Gibbs Free Energy Thermodynamics Lecture 3 Gibbs Free

Gibb’s Free Energy Thermodynamics Lecture 3

Gibb’s Free Energy (ΔG) • Energy associated with a chemical reaction that can be used to do work • Value tells whether a reaction is spontaneous or not – Spontaneous: reaction that occurs without being driven by some outside force – Two driving forces for chemical reactions: • ΔS= entropy (disorder) • ΔH= enthalpy (heat)

ΔG= ΔH- TΔS • If ΔG is negative – Reaction is spontaneous • If ΔG is positive – Reaction is non spontaneous (work must be done to make the reaction occur)

Question 1 Is ice melting at a temperature less than 0°C a spontaneous reaction? H 2 O (s) H 2 O (l) ΔG= ΔH- TΔS • Melting is endothermic reaction so ΔH is positive • T is low • Entropy increases as you go from solid to liquid

Question 2 • Is condensation at a temperature less than 100 °C a spontaneous reaction? H 2 O (g) H 2 O (l) ΔG= ΔH- TΔS • Condensation is exothermic reaction so ΔH is negative • T is low • Entropy decreases as you go from gas to liquid so it is a negative value
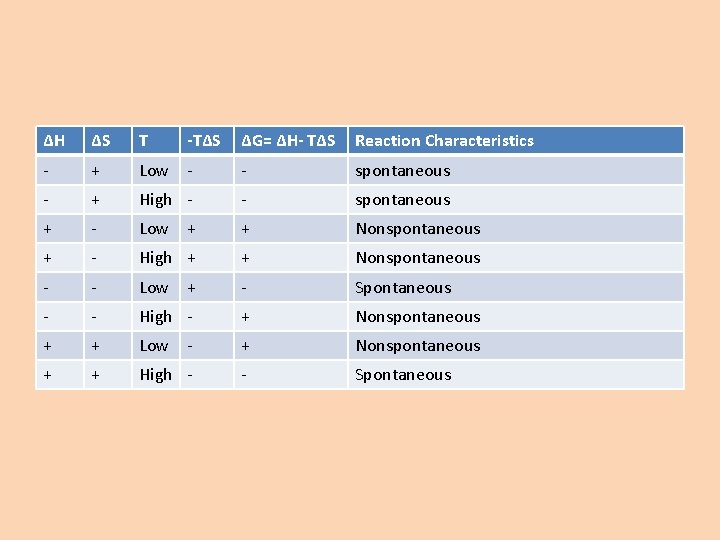
ΔH ΔS T -TΔS ΔG= ΔH- TΔS Reaction Characteristics - + Low - - spontaneous - + High - - spontaneous + - Low + + Nonspontaneous + - High + + Nonspontaneous - - Low + - Spontaneous - - High - + Nonspontaneous + + Low - + Nonspontaneous + + High - - Spontaneous
- Slides: 6