GJHeducation nfs Edexcel GCSE Combined Science FT PAPER

GJHeducation nfs Edexcel GCSE Combined Science FT PAPER 3 REVISION Topics C 1 – C 4

Fill the VOID! Get this PAPER 3 revision lesson started by doing just as the title says and filling the void with the name of the substance that will be formed! sodium + oxygen sodium oxide

Understanding check #1 sodium + oxygen sodium oxide By verbalising your answers with a partner, state whether the substances in the word equation above are elements, compounds or mixtures (3 marks) Element Compound sodium oxide oxygen Mixture

Understanding check #2 Which of the following is the definition of an element? (1 mark) A B The smallest unit into which matter can be divided A substance that is made of only 1 type of atom A substance that is made of 2 or more of a So what is being described in A? types Take atom chemically bonded together couple of minutes to discuss what you think D A substance that is made of 2 or more types of it is atom and which then are to try to recall what is found not all chemically bonded inside these structures together C

An atom is the smallest unit into which matter can be divided and atoms contain different numbers of protons, electrons and neutrons…

EDEXCEL GCSE CS (FT) PAPER 3 REVISION QUIZ R 1 Round 1 is going to run and it will test your ability to spot the answers to the quantitative parts of topics C 1 – C 4. This is “Number Crazy”

3 0 19 22 17 4 20 72 48 11 40 9 This round of the competition will run for the duration of the lesson so keep your eyes peeled for this background. A statement will appear in this circle which has a numerical answer that is shown somewhere on this slide. Buzz in when you’ve spotted the number and if you’re 1 st and correct, you’ll get 2 TEAM POINTS! 2 6 16 8 28 64 1 23 21 7 12 18 10
3 0 19 22 17 4 72 48 20 21 11 6 Protons in a sodium atom 40 9 16 8 28 10 1 23 2 7 18 64 12
Understanding check #3 By verbalising your answer to a partner, state the word which could replace “protons” in this statement and would still give 11 as the answer electrons

KEY POINT In an atom, the number of positively-charged protons is always the same as the number of negatively-charged electrons

Understanding check #4 In an atom, the number of positively-charged protons is always the same as the number of negatively-charged electrons State the overall charge of a sodium atom. You should make reference to the sub-atomic particles in a sodium atom in your answer (2 marks) The overall charge of a sodium atom is neutral because it contains 11 positively-charged protons and 11 negatively-charged electrons

1 TO THINK ABOUT You have 4 minutes to discuss with 1 person The nucleus of an atom is Another student wrongly putbecause this as their positively-charged answer. A particular part of an atom is it containsbutpositively-charged it is not the overall charge. Discuss which partand of thealso atom charged protons the student must have been thinking of neutrons which have and make sure you can explain whyno this part is positively-charged charge The overall charge of a sodium atom is positive

KEY POINT The nucleus of an atom is positively-charged but the overall charge of an atom is neutral
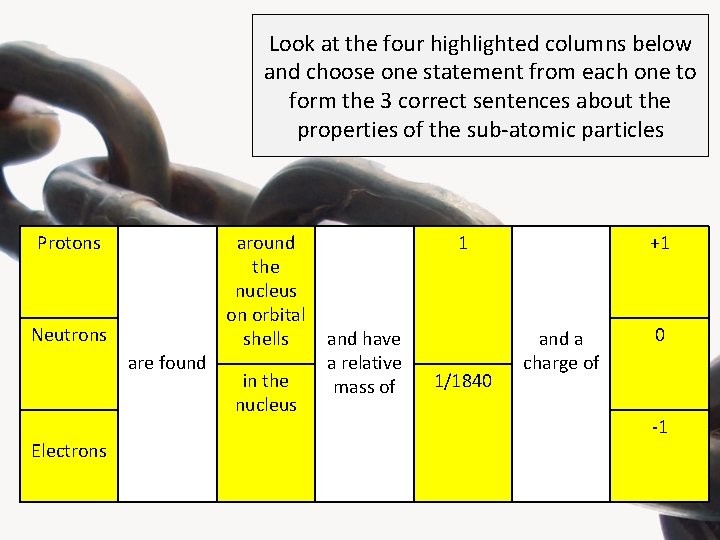
Look at the four highlighted columns below and choose one statement from each one to form the 3 correct sentences about the properties of the sub-atomic particles Protons Neutrons are found Electrons around the nucleus on orbital shells in the nucleus 1 and have a relative mass of 1/1840 +1 and a charge of 0 -1
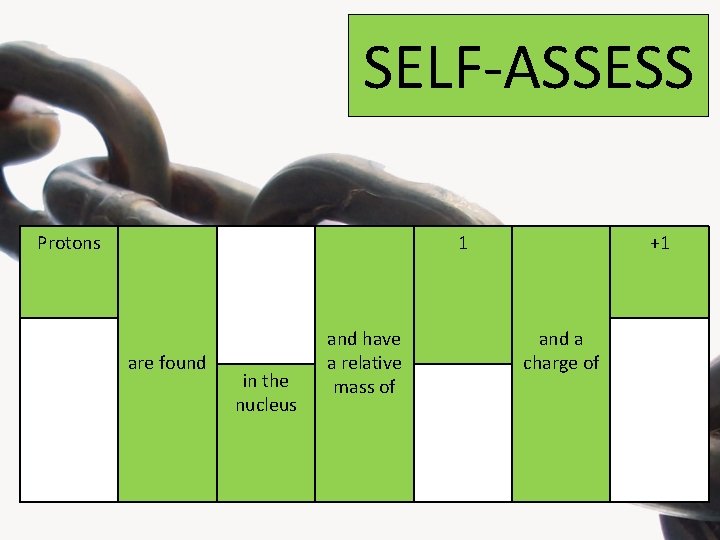
SELF-ASSESS Protons 1 are found in the nucleus and have a relative mass of +1 and a charge of
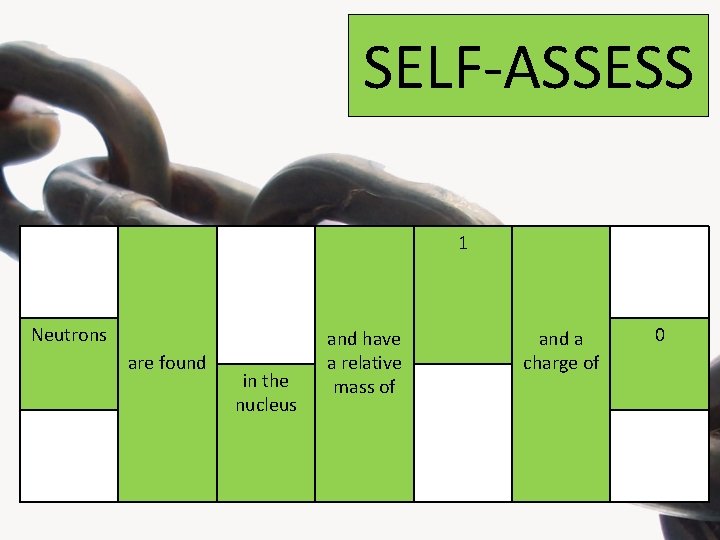
SELF-ASSESS 1 Neutrons are found in the nucleus and have a relative mass of and a charge of 0
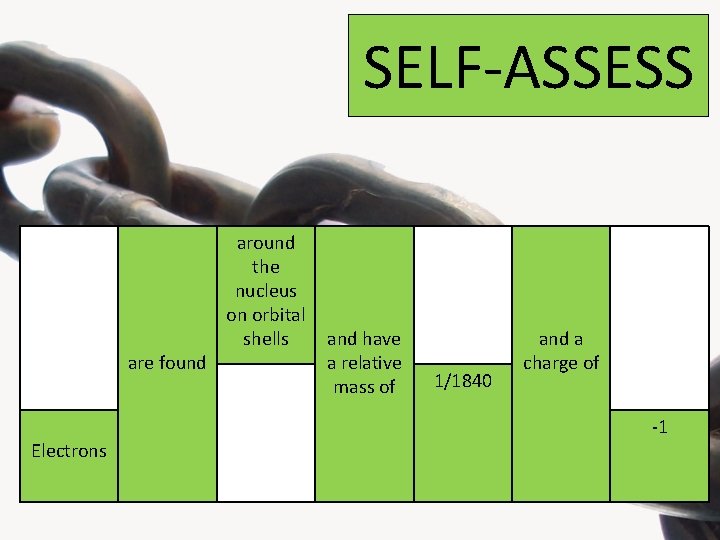
SELF-ASSESS are found Electrons around the nucleus on orbital shells and have a relative mass of 1/1840 and a charge of -1

