Gibbs Free Energy Temperature and Spontaneity Free Energy

Gibbs Free Energy Temperature and Spontaneity Free Energy and Equilibrium Thermodynamics of Living Systems 1

Thermodynamics Second Law of Thermodynamics Nonspontaneous process: Suniv < 0 tem Suniv = 0 sy s Equilibrium : un d Suniv > 0 su rro Spontaneous process: ing The entropy of the universe increases in a spontaneous process and remains unchanged in an equilibrium process. If something happens the total entropy of the universe increases! All we need to know is Suniv 2
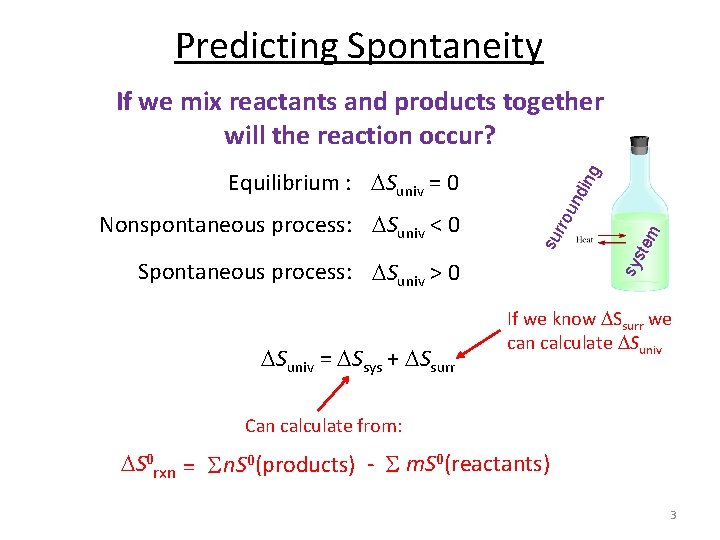
Predicting Spontaneity sy s Spontaneous process: Suniv > 0 Suniv = Ssys + Ssurr tem din rro un Nonspontaneous process: Suniv < 0 su Equilibrium : Suniv = 0 g If we mix reactants and products together will the reaction occur? If we know Ssurr we can calculate Suniv Can calculate from: S 0 rxn = S n. S 0(products) - S m. S 0(reactants) 3
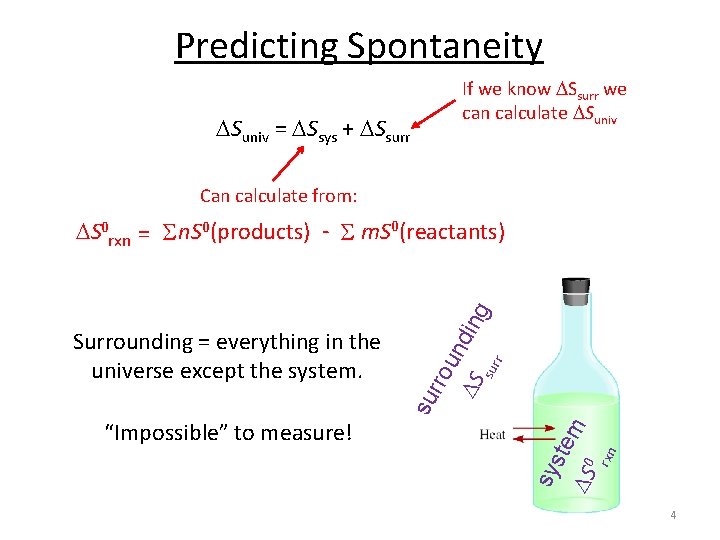
Predicting Spontaneity If we know Ssurr we can calculate Suniv = Ssys + Ssurr Can calculate from: r sur rxn sy ste S 0 m “Impossible” to measure! su Surrounding = everything in the universe except the system. rro un S ding S 0 rxn = S n. S 0(products) - S m. S 0(reactants) 4
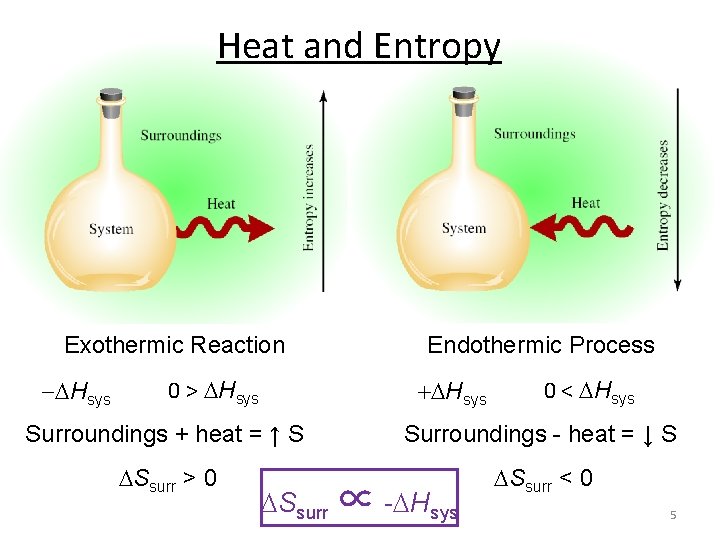
Heat and Entropy Exothermic Reaction Hsys 0 > Hsys Endothermic Process + Hsys 0 < Hsys Surroundings + heat = ↑ S Surroundings - heat = ↓ S Ssurr > 0 Ssurr < 0 Ssurr ∝ - Hsys 5

