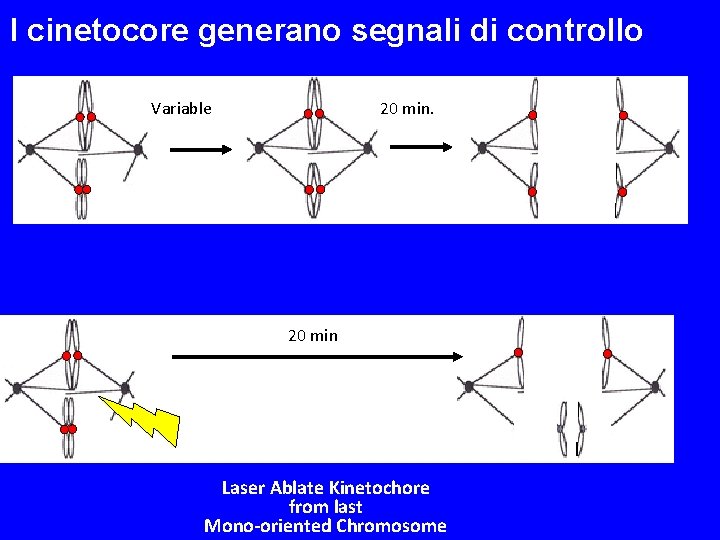
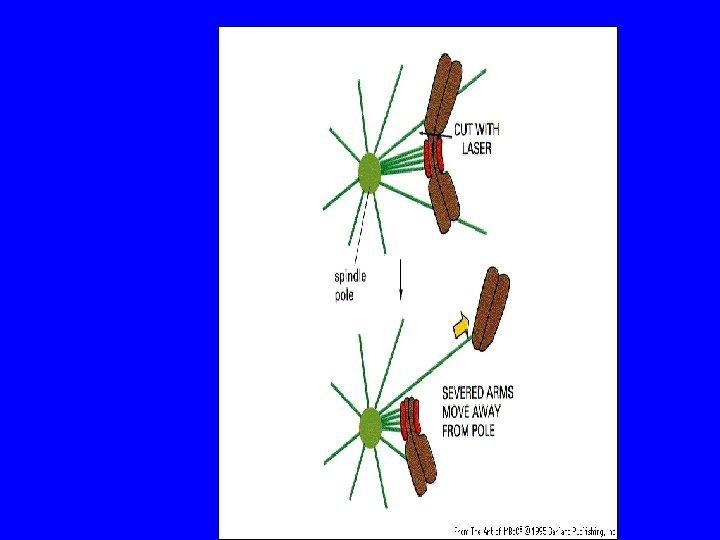
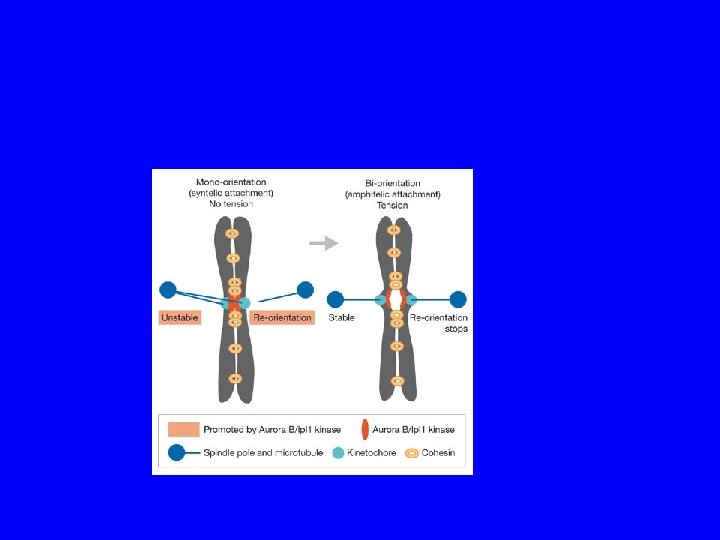
I cinetocore generano segnali di controllo Variable 20 min Laser Ablate Kinetochore from last Mono-oriented Chromosome
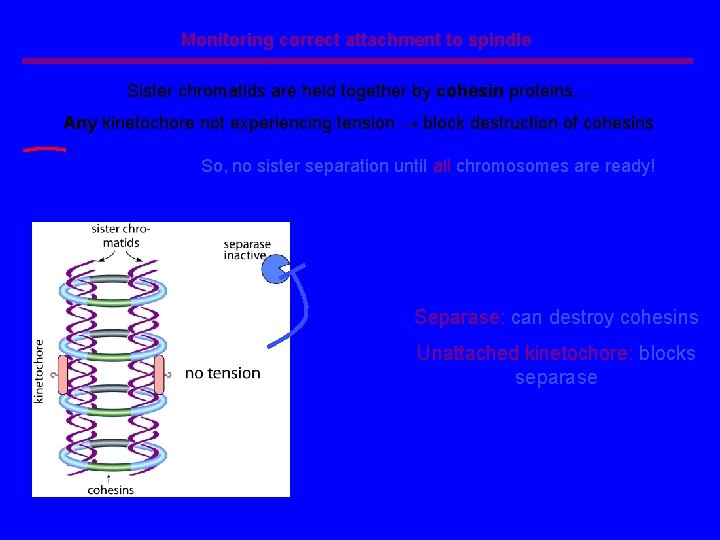
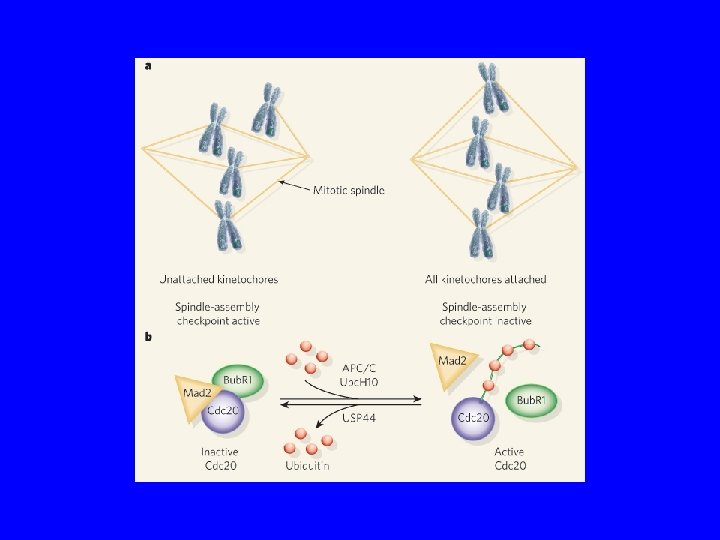
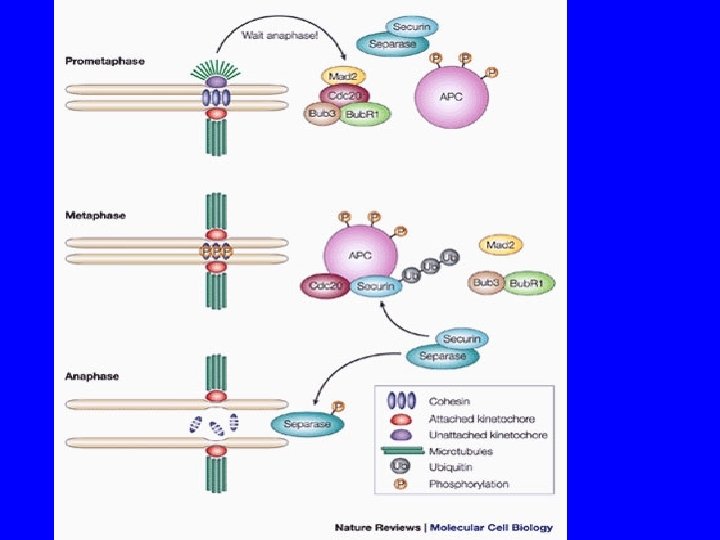
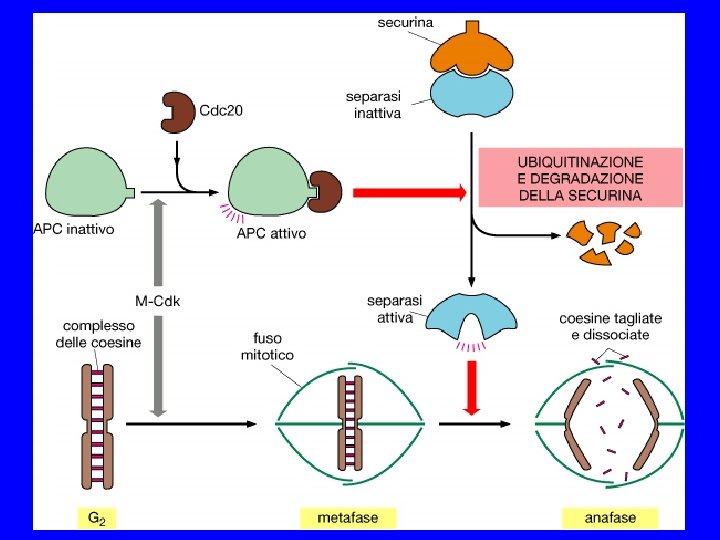
Monitoring correct attachment to spindle Sister chromatids are held together by cohesin proteins… Any kinetochore not experiencing tension block destruction of cohesins So, no sister separation until all chromosomes are ready! Separase: can destroy cohesins Unattached kinetochore: blocks separase
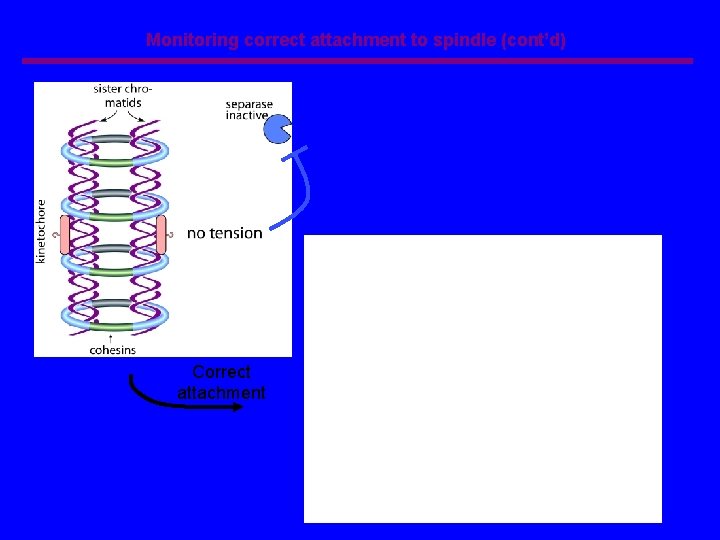
Monitoring correct attachment to spindle (cont’d) Correct attachment
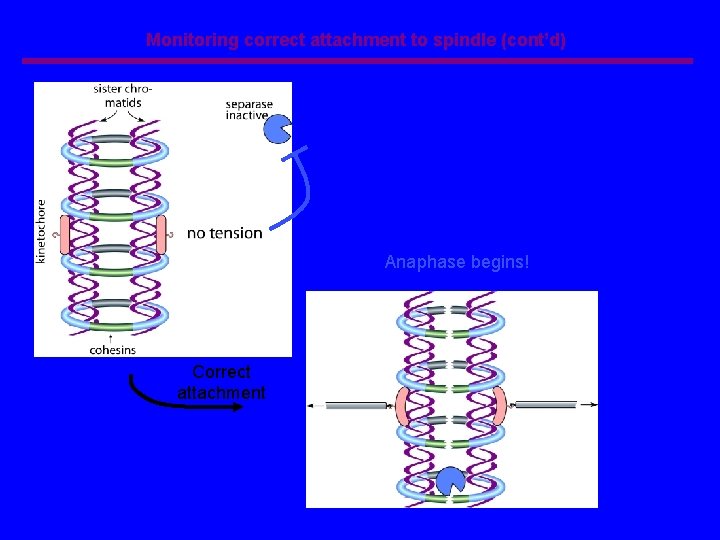