3 0 19 22 17 4 72 48 20 21 11 6 Neutrons in a sodium atom 40 9 16 8 28 10 1 23 2 7 18 64 12

Using the Periodic Table Mass number 23 Na Number of protons + neutrons To calculate the number of neutrons, subtract the atomic number from the mass number Atomic number 11 Number of protons Number of electrons

EXAM QUESTION “State the number of the different subatomic particles that are found in an aluminium atom”
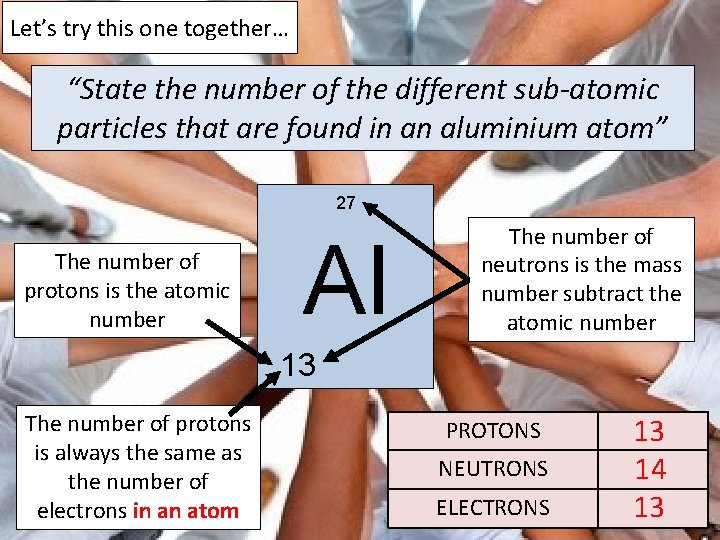
Let’s try this one together… “State the number of the different sub-atomic particles that are found in an aluminium atom” 27 The number of protons is the atomic number Al The number of neutrons is the mass number subtract the atomic number 13 The number of protons is always the same as the number of electrons in an atom PROTONS NEUTRONS ELECTRONS 13 14 13

Understanding check #5 State the number of each of the sub-atomic particles in the following atoms (4 marks): 1. Lithium 3 protons, 4 neutrons, 3 electrons 2. Fluorine 9 protons, 10 neutrons, 9 electrons 3. Hydrogen 1 proton, 0 neutrons, 1 electron 4. Potassium 19 protons, 20 neutrons, 19 electrons

This is actually fairly important…… So each of the 100 different elements that can be found in the Periodic Table consist of only 1 type of atom. When these elements have to be written in a chemical symbol equation, most of them are written as a single atom (e. g. Li) because they are said to be monatomic. However, there are 7 elements which are diatomic and must be written as a pair of atoms…. .

EDEXCEL GCSE CS (FT) PAPER 3 REVISION QUIZ R 2 Round 2 is a good old version of SNAP! It’s very simple – can you spot when an “element” MATCH has been made or not?

THE RULES The name of an element and a formula will appear on the board at the same time. If they match, the first person to shout “SNAP” gets 2 TEAM POINTS. If they don’t match, then you must stay completely silent. Anyone who shouts SNAP when it isn’t a match loses 1 TEAM POINT.

LITHIUM Li 2
OXYGEN O 2

POTASSIUM K
HYDROGEN H
NITROGEN N

CHLORINE Cl 2

Of the 100+ elements in the Periodic Table, 7 of them exist as a pair of atoms as they are diatomic. Therefore, they need to be written like this in the symbol equation If the element isn’t one of the 7, then it is monatomic and only exists as a single atom and should be written accordingly Diatomic molecules hydrogen H 2 nitrogen N 2 oxygen O 2 fluorine F 2 chlorine Cl 2 bromine Br 2 iodine I 2 magnesium would be written Mg

Understanding check #6 H 2 N 2 O 2 F 2 Cl 2 Br 2 I 2 The atoms in each of these diatomic molecules are held together by the same type of chemical bond. By verbalising your answer with a partner, identify the chemical bond found in all of these structures (1 mark) covalent

1 TO THINK ABOUT You have 2 minutes to discuss with 1 person Covalent bonds occur in mostonnon-metallic By focusing the type of atoms that are found in these substances, discuss elements and in how you can spot whether a structure is heldcompounds together by covalent if you ofbonds nonare given the chemical formula in an metals assessment The atoms in H 2 N 2 O 2 F 2 Cl 2 Br 2 and I 2 are all held together by covalent bonds.
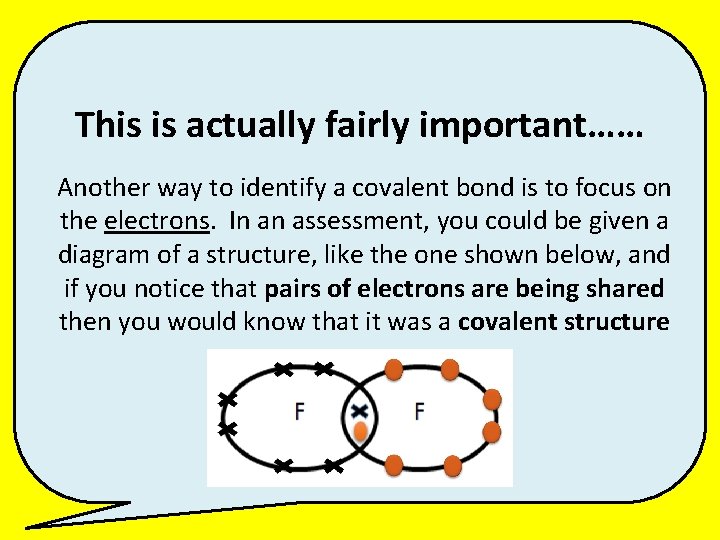
This is actually fairly important…… Another way to identify a covalent bond is to focus on the electrons. In an assessment, you could be given a diagram of a structure, like the one shown below, and if you notice that pairs of electrons are being shared then you would know that it was a covalent structure

EDEXCEL GCSE CS (FT) PAPER 3 REVISION QUIZ R 3 There a huge number of key words in topics C 1 – C 4 and during round 3, the letters of a key word will be revealed one at a time – simply buzz in when you know the word

THE BIG REVEAL Intermolecular forces are the weak forces of attraction that exist between simple molecules and U low N ___ Ithese R Rused Amelting Tcan Cthe L ___ E be___ M___ Oto___ Lexplain E ___ ___ ___ and boiling points of the simple covalent structures like O 2 and CO 2 Will you be the first to get it?
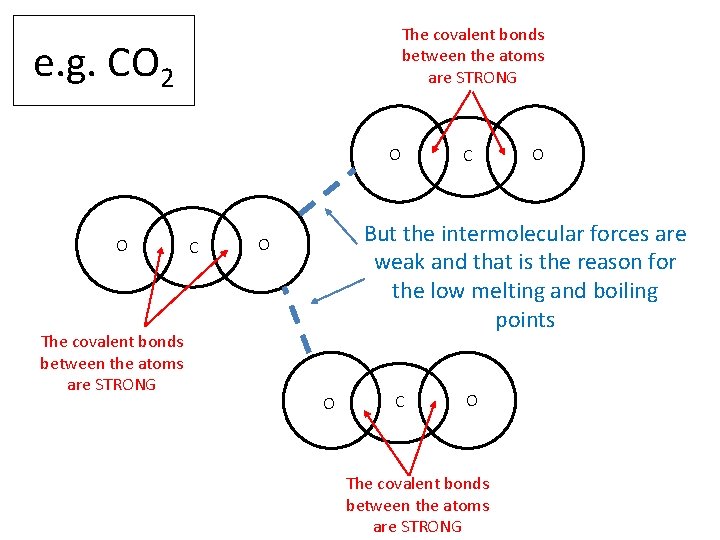
The covalent bonds between the atoms are STRONG e. g. CO 2 O O The covalent bonds between the atoms are STRONG C C O But the intermolecular forces are weak and that is the reason for the low melting and boiling points O O C O The covalent bonds between the atoms are STRONG

But not all covalent structures are simple. The giant covalent structures, like diamond and graphite, have high melting and boiling points as a result of strong intermolecular forces and one of them can even conduct electricity…

A POTENTIAL EXAM QUESTION Explain why diamond is unable to conduct electricity. You should make reference to electrons in your answer

Let’s try this together… Explain why diamond is unable to conduct electricity. You should make reference to electrons in your answer

3 0 19 22 17 4 72 48 20 21 11 Electrons in the outer shell of a carbon atom 40 9 6 16 8 28 10 1 23 2 7 18 64 12

Let’s try this together… Explain why diamond is unable to conduct electricity. You should make reference to electrons in your answer § Each carbon atom has four electrons in its outer shell.

