GI SLIDE DECK 2018 Selected abstracts from 20

GI SLIDE DECK 2018 Selected abstracts from: 20 th World Congress on Gastrointestinal Cancer 20– 23 June 2018 | Barcelona, Spain Supported by Eli Lilly and Company has not influenced the content of this publication

Letter from ESDO DEAR COLLEAGUES It is our pleasure to present this ESDO slide set which has been designed to highlight and summarise key findings in digestive cancers from the major congresses in 2018. This slide set specifically focuses on the 20 th World Congress on Gastrointestinal Cancer and is available in English, French, Japanese and Chinese. The area of clinical research in oncology is a challenging and ever changing environment. Within this environment, we all value access to scientific data and research which helps to educate and inspire further advancements in our roles as scientists, clinicians and educators. I hope you find this review of the latest developments in digestive cancers of benefit to you in your practice. If you would like to share your thoughts with us we would welcome your comments. Please send any correspondence to info@esdo. eu. Finally, we are also very grateful to Lilly Oncology for their financial, administrative and logistical support in the realisation of this activity. Yours sincerely, Eric Van Cutsem Thomas Seufferlein Côme Lepage Wolff Schmiegel Phillippe Rougier (hon. ) (ESDO Governing Board) Ulrich Güller Thomas Grünberger Tamara Matysiak-Budnik Jaroslaw Regula Jean-Luc Van Laethem

ESDO Medical Oncology Slide Deck Editors 2018 COLORECTAL CANCERS Prof Eric Van Cutsem Digestive Oncology, University Hospitals, Leuven, Belgium Prof Wolff Schmiegel Department of Medicine, Ruhr University, Bochum, Germany Prof Thomas Gruenberger Department of Surgery, Kaiser-Franz-Josef Hospital, Vienna, Austria Prof Jaroslaw Regula Department of Gastroenterology and Hepatology, Institute of Oncology, Warsaw, Poland PANCREATIC CANCER AND HEPATOBILIARY TUMOURS Prof Jean-Luc Van Laethem Digestive Oncology, Erasme University Hospital, Brussels, Belgium Prof Thomas Seufferlein Clinic of Internal Medicine I, University of Ulm, Germany Prof Ulrich Güller Medical Oncology & Hematology, Kantonsspital St Gallen, Switzerland GASTRO-OESOPHAGEAL AND NEUROENDOCRINE TUMOURS Prof Côme Lepage University Hospital & INSERM, Dijon, France Prof Tamara Matysiak Hepato-Gastroenterology & Digestive Oncology, Institute of Digestive Diseases, Nantes, France BIOMARKERS Prof Eric Van Cutsem Digestive Oncology, University Hospitals, Leuven, Belgium Prof Thomas Seufferlein Clinic of Internal Medicine I, University of Ulm, Germany
