Gibbs Phase Rule Eutectic Phase Diagram Ag Cu
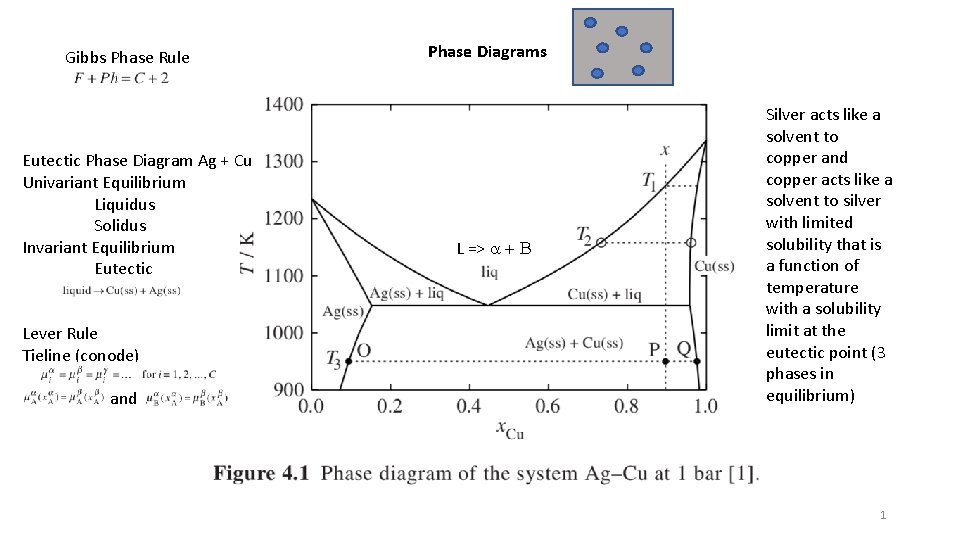
Gibbs Phase Rule Eutectic Phase Diagram Ag + Cu Univariant Equilibrium Liquidus Solidus Invariant Equilibrium Eutectic Lever Rule Tieline (conode) and Phase Diagrams L => a + B Silver acts like a solvent to copper and copper acts like a solvent to silver with limited solubility that is a function of temperature with a solubility limit at the eutectic point (3 phases in equilibrium) 1
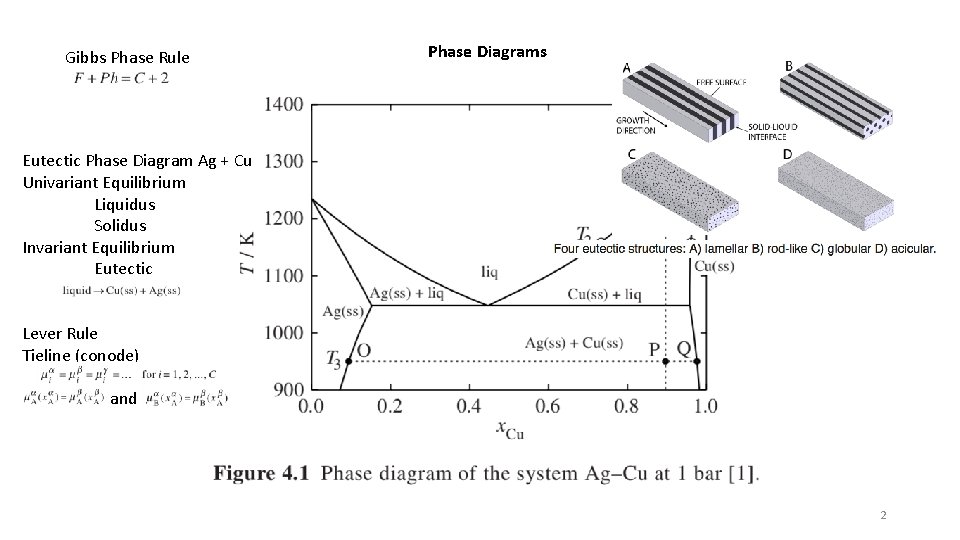
Gibbs Phase Rule Phase Diagrams Eutectic Phase Diagram Ag + Cu Univariant Equilibrium Liquidus Solidus Invariant Equilibrium Eutectic Lever Rule Tieline (conode) and 2
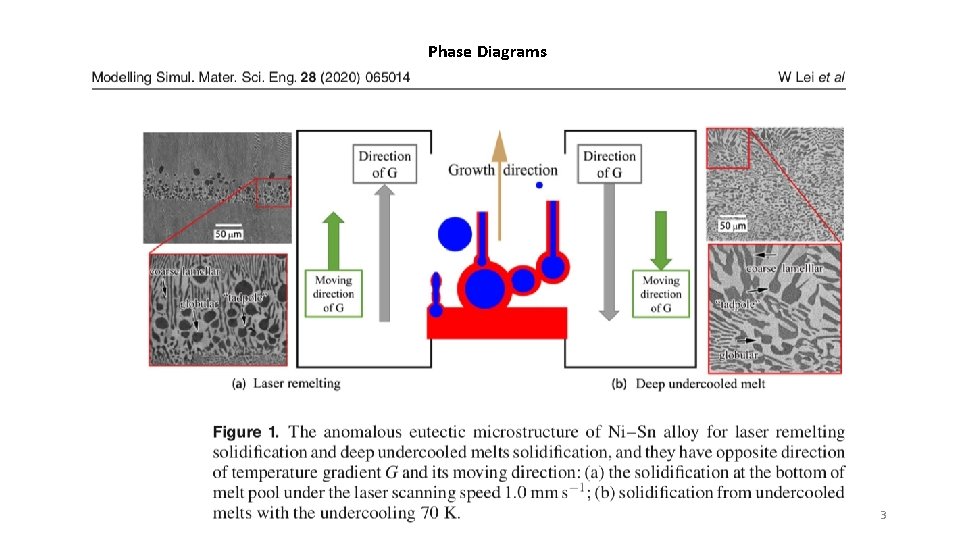
Phase Diagrams 3
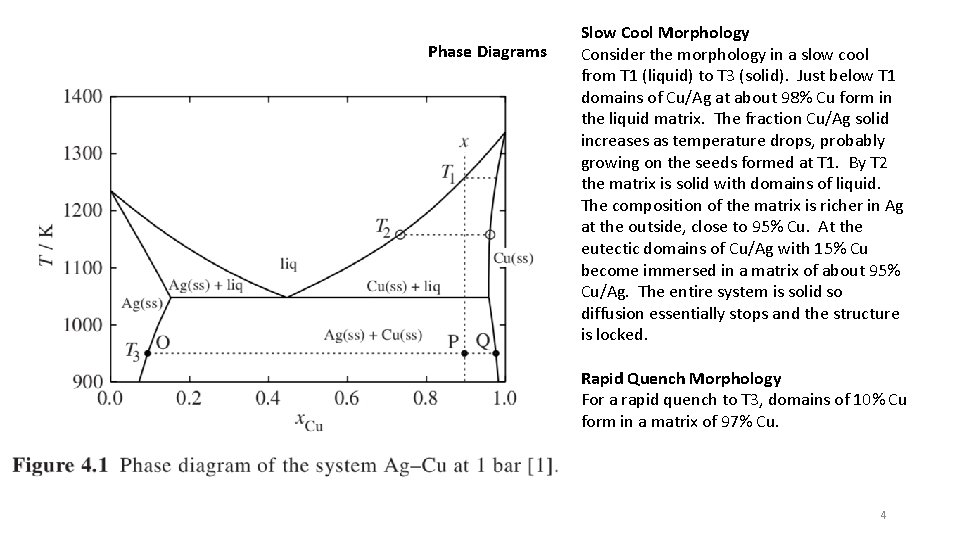
Phase Diagrams Slow Cool Morphology Consider the morphology in a slow cool from T 1 (liquid) to T 3 (solid). Just below T 1 domains of Cu/Ag at about 98% Cu form in the liquid matrix. The fraction Cu/Ag solid increases as temperature drops, probably growing on the seeds formed at T 1. By T 2 the matrix is solid with domains of liquid. The composition of the matrix is richer in