Heat and Entropy Heat released by the system increases the disorder of the surroundings. Ssurr ∝ - Hsys The effect of - Hsys on the surroundings depends on temperature: – At high temperature, where there is already considerable disorder, the effect is muted – At low temperature the effect is much more significant – The difference between tossing a rock into a calm pool (low T) and a storm-tossed ocean (high T) Ssurr = - Hsys T 6

Predicting Spontaneity Suniv = Ssys + Ssurr Substitution: Suniv = Ssys + - Hsys Ssurr = T Multiply by -T: -T Suniv = -T Ssys + Hsys Rearrange: -T Suniv = Hsys - T Ssys - Hsys T This equation relates Suniv to Hsys and Ssys. Both in terms of the system. 7

Gibbs Free Energy -T Suniv = Hsys - T Ssys Gibbs free energy ( G)- • AKA Free energy. • Relates S, H and T of a system. • Can be used to predict spontaneity. • G is a state function. Josiah Willard Gibbs (1839 -1903) First American Ph. D. in Engineering (Yale, 1863) Praised by Albert Einstein as "the greatest mind in American history" 8

Gibbs Free Energy For a constant temperature and constant pressure process: -T Suniv = Hsys - T Ssys Gibbs free energy ( G)- Can be used to predict spontaneity. G < 0 The reaction is spontaneous in the forward direction. G = -T(+ Suniv) G > 0 Suniv > 0 The reaction is nonspontaneous as written. The reaction is spontaneous in the reverse direction. + G = -T(- Suniv) S < 0 univ G = 0 The reaction is at equilibrium. G = -T(- Suniv) = 0 Suniv = 0 9

Gibbs Free Energy G = H - T S • If you know G for reactants and products then you can calculate if a reaction is spontaneous. • If you know G for two reaction then you can calculate if the sum is spontaneous. • If you know S, H and T then you can calculate spontaneity. • Can predict the temperature when a reaction becomes spontaneous. • If you have Hvap or Hfus and S you can predict boiling and freezing points. • If you have Hvap or Hfus and T you can predict the entropy change during a phase change. • Can predict equilibrium shifts. 10

Standard Free Energy Changes 0 ) is the free-energy The standard free-energy of reaction (DGrxn change for a reaction when it occurs under standard-state conditions. a. A + b. B c. C + d. D 0 Grxn = S n G 0 f (products) - S m G 0 f (reactants) Standard free energy of formation ( Gf 0) is the free-energy change that occurs when 1 mole of the compound is formed from its elements in their standard states. 0 Grxn = [ c G 0 f (C) + d G 0 f (D) ] - [ a G 0 f (A) + b G 0 f (B) ] G 0 f of any element in its stable form is zero. 11

Standard Free Energy Changes G is a state function so free energy can be calculated from the table of standard values just as enthalpy and entropy changes. a. A + b. B c. C + d. D 0 Grxn = S n G 0 f (products) - S m G 0 f (reactants) Standard free energy of formation ( Gf 0) is the free-energy change that occurs when 1 mole of the compound is formed from its elements in their standard states. 12
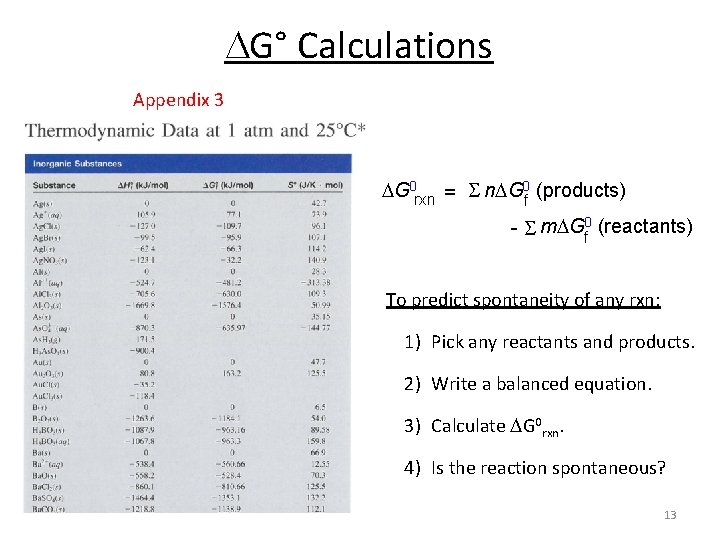
G° Calculations Appendix 3 G 0 rxn = S n G 0 f (products) - S m Gf 0 (reactants) To predict spontaneity of any rxn: 1) Pick any reactants and products. 2) Write a balanced equation. 3) Calculate G 0 rxn. 4) Is the reaction spontaneous? 13