Monitoring correct attachment to spindle (cont’d) Anaphase begins! Correct attachment
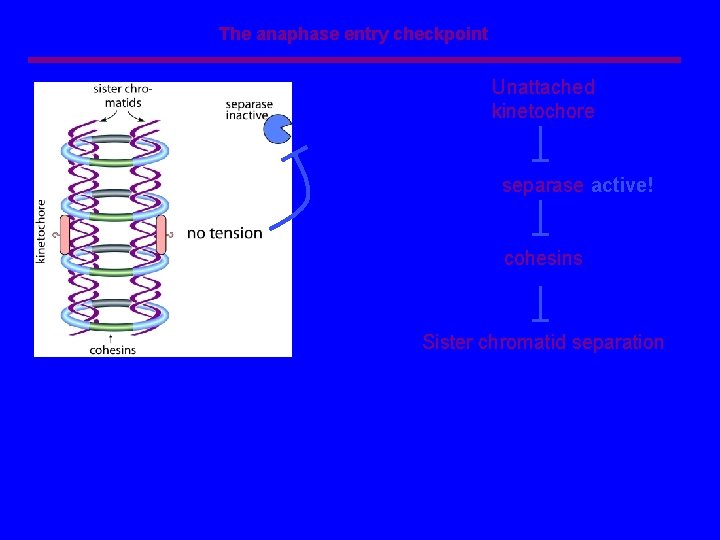
The anaphase entry checkpoint Unattached kinetochore separase active! cohesins Sister chromatid separation
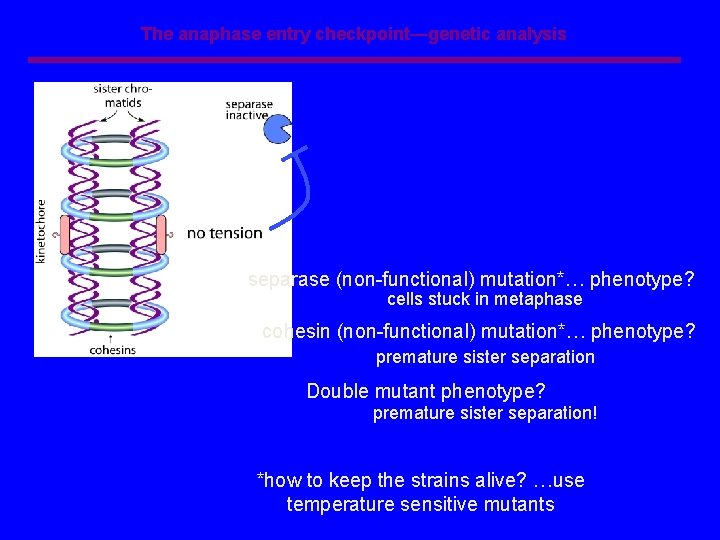
The anaphase entry checkpoint—genetic analysis separase (non-functional) mutation*… phenotype? cells stuck in metaphase cohesin (non-functional) mutation*… phenotype? premature sister separation Double mutant phenotype? premature sister separation! *how to keep the strains alive? …use temperature sensitive mutants

Checkpoints Cellular surveillance systems to monitor the integrity of the genome and of cellular structures Enforce the correct order of execution of cellular events. Examples: - Chromosomes not attached to spindle block onset of anaphase - DNA is damaged halt the cell cycle to allow repair - Irreparable DNA damage trigger cell death