Ag at the outside, close to 95% Cu. At the eutectic domains of Cu/Ag with 15% Cu become immersed in a matrix of about 95% Cu/Ag. The entire system is solid so diffusion essentially stops and the structure is locked. Rapid Quench Morphology For a rapid quench to T 3, domains of 10% Cu form in a matrix of 97% Cu. 4
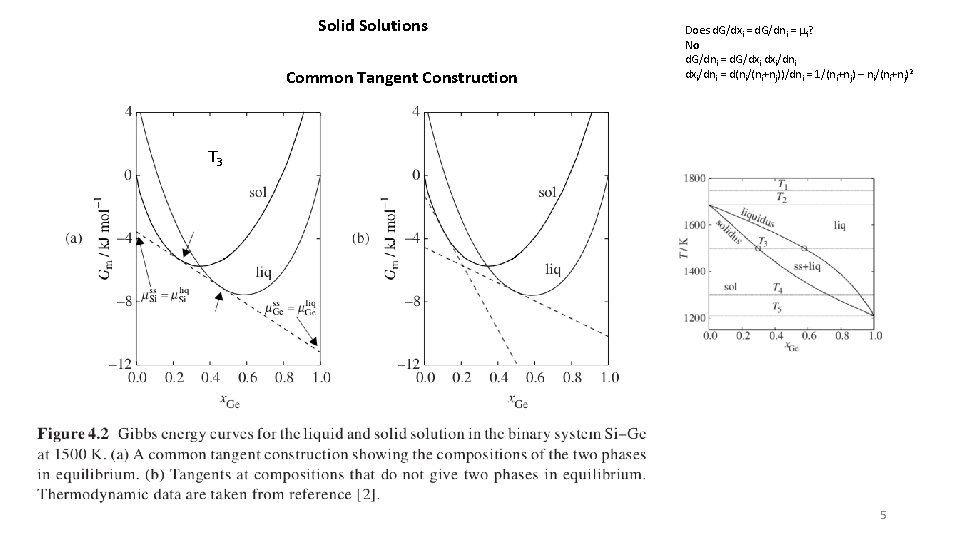
Solid Solutions Common Tangent Construction Does d. G/dxi = d. G/dni = mi? No d. G/dni = d. G/dxi dxi/dni = d(ni/(ni+nj))/dni = 1/(ni+nj) – ni/(ni+nj)2 T 3 5
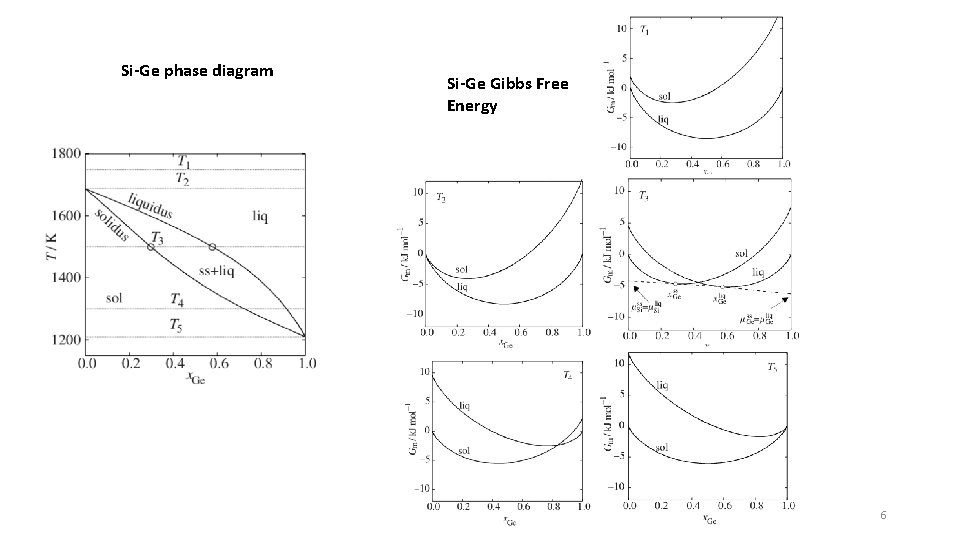
Si-Ge phase diagram Si-Ge Gibbs Free Energy 6
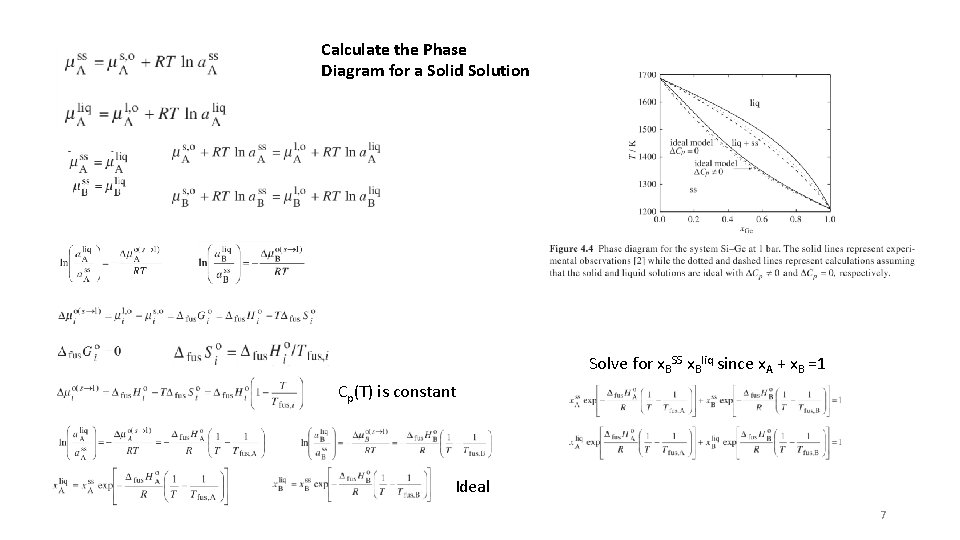
Calculate the Phase Diagram for a Solid Solution Solve for x. BSS x. Bliq since x. A + x. B =1 Cp(T) is constant Ideal 7
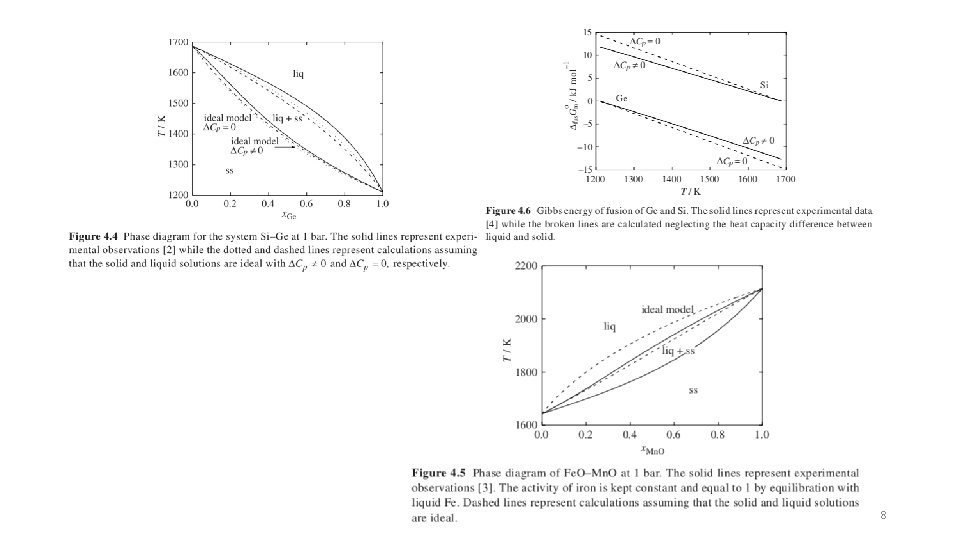
8
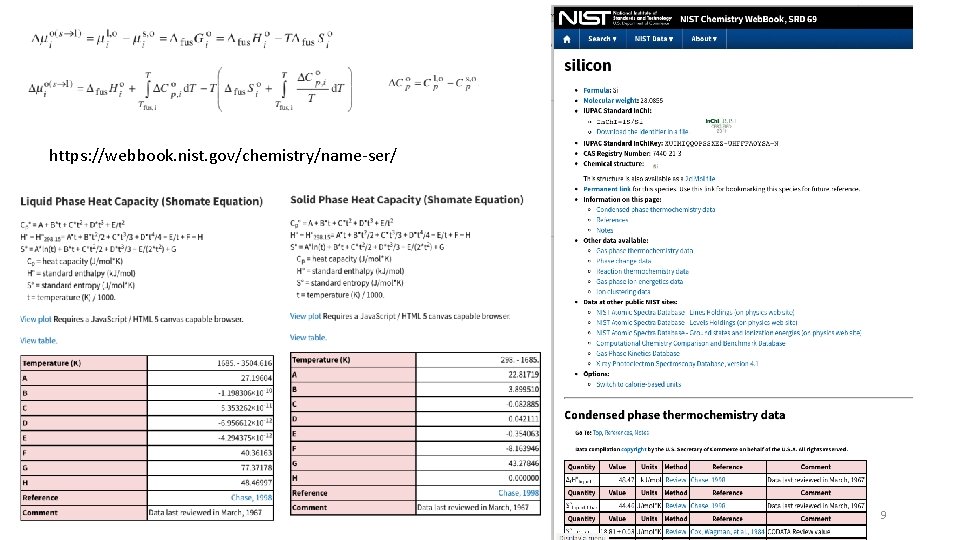
https: //webbook. nist. gov/chemistry/name-ser/ 9
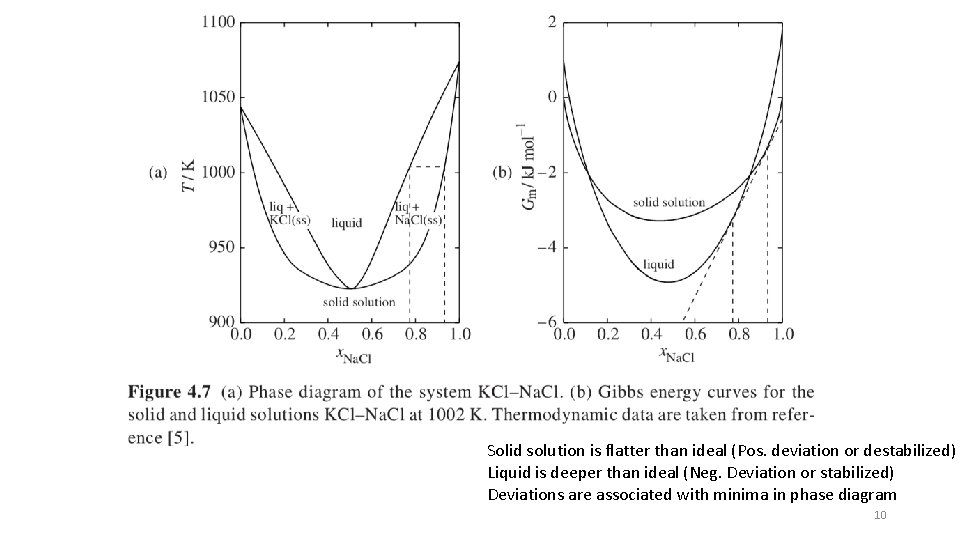
Solid solution is flatter than ideal (Pos. deviation or destabilized) Liquid is deeper than ideal (Neg. Deviation or stabilized) Deviations are associated with minima in phase diagram 10
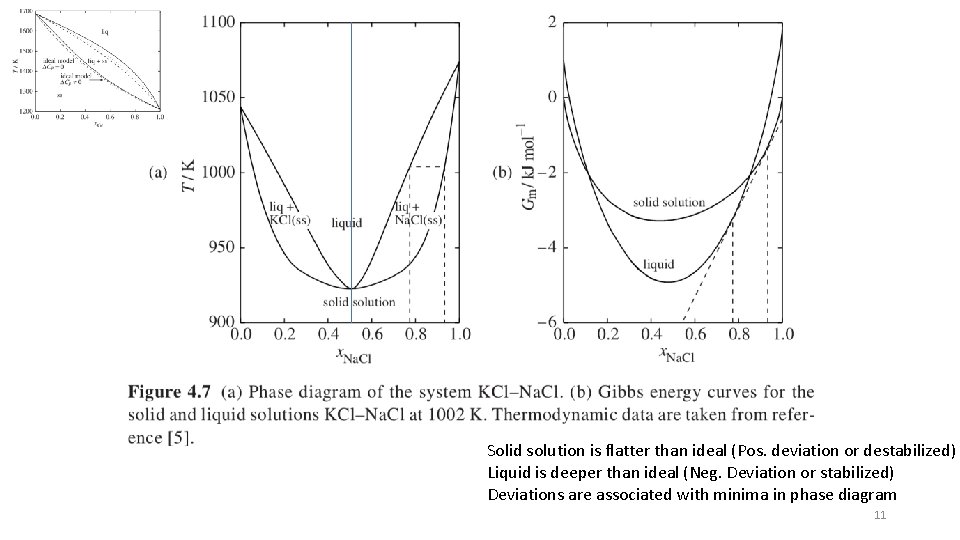