Standard Free Energy Changes Calculate the standard free-energy change for the following reaction: 2 KCl. O 3(s) 2 KCl(s) + 3 O 2(g) 0 Grxn = S n G 0 f (products) - S m G 0 f (reactants) G 0 rxn = [2( 408. 3 k. J/mol) + 3(0)] [2( 289. 9 k. J/mol)] G 0 rxn = 816. 6 ( 579. 8) G 0 rxn = 236. 8 k. J/mol Is the reaction spontaneous? G 0 rxn < 0 From appendix 3: KCl. O 3(s) G f = -289. 9 k. J/mol KCl(s) G f = -408. 3 k. J/mol O 2(g) G f = 0 k. J/mol Yes! 14
![Another Example C(s, diamond) + O 2(g) CO 2(g) ΔG°rxn = [ΔG°f (CO 2)] Another Example C(s, diamond) + O 2(g) CO 2(g) ΔG°rxn = [ΔG°f (CO 2)]](http://slidetodoc.com/presentation_image_h2/b7303f68d02b291cd54b665c59bb685b/image-15.jpg)
Another Example C(s, diamond) + O 2(g) CO 2(g) ΔG°rxn = [ΔG°f (CO 2)] - [ΔG°f (C, diamond) + ΔG°f (O 2)] From appendix 3: G°rxn = -397. 3 k. J Is the reaction spontaneous? C, diamond(s) G f = 2. 9 k. J/mol O 2(g) G f = 0 k. J/mol CO 2(g) G f = -394. 4 k. J/mol + O 2 Therefore, diamonds are contributing to climate change! very slowly

More G° Calculations Similar to H°, one can use the G° for various reactions to determine G° for the reaction of interest (a “Hess’ Law” for G°) Hess’ Law- states that regardless of the multiple stages or steps of a reaction, the total enthalpy change for the reaction is the sum of all changes. What is the G° for this reaction: Given: C(s, diamond) + O 2(g) CO 2(g) G° = -397 k. J C(s, graphite) + O 2(g) CO 2(g) G° = -394 k. J 16

More G° Calculations What is the G° for this reaction: Given: C(s, diamond) + O 2(g) C(s, graphite) + O 2(g) CO 2(g) C(s, diamond) CO 2(g) G° = -397 k. J G° = -394 k. J C(s, graphite) + O 2(g) G° = +394 k. J CO 2(g) G° = -397 k. J C(s, graphite) + O 2(g) G° = +394 k. J C(s, graphite) G° = -3 k. J G°rxn < 0…. . rxn is spontaneous 17
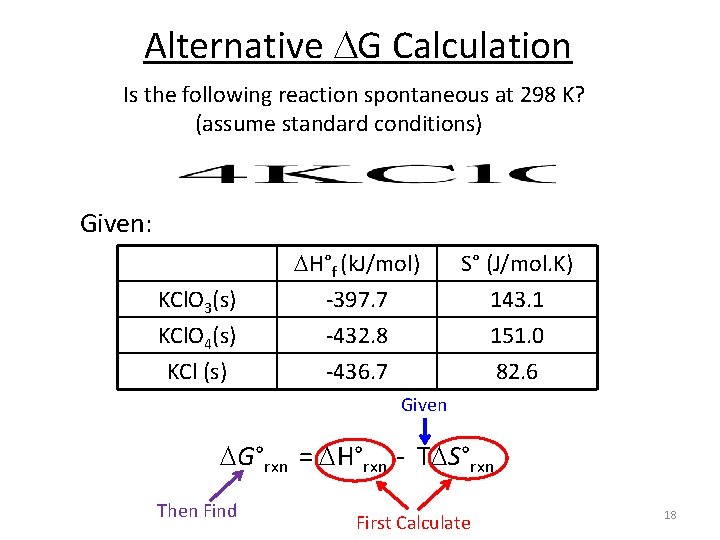
Alternative G Calculation Is the following reaction spontaneous at 298 K? (assume standard conditions) Given: KCl. O 3(s) KCl. O 4(s) KCl (s) H°f (k. J/mol) -397. 7 -432. 8 -436. 7 S° (J/mol. K) 143. 1 151. 0 82. 6 Given G°rxn = H°rxn - T S°rxn Then Find First Calculate 18
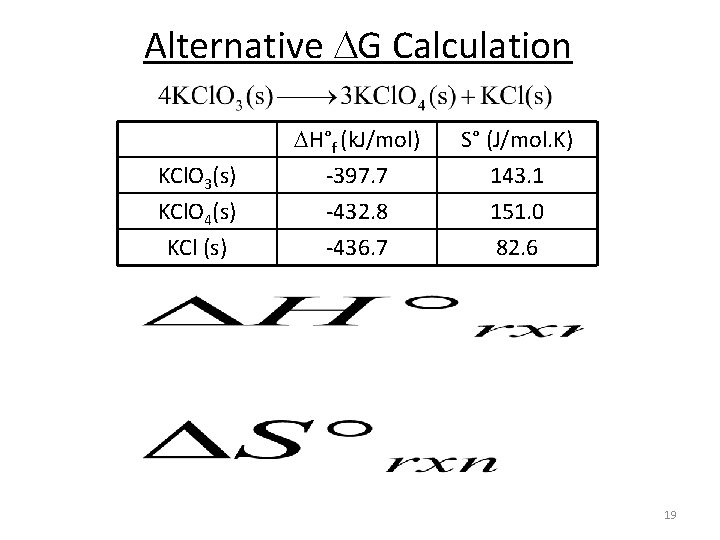
Alternative G Calculation KCl. O 3(s) KCl. O 4(s) KCl (s) H°f (k. J/mol) -397. 7 -432. 8 -436. 7 S° (J/mol. K) 143. 1 151. 0 82. 6 19
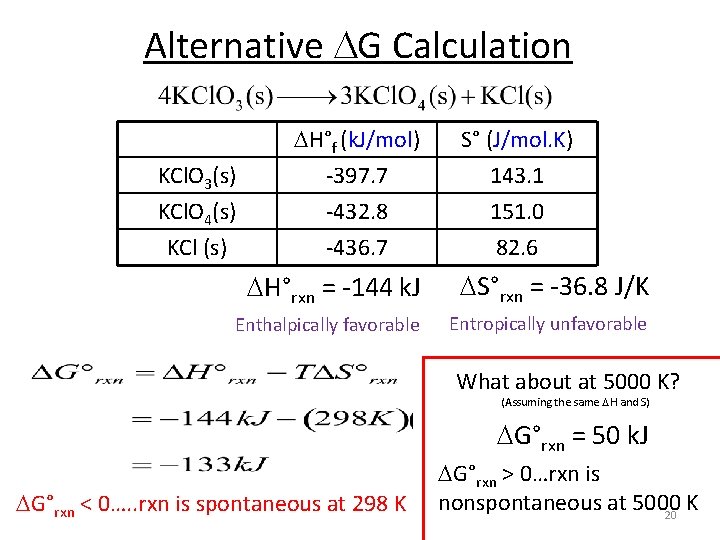
Alternative G Calculation KCl. O 3(s) KCl. O 4(s) KCl (s) H°f (k. J/mol) -397. 7 -432. 8 -436. 7 S° (J/mol. K) 143. 1 151. 0 82. 6 H°rxn = -144 k. J S°rxn = -36. 8 J/K Enthalpically favorable Entropically unfavorable What about at 5000 K? (Assuming the same H and S) G°rxn = 50 k. J G°rxn < 0…. . rxn is spontaneous at 298 K G°rxn > 0…rxn is nonspontaneous at 5000 K 20

