Gibbs Free Energy G See pages 13 15

Gibbs Free Energy: G (See pages 13 -15; 309 - 318 Horton) G = H –TS H = heat of system S = entropy of system Determine the change in free energy of a reaction DG DG = DH –T/DS DG = amount of energy available to do work DG = Gproducts - Greactants A+B C + D DG = (GC + GD ) - (GA + GB) DG < 0 exergonic, rx tends to be spontaneous DG > 0 endergonic, rs requires input of energy DG = 0 at equilibrium Units: Joules or kilo. Joules Joule = amount of energy required to apply 1 newton of force over 1 m

Standard free energy change: DGo’ Reactants and products present at Concentration of 1 M and p. H = 7. 0 A B Keq = [B] / [A] Reaction may be: exergonic and A DGo’ < 0 Reaction may be: endergonic and B DGo’ > 0 B A Reaction may be at equilibrium DGo’ = 0

• DGo’ is independent of pathway A A B B or E F C G C Both pathways have the same DGo’

Calculation of DG: change in Gibbs free energy 1. DGo’ = -RTln. Keq R = 8. 315 J/mol/o. K T = 298 o. K (25 o. C) = 2. 48 ln. Keq (k. J/mol) i. A B DGo’ = +16. 7 KJ/mol What is Keq for this reaction? DGo’ = -RTln. Keq 16. 7 = -2. 48 ln. Keq = -(16. 7/2. 48) = -6. 73 Keq = 1. 19 x 10 -3 = [B] / [A]

• small changes in DGo’ produce large changes in Keq If DGo’ = +22. 4 k. J/mol 22. 4 = -2. 48 ln. Keq = -(22. 4/2. 48) = -9. 03 Keq = 1. 19 x 10 -4 Increase in DGo’ from 16. 7 to 22. 4, a 35% increase, results in a 10 -fold change in Keq.
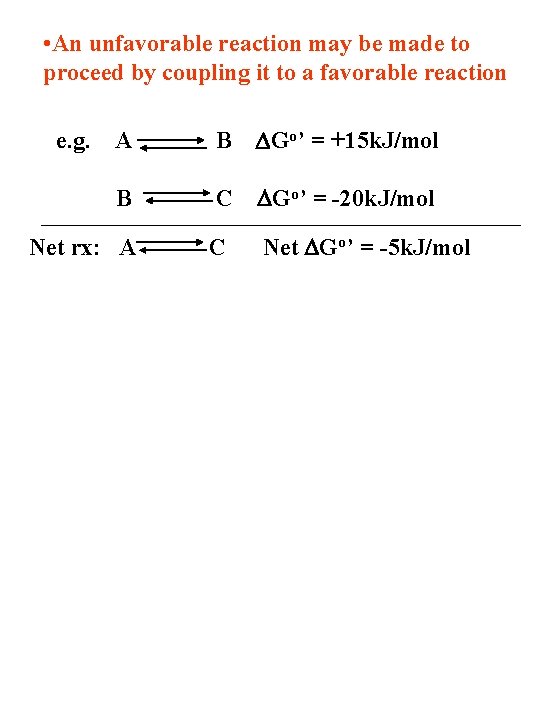
• An unfavorable reaction may be made to proceed by coupling it to a favorable reaction e. g. A B DGo’ = +15 k. J/mol B C DGo’ = -20 k. J/mol Net rx: A C Net DGo’ = -5 k. J/mol
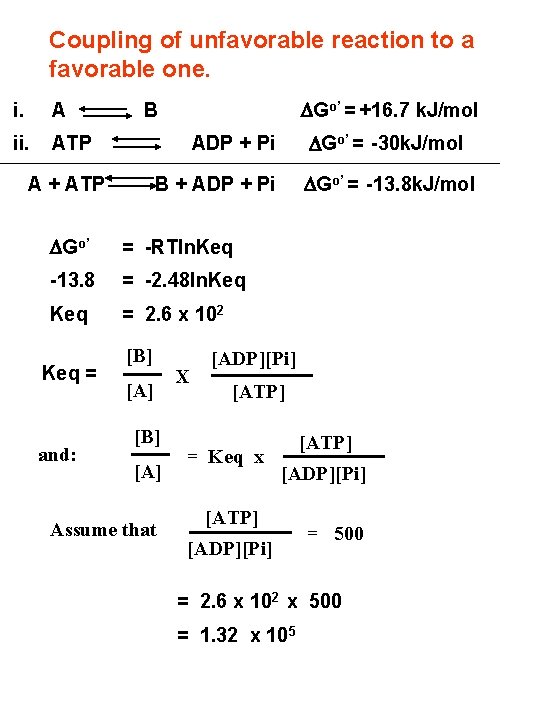
Coupling of unfavorable reaction to a favorable one. i. A ii. ATP DGo’ = +16. 7 k. J/mol B A + ATP = -RTln. Keq -13. 8 = -2. 48 ln. Keq = 2. 6 x 102 and: DGo’ = -13. 8 k. J/mol B + ADP + Pi DGo’ Keq = DGo’ = -30 k. J/mol ADP + Pi [B] [A] Assume that X [ADP][Pi] [ATP] = Keq x [ATP] [ADP][Pi] = 500 = 2. 6 x 102 x 500 = 1. 32 x 105

Keq in the absence of ATP hydrolysis: = 1. 19 x 10 -3 Keq in the presence of ATP hydrolysis: = 1. 32 x 105 An increase of 108 -fold
![Actual free energy change 2. DGo’ DG = [B] + RTln [B] [A] = Actual free energy change 2. DGo’ DG = [B] + RTln [B] [A] =](http://slidetodoc.com/presentation_image_h/18d699f1fe1cfebf641c11772312e239/image-9.jpg)
Actual free energy change 2. DGo’ DG = [B] + RTln [B] [A] = Q, the mass action ratio [A] e. g. A B Keq = 0. 0475 from DGo’ = -RTln. Keq DGo’ = 7. 55 k. J/mol standard conditions BUT If [A] = 2 x 10 -4 M , and [B] = 3 x 10 -6 M Then DG = DGo’ + RTln = 7. 55 + RTln [B] [A] 3 x 10 -6 M 2 x 10 -4 M = -2. 86 k. J/mol actual conditions

Control of metabolic flux • Reactions that operate near equilibrium are readily reversible - rate and direction of reaction effectively controlled by concentrations of substrate and products • Reactions that operate far from equilibrium are metabolically irreversible – rate can only be altered by changing enzyme activity

e. g. Phosphofructokinase F-6 -P + ATP F-1, 6 -bis. P + ADP Keq = 300 But under intracellular conditions Q = 0. 03 Insuffcient enzyme activity to equilibrate reaction and enzyme operates near Vmax at all times Can only increase rate of product formation by increasing enzyme activity This a potential control point

A 1 B 2 C 3 D 4 E In metabolic pathways intermediates are not allowed to “pile up” All reactions in a sequence proceed at the same rate and concentration of intermediates is constant – the steady state condition This is achieved by having several points of control First enzyme of a pathway does not feed substrate into the pathway at a rate that is faster than the slowest enzyme downstream
- Slides: 12