Solid solution is flatter than ideal (Pos. deviation or destabilized) Liquid is deeper than ideal (Neg. Deviation or stabilized) Deviations are associated with minima in phase diagram 11
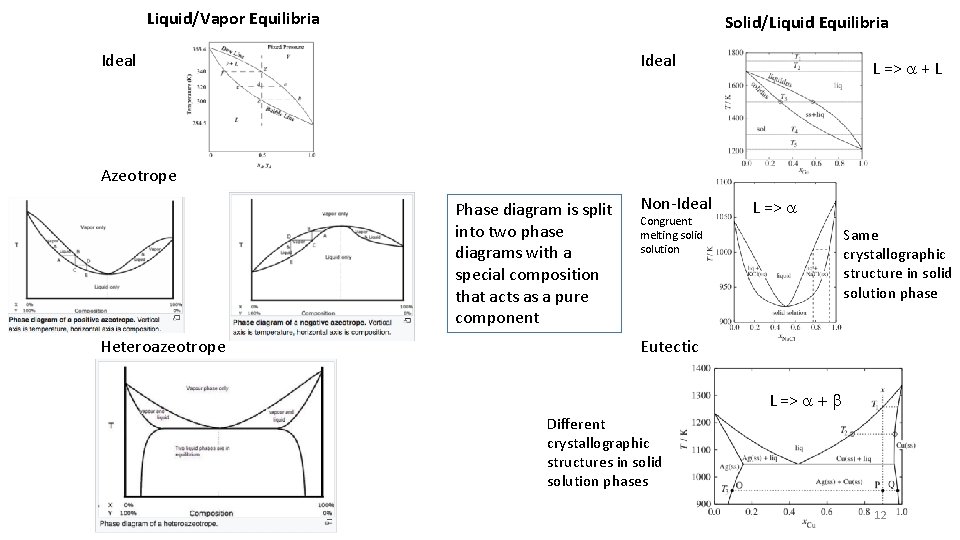
Liquid/Vapor Equilibria Solid/Liquid Equilibria Ideal L => a + L Azeotrope Phase diagram is split into two phase diagrams with a special composition that acts as a pure component Heteroazeotrope Non-Ideal Congruent melting solid solution L => a Same crystallographic structure in solid solution phase Eutectic L => a + b Different crystallographic structures in solid solution phases 12
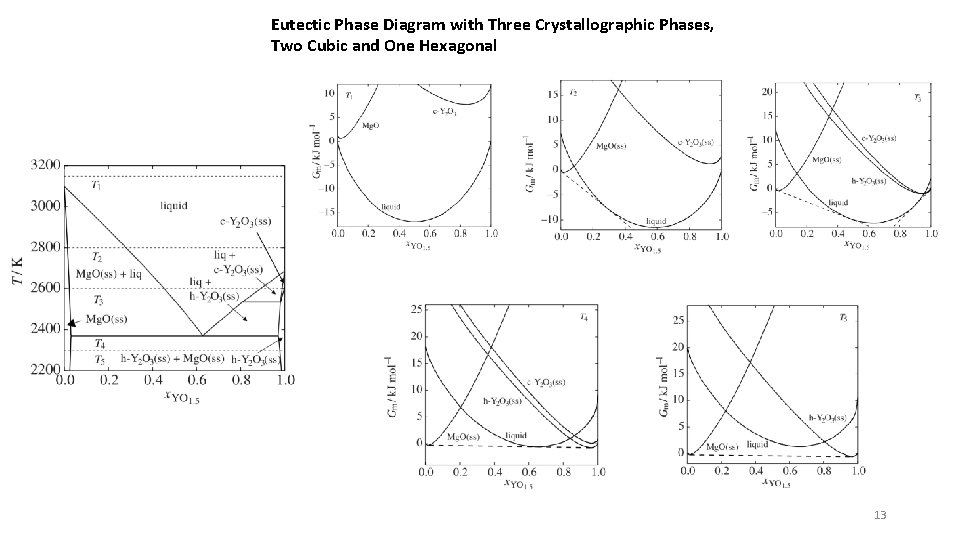
Eutectic Phase Diagram with Three Crystallographic Phases, Two Cubic and One Hexagonal 13

Regular Solution Modeling for Phase Diagrams GE/RT = x. A ln g. A + x. B ln g. B Generic expression using activity coefficient GE/RT = W x. A x. B Hildebrand Regular Solution expression Two unknowns & Two equations: We found that a solution of RT ln g. A = W x. B 2 and RT ln g. B = W x. A 2 worked Two unknowns, x. BSS, x. Bliq & Two equations Solve numerically Generate Phase diagram since Dµ is a function of T Positive W is necessary for phase separation DGm = RT(x. A ln(x. A) + x. B ln(x. B)) + W x. Ax. B 14