Glossary 1/2/3 L 5 FU AE AFP ALT AST BCLC BICR bid BOR BSC CI CPS CR CRC CT ct. DNA DCR DFS d. MMR (m)Do. R ECOG EGFR ESCC ESMO first/second/third line 5 -fluorouracil adverse event alpha-fetoprotein alkaline phosphatase alanine aminotransferase aspartate aminotransferase Barcelona clinic liver cancer blinded-independent central review twice daily best overall response best supportive care confidence interval combined positive score complete response colorectal cancer chemotherapy circulating tumour DNA disease control rate disease-free survival deficient mismatch repair (median) duration of response Eastern Cooperative Oncology Group epidermal growth factor receptor oesophageal squamous cell cancer European Society of Medical Oncology FAS full analysis set FOLFIRINOX leucovorin + 5 -fluorouracil + irinotecan + oxaliplatin FOLFOX leucovorin + 5 -fluorouracil + oxaliplatin (m)FOLFOXIRI GEJ GI GIST HBV HCC HCV HIF-1α HR IHC IQR ITT iv KM LV m. Ab m. CRC met m. PDAC MSI(-H) Mut NA NE NR OR ORR (m)OS PCR PD (modified) leucovorin + 5 -fluorouracil + oxaliplatin + irinotecan gastro-oesophageal junction gastrointestinal stromal tumour hepatitis B virus hepatocellular carcinoma hepatitis C virus hypoxia-inducible factor-1α hazard ratio immunohistochemistry interquartile range intent-to-treat intravenous Kaplan-Meier leucovorin monoclonal antibody metastatic colorectal cancer metastasis metastatic pancreatic ductal adenocarcinoma (high) microsatellite instability mutant not available not evaluable not reached odds ratio overall/objective response rate (median) overall survival polymerase chain reaction progressive disease PD-L 1 PI 3 KCA (m)PFS PPES PR PS q(2/3/4/6)w Qo. L R RCT RECIST RT SAE SD Tid TRAE TRK TRR TTP (m)TTR tx VEGF WT programmed death-ligand 1 phosphatidylinositol 3 -kinase (median) progression-free survival palmar-plantar erythrodysesthesia syndrome partial response performance status every (2/3/4/6) week(s) quality of life randomised controlled trial Response Evaluation Criteria In Solid Tumors radiotherapy serious adverse event stable disease three times daily treatment-related adverse event tropomyosin receptor kinase tumour resection rate time-to-progression (median) time-to-response treatment vascular endothelial growth factor wild-type

Contents • Cancers of the oesophagus and stomach • Cancers of the pancreas, small bowel and hepatobiliary tract 6 15 – Pancreatic cancer 16 – Hepatocellular carcinoma 21 • Cancers of the colon, rectum and anus Note: To jump to a section, right click on the number and ‘Open Hyperlink’ 30

