Glass By Dr S John Mary Glass 1

Glass By Dr. S. John Mary

Glass 1. Glass is a state of matter. It is a solid produced by cooling molten material so that the internal arrangment of atoms or molecule remains in a random or distorted state, similar to the arangement in a liquid 2. Glasses belong to the amorphous material 3. Glass is a super cooled liquid

BASIC MATERIAL FOR GLASS 1. The excellent glass can be produced by using pure silica 2. Silica is the basic material for the glass 3. It requires the temperature of 1723 oc. So it is very difficult to produce pure silica glass as it requires high temperature


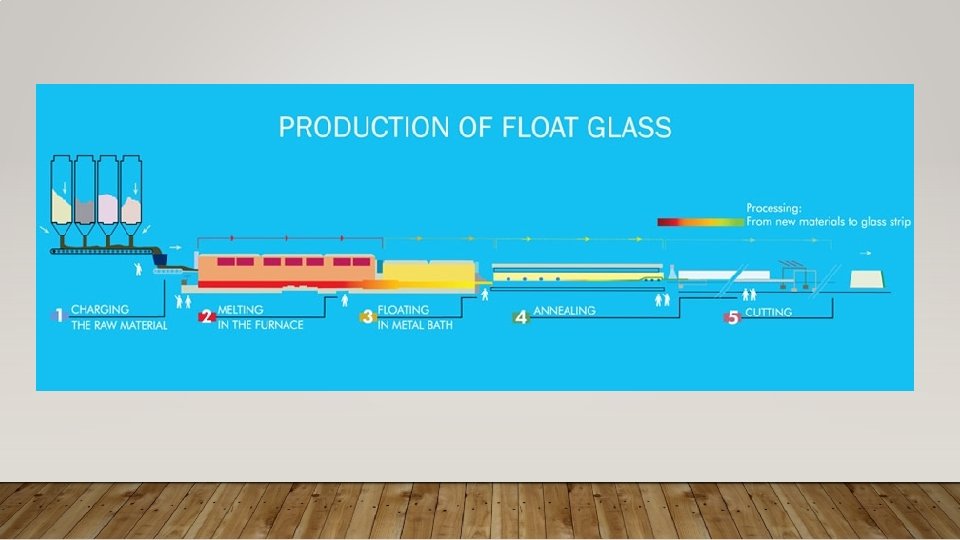


TYPES OF GLASS 1. Natural glass 2. Synthetic glass

NATURAL GLASSES 1. Obsidian • When the heat of the volcanoes melts the rocks • Which become glassy upon cooling. 1. Fulgurities • Glass tubes formed by the lightining strikes on road 1. Pumice • It is a glass foam produced from lava

SYNTHETIC GLASS 1. In 1964 englishman Ravenshroft found that adding lead oxide to glass produce brilliant glass 2. Pb. O makes much easier to melt silica glass 3. This glass is used to make fine crystal bowls and gobblets

Types Of Synthetic Glasses Borosilicat e Glass Soda Lime Glass Alumino- silicate Glass Pure Silica Glass

Types of synthetic glass 1. • These glasses are produced by replacing the soda in the sodalime glass with the boron trioxide, it is called as borosilicate glasses • These glass contains 12℅ of boron trioxide and 81℅ of silica • It is used to make glasses which was used in lab • It has greater durability, greater heat shock resistance

Composition of borosilicate glasses 1. SILICA-81℅ 2. BORON OXIDE-13℅ 3. SODIUM OXIDE-4℅ 4. ALUMINIUM OXIDE-2℅


SODA LIME GLASS 1. Soda lime glass relatively inexpensive, chemically stable, resonably hard, and extremely workable. 2. Silica glass is difficult to melt because of its high melting point. 3. In order to solve this problem, sodium carbonate is added to silica in order to reduce its melting point. 4. To provide for better durability cao is added. Cao is generally obtained from lime stone. 5. Soda lime glass conatins 70 -74℅ of silica


1. Alumina can be used instead of boron oxide for even greater heat shock resistance Chemical durability 2. Alumino silicate glass has high resistance to heat shock, so it is used directly on heating element of the kitchen stove stop 3. It is also used to make Special bottles used for liquid pharmaceutical 4. Aluminosilicate glasses contain 16℅ of alumina and 60℅ of silica 5. Usually Tempored glass are made from this alumina glass

Silica-60℅ Aluminium oxide-16℅ Calcium oxide-7℅ Magnesium oxide-6℅ Barium oxide-6℅ Boron trioxide-4℅ Sodium oxide-1℅