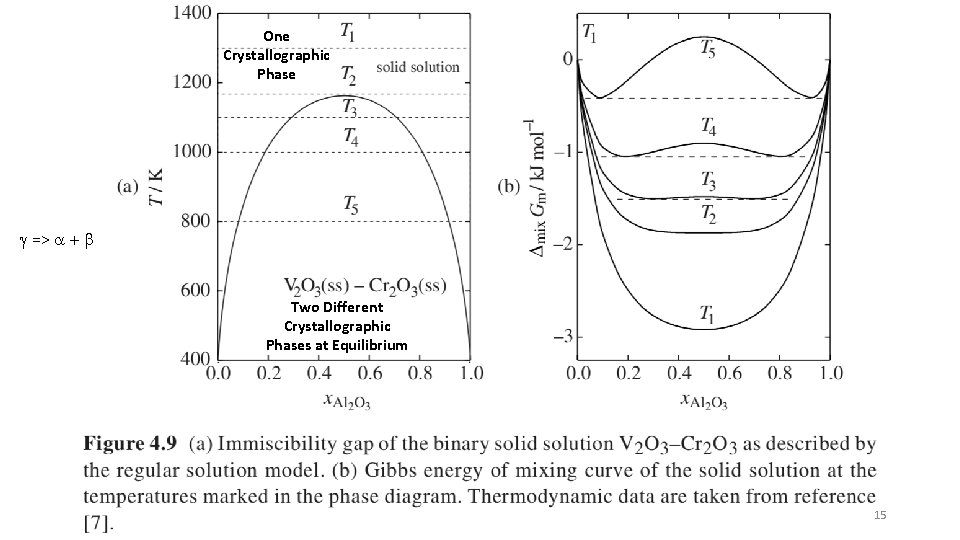
One Crystallographic Phase g => a + b Two Different Crystallographic Phases at Equilibrium 15
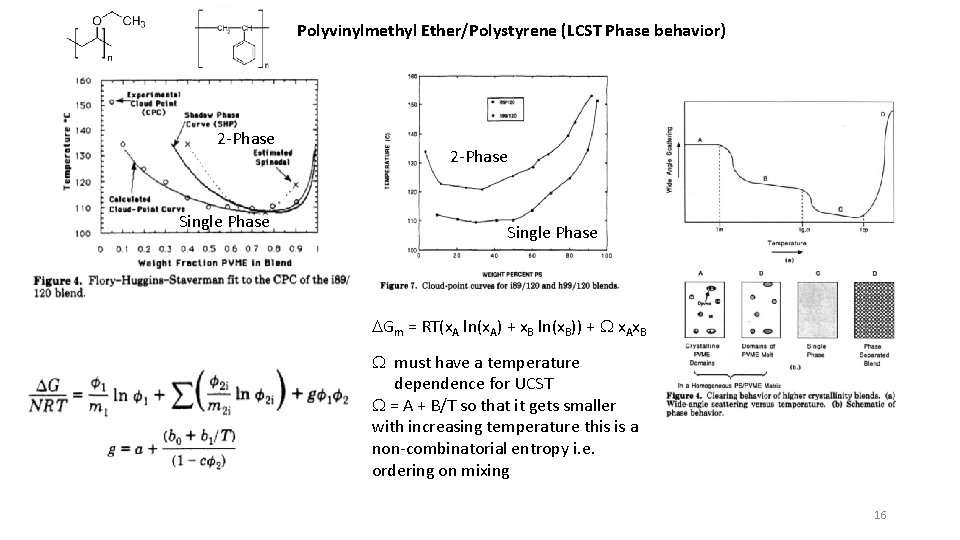
Polyvinylmethyl Ether/Polystyrene (LCST Phase behavior) 2 -Phase Single Phase DGm = RT(x. A ln(x. A) + x. B ln(x. B)) + W x. Ax. B W must have a temperature dependence for UCST W = A + B/T so that it gets smaller with increasing temperature this is a non-combinatorial entropy i. e. ordering on mixing 16
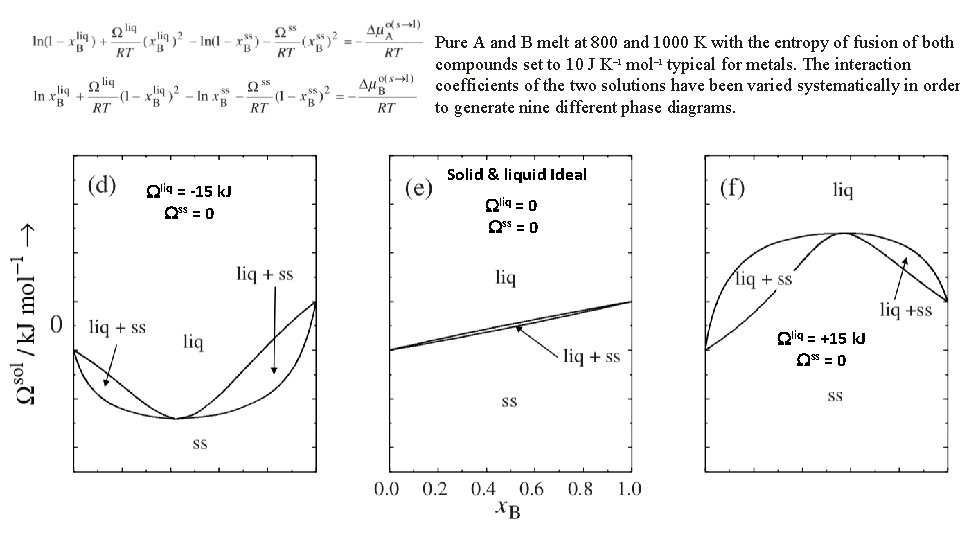
Pure A and B melt at 800 and 1000 K with the entropy of fusion of both compounds set to 10 J K– 1 mol– 1 typical for metals. The interaction coefficients of the two solutions have been varied systematically in order to generate nine different phase diagrams. Wliq = -15 k. J Wss = 0 Solid & liquid Ideal Wliq = 0 Wss = 0 Wliq = +15 k. J Wss = 0 17
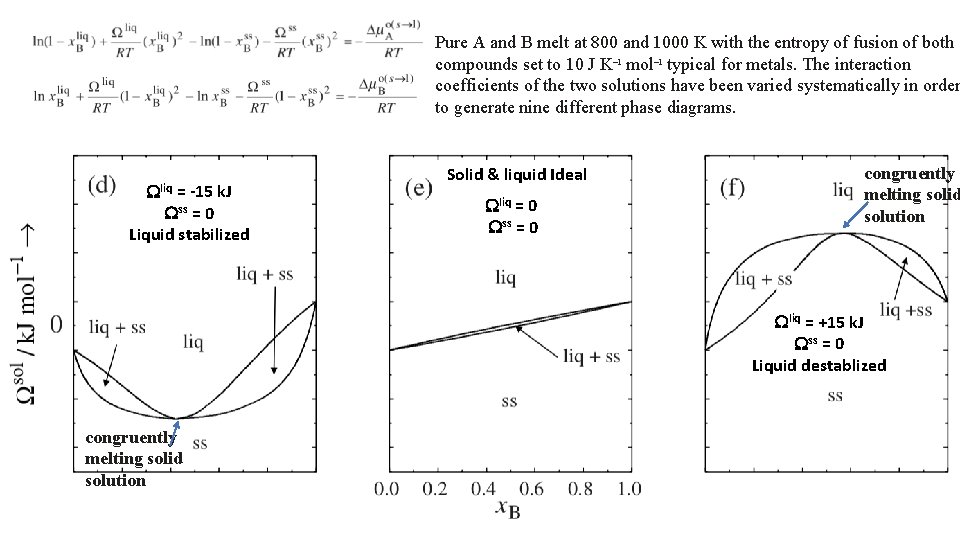