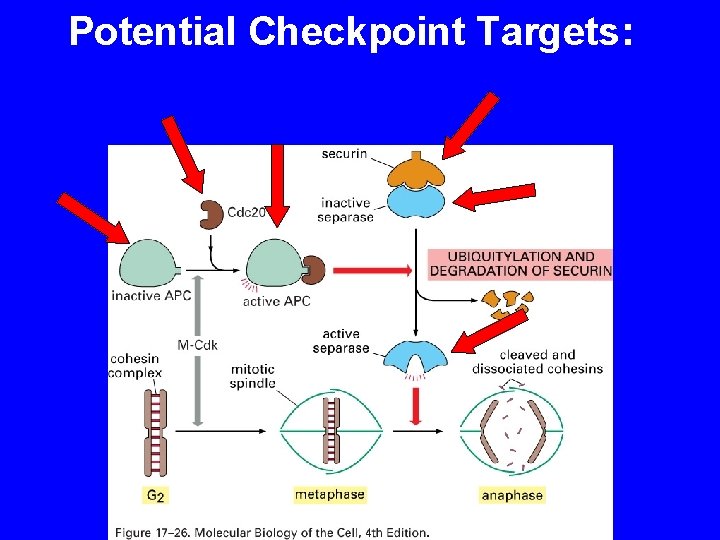
Potential Checkpoint Targets:
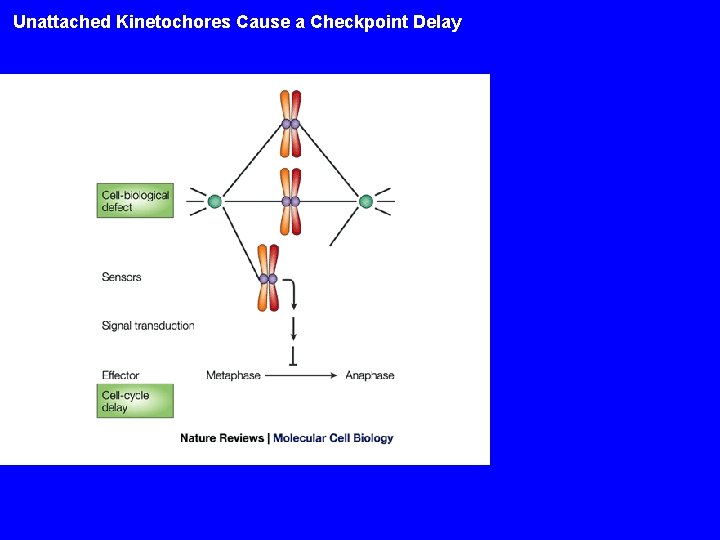
Unattached Kinetochores Cause a Checkpoint Delay
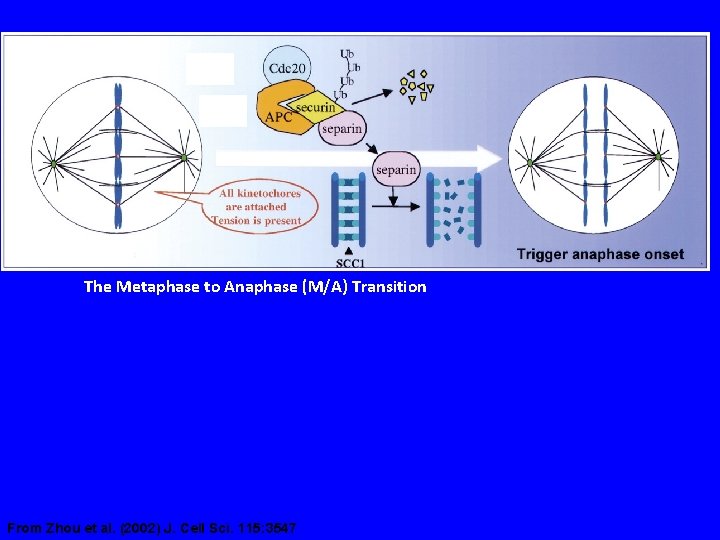
The Metaphase to Anaphase (M/A) Transition From Zhou et al. (2002) J. Cell Sci. 115: 3547
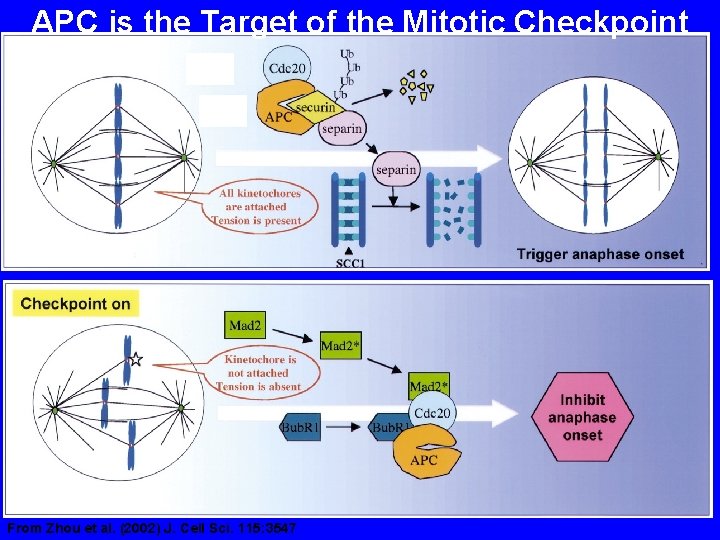
APC is the Target of the Mitotic Checkpoint From Zhou et al. (2002) J. Cell Sci. 115: 3547
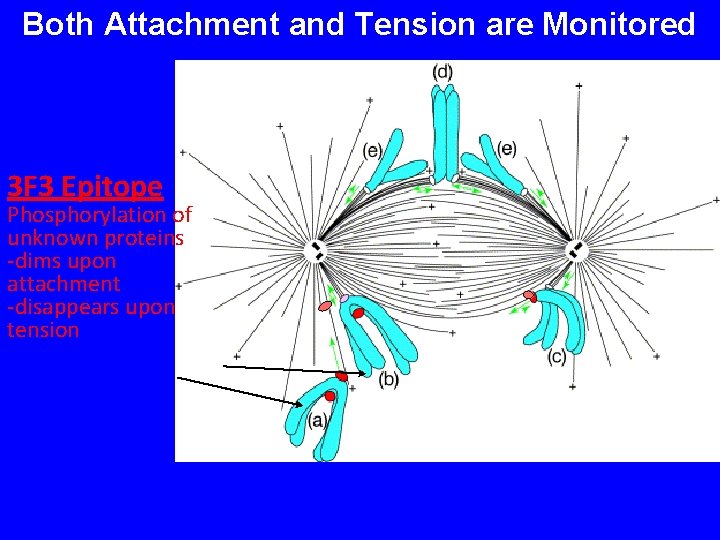
Both Attachment and Tension are Monitored 3 F 3 Epitope Phosphorylation of unknown proteins -dims upon attachment -disappears upon tension
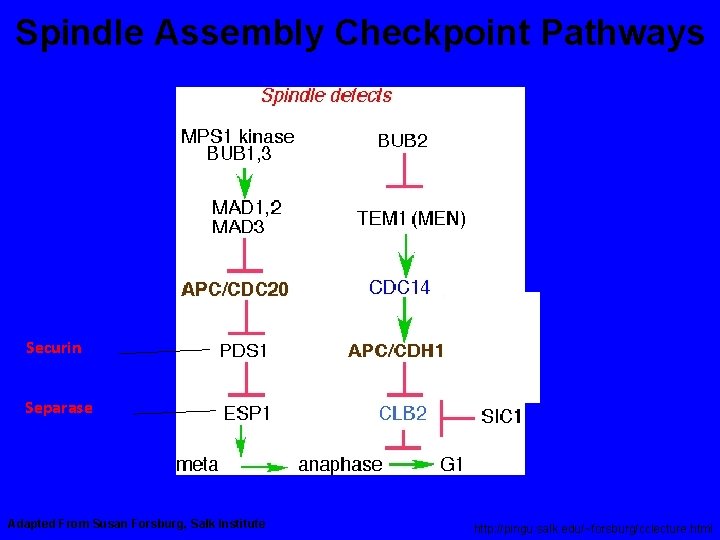
Spindle Assembly Checkpoint Pathways Securin Separase Adapted From Susan Forsburg, Salk Institute http: //pingu. salk. edu/~forsburg/cclecture. html
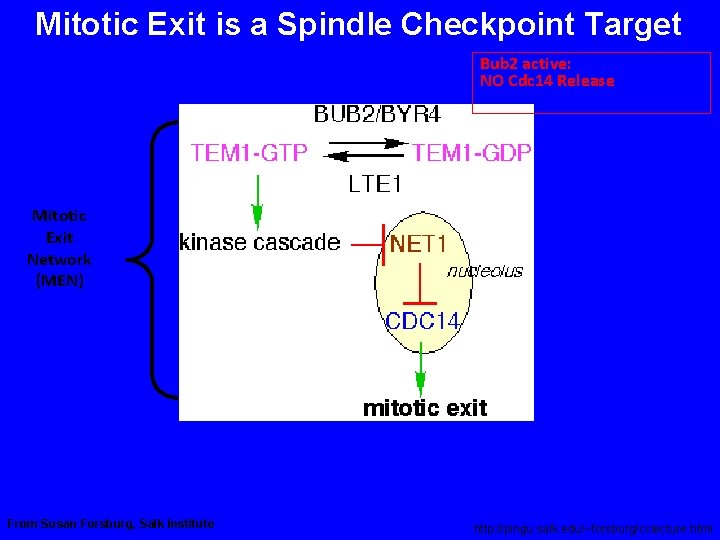