3 0 19 22 17 4 72 48 20 21 Other carbon atoms that each carbon atom bonds with in diamond 11 40 9 10 1 23 2 7 18 6 16 8 28 64 12

Let’s try this together… Explain why diamond is unable to conduct electricity. You should make reference to electrons in your answer § Each carbon atom has four electrons in its outer shell. § In diamond, all four of these electrons form covalent bonds with four other carbon atoms.

Let’s try this together… Explain why diamond is unable to conduct electricity. You should make reference to electrons in your answer § Each carbon atom has four electrons in its outer shell. § In diamond, all four of these electrons form covalent bonds with four other carbon atoms. § This means that all of the electrons are involved in the bonding and there are no delocalised electrons

Let’s try this together… Explain why diamond is unable to conduct electricity. You should make reference to electrons in your answer § Each carbon atom has four electrons in its outer shell. § In diamond, all four of these electrons form covalent bonds with four other carbon atoms. § This means that all of the electrons are involved in the bonding and there are no delocalised electrons § As a result, these electrons are not free to carry charge through the structure and so diamond cannot conduct electricity

3 0 19 22 17 4 72 48 20 21 Other carbon atoms that each carbon atom bonds with in graphite 11 40 9 10 1 23 2 7 18 6 16 8 28 64 12

Understanding check #7 “Each carbon atom has four electrons in its’ outer shell” “In graphite, each carbon atom bonds to three other carbon atoms” By considering the two facts above and using the passage about diamond that you’ve just seen, answer the following: Explain why graphite is able to conduct electricity (4 marks)

Each carbon atom has four electrons in its outer shell. In graphite, three of these electrons form covalent bonds with the three atoms that each atom connects with in its layer. This leaves a fourth electron, which is not involved in bonding but is delocalised instead. Delocalised electrons are free to move through the structure and to carry the charge so that graphite and graphene can conduct electricity.

In addition to the strong covalent bonds that are found between most non-metallic elements and in compounds of non-metals, you need to be able to recognise and know the details of ionic bonding and metallic bonding….

EDEXCEL GCSE CS (FT) PAPER 3 REVISION QUIZ R 4 Round 4 makes use of one of the all time film quotes and says “The name’s bond…”

In this round, a clue will appear on the board to the identity of a type of BOND. Buzz in when you recognise the BOND and if you’re right, you’ll get 2 TEAM POINTS. Of course, there are 007 to do… The name’s bond, _____ bond

#001 Occur between most nonmetallic elements The name’s bond, COVALENT _____ bond

#002 Occurs in compounds formed from metals combined with non-metals IONIC bond The name’s bond, _____

#003 Electrons are transferred from the outer shell of the metal atom to the outer shell of the non-metal atom IONIC bond The name’s bond, _____

#004 Explains why all metals* are solids at room temperature METALLIC bond The name’s bond, _____

#005 Occurs in alloys METALLIC bond The name’s bond, _____
#006 The name’s bond, COVALENT _____ bond

#007 Occur in a compound like lithium fluoride (Li. F) IONIC bond The name’s bond, _____

So that latest competition reminded us that ionic bonds occur in compounds formed from metals combined with non-metals, like lithium fluoride (Li. F), and that the electrons are transferred from the outer shell of the metal atom to the outer shell of the non-metal atom. In a similar way to covalent bonding, the result is two atoms with full outer shells of electrons….

A POSSIBLE EXAM QUESTION “Draw a dot and cross diagram to represent the ionic compound, lithium fluoride (Li. F) and describe how it is formed”

This might prove useful…… It will probably prove useful to recall from earlier in the lesson that a lithium atom has 3 protons and 3 electrons and that a fluorine atom has 9 protons and 9 electrons
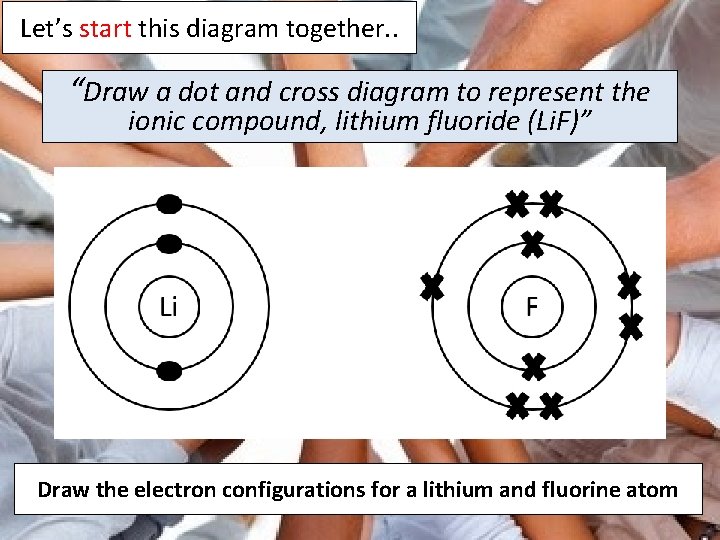
Let’s start this diagram together. . “Draw a dot and cross diagram to represent the ionic compound, lithium fluoride (Li. F)” Draw the electron configurations for a lithium and fluorine atom
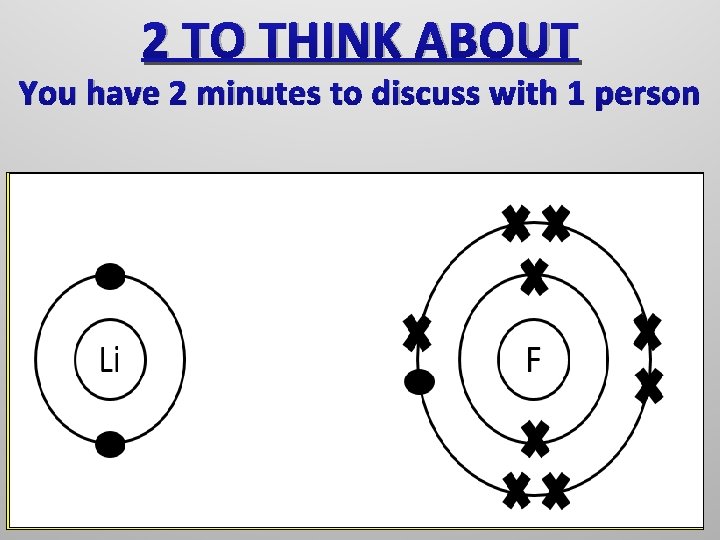
2 TO THINK ABOUT You have 2 minutes to discuss with 1 person The electron in the outer shell of lithium is By reminding yourself of the point above and transferred toconsider the how by studying your configurations, it is possible for both the lithium and the outer shell ofouter fluorine to get full shells All of the elements that are found to the left of the noble gases do not have full outer shells and therefore will react and bond with other elements in order to get a full outer shell.