Gibbs Free Energy Temperature and Spontaneity Free Energy and Equilibrium Thermodynamics of Living Systems 21

G Temperature Dependence G°rxn = -133 k. J at 298 K Spontaneous G°rxn = 50 k. J at 5000 K Nonspontaneous Reaction spontaneity is a temperature dependent phenomenon! H 2 O(s) Grxn = Hrxn - H 2 O(l) T Srxn 22

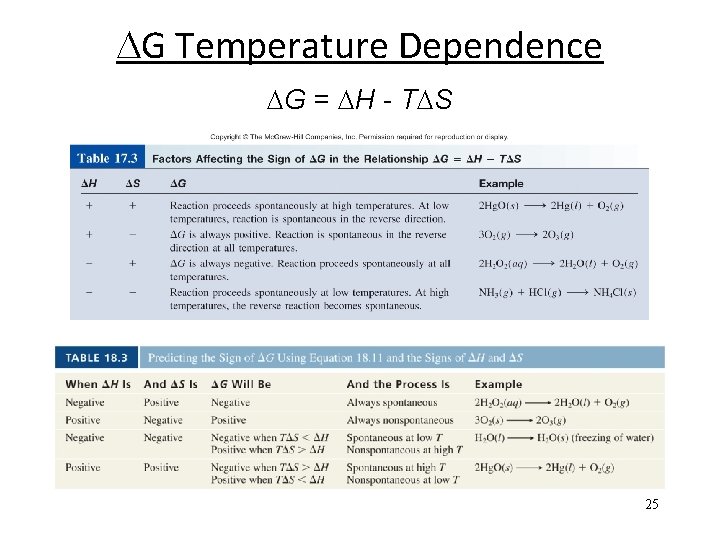
G Temperature Dependence Enthalpy: Entropy: Hrxn < 0 The reaction is enthalpically favorable. Srxn > 0 The reaction is entropically favorable. Need both to predict spontaneity. And sometimes temperature! G = H - T S G < 0 G > 0 G = 0 Spontaneous Nonspontaneous Equilibrium 1) If H < 0 and S > 0, then G is negative at all T 2) If H > 0 and S > 0, then G depends on T 3) If H < 0 and S < 0, then G depends on T 4) If H > 0 and S < 0, then G is positive at all T 23

G Temperature Dependence G = H - T S 3) H < 0 and S < 0 G < 0 G > 0 G = 0 Spontaneous Nonspontaneous Equilibrium (Enthalpically favorable, entropically unfavorable) If H < T S, then G is positive. If H > T S, then G is negative. Nonspontaneous at high T Spontaneous at low T 2) H > 0 and S > 0 (Enthalpically unfavorable, entropically favorable) If H < T S, then G is negative. If H > T S, then G is positive. Spontaneous at high T Nonspontaneous at low T 24
G Temperature Dependence G = H - T S 25