Mitotic Exit is a Spindle Checkpoint Target Bub 2 active: NO Cdc 14 Release Mitotic Exit Network (MEN) Bub 2 inactive: Cdc 14 Released From Susan Forsburg, Salk Institute http: //pingu. salk. edu/~forsburg/cclecture. html
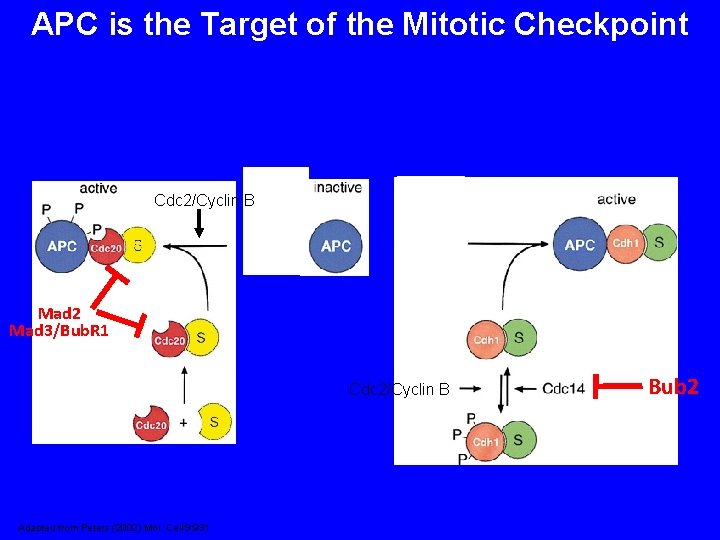
APC is the Target of the Mitotic Checkpoint Cdc 2/Cyclin B Mad 2 Mad 3/Bub. R 1 Cdc 2/Cyclin B Adapted from Peters (2002) Mol. Cell 9: 931 Bub 2
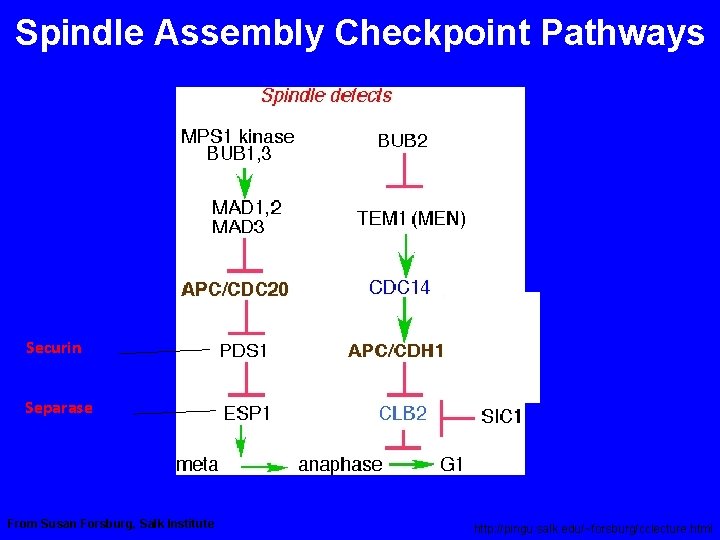
Spindle Assembly Checkpoint Pathways Securin Separase From Susan Forsburg, Salk Institute http: //pingu. salk. edu/~forsburg/cclecture. html
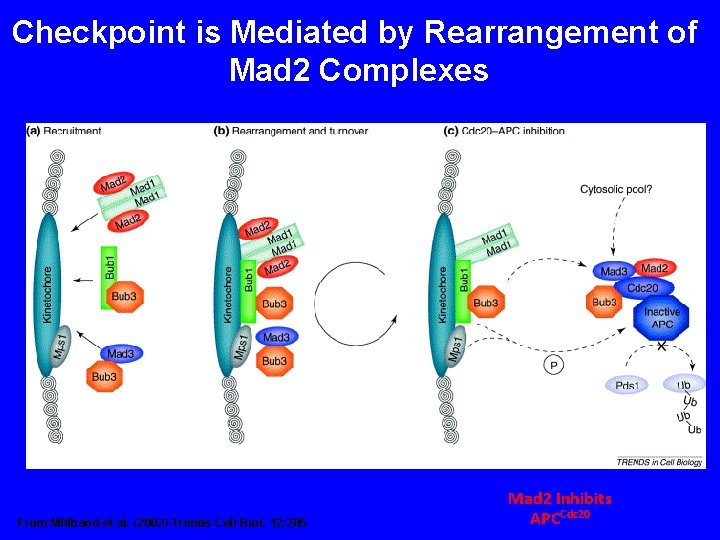
Checkpoint is Mediated by Rearrangement of Mad 2 Complexes From Millband et al. (2002) Trends Cell Biol. 12: 205 Mad 2 Inhibits APCCdc 20
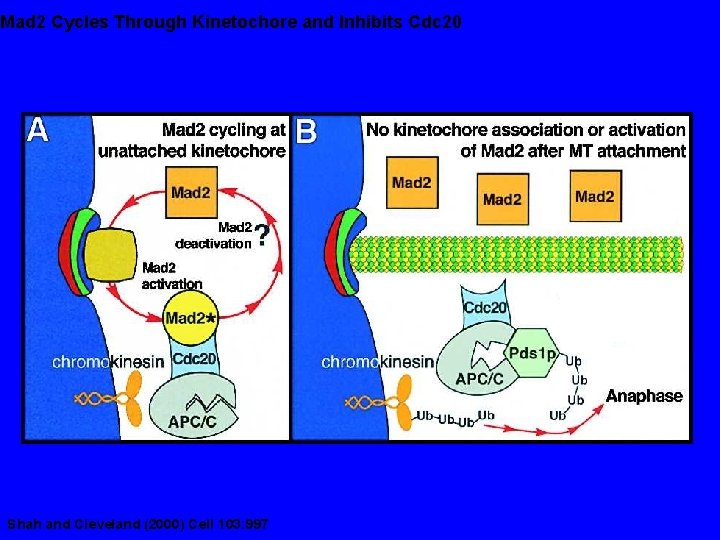
Mad 2 Cycles Through Kinetochore and Inhibits Cdc 20 Shah and Cleveland (2000) Cell 103: 997
