Gibbs Free Energy G Suniv Ssurr Ssys Multiply

Gibbs Free Energy, G ∆Suniv = ∆Ssurr + ∆Ssys Multiply through by -T -T∆Suniv = ∆Hsys - T∆Ssys -T∆Suniv = change in Gibbs free energy for the system = ∆Gsystem Under standard conditions — ∆Gosys = ∆Hosys - T∆Sosys 1

o ∆G = o ∆H - o T∆S 2 Gibbs free energy change = total energy change for system - energy lost in disordering the system If reaction is • exothermic (negative ∆ Ho) (energy dispersed) • and entropy increases (positive ∆So) (matter dispersed) • then ∆Go must be NEGATIVE • reaction is spontaneous (and productfavored).

o ∆G = o ∆H - o T∆S 3 Gibbs free energy change = total energy change for system - energy lost in disordering the system If reaction is • endothermic (positive ∆Ho) • and entropy decreases (negative ∆So) • then ∆Go must be POSITIVE • reaction is not spontaneous (and is reactant- favored).

Gibbs Free Energy, G o ∆G = o ∆H - o T∆S ∆Ho ∆So ∆Go Reaction exo(–) increase(+) – Prod-favored endo(+) decrease(-) + React-favored exo(–) decrease(-) ? T dependent endo(+) increase(+) ? T dependent 4

Gibbs Free Energy, G o ∆G = o ∆H - 5 o T∆S Two methods of calculating ∆Go a) Determine ∆Horxn and ∆Sorxn and use GIbbs equation. b) Use tabulated values of free energies of formation, ∆Gfo. ∆Gorxn = ∆Gfo (products) - ∆Gfo (reactants)

Free Energies of Formation Note that ∆G˚f for an element = 0 6

Calculating ∆Gorxn 7 Combustion of acetylene C 2 H 2(g) + 5/2 O 2(g) --> 2 CO 2(g) + H 2 O(g) Use enthalpies of formation to calculate ∆Horxn = -1238 k. J Use standard molar entropies to calculate ∆Sorxn = -97. 4 J/K or -0. 0974 k. J/K ∆Gorxn = -1238 k. J - (298 K)(-0. 0974 J/K) = -1209 k. J Reaction is product-favored in spite of negative ∆Sorxn. Reaction is “enthalpy driven”

Calculating ∆Gorxn NH 4 NO 3(s) + heat ---> NH 4 NO 3(aq) Is the dissolution of ammonium nitrate productfavored? If so, is it enthalpy- or entropy-driven? 8

Calculating ∆Gorxn NH 4 NO 3(s) + heat ---> NH 4 NO 3(aq) From tables of thermodynamic data we find ∆Horxn = +25. 7 k. J ∆Sorxn = +108. 7 J/K or +0. 1087 k. J/K ∆Gorxn = +25. 7 k. J - (298 K)(+0. 1087 J/K) = -6. 7 k. J Reaction is product-favored in spite of negative ∆Horxn. Reaction is “entropy driven” 9

Gibbs Free Energy, G o ∆G = o ∆H - 10 o T∆S Two methods of calculating ∆Go a) Determine ∆Horxn and ∆Sorxn and use GIbbs equation. b) Use tabulated values of free energies of formation, ∆Gfo. ∆Gorxn = ∆Gfo (products) - ∆Gfo (reactants)

Calculating ∆Gorxn = ∆Gfo (products) - ∆Gfo (reactants) 11 Combustion of carbon C(graphite) + O 2(g) --> CO 2(g) ∆Gorxn = ∆Gfo(CO 2) - [∆Gfo(graph) + ∆Gfo(O 2)] ∆Gorxn = -394. 4 k. J - [ 0 + 0] Note that free energy of formation of an element in its standard state is 0. ∆Gorxn = -394. 4 k. J Reaction is product-favored as expected.