Predicting T from Gibbs Equation G°rxn = -133 k. J at 298 K Spontaneous G°rxn = 50 k. J at 5000 K Nonspontaneous Reaction spontaneity is a temperature dependent phenomenon! At what temperature will the reaction become spontaneous? Find T where G changes from positive to negative. I. e. when G =0. G = H – T S = 0 T = H/ S 26

Predicting T from Gibbs Equation At what T is the following reaction spontaneous? Br 2(l) Br 2(g) Given: H°= 30. 91 k. J/mol S°= 93. 2 J/mol. K G = H – T S = 0 T = H/ S T = (30. 91 k. J/mol) /(93. 2 J/mol. K) T = 331. 7 K H > 0, S > 0 The reaction will be spontaneous when T > 331. 7 K 27

Gibbs Free Energy Temperature and Spontaneity Free Energy and Equilibrium Thermodynamics of Living Systems 28

Free Energy and Equilibrium 0 ) is the free-energy The standard free-energy of reaction (DGrxn change for a reaction when it occurs under standard-state conditions. a. A + b. B c. C + d. D 0 Grxn = S n G 0 f (products) - S m G 0 f (reactants) G° < 0 favors products spontaneously G° > 0 favors reactants spontaneously Does not tell you it will go to completion! a. A + b. B G° fwd G° rev c. C + d. D G° fwd = - G° rev The value of G° calculated under the standard conditions characterizes the “driving force” of the reaction towards equilibrium. 29

Free Energy and Equilibrium G° = -R T ln. K standard free-energy (k. J/mol) gas constant (8. 314 J/K mol) equilibrium constant (Kp, Kc, Ka, Ksp, etc. ) temperature (K) • Arguably most important equation in chemical thermodynamics! • It allows us to calculate the extent of a chemical reaction if its enthalpy and entropy changes are known. • The changes in enthalpy and entropy can be evaluated by measuring the variation of the equilibrium constant with temperature. • This relationship is only valid for the standard conditions, i. e. when the activities of all reactants and products are equal to 1. 30

Free Energy and Equilibrium G° = -R T ln. K R is constant so at a given temperature: 0 DG (k. J) K 100 3 x 10 -18 50 2 x 10 -9 10 2 x 10 -2 1 7 x 10 -1 0 1 -1 1. 5 -10 5 x 101 -50 6 x 108 -100 3 x 1017 -200 1 x 1035 Essentially no forward reaction; reverse reaction goes to completion Forward and reverse reactions proceed to same extent Forward reaction goes to completion; essentially no reverse reaction REVERSE REACTION 9 x 10 -36 FORWARD REACTION 200 Significance 31

Using G° and K Using the table of standard free energies, calculate the equilibrium constant, KP, for the following reaction at 25 C. 2 HCl(g) H 2(g) + Cl 2(g) G 0 rxn = S n G 0 (products) - S m G 0 f (reactants) f G 0 rxn = [1(0) + 1(0)] - [2( 95. 3 k. J/mol)] G 0 rxn = 190. 6 k. J/mol Non-spontaneous! Favors reactants! G° = -R T ln K From appendix 3: H 2(g) G f = 0 k. J/mol Cl 2(s) G f = 0 k. J/mol HCl(g) G f = -95. 3 k. J/mol 32

Using G° and K Using the table of standard free energies, calculate the equilibrium constant, KP, for the following reaction at 25 C. 2 HCl(g) H 2(g) + Cl 2(g) G 0 rxn = S n G 0 (products) - S m G 0 f (reactants) f G 0 rxn = [1(0) + 1(0)] - [2( 95. 3 k. J/mol)] G 0 rxn = 190. 6 k. J/mol Non-spontaneous! Favors reactants! G° = -R T ln K 190. 6 k. J/mol = (8. 314 J/K·mol)(25 C) ln KP correct units 190. 6 k. J/mol = (8. 314 x 10 3 k. J/K·mol)(298 K) ln KP KP = 3. 98 x 10 34 Favors reactants! 33

Free Energy and Equilibrium G° = -R T ln. K G° Rearrange: ln K = RT Substitution: ln K = - H - T S RT G = H - T S H T S Rearrange: ln K = + RT RT ln K = - H 1 R T () + S R 34
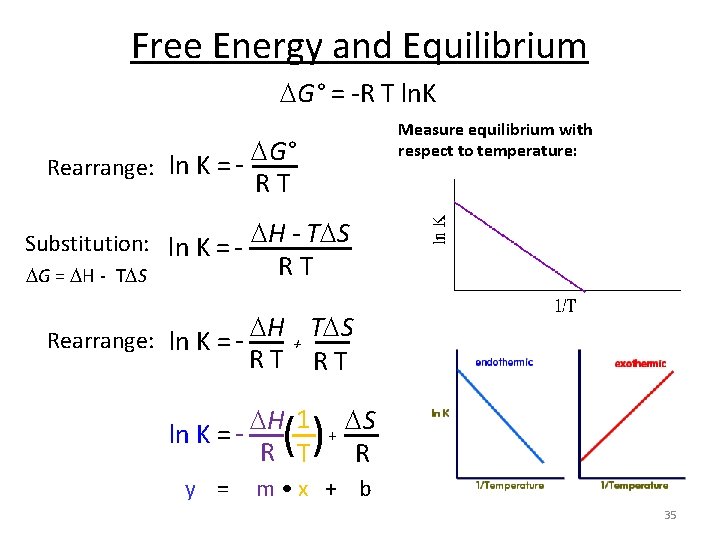
Free Energy and Equilibrium G° = -R T ln. K Measure equilibrium with respect to temperature: G° Rearrange: ln K = RT Substitution: ln K = - H - T S RT G = H - T S H T S Rearrange: ln K = + RT RT ln K = y = H 1 R T () + S R m • x + b 35