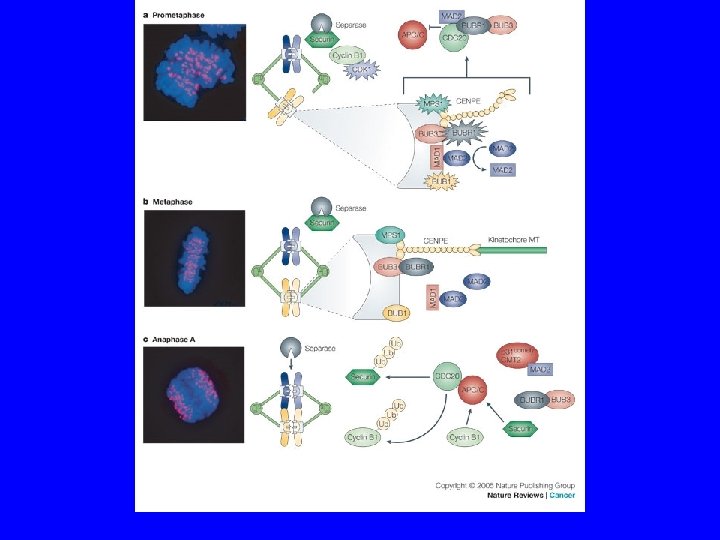
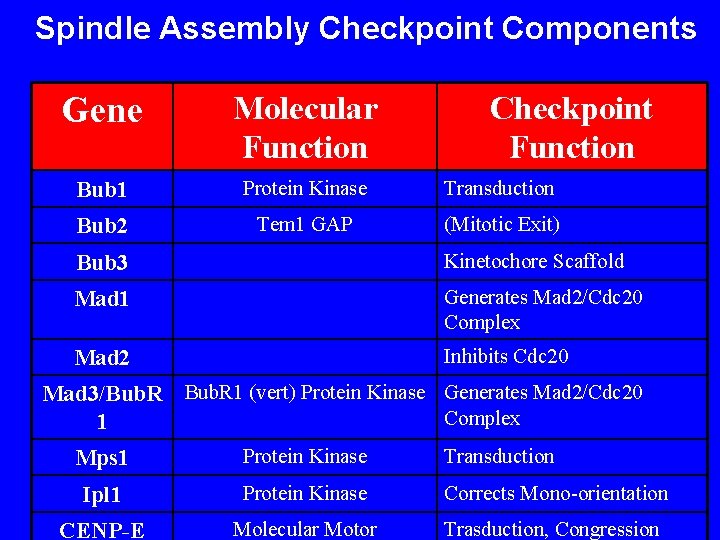
Spindle Assembly Checkpoint Components Gene Molecular Function Checkpoint Function Bub 1 Protein Kinase Transduction Bub 2 Tem 1 GAP (Mitotic Exit) Bub 3 Kinetochore Scaffold Mad 1 Generates Mad 2/Cdc 20 Complex Mad 2 Inhibits Cdc 20 Mad 3/Bub. R 1 (vert) Protein Kinase Generates Mad 2/Cdc 20 Complex 1 Mps 1 Protein Kinase Transduction Ipl 1 Protein Kinase Corrects Mono-orientation CENP-E Molecular Motor Trasduction, Congression

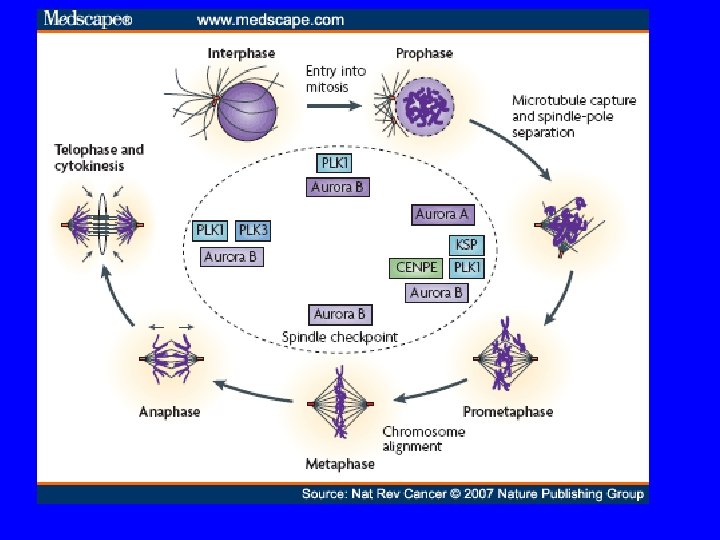
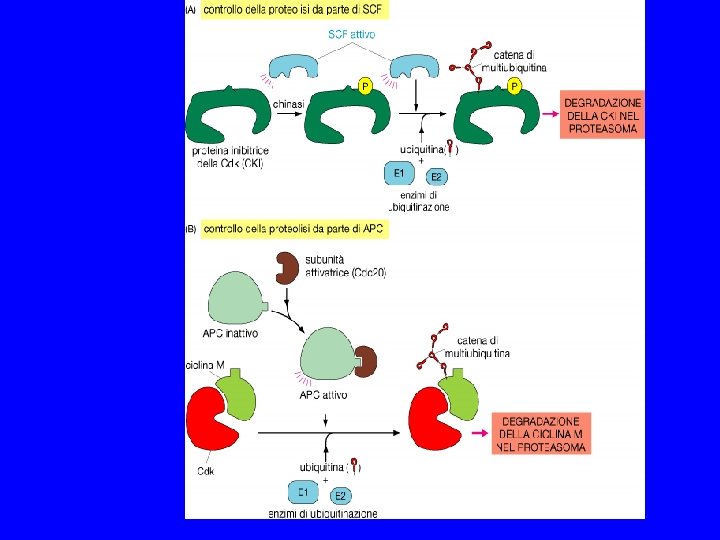
SPINDLE CHEKPOINT In profase le proteine del cinetocore sono fosforilate Bub 1 e 3 (proteine del checkpoint) si legano al cinetocore Mad 2 si lega alle pp del cinetocore Mad 2 inibisce CDC 20 attiva APC degrada Pds 1(Cut 2) (securina) Pds 1 inibisce Esp 1(Cut 1) (separasi) Esp 1 degrada Scc 1 (coesine) Scc 1 tiene uniti i cromatidi