3 0 19 22 17 4 72 48 20 21 Electrons remaining in lithium after the transfer of the electron 11 40 9 10 1 23 2 7 18 6 16 8 28 64 12

3 0 19 22 17 4 72 48 20 21 Electrons in “fluorine” after it receives an electron from lithium 11 40 9 10 1 23 2 7 18 6 16 8 28 64 12

Understanding check #8 As neither lithium nor fluorine will have the same number of protons as electrons (once an electron has been transferred from the lithium to the fluorine), they are no longer considered to be atoms State the name which is given to substances which are formed from atoms and have different numbers of protons to electrons (1 mark) ions

KEY POINT Ions contain a different number of protons when compared to electrons and as a result, ions are particles with a charge

KEY POINT During ionic bonding, metal atoms lose electrons and become positivelycharged ions whereas nonmetal atoms gain electrons and become negativelycharged ions

This is actually fairly important…… When a lithium atom loses 1 electron, it becomes a lithium ion with a 1+ charge due to the fact that it contains 3 protons but only 2 electrons

Understanding check #9 When a lithium atom loses 1 electron, it becomes a lithium ion with a 1+ charge due to the fact that it contains 3 protons but only 2 electrons Use the description above as a guide to answer the following question: State the charge of a fluoride ion and explain your answer with reference to the specific number of protons and electrons (3 marks)

When a fluorine atom gains 1 electron, it becomes a fluoride ion with a 1 - charge due to the fact that it contains 9 protons and 10 electrons
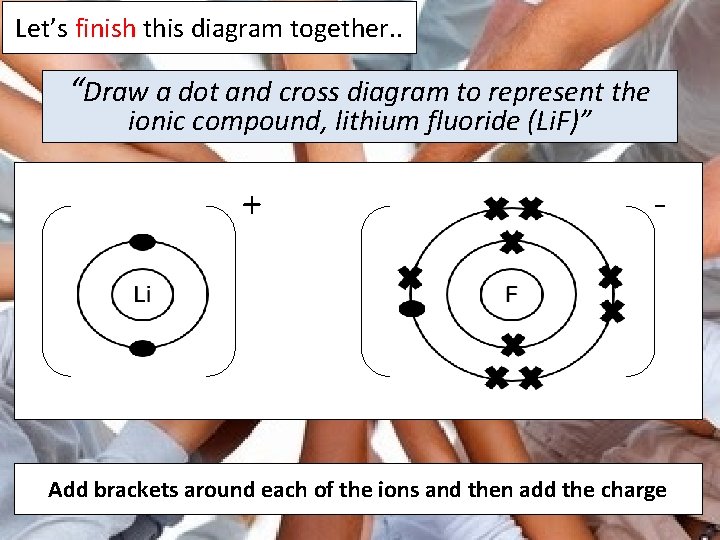
Let’s finish this diagram together. . “Draw a dot and cross diagram to represent the ionic compound, lithium fluoride (Li. F)” + - Add brackets around each of the ions and then add the charge

The ionic compound, lithium fluoride, is formed when the single electron in the outer shell of lithium is transferred to the outer shell of fluorine. As a result, both atoms get full outer shells and become ions. Lithium becomes a 1+ ion and fluoride is a 1 - ion A STAR DESCRIPTION!

This is actually fairly important…… You may have noticed that both the positive and negative ions have full outer shells of electrons. This means that an ion will have the same electronic structure as a noble gas (group 0)

Understanding check #10 Of the remaining noble gases, which structure one has the A fluoride ion has 3 the same electronic as same electronic structure noble as a lithium (1 mark) which of the following gases? ion (1 mark) A lithium ion =helium (2) and helium = (2) B neon and neon = (2. 8) fluoride ion = (2. 8) C argon D krypton

If you feeling 100% confident with this topic, draw a dot and cross diagram to represent sodium chloride (Na. Cl) and then use your diagram to describe how this ionic compound was formed • If you would like a little assistance with drawing the dot and cross diagram and the description, ask for a worksheet labelled SQUARES • If you simply cannot begin, ask for a STARS worksheet which gives more support
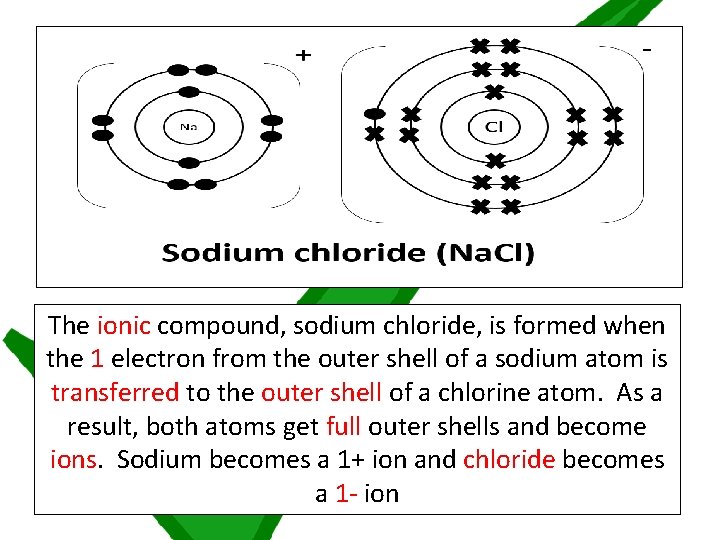
The ionic compound, sodium chloride, is formed when the 1 electron from the outer shell of a sodium atom is transferred to the outer shell of a chlorine atom. As a result, both atoms get full outer shells and become ions. Sodium becomes a 1+ ion and chloride becomes a 1 - ion

There is one more task to check on your understanding of ionic compounds. Read through the passage about the formation of magnesium oxide, identify the four errors and then make the appropriate corrections Once you’ve read through the passage, if you are unable to find the errors, ask for a worksheet labelled STARS which will provide some support
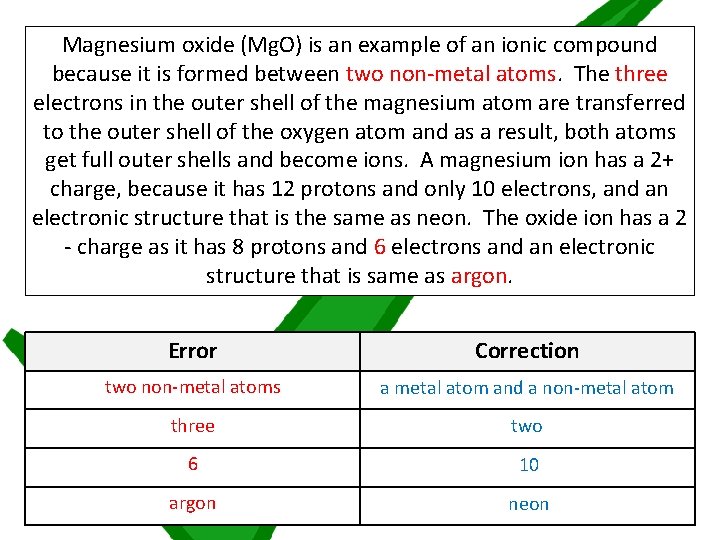
Magnesium oxide (Mg. O) is an example of an ionic compound because it is formed between two non-metal atoms. The three electrons in the outer shell of the magnesium atom are transferred to the outer shell of the oxygen atom and as a result, both atoms get full outer shells and become ions. A magnesium ion has a 2+ charge, because it has 12 protons and only 10 electrons, and an electronic structure that is the same as neon. The oxide ion has a 2 - charge as it has 8 protons and 6 electrons and an electronic structure that is same as argon. Error Correction two non-metal atoms a metal atom and a non-metal atom three two 6 10 argon neon

1 TO THINK ABOUT You have 5 minutes to discuss with 2 people The charge on on the ions formed by the By focusing that wemetals have inencountered group 1 and 2 and the non-metals in group in our three examples 6 and 7 relates to the group number of the and by studyingelement: the positions of their respective atoms in the Periodic Group 1 atoms become 1+ ions Table, Group discuss whether you can see a 2 atoms become 2+ ions connection between the group Group 6 atoms become 2 -ions number andbecome ion charge Group 7 atoms 1 -ions