Pure A and B melt at 800 and 1000 K with the entropy of fusion of both compounds set to 10 J K– 1 mol– 1 typical for metals. The interaction coefficients of the two solutions have been varied systematically in order to generate nine different phase diagrams. Wliq = -15 k. J Wss = 0 Liquid stabilized Solid & liquid Ideal Wliq = 0 Wss = 0 congruently melting solid solution Wliq = +15 k. J Wss = 0 Liquid destablized congruently melting solid solution 18
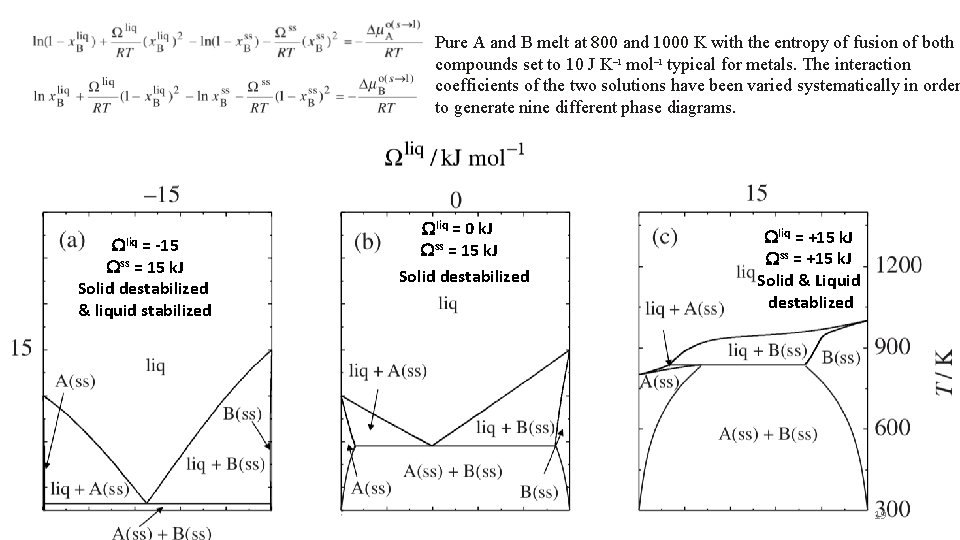
Pure A and B melt at 800 and 1000 K with the entropy of fusion of both compounds set to 10 J K– 1 mol– 1 typical for metals. The interaction coefficients of the two solutions have been varied systematically in order to generate nine different phase diagrams. Wliq = -15 Wss = 15 k. J Solid destabilized & liquid stabilized Wliq = 0 k. J Wss = 15 k. J Solid destabilized Wliq = +15 k. J Wss = +15 k. J Solid & Liquid destablized 19
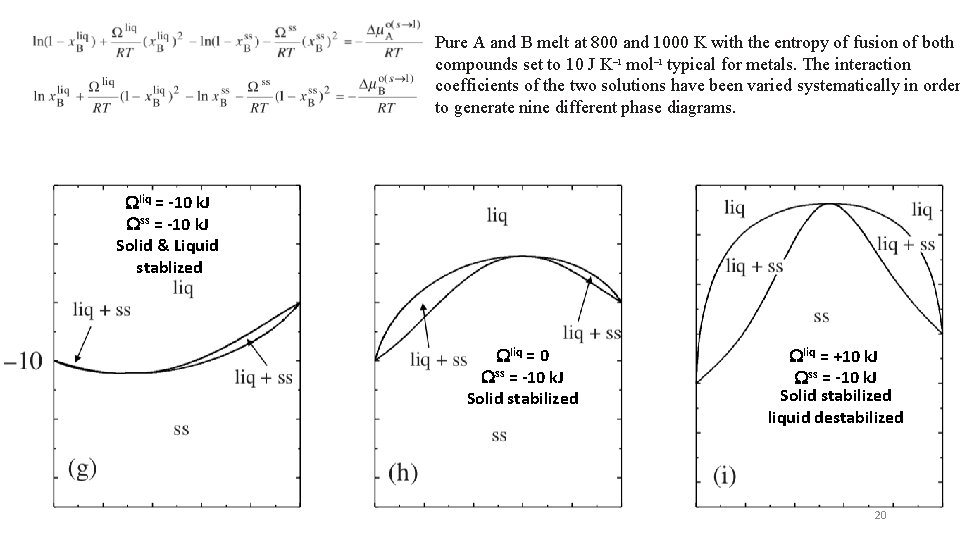
Pure A and B melt at 800 and 1000 K with the entropy of fusion of both compounds set to 10 J K– 1 mol– 1 typical for metals. The interaction coefficients of the two solutions have been varied systematically in order to generate nine different phase diagrams. Wliq = -10 k. J Wss = -10 k. J Solid & Liquid stablized Wliq = 0 Wss = -10 k. J Solid stabilized Wliq = +10 k. J Wss = -10 k. J Solid stabilized liquid destabilized 20