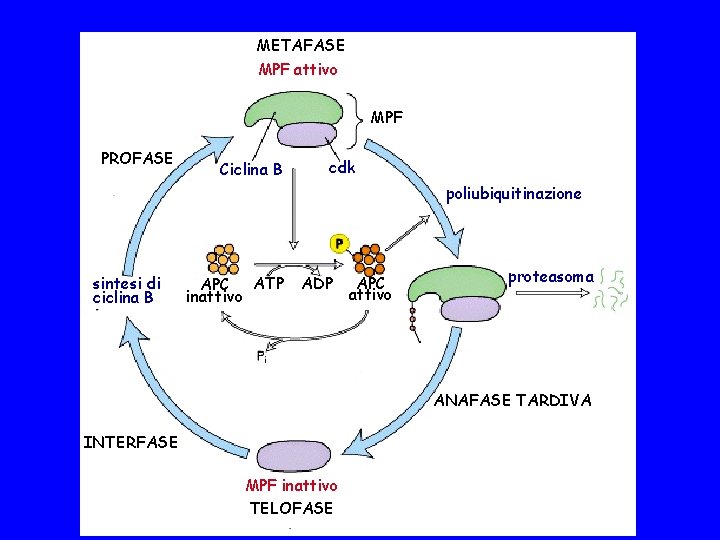
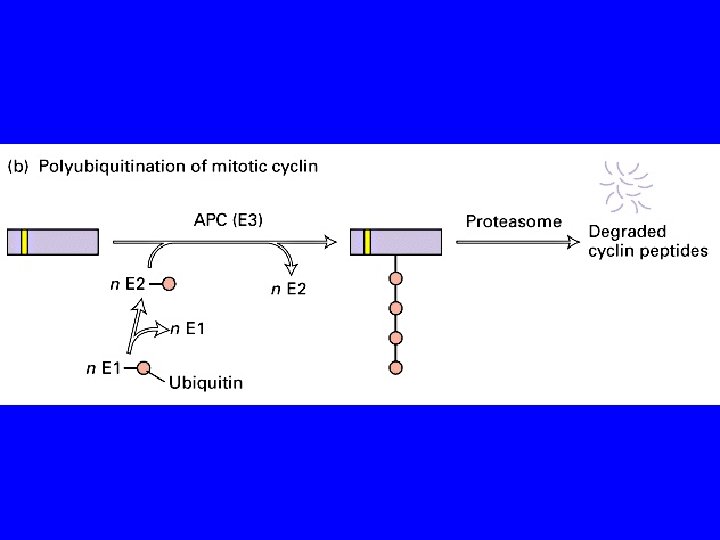
METAFASE MPF attivo MPF PROFASE Ciclina B cdk poliubiquitinazione sintesi di ciclina B APC ATP inattivo ADP APC attivo proteasoma ANAFASE TARDIVA INTERFASE MPF inattivo TELOFASE
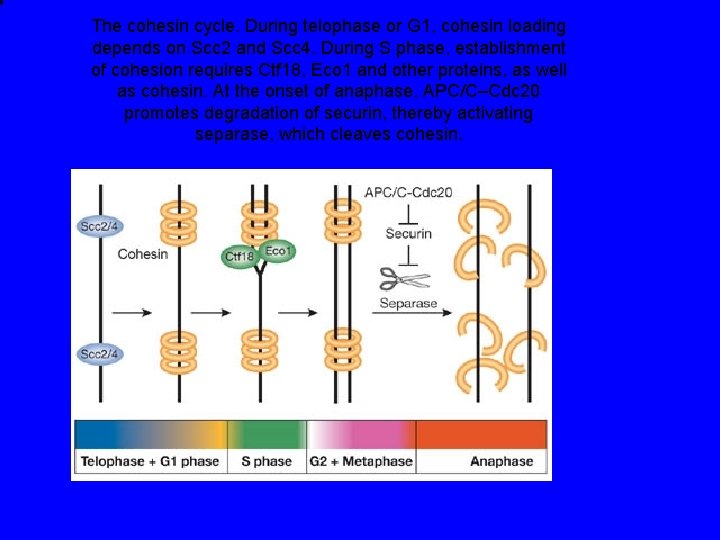
The cohesin cycle. During telophase or G 1, cohesin loading depends on Scc 2 and Scc 4. During S phase, establishment of cohesion requires Ctf 18, Eco 1 and other proteins, as well as cohesin. At the onset of anaphase, APC/C–Cdc 20 promotes degradation of securin, thereby activating separase, which cleaves cohesin.
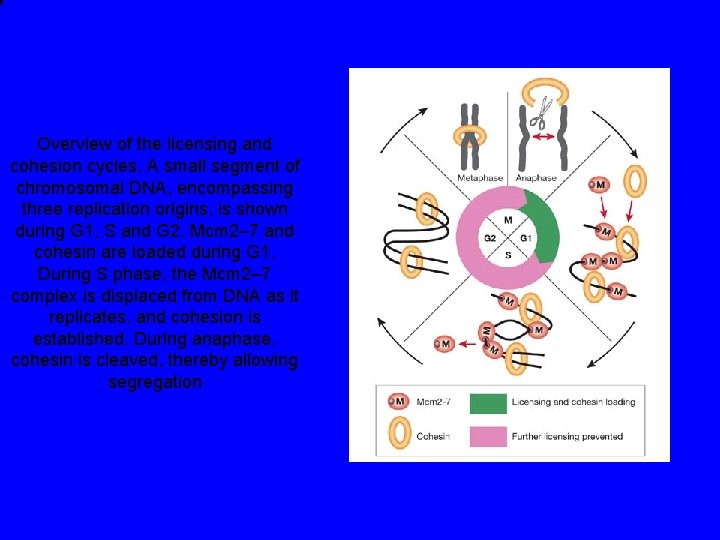