Free Energy and Equilibrium Find the S and H of the following: kobs kf A B rate. A B = kobs [A] rate. B A = kf [B] At equilibrium: rate. A B = rate. B A kobs [A] = kf [B] kobs kf = [B] [A] = Ku 36
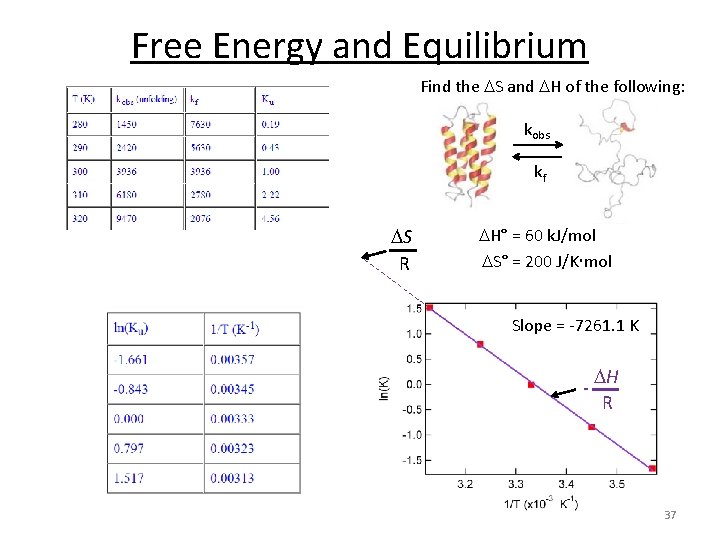
Free Energy and Equilibrium Find the S and H of the following: kobs kf S R H° = 60 k. J/mol S° = 200 J/K mol Slope = -7261. 1 K - H R 37

G° vs G You have already delta ΔG°, ΔS° and ΔH° in which the ° indicates that all components are in their standard states. Definition of “standard”: • Even if we start a reaction at standard conditions (1 M) the reaction will quickly deviate from standard. • G° indicates whether reactants or products are favored at equilibrium. • G at any give time is used to predict the direction shift to reach equilibrium. • If a mixture is not at equilibrium, the liberation of the excess Gibbs free energy ( G) is the “driving force” for the composition of the mixture to *There is no "standard temperature", change until equilibrium is reached. 38 but we usually use 298. 15 K (25° C).

Free Energy and Equilibrium At equilibrium: At any time: G° = -R T ln K G = G° + R T ln Q reaction quotient temperature standard free-energy (K) non-standard free-energy gas constant (k. J/mol) (8. 314 J/K mol) The sign of G tells us that the reaction would have to shift to the left to reach equilibrium. G < 0, reaction will shift right G > 0, reaction will shift left G = 0, the reaction is at equilibrium The magnitude of G tells us how far it has to go to reach equilibrium. 39

Free Energy and Equilibrium G = G° + R T ln Q reaction quotient temperature standard free-energy (K) non-standard free-energy gas constant (k. J/mol) (8. 314 J/K mol) If Q/K < 1, then ln Q/K < 0; the reaction proceeds to the right ( G < 0) If Q/K > 1, then ln Q/K > 0; the reaction proceeds to the left ( G > 0) If Q/K = 1, then ln Q/K = 0; the reaction is at equilibrium ( G = 0) 40

Another Example For the following reaction at 298 K: H 2(g) + Cl 2(g) 2 HCl(g) Given: From appendix 3: H 2 = 0. 25 atm Cl 2 = 0. 45 atm HCl = 0. 30 atm H 2(g) G f = 0 k. J/mol Cl 2(s) G f = 0 k. J/mol HCl(g) G f = -95. 3 k. J/mol Which way will the reaction shift to reach equilibrium? G = G° + R T ln Q calculate constant calculate given 41

Another Example For the following reaction at 298 K: H 2(g) + Cl 2(g) 2 HCl(g) Given: H 2 = 0. 25 atm Cl 2 = 0. 45 atm HCl = 0. 30 atm From appendix 3: H 2(g) G f = 0 k. J/mol Cl 2(s) G f = 0 k. J/mol HCl(g) G f = -95. 3 k. J/mol Which way will the reaction shift to reach equilibrium? G = G° + R T ln Q G° = [2( 95. 27 k. J/mol)] [0 + 0] = 190. 54 k. J/mol 42

Another Example For the following reaction at 298 K: H 2(g) + Cl 2(g) 2 HCl(g) Given: From appendix 3: H 2 = 0. 25 atm Cl 2 = 0. 45 atm HCl = 0. 30 atm H 2(g) G f = 0 k. J/mol Cl 2(s) G f = 0 k. J/mol HCl(g) G f = -95. 3 k. J/mol Which way will the reaction shift to reach equilibrium? G = G° + R T ln Q G° = 190. 54 k. J/mol constant Q = 0. 80 given G = 190, 540 J/mol + (8. 314 J/K·mol)(298 K) ln (0. 80) G = 191. 09 k. J/mol Because ΔG < 0, the net reaction proceeds from left to right to reach equilibrium. 43