If it is assumed that Then you need only one W (The solid state doesn’t mix) Regular solution model has only a few parameters This means that experimentally you need only determine a few points of equilibrium composition to predict the entire phase diagram. 21
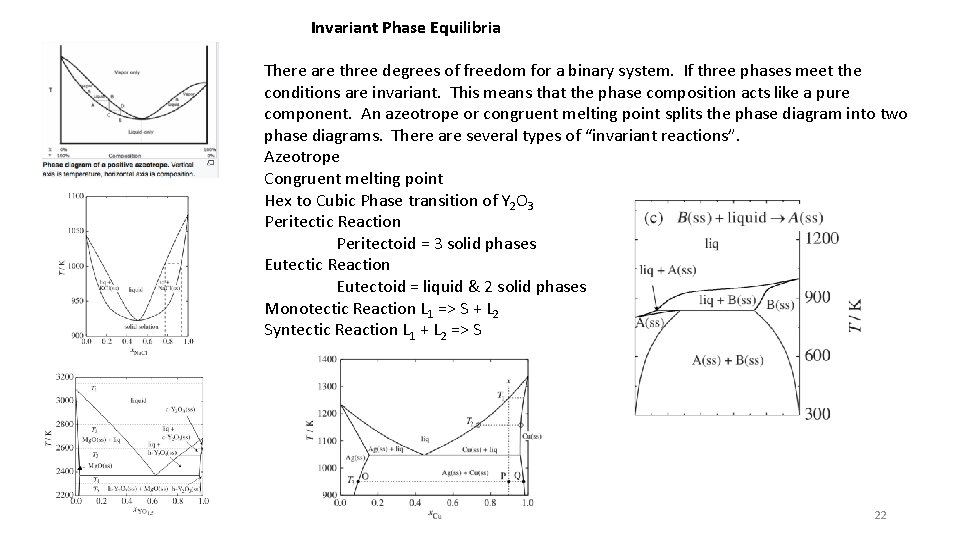
Invariant Phase Equilibria There are three degrees of freedom for a binary system. If three phases meet the conditions are invariant. This means that the phase composition acts like a pure component. An azeotrope or congruent melting point splits the phase diagram into two phase diagrams. There are several types of “invariant reactions”. Azeotrope Congruent melting point Hex to Cubic Phase transition of Y 2 O 3 Peritectic Reaction Peritectoid = 3 solid phases Eutectic Reaction Eutectoid = liquid & 2 solid phases Monotectic Reaction L 1 => S + L 2 Syntectic Reaction L 1 + L 2 => S 22
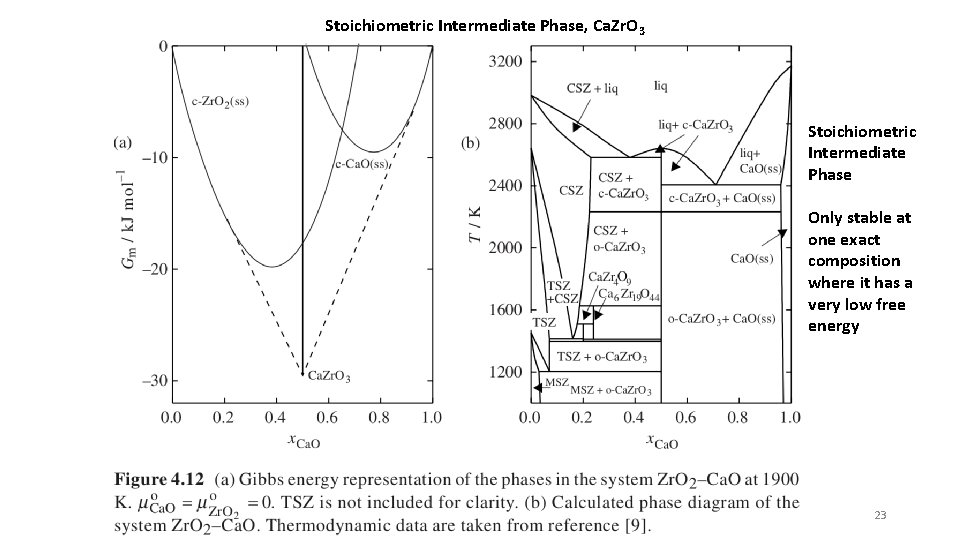
Stoichiometric Intermediate Phase, Ca. Zr. O 3 Stoichiometric Intermediate Phase Only stable at one exact composition where it has a very low free energy 23