1. These glasses contain (96. 5 -100℅) of silica but these glasses are difficult to make because of its very high melting point 2. However it has been made for special purposes because of its outstanding durability, chemical attack, ability to transmit uv light 3. It is used in space craft windows, made up of 100℅ silica which can withstand temperature as high as 1200°c
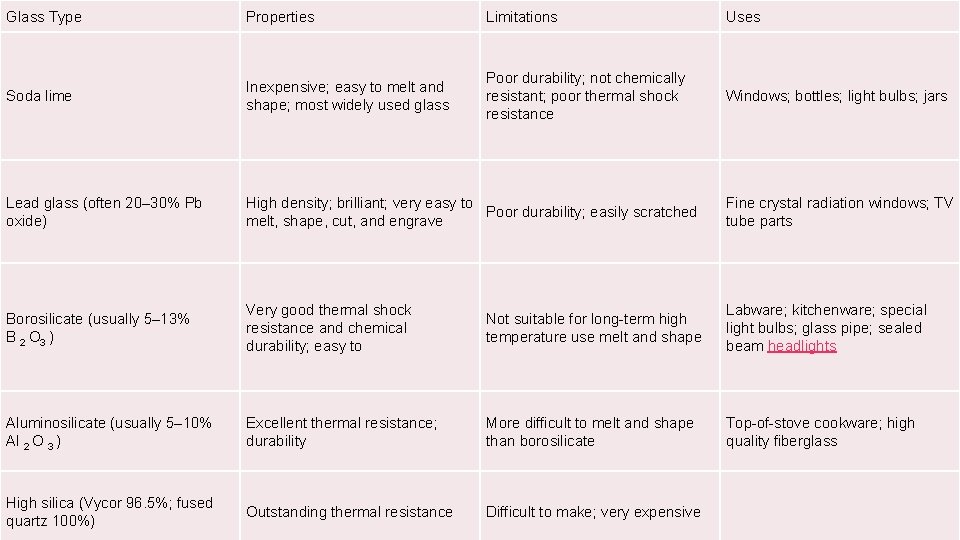
Glass Type Properties Limitations Uses Soda lime Inexpensive; easy to melt and shape; most widely used glass Poor durability; not chemically resistant; poor thermal shock resistance Windows; bottles; light bulbs; jars Lead glass (often 20– 30% Pb oxide) High density; brilliant; very easy to Poor durability; easily scratched melt, shape, cut, and engrave Fine crystal radiation windows; TV tube parts Borosilicate (usually 5– 13% B 2 O 3 ) Very good thermal shock resistance and chemical durability; easy to Not suitable for long-term high temperature use melt and shape Labware; kitchenware; special light bulbs; glass pipe; sealed beam headlights Aluminosilicate (usually 5– 10% Al 2 O 3 ) Excellent thermal resistance; durability More difficult to melt and shape than borosilicate Top-of-stove cookware; high quality fiberglass High silica (Vycor 96. 5%; fused quartz 100%) Outstanding thermal resistance Difficult to make; very expensive

Glass composition 1. 2. 3.

FORMERS • • •

1. 2. 3. 4.


• The natural glass used by the ancient people were, all dark in colour. Usually ranging from olivegreen or brown to jetblack • The colour was due to the presence of significant amounts, of metal impurities, especially iron. Due to the presence of iron in nature causes most ordinary glass to have slight green colour • Major glass colorants are oxides of metals such as cobalt ( blue), chromium ( green), and manganese violet • Yellow glass is usually made with calcium sulfide, red or pink colour usually contains Se.

• Red colour glass had gold added. • Glass colour not only depend on which element are added, but also on the composition of glass • And also depend on, whether the furnace used was in an oxidizing or reduced medium • For example, copper can produce blue or green colour or opaque red colour depend on the melting conditions.


Dichroic glass • The egyptian of 1500 B. C knew that they could make brightly coloured glassby adding certain impurities to the glass melt • The roman expanded it into next level. By 4 th century the Roman learned how to produce a dichroic ( two colour) • The most famous dichroic glass article left by the romans is the lycurgus cup ( now at the British Museum) • It is green in reflected light, but red in transmitted light • This is unusual glass contains microscopic particles of gold and silver.

Pictures of dichroic glass
1. Fiber glass 2. Photochromic glass

• Although, it is not a new product, owes in enhanced usefulness to extreme fitness often about 10 micro metre, but it may be down to 5 micro meter or less • It can drawn into thread or blown intova mat for insulation, tape and air filters • The drawn fibres is used to reinforce various plasticsand composite product is fabricated into pipes, tanks, sporting goods, and serves as fishing fibre and skis • The most common resin are epoxide and polyester

• A special glass Low in silica is used for production of fibres • It have been made to provide fibre for the reinforcement of concrete that would not be attacked by alkali • The most successful, alkali-resistant fibres are made from glass containing 17℅ of zro • The additives make glass very expensive, it is difficult to produce fibre from it • Fibre produced from limestone glass are claimed to be easier to produce than zirconia glass fibre and to posses better alkali resistance


• Photochromic silicate glass posses the following unusual properties: optical darkening in light from ultraviolet through the visible spectrum • Optical bleaching, or fading in the dark and thermal bleaching at higher temperature • The photochromic glasses are truly reversible • The explanation of this photochromic process is a manufacture of glass on which submicroscopic silver halide particle exists which reacts differently from ordinary photographic silver halide when exposed to light • Such tiny halide are about 5 nm in diameter and have concentration of 1015 per cubic cm


• The main aim of this presentation is to explain about the existence of glass in this world and its types. • In our day to day life we use glasses everywhere. • Even in this area we have glasses. • This very attitude of usage of a transparent material like a Glass may give a attractive look and also a great decorative artwork too. • Henceforth this glass has multiple application and various properties and one of the existing industry.

REFERENCES • Shreves chemical process industries Links: https: //theconstructor. org/building/types-of-glass-properties-usesconstruction/14755/ https: //www. sciencedirect. com/topics/earth-and-planetary-sciences/glass-industry https: //www. youtube. com/watch? v=-z 1 P 2 c. K 9 Rro https: //en. wikipedia. org/wiki/Glass_production https: //www. aisglass. com/glass-manufacturing-process

- Slides: 39