This might prove useful…… At the start of your GCSE Chemistry exam, you could write 1+, 2 - and 1 - at the top of groups 1, 2, 6 and 7 of the Periodic table. These charges can come in very handy in a number of topics and one such topic is the writing of chemical formulae for ionic compounds…. .
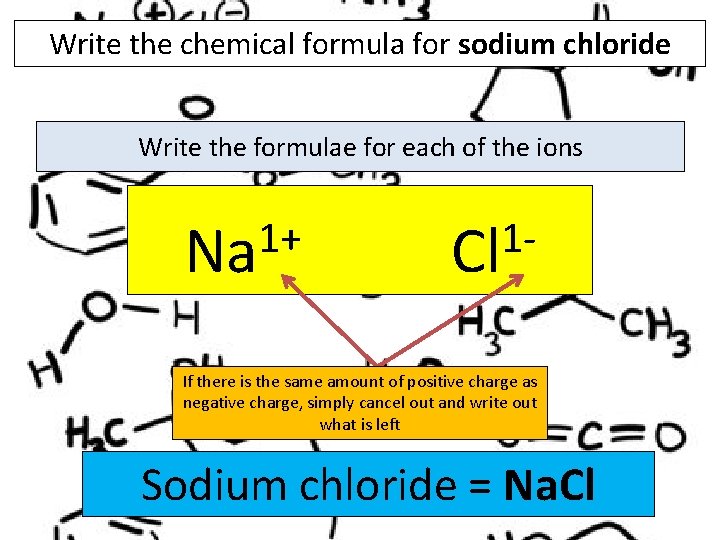
Write the chemical formula for sodium chloride Write the formulae for each of the ions 1+ Na 1 Cl If there is the same amount of positive charge as negative charge, simply cancel out and write out what is left Sodium chloride = Na. Cl
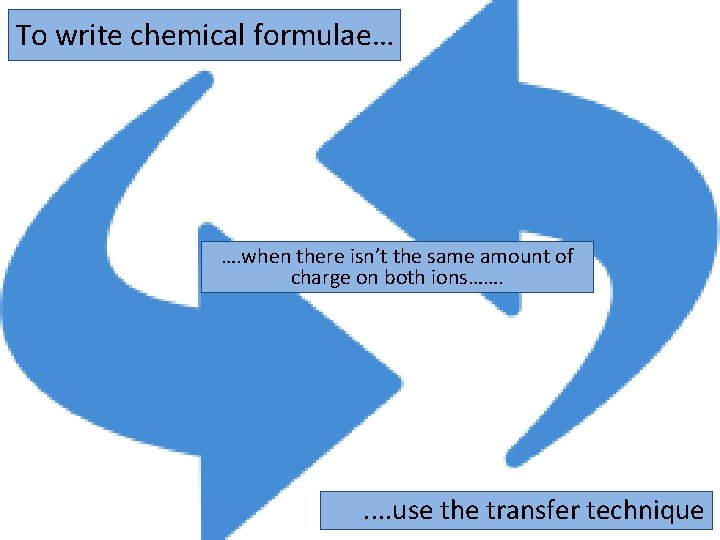
To write chemical formulae… …. when there isn’t the same amount of charge on both ions……. . . use the transfer technique
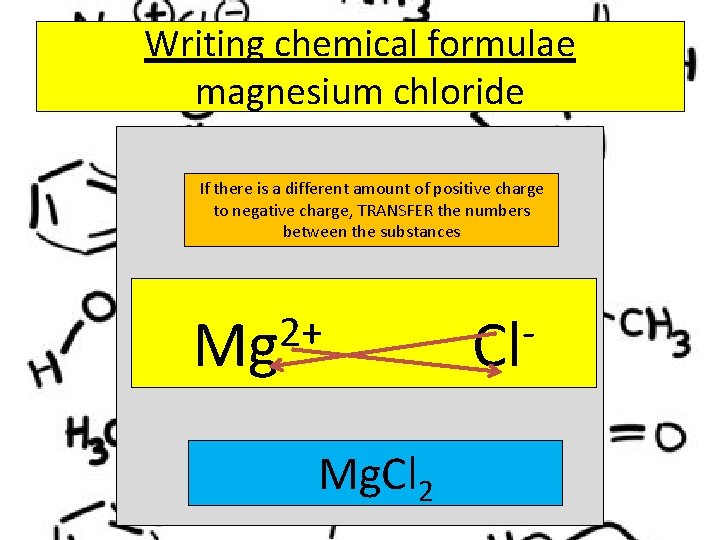
Writing chemical formulae magnesium chloride If there is a different amount of positive charge to negative charge, TRANSFER the numbers between the substances 2+ Mg Mg. Cl 2 Cl

EDEXCEL GCSE CS (FT) PAPER 3 REVISION QUIZ R 5 Round 5 will challenge you to apply what you’ve just recalled. So let’s see who is on FORM

“Are you on FORM? ” The name or formula of an ionic compound will appear on the board. • If the name appears, the 1 st person to buzz in with the correct formula will get the 2 TEAM POINTS! • If the chemical formula appears, the 1 st person to buzz in with the correct name will get the 2 TEAM POINTS!

“Are you on FORM? ” Ca. F 2 Calcium fluoride

“Are you on FORM? ” KBr Potassium bromide

“Are you on FORM? ” Na. I Sodium iodide

“Are you on FORM? ” Mg. Br 2 Magnesium bromide

“Are you on FORM? ” Li 2 O lithium oxide

Understanding check #11 Using the technique that you’ve just seen, work out the chemical formula for sodium oxide (1 mark)

“Are you on FORM? ” Na 2 O Sodium oxide

Understanding check #12 The chemical formula for sodium oxide is Na 2 O Which of the following is the correct number of atoms of each element in this compound? (1 mark) A 2 atoms of sodium and 1 atom of oxygen B 1 atom of sodium and 2 atoms of oxygen C 2 atoms of sodium and 2 atoms of oxygen D 2 atoms of sodium

KEY POINT The subscript number after a chemical symbol represents the number of atoms of that element in the compound

This is actually fairly important…… As you saw with the correct answer to the multiple-choice question, if there is no subscript number after the symbol then there is only 1 atom of that particular element in the compound

This police officer was laying down a (Chemistry) law when she was interrupted by a call on her radio… “No atoms are lost or made…. . Make sure that this PAPER 3 revision lesson keeps moving along just as it was by identifying this Chemistry law and trying to finish it off if

The law of conservation of mass states that no atoms are lost or made during a chemical reaction so the mass of the products equals the mass of the reactants

MAKE THE LINK! You have 5 minutes to discuss as a class The law of conservation of mass states that no atoms are lost or made during a chemical reaction so the mass of the products equals the mass of the reactants The law of the conservation of mass means that chemical symbol equations must balance What is the link between the law above ofsymbol the numbers of andin theterms chemical equation below? atoms of each element involved on both. Na sides equation + O 2 of the Na 2 O

KEY POINT To balance an equation, you put a number in front of a substance (written in normal script) and this is known as a multiplier

KEY POINT This multiplies all of the atoms in that substance by that number

There are 2 atoms of oxygen on the left hand side of this equation And only 1 atom of sodium on the left There is 1 atom of oxygen on the right hand side of this equation Now there are 4 atoms of sodium on the right 4 2 2 O …Na + …O 2 …Na So where should we put a number? And which number is needed to balance the equation?
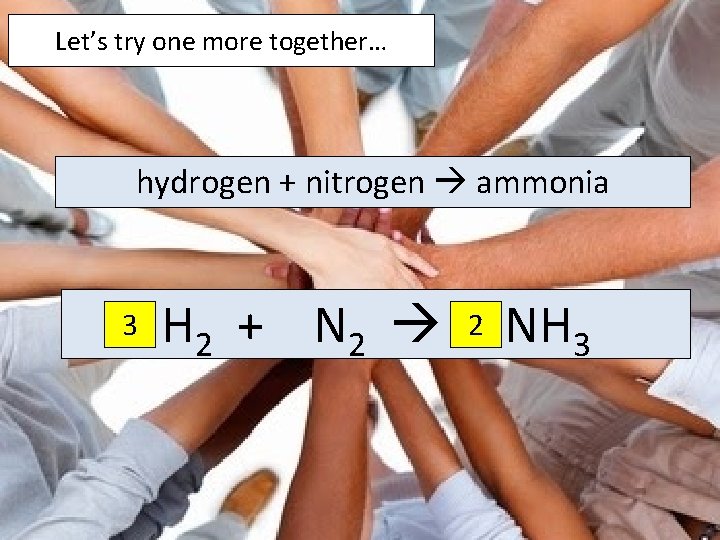
Let’s try one more together… hydrogen + nitrogen ammonia 3 H 2 + N 2 2 NH 3