Gibbs Free Energy Temperature and Spontaneity Free Energy and Equilibrium Thermodynamics of Living Systems 44
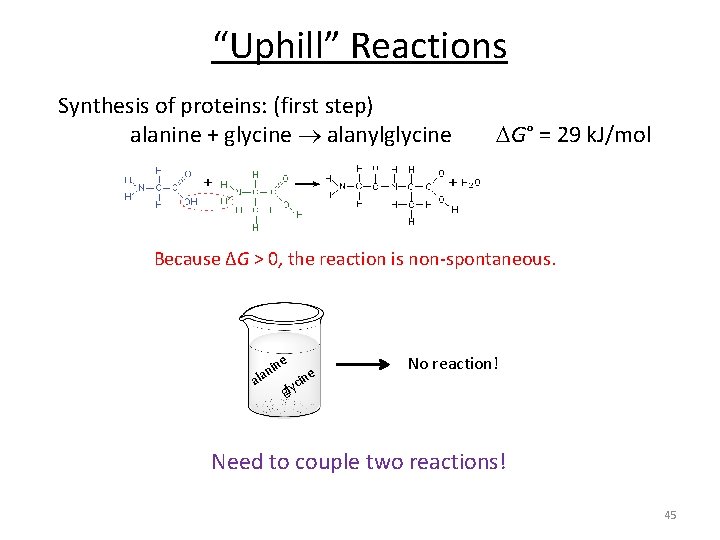
“Uphill” Reactions Synthesis of proteins: (first step) alanine + glycine alanylglycine G° = 29 k. J/mol Because ΔG > 0, the reaction is non-spontaneous. e in lan a e cin No reaction! gly Need to couple two reactions! 45

Coupled Reactions- using a thermodynamically favorable reaction ( G° < 0) to drive an unfavorable one ( G° > 0). Example: Industrial ore separation- Zinc Metal Sphalerite ore Major applications in the US 1) Galvanizing (55%) 2) Alloys (21%) 3) Brass and bronze (16%) 4) Miscellaneous (8%) White pigment (Zn. O) Fire retardant (Zn. Cl 2) Vitamin supplement (Zn 2+) Reducing agent (Zn(s)) We need 2000 tones of the zinc metal per year!46