Free Energy and Temperature 2 Fe 2 O 3(s) + 3 C(s) ---> 4 Fe(s) + 3 CO 2(g) ∆Horxn = +467. 9 k. J ∆Sorxn = +560. 3 J/K ∆Gorxn = +300. 8 k. J Reaction is reactant-favored at 298 K At what T does ∆Gorxn just change from being (+) to being (-)? When ∆Gorxn = 0 = ∆Horxn - T∆Sorxn 12

13 More thermo? You betcha!

Thermodynamics and Keq u FACT: ∆Gorxn is the change in free energy when pure reactants convert COMPLETELY to pure products. u FACT: Product-favored systems have Keq > 1. u Therefore, both ∆G˚rxn and Keq are related to reaction favorability. 14

Thermodynamics and Keq is related to reaction favorability and so to ∆Gorxn. The larger the value of K the more negative the value of ∆Gorxn o ∆G rxn = - RT ln. K where R = 8. 31 J/K • mol 15

Thermodynamics and Keq ∆Gorxn = - RT ln. K Calculate K for the reaction N 2 O 4 --->2 NO 2 ∆Gorxn = +4. 8 k. J ∆Gorxn = +4800 J = - (8. 31 J/K)(298 K) ln K K = 0. 14 When ∆Gorxn > 0, then K < 1 16