Chemical symbol equations must balance by having the same numbers of atoms of each element on both sides of the equation. If an equation is not balanced, place multipliers in front of substances to multiply all of the atoms in that substance by that number until the equation balances Balance the 7 symbol equations on the worksheet The equations get slightly harder and harder as you go along. Start at number 1 but if you find that you are unable to do any, ask for a worksheet labelled STARS which has some of the multipliers added already
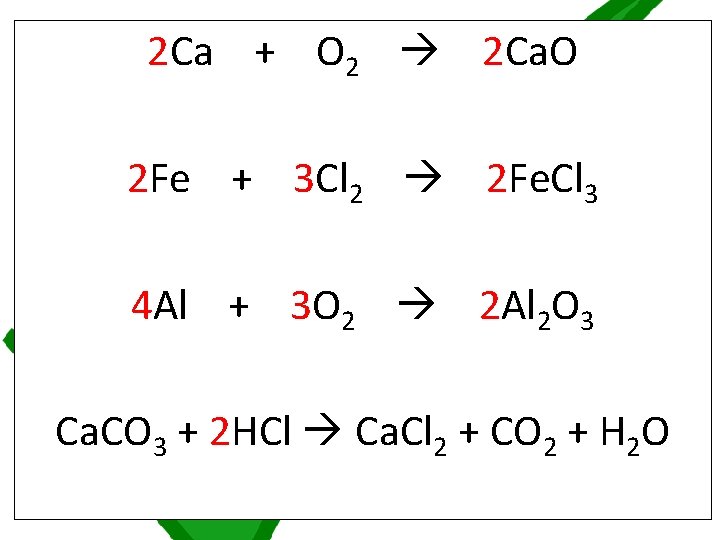
2 Ca + O 2 2 Ca. O 2 Fe + 3 Cl 2 2 Fe. Cl 3 4 Al + 3 O 2 2 Al 2 O 3 Ca. CO 3 + 2 HCl Ca. Cl 2 + CO 2 + H 2 O
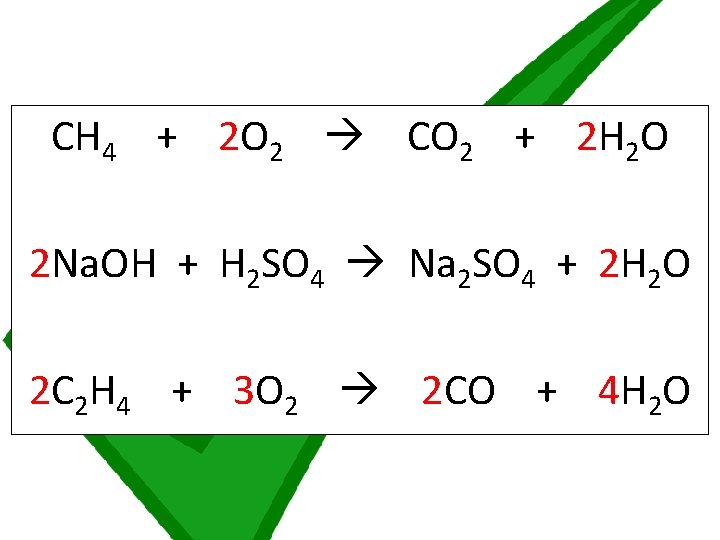
CH 4 + 2 O 2 CO 2 + 2 H 2 O 2 Na. OH + H 2 SO 4 Na 2 SO 4 + 2 H 2 O 2 C 2 H 4 + 3 O 2 2 CO + 4 H 2 O

3 0 19 62 17 4 72 48 20 21 11 6 Relative formula mass of Na 2 O 40 9 16 8 28 10 1 23 2 7 18 64 12

You need to use the mass number for each of the elements There is 1 atom of oxygen so O = 16 There are 2 atoms of sodium so Na 2 = 23 x 2 = 46 Na 2 O Mr of Na 2 O = (23 x 2) + 16 = 62

This is actually fairly important…… When you are normally asked to calculate a mass, your teacher will be screaming at you to remember to add a unit (g or kg). However, this is a relative formula mass and therefore no units are required.
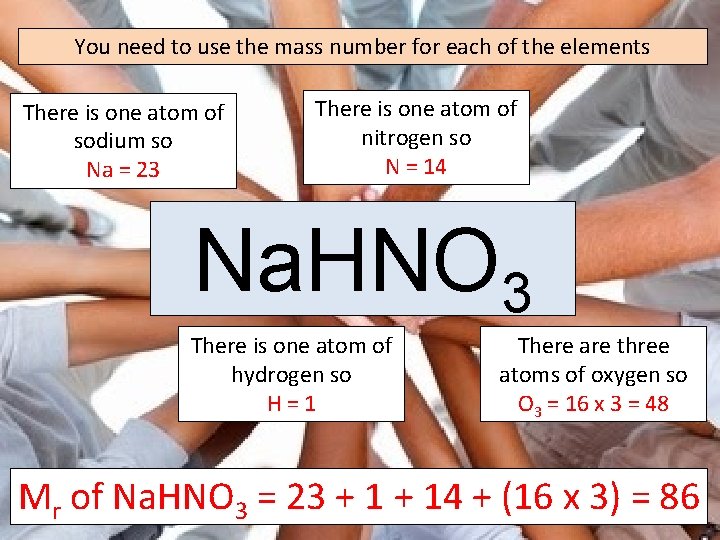
You need to use the mass number for each of the elements There is one atom of sodium so Na = 23 There is one atom of nitrogen so N = 14 Na. HNO 3 There is one atom of hydrogen so H=1 There are three atoms of oxygen so O 3 = 16 x 3 = 48 Mr of Na. HNO 3 = 23 + 14 + (16 x 3) = 86

Welcome to the RELATIVE FORMULA MASS OLYMPICS Calculate the RELATIVE FORMULA MASS of the compounds on the next slide and put them in order, from the lowest to the highest. For an extra challenge, try to name each of the compounds
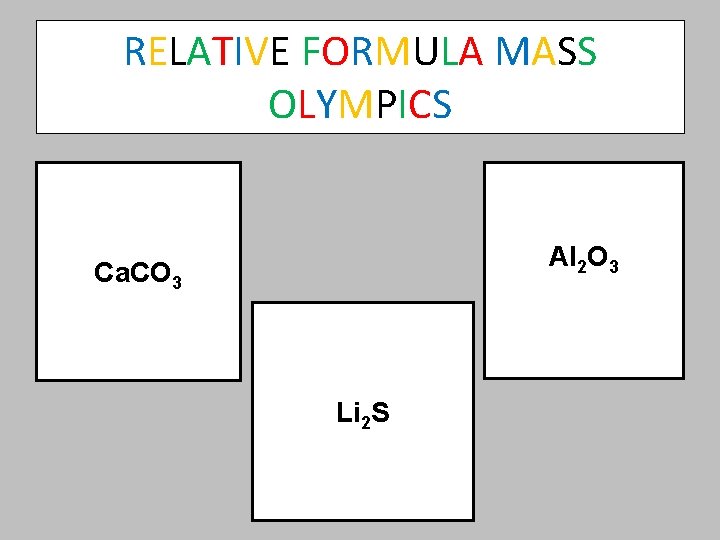
RELATIVE FORMULA MASS OLYMPICS Al 2 O 3 Ca. CO 3 Li 2 S
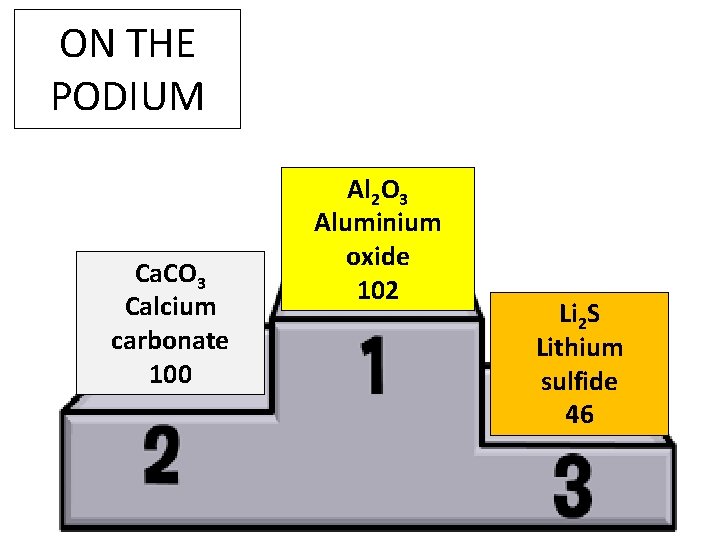
ON THE PODIUM Ca. CO 3 Calcium carbonate 100 Al 2 O 3 Aluminium oxide 102 Li 2 S Lithium sulfide 46