CANCERS OF THE OESOPHAGUS AND STOMACH
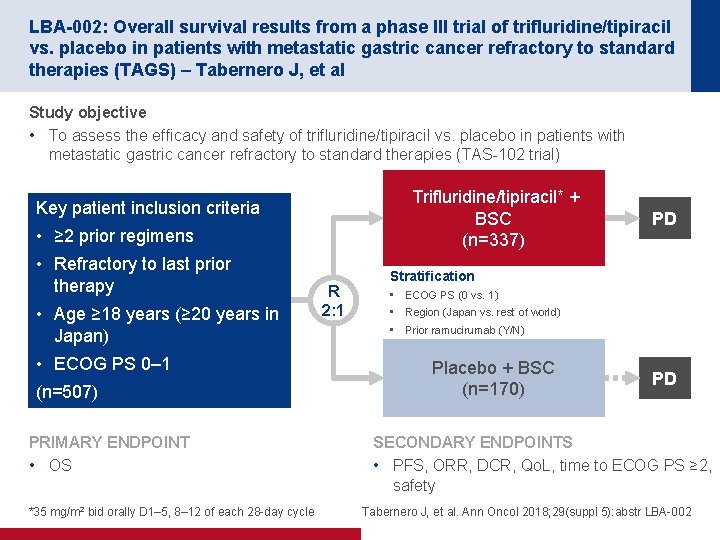
LBA-002: Overall survival results from a phase III trial of trifluridine/tipiracil vs. placebo in patients with metastatic gastric cancer refractory to standard therapies (TAGS) – Tabernero J, et al Study objective • To assess the efficacy and safety of trifluridine/tipiracil vs. placebo in patients with metastatic gastric cancer refractory to standard therapies (TAS-102 trial) Trifluridine/tipiracil* + BSC (n=337) Key patient inclusion criteria • ≥ 2 prior regimens • Refractory to last prior therapy • Age ≥ 18 years (≥ 20 years in Japan) • ECOG PS 0– 1 (n=507) PRIMARY ENDPOINT • OS *35 mg/m 2 bid orally D 1– 5, 8– 12 of each 28 -day cycle R 2: 1 PD Stratification • • • ECOG PS (0 vs. 1) Region (Japan vs. rest of world) Prior ramucirumab (Y/N) Placebo + BSC (n=170) PD SECONDARY ENDPOINTS • PFS, ORR, DCR, Qo. L, time to ECOG PS ≥ 2, safety Tabernero J, et al. Ann Oncol 2018; 29(suppl 5): abstr LBA-002
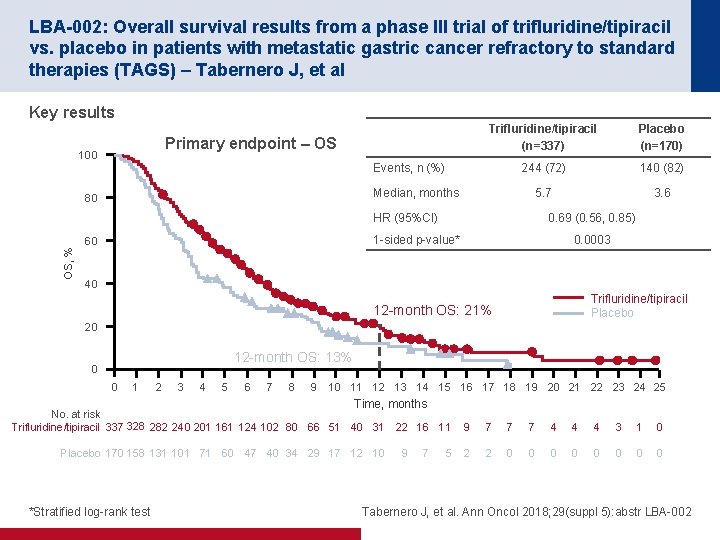
LBA-002: Overall survival results from a phase III trial of trifluridine/tipiracil vs. placebo in patients with metastatic gastric cancer refractory to standard therapies (TAGS) – Tabernero J, et al Key results Primary endpoint – OS 100 Trifluridine/tipiracil (n=337) Placebo (n=170) 244 (72) 140 (82) 5. 7 3. 6 Events, n (%) Median, months 80 OS, % HR (95%CI) 0. 69 (0. 56, 0. 85) 1 -sided p-value* 60 0. 0003 40 Trifluridine/tipiracil Placebo 12 -month OS: 21% 20 12 -month OS: 13% 0 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 Time, months No. at risk Trifluridine/tipiracil 337 328 282 240 201 161 124 102 80 66 51 40 31 22 16 11 Placebo 170 158 131 101 71 60 47 40 34 29 17 12 10 *Stratified log-rank test 9 7 5 9 7 7 7 4 4 4 3 1 0 2 2 0 0 0 0 Tabernero J, et al. Ann Oncol 2018; 29(suppl 5): abstr LBA-002
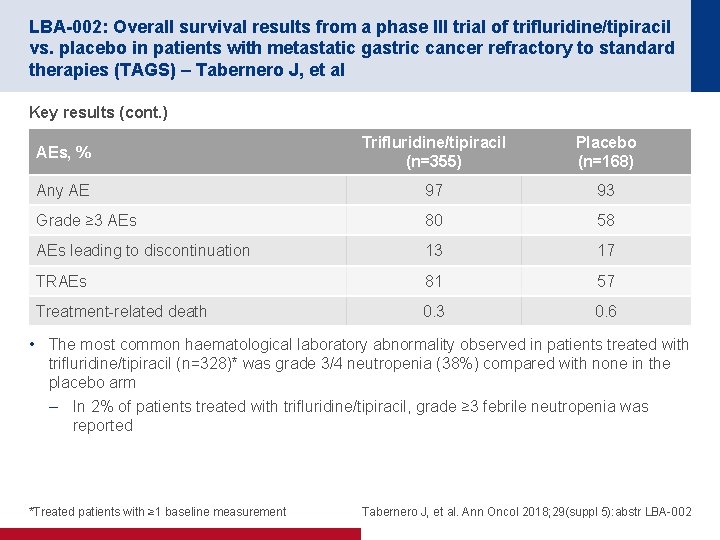
LBA-002: Overall survival results from a phase III trial of trifluridine/tipiracil vs. placebo in patients with metastatic gastric cancer refractory to standard therapies (TAGS) – Tabernero J, et al Key results (cont. ) AEs, % Trifluridine/tipiracil (n=355) Placebo (n=168) Any AE 97 93 Grade ≥ 3 AEs 80 58 AEs leading to discontinuation 13 17 TRAEs 81 57 Treatment-related death 0. 3 0. 6 • The most common haematological laboratory abnormality observed in patients treated with trifluridine/tipiracil (n=328)* was grade 3/4 neutropenia (38%) compared with none in the placebo arm – In 2% of patients treated with trifluridine/tipiracil, grade ≥ 3 febrile neutropenia was reported *Treated patients with ≥ 1 baseline measurement Tabernero J, et al. Ann Oncol 2018; 29(suppl 5): abstr LBA-002