∆G, ∆G˚, and Keq • ∆G is change in free energy at nonstandard conditions. • ∆G is related to ∆G˚ • ∆G = ∆G˚ + RT ln Q where Q = reaction quotient • When Q < K or Q > K, reaction is spontaneous. • When Q = K reaction is at equilibrium • • When ∆G = 0 reaction is at equilibrium Therefore, ∆G˚ = - RT ln K 17
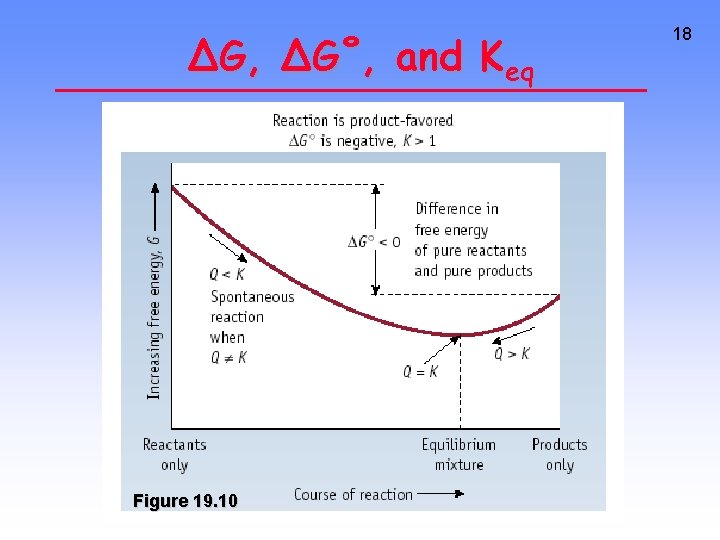
∆G, ∆G˚, and Keq Figure 19. 10 18
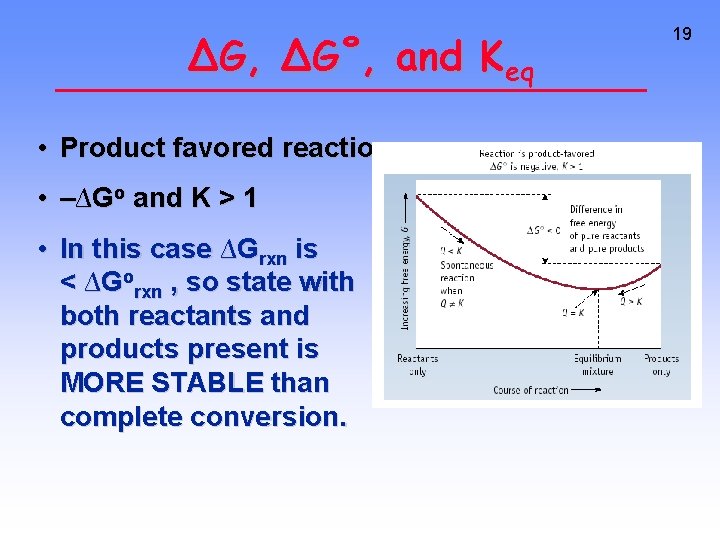
∆G, ∆G˚, and Keq • Product favored reaction • –∆Go and K > 1 • In this case ∆Grxn is < ∆Gorxn , so state with both reactants and products present is MORE STABLE than complete conversion. 19
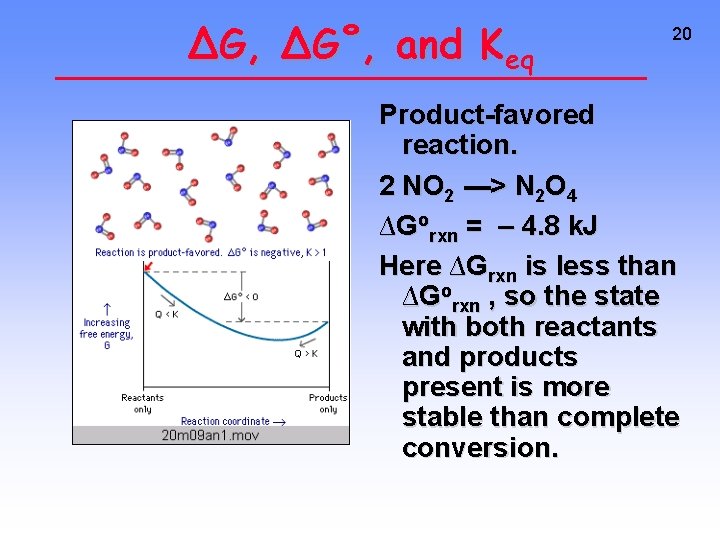
∆G, ∆G˚, and Keq 20 Product-favored reaction. 2 NO 2 ---> N 2 O 4 ∆Gorxn = – 4. 8 k. J Here ∆Grxn is less than ∆Gorxn , so the state with both reactants and products present is more stable than complete conversion.
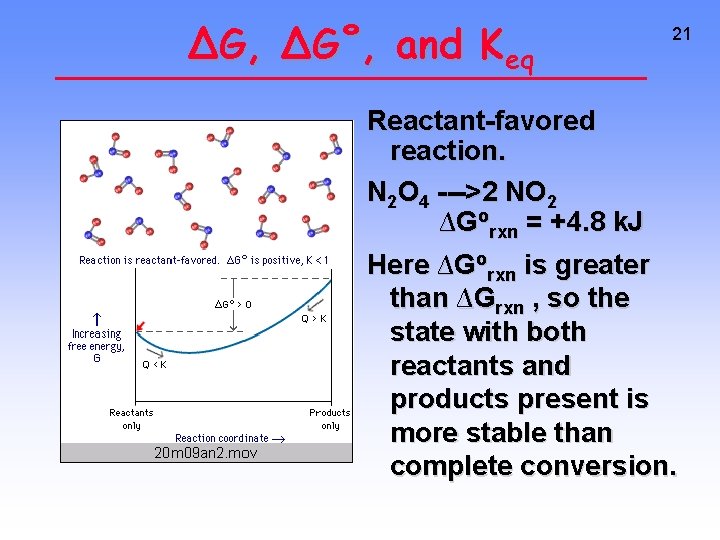
∆G, ∆G˚, and Keq 21 Reactant-favored reaction. N 2 O 4 --->2 NO 2 ∆Gorxn = +4. 8 k. J Here ∆Gorxn is greater than ∆Grxn , so the state with both reactants and products present is more stable than complete conversion.

Thermodynamics and Keq u Keq is related to reaction favorability. u When ∆Gorxn < 0, reaction moves energetically “downhill” u ∆Gorxn is the change in free energy when reactants convert COMPLETELY to products. 22
- Slides: 22