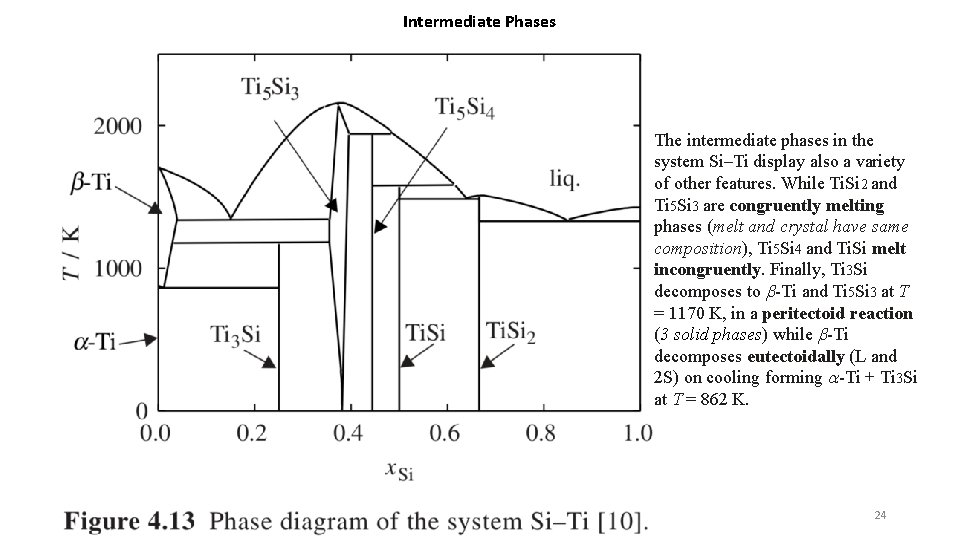
Intermediate Phases The intermediate phases in the system Si–Ti display also a variety of other features. While Ti. Si 2 and Ti 5 Si 3 are congruently melting phases (melt and crystal have same composition), Ti 5 Si 4 and Ti. Si melt incongruently. Finally, Ti 3 Si decomposes to b-Ti and Ti 5 Si 3 at T = 1170 K, in a peritectoid reaction (3 solid phases) while b-Ti decomposes eutectoidally (L and 2 S) on cooling forming a-Ti + Ti 3 Si at T = 862 K. 24
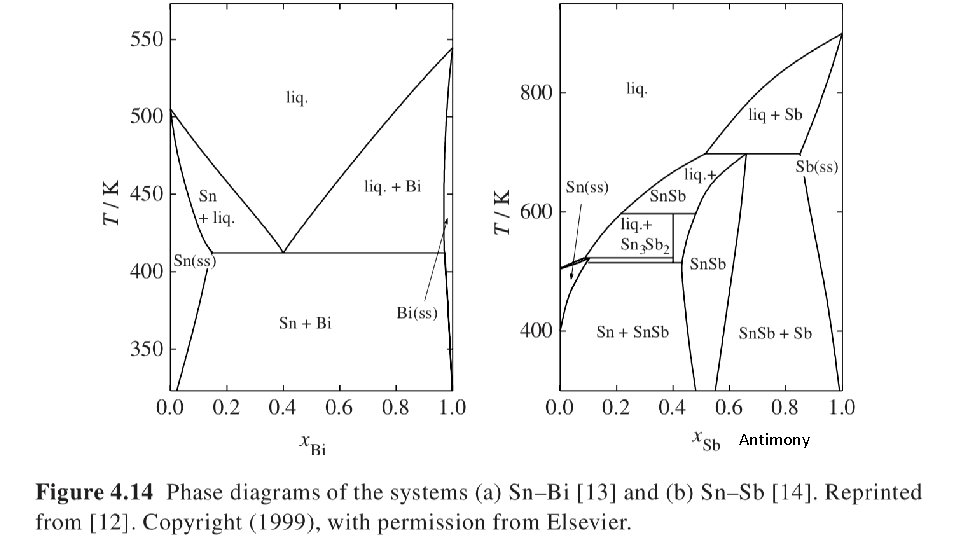
Antimony 25
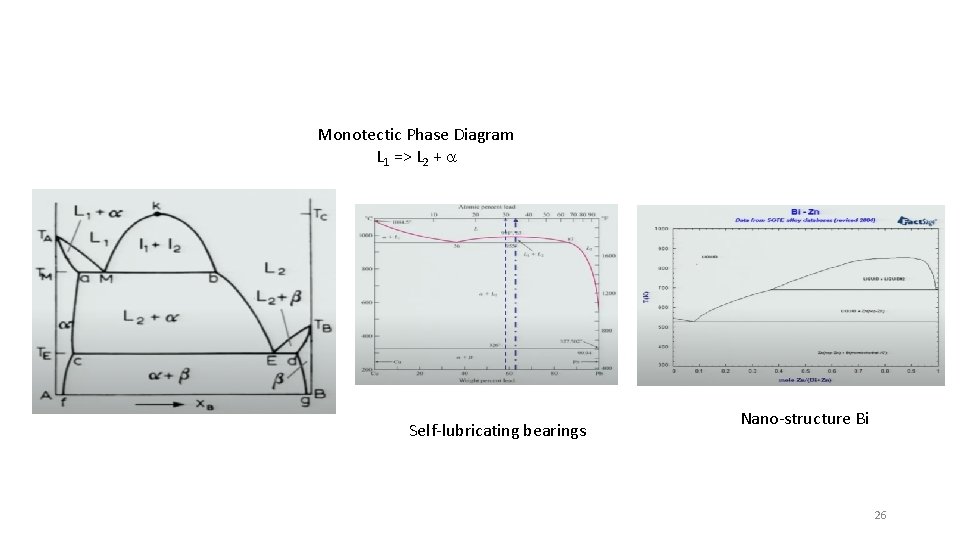
Monotectic Phase Diagram L 1 => L 2 + a Self-lubricating bearings Nano-structure Bi 26

Freezing Point Depression Pure solid in equilibrium with a binary solution following Henry’s Law dµB, Solid = Vm. B, Solid d. P – Sm. B, Solid d. T + RT d(lna. B, Solid) Isobaric, pure component B so lna. B, Solid = 0 dµB, Solid = – Sm. B, Solid d. Tfp Binary solution following Henry’s Law dµB, Solution = – Sm. B, Solultion d. Tfp + RTfp d(lny. B, Solution)P, T For small x: e-x = 1 - x + … or ln(1 -x) = -x So for small y. B, Solution: lny. B, Solution ~ -y. A, Solution