Coupled Reactions- using a thermodynamically favorable ( G° < 0) reaction ( G° < 0) to drive an unfavorable one ( G° > 0). Example: Industrial ore separation- Unfavorable reaction ( G° > 0) 95 % of Zinc is produced by this method 47
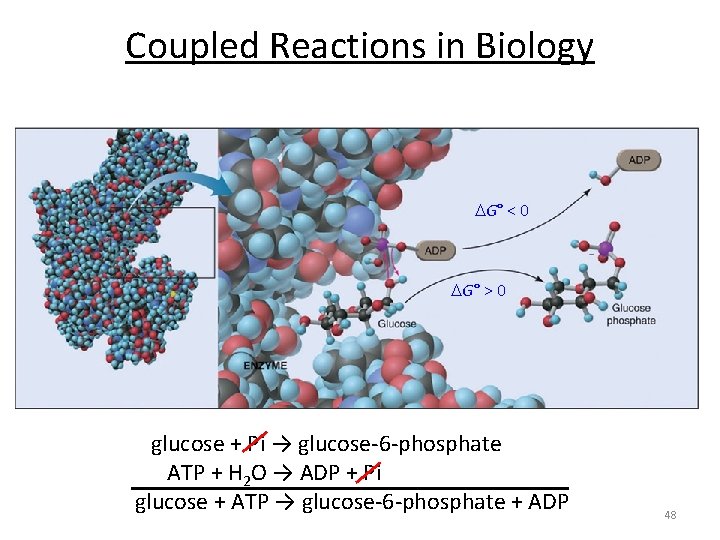
Coupled Reactions in Biology G° < 0 G° > 0 glucose + Pi → glucose-6 -phosphate ATP + H 2 O → ADP + Pi glucose + ATP → glucose-6 -phosphate + ADP 48
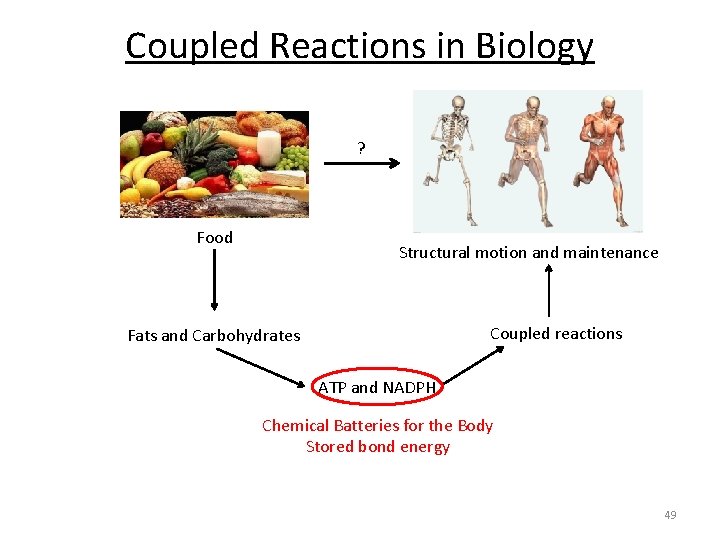
Coupled Reactions in Biology ? Food Structural motion and maintenance Coupled reactions Fats and Carbohydrates ATP and NADPH Chemical Batteries for the Body Stored bond energy 49
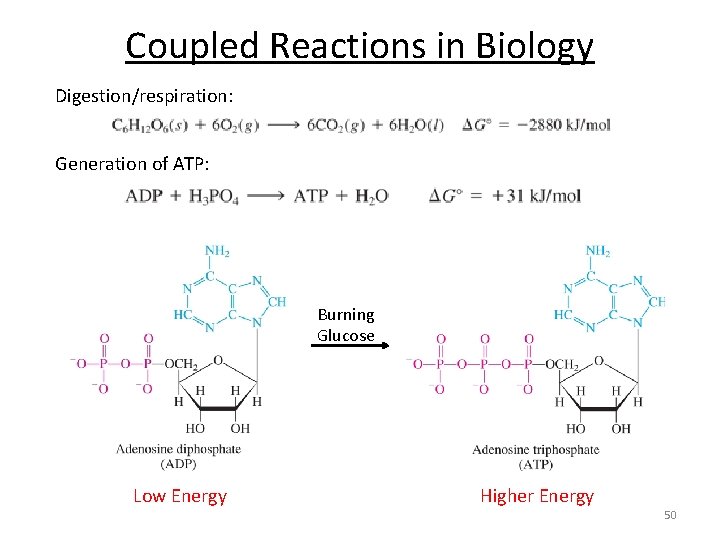
Coupled Reactions in Biology Digestion/respiration: Generation of ATP: Burning Glucose Low Energy Higher Energy 50
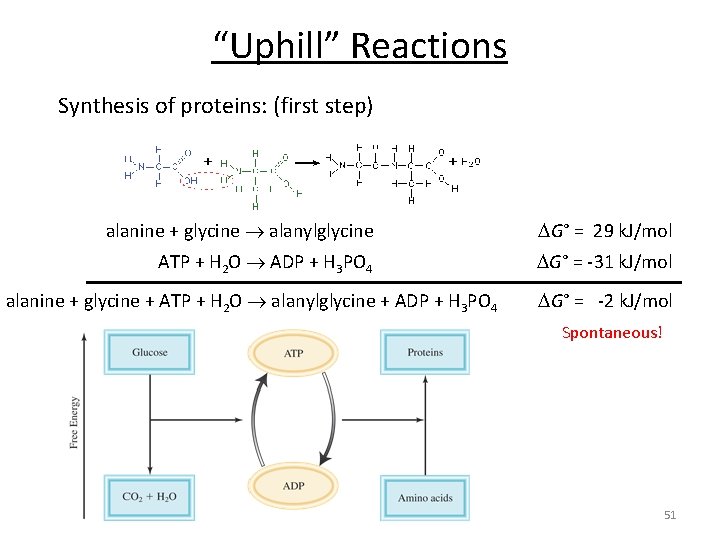
“Uphill” Reactions Synthesis of proteins: (first step) alanine + glycine alanylglycine G° = 29 k. J/mol ATP + H 2 O ADP + H 3 PO 4 G° = -31 k. J/mol alanine + glycine + ATP + H 2 O alanylglycine + ADP + H 3 PO 4 G° = -2 k. J/mol Spontaneous! 51
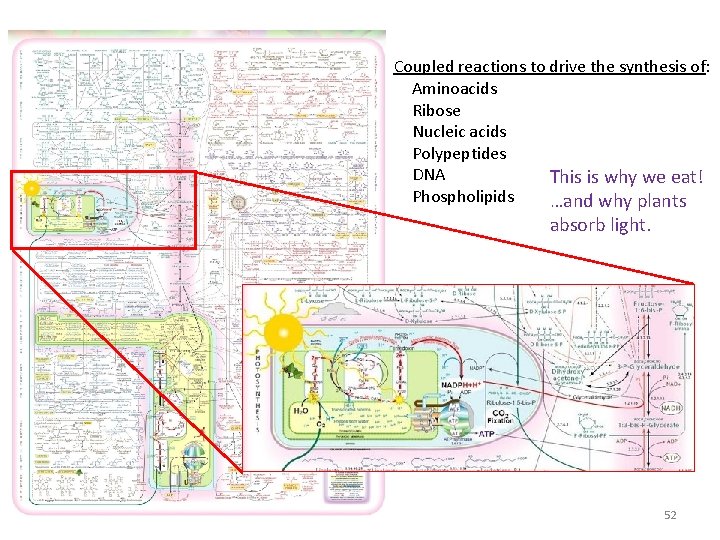
Coupled reactions to drive the synthesis of: Aminoacids Ribose Nucleic acids Polypeptides DNA This is why we eat! Phospholipids …and why plants absorb light. 52

53
- Slides: 53