This might prove useful…… When trying to name compounds, counting the number of elements and remembering these rules could help: • If the compound contains only 2 elements, the name of the compound ends in –ide (e. g. Li 2 S was lithium sulphide) • If the compound contains more than 2 elements, it ends in –ate (e. g. Ca. CO 3 was calcium carbonate)

EDEXCEL GCSE CS (FT) PAPER 3 REVISION QUIZ R 6 The next round is an absolute classic that never gets old – it’s “HIGHER or LOWER”

HIGHER or LOWER Each team has a mini-whiteboard with “HIGHER” written on one side and “LOWER” written on the other side. A chemical formula will appear on the board and as a team, you have just 30 seconds to calculate the relative formula mass (Mr). At the end of the teacher’s countdown, each team with the correct side showing (HIGHER or LOWER) will get 1 POINT and the 2 nd point will be awarded to any teams with the exact mass also written on the whiteboard! HIGHER or LOWER

The starting number is 50

HIGHER than 50? 30 Li O Mr of 2 LOWER than 50?

HIGHER than 30? 18 H O Mr of 2 LOWER than 30?

HIGHER than 18? 44 CO Mr of 2 LOWER than 18?

HIGHER than 44? Mr of 160 Fe 2 O 3 LOWER than 44?

HIGHER than 160? Mr of 367 Pb. Br 2 LOWER than 160?

Understanding check #13 In the last competition, you found out that the relative formula mass of Pb. Br 2 was 367. By verbalising your answer to a partner, state the name of the compound with the chemical formula Pb. Br 2 and state whether it is an ionic or a covalent compound (2 marks) lead bromide is an ionic compound

EDEXCEL GCSE CS (FT) PAPER 3 REVISION QUIZ R 7 Round 7 and we are back for another BIG REVEAL. Just remember, if you’re the 1 st to recognise this key term, 2 TEAM POINTS will be yours!

THE BIG REVEAL When solid lead bromide is melted into a liquid, it forms molten lead bromide. This Eliquid R ____ Oelectricity L ____ Y and Tconduct L is____ Eable T ____ ____ Cto____ is. E therefore an example of an electrolyte Will you be the first to get it?

Understanding check #14 1. solid aluminium oxide 2. molten lead bromide 3. sodium chloride solution Which of these ionic compounds are electrolytes? (1 mark) A 1 only B 1 and 2 only C 2 and 3 only D 1, 2 and 3

This is actually fairly important…… As the ions are unable to move to the electrodes in a solid ionic compound, these substances cannot be split by electrolysis and therefore are not considered to be electrolytes.

KEY POINT An ionic compound must be melted (to form a molten ionic compound) or be dissolved in water (to form a solution) in order for the ions to be free to move to the cathode and anode during electrolysis

2 TO AGREE ON! You have 2 minutes to debate as a pair! The negative electrode The cathode and anode are the names for is the cathode the individual electrodes used in Once an ionic compound has been melted or dissolved in water, the ions are free to move to the cathode and anode electrolysis, but which one is which? Take a few minutes to make sure that you and your partner agree on the identity of the positive and the negative electrode The positive electrode is the anode

This might prove useful…… There are many ways to remember the key points in Science, and you have to find those that work for you. Here’s one that might help you to remember the different electrodes: • Cathy in Eastenders is forever moaning and therefore some would say she is a negative person (cathode = negative) • The musical Annie is about a little girl who has a difficult life but is always singing very positive songs (anode = positive)

GET MOVING! You have 2 minutes to discuss as a pair! In an electrolysis experiment, the negative electrode is the cathode and the positive electrode is the anode So once the electric current passes through the electrolyte, the ions will move to the electrodes. But which ions are will move to which electrode? You only have 2 minutes to discuss this question so get moving!

KEY POINT Passing an electric current through an electrolyte causes the positively-charged ions to move to the cathode and the negatively-charged ions to move to the anode

As you’ve just seen, knowing the charge of an ion will enable you to know which electrode it is attracted to, but it doesn’t end there…. . Example: 2+ Pb • As a lead ion is a positive ion, it moves to the negative electrode (cathode) • As it is a positive ion, it will gain electrons at the cathode • As it is a 2+ ion, it will gain 2 electrons at the cathode • As a result, the lead ions are discharged and the metal lead forms on the cathode

Understanding check #15 Bromide ions are found in the electrolyte during the electrolysis of molten lead bromide. Answer the following questions about these ions: 1. 2. 3. 4. 5. What is the charge of a bromide ion? Which electrode will the bromide ions move to? Will the bromide ions gain or lose electrons? How many electrons will the bromide ions gain or lose? What is final product at this electrode? CHALLENGE Try to write one sentence that will answer all five questions

During the electrolysis of molten lead bromide, the bromide ions (Br-) move to the anode (or positive electrode) where they will each lose 1 electron and the result is that the non-metal bromine (Br 2) forms

KEY POINT The products of the electrolysis of a molten ionic compound are a metal element at the cathode and a non-metal element at the anode

Answer question 1 on the electrolysis and extracting metals worksheet by completing the table with the missing ions, products or molten electrolyte If you have read through question 1 and don’t feel like you are able to begin, ask for a worksheet labelled STARS which gives some extra assistance
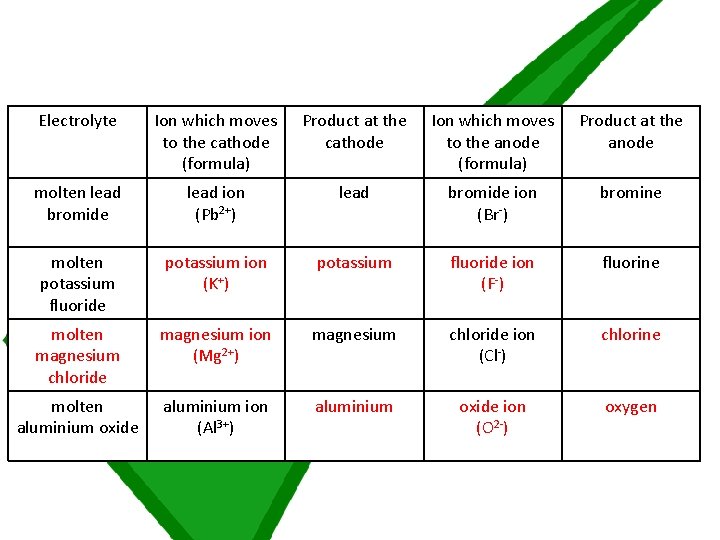
Electrolyte Ion which moves to the cathode (formula) Product at the cathode Ion which moves to the anode (formula) Product at the anode molten lead bromide lead ion (Pb 2+) lead bromide ion (Br-) bromine molten potassium fluoride potassium ion (K+) potassium fluoride ion (F-) fluorine molten magnesium chloride magnesium ion (Mg 2+) magnesium chloride ion (Cl-) chlorine molten aluminium oxide aluminium ion (Al 3+) aluminium oxide ion (O 2 -) oxygen