So, Sm. B, Solid d. Tfp = Sm. B, Solution d. Tfp + RTfp dy. A, Solution = (Sm. B, Solid - Sm. B, Solution)/(RTfp) d. Tfp ~ -DSm. B/(RTfp) d. Tfp = -DHm. B/(RTF) (d. Tfp)/Tfp y. A, Solution = -DHm. B/(RTF) ln(Tfp/TF) For small x: ln(x) = x – 1 y. A, Solution = -DHm. B/(RTF) (Tfp/TF - 1) = -DHm. B/(RT 2 F) DT Tfp = TF - y. A, Solution. RT 2 F/DHm. B 27

DT = -RT 2 Fy. A, Solution/ DHm. B 28
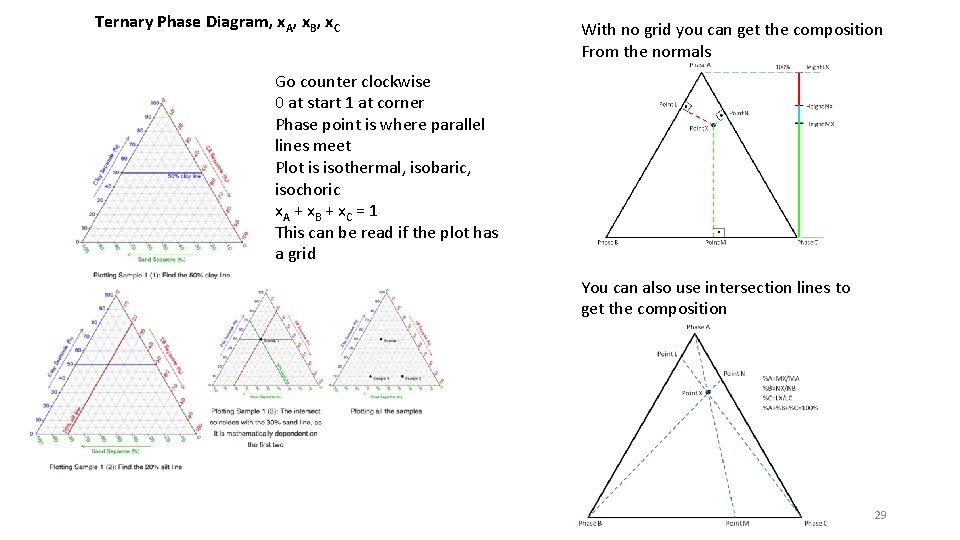
Ternary Phase Diagram, x. A, x. B, x. C With no grid you can get the composition From the normals Go counter clockwise 0 at start 1 at corner Phase point is where parallel lines meet Plot is isothermal, isobaric, isochoric x. A + x B + x C = 1 This can be read if the plot has a grid You can also use intersection lines to get the composition 29
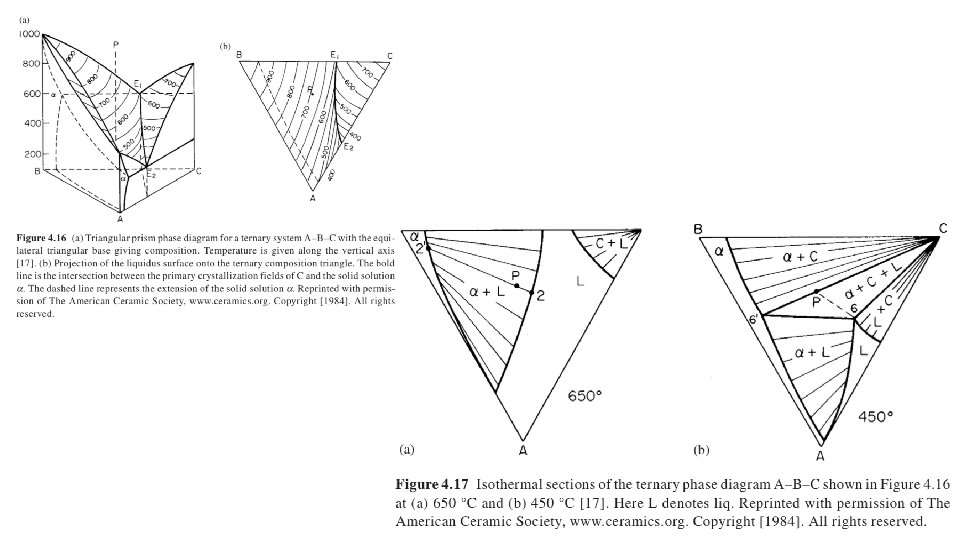
30
![Predominance Diagram (Which species is dominant) p. H = -log [H+] p. Cr = Predominance Diagram (Which species is dominant) p. H = -log [H+] p. Cr =](http://slidetodoc.com/presentation_image_h2/522848925e3454ecd2efb56ce04b7aea/image-31.jpg)
Predominance Diagram (Which species is dominant) p. H = -log [H+] p. Cr = -log [Cr] 31

32
- Slides: 32