Overview of the licensing and cohesion cycles. A small segment of chromosomal DNA, encompassing three replication origins, is shown during G 1, S and G 2. Mcm 2– 7 and cohesin are loaded during G 1. During S phase, the Mcm 2– 7 complex is displaced from DNA as it replicates, and cohesion is established. During anaphase, cohesin is cleaved, thereby allowing segregation
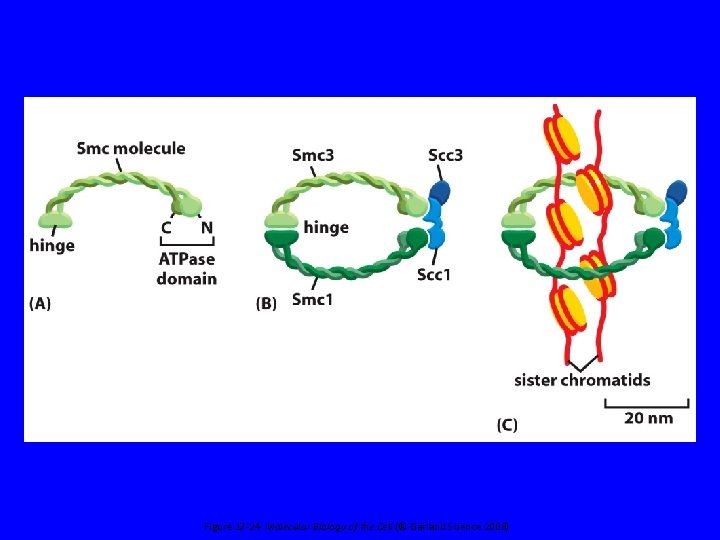
Figure 17 -24 Molecular Biology of the Cell (© Garland Science 2008)
COHESIN
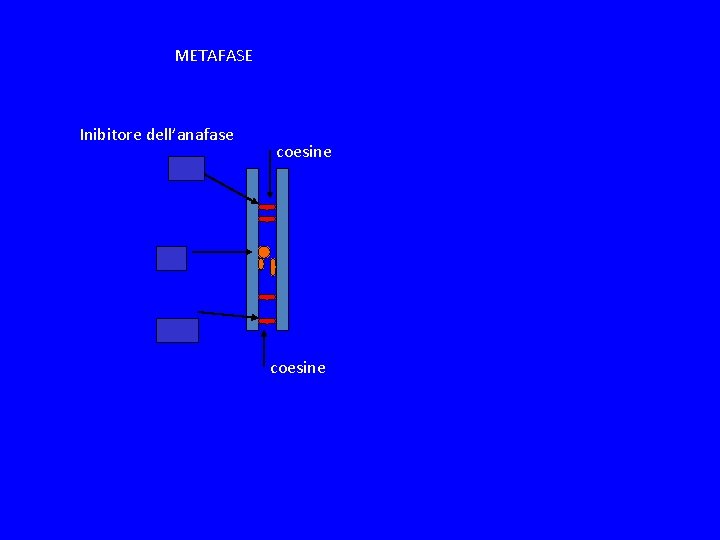
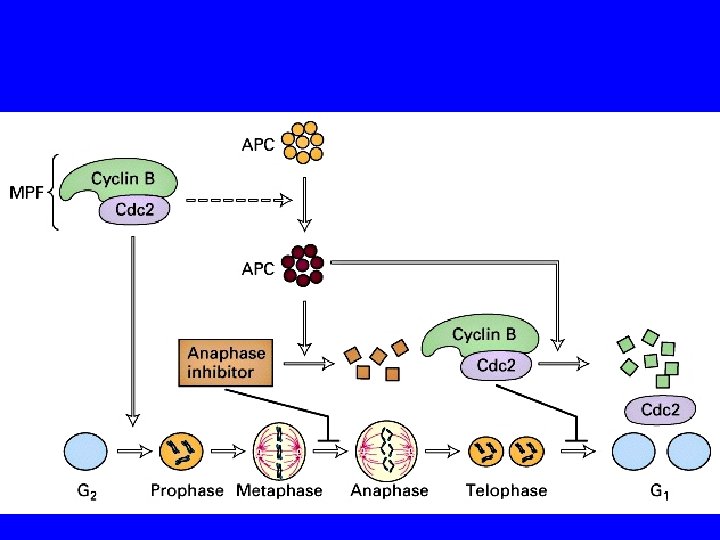
METAFASE Inibitore dell’anafase coesine
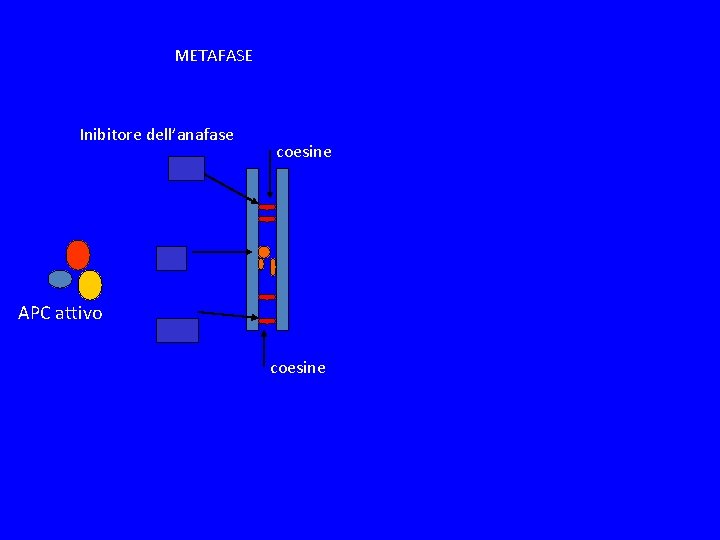
METAFASE Inibitore dell’anafase coesine APC attivo coesine
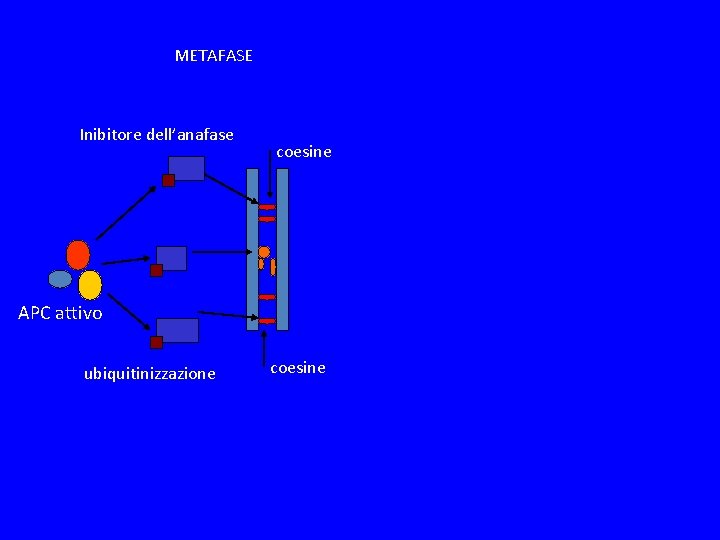
METAFASE Inibitore dell’anafase coesine APC attivo ubiquitinizzazione coesine
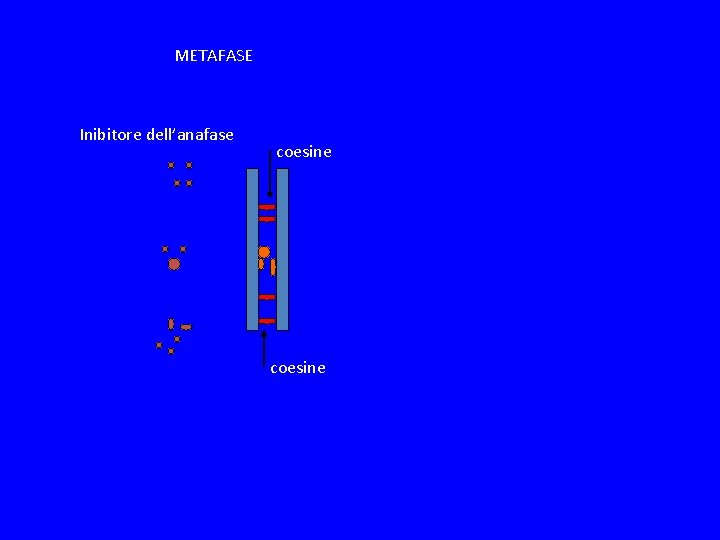
METAFASE Inibitore dell’anafase coesine
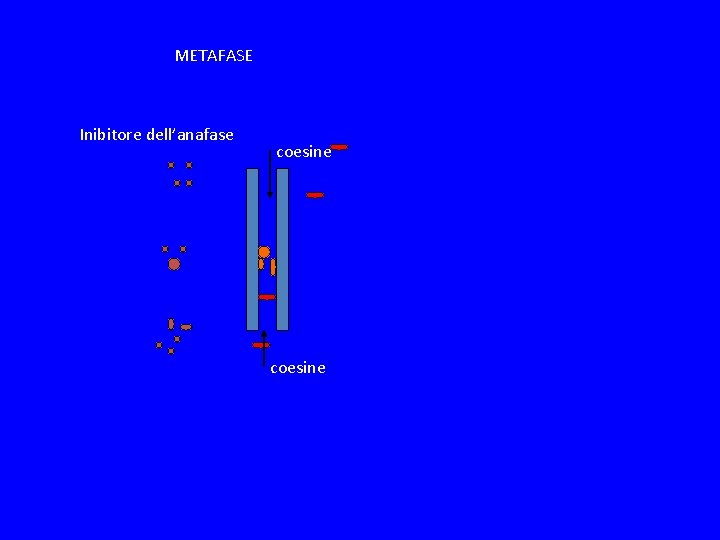
METAFASE Inibitore dell’anafase coesine