EDEXCEL GCSE CS (FT) PAPER 3 REVISION QUIZ R 8 Can you believe that it’s already time for ROUND 8? Well even if you don’t trust your memory, you had better trust your eyes as we play “WHAT EXACTLY IS THAT? ”. The 1 st team to name it gets the 2 TEAM POINTS
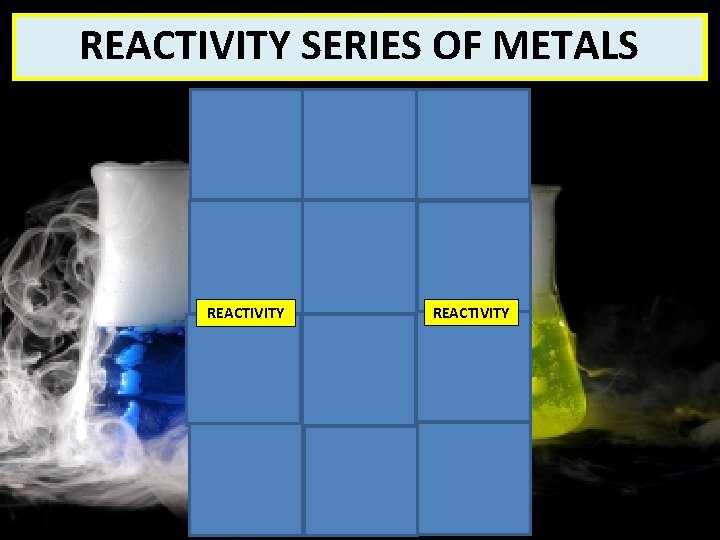
REACTIVITY METALS What SERIES exactly. OF is it? REACTIVITY Potassium Sodium Lithium Calcium Magnesium Aluminium Carbon REACTIVITY Zinc Iron Hydrogen Copper Silver Gold

Understanding check #16 By verbalising your answers with a partner, identify the two non-metals which are included in the reactivity series of metals shown to the right (2 marks) Potassium Sodium Lithium Calcium Magnesium Aluminium CARBON Carbon Zinc Iron HYDROGEN Hydrogen Copper Silver Gold

The non-metal, carbon, is included in the reactivity series because of its importance for the extraction of metals from their oxides Copy the reactivity series onto your worksheet and then challenge your understanding of the statement at the top by answering questions 2 and 3

Zinc can be extracted from zinc oxide by reacting it with carbon because zinc is less reactive than carbon. This is a reduction reaction because the carbon reduces the zinc oxide to zinc. As aluminium is above carbon in the reactivity series, it is more reactive than the non-metal which means that the carbon is unable to reduce the aluminium oxide to aluminium and so it has to be extracted by electrolysis

KEY POINT The metals found above carbon in the reactivity series (potassium, sodium, lithium, calcium, magnesium, aluminium) have to be extracted from their metal oxides by electrolysis

KEY POINT The metals found below carbon in the reactivity series can be extracted from their metal oxides by reacting them with carbon. These are reduction reactions as the metal oxide loses oxygen

This might prove useful…… Zinc could be extracted from zinc oxide by electrolysis but this extraction process requires large amounts of energy to melt the compounds and to produce the electrical current so the alternative of reduction with carbon is preferred when possible
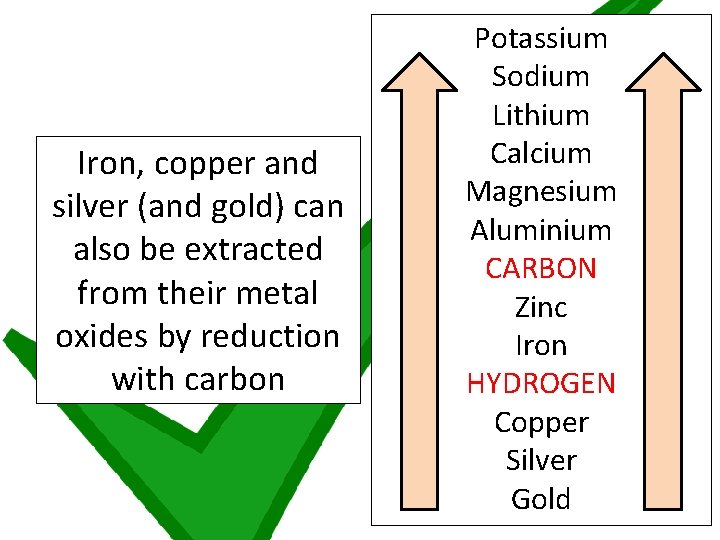
Iron, copper and silver (and gold) can also be extracted from their metal oxides by reduction with carbon Potassium Sodium Lithium Calcium Magnesium Aluminium CARBON Zinc Iron HYDROGEN Copper Silver Gold

1 TO THINK ABOUT! You have 4 minutes to discuss with 1 person! Iron, copper and silver (and gold) can also be extracted from their metal oxides by reduction with carbon Gold was written in brackets because it doesn’t tend to need to be extracted from its oxide. Use your understanding of the reactivity series to discuss why this word has been underlined

KEY POINT Gold is so unreactive that it tends to be found in the ground as the metal itself (and not as gold oxide)

EDEXCEL GCSE CS (FT) PAPER 3 REVISION QUIZ R 9 Simon Cowell knows that his famous singing contest is coming to an end so we thought we would make a slight alteration and call round 9, THE XTRACTION FACTOR!

THE TRACTION FACTOR

This is a face to face battle where a member of one team will battle against a member of another team. There will be three mini-whiteboards on the table between the competitors which say either “Electrolysis” or “Reduce with carbon” or “Found as itself in the ground”. The competitors must sit on their hands whilst they wait for the name of a metal to appear on the board. It is then the individual who is 1 st to pick up the correct sign and hold it aloft that gets the 2 TEAM POINTS. However, be wary of predicting the answer because anyone who holds a wrong sign aloft will lose 2 TEAM POINTS!

THE Reduced with ZINC carbon FACTOR

THE Electrolysis ALUMINIUM FACTOR

THE Reduced IRON with carbon FACTOR

THE Found as itself in the GOLD ground FACTOR

THE Reduced with COPPER carbon FACTOR

THE Electrolysis POTASSIUM FACTOR

THE Electrolysis LITHIUM FACTOR

THE Reduced with SILVER carbon FACTOR

The other non-metal in the reactivity series is hydrogen and its position is important when predicting the product at the cathode during the electrolysis of an aqueous solution. If the metal is above hydrogen in the reactivity series, hydrogen is produced at the cathode If the metal is below hydrogen in the reactivity series, the metal is produced at the cathode Potassium Sodium Lithium Calcium Magnesium Aluminium Carbon Zinc Iron HYDROGEN Copper Silver Gold

This is also fairly important…… During the electrolysis of an aqueous solution, oxygen is produced at the anode unless the solution contains halide ions and then the halogen will be produced

Challenge your ability to apply these rules about aqueous solutions by completing the table in question 4 and predicting the products at the cathode and anode

Electrolyte Predicted product at the cathode Predicted product at the anode copper sulfate solution copper oxygen sodium chloride solution hydrogen chlorine In addition to hydrogen and chlorine at the electrodes, the electrolysis of sodium chloride solution also produces an alkali called sodium hydroxide

3 0 19 62 17 14 72 48 20 21 Range on the p. H scale which represent the alkali solutions 11 40 9 10 1 23 2 7 18 6 16 8 28 64 12

EDEXCEL GCSE CS (FT) PAPER 3 REVISION QUIZ R 10 The final round of this PAPER 3 REVISION QUIZ is BLOCKBUSTERS and there a huge number of TEAM POINTS available so all is still to play for!

B L O C K U S T E R S On the next slide, 18 hexagons are filled with different letters and these represent the 1 st letter of the answer to a PAPER 3 based question. Beginning with the hexagon in the top left corner, the teacher will read out a clue and it’s the 1 st team to buzz in with the correct answer that wins the POINTS as shown by their colour on the right. The student who provided the correct answer can then choose the next hexagon and all teams will compete to get those POINTS. Of course, speed is important but if you interrupt the clue before it has finished and get it wrong, you will lose that number of POINTS! 1 2 3
B L O U S T E M R S AM C GC K H PI FOSOE MC M D G CE R N S IS PPM C U. I 1 2 3

So that’s it, the competition is over and the points have been tallied. This year’s PAPER 3 REVISION QUIZ CHAMPIONS are. . . PAPER 3 CHAMPIONS

Whether you are part of the winning team or in a team that came slightly lower down the ladder, the real winners will be those who take note of all of the KEY POINTS and the helpful hints and uses them in their revision! Best of luck for your exams!
- Slides: 172