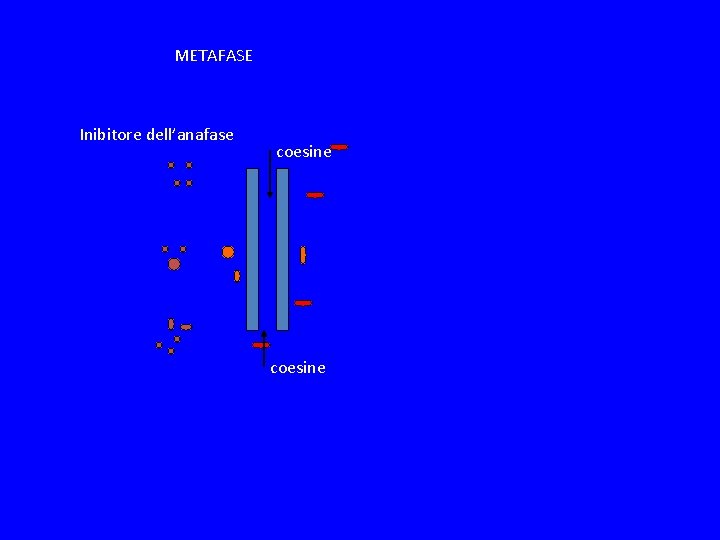
METAFASE Inibitore dell’anafase coesine
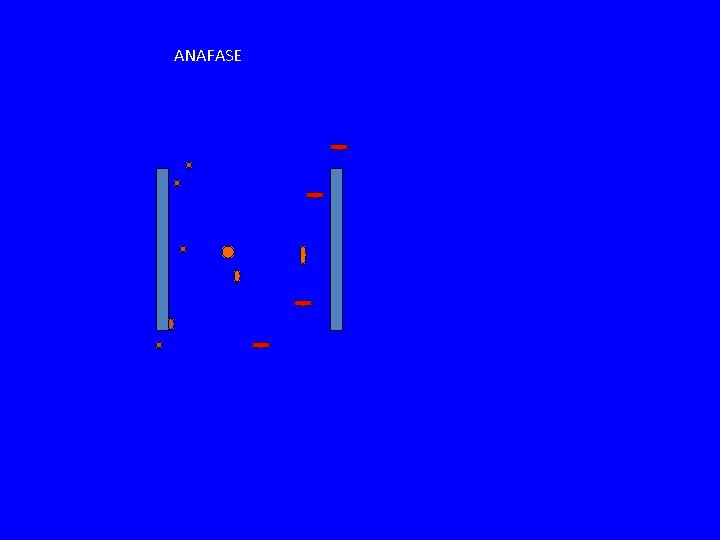
ANAFASE
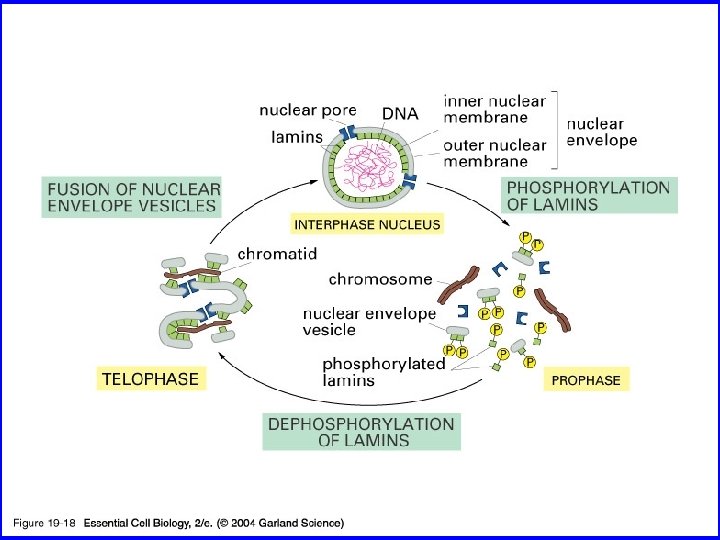
19_18_envelope breaks. jpg

CYTOKINESIS - The plasma membrane begins to invaginate due to the action of the contractile ring of actin and myosin fibers inside the cell. This ring always orientates itself along the metaphase plate 19_19_cleavage_furrow. jpg
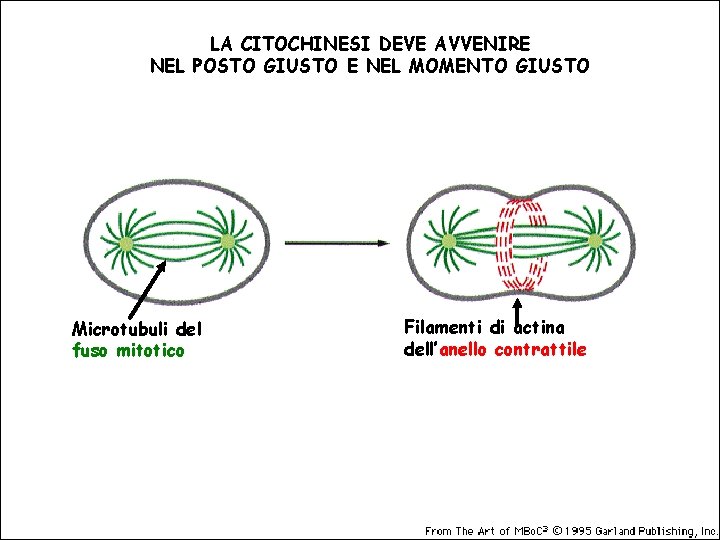
LA CITOCHINESI DEVE AVVENIRE NEL POSTO GIUSTO E NEL MOMENTO GIUSTO Microtubuli del fuso mitotico Filamenti di actina dell’anello contrattile
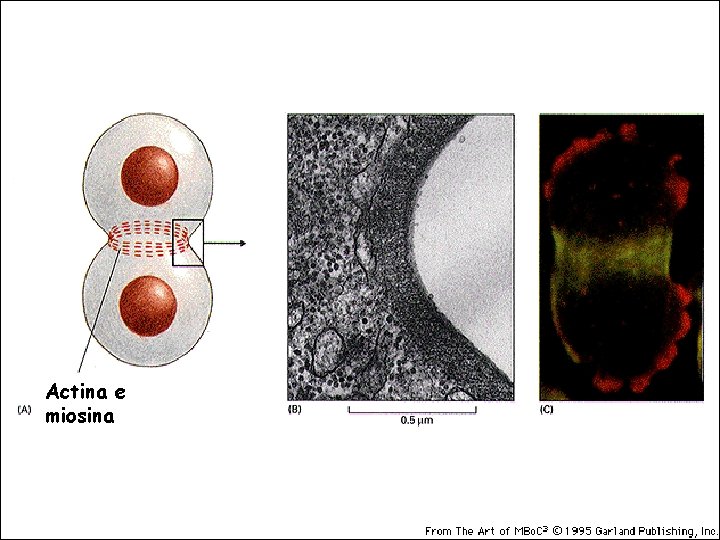
Actina e miosina

COME SONO RIPARTITI GLI ORGANELLI TRA LE CELLULE FIGLIE? Gli organelli della via secretoria vengono frammentati in vescicole che formano clusters e vengono ripartiti tra le cellule figlie.
- Slides: 89