LBA-002: Overall survival results from a phase III trial of trifluridine/tipiracil vs. placebo in patients with metastatic gastric cancer refractory to standard therapies (TAGS) – Tabernero J, et al Conclusions • In heavily pre-treated patients with metastatic gastric cancer, trifluridine/tipiracil was associated with a clinically meaningful and statistically significant improvement in survival vs. placebo • No new safety signals were noted and the safety profile was consistent with that previously seen in other patient populations Tabernero J, et al. Ann Oncol 2018; 29(suppl 5): abstr LBA-002
O-010: Cisplatin/5 -fluorouracil +/- panitumumab for patients with nonresectable, advanced or metastatic esophageal squamous cell cancer: A randomized phase III AIO/EORTC trial with an extensive biomarker program – Moehler M, et al Study objective • To assess the efficacy and safety of cisplatin + 5 FU with or without panitumumab in patients with ESCC in an AIO/EORTC study* Key patient inclusion criteria • Unresectable locally advanced or metastatic ESCC Panitumumab† + cisplatin + 5 FU‡ (n=73) PD Cisplatin + 5 FU‡ (n=73) PD R 1: 1 • ECOG PS 0– 1 (n=146) ENDPOINTS • BOR, OS, PFS and safety *Study stopped early due to futility and potential safety concerns; †panitumumab 9 mg/kg D 1 of each cycle prior to CT q 3 w; ‡cisplatin 100 mg/m 2 iv infusion over 2 hours D 1 + 5 FU 1000 mg/m 2 iv infusion over 24 hours D 1– 4 q 3 w Moehler M, et al. Ann Oncol 2018; 29(suppl 5): abstr O-010
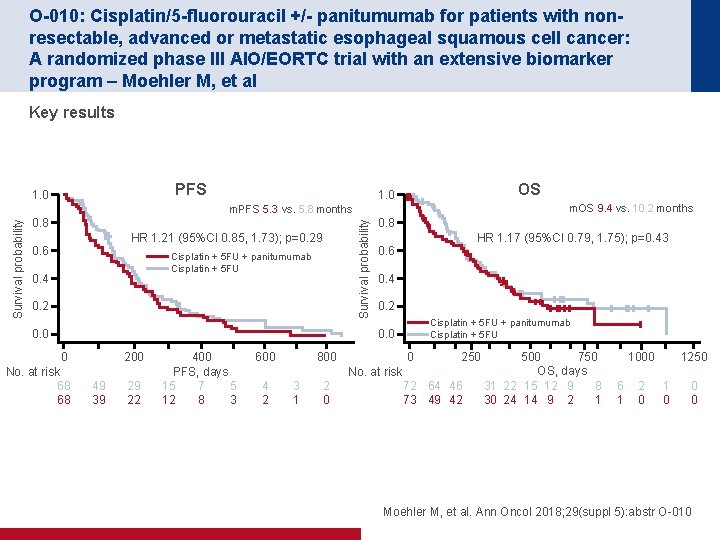
O-010: Cisplatin/5 -fluorouracil +/- panitumumab for patients with nonresectable, advanced or metastatic esophageal squamous cell cancer: A randomized phase III AIO/EORTC trial with an extensive biomarker program – Moehler M, et al Key results PFS 1. 0 OS 1. 0 m. OS 9. 4 vs. 10. 2 months Survival probability m. PFS 5. 3 vs. 5. 8 months 0. 8 HR 1. 21 (95%CI 0. 85, 1. 73); p=0. 29 0. 6 Cisplatin + 5 FU + panitumumab Cisplatin + 5 FU 0. 4 0. 2 0. 0 0 No. at risk 68 68 0. 8 HR 1. 17 (95%CI 0. 79, 1. 75); p=0. 43 0. 6 0. 4 0. 2 Cisplatin + 5 FU + panitumumab Cisplatin + 5 FU 0. 0 49 39 200 29 22 400 PFS, days 5 15 7 3 12 8 600 0 800 4 2 3 1 2 0 500 750 OS, days 31 22 15 12 9 8 30 24 14 9 2 1 250 No. at risk 72 64 46 73 49 42 1000 6 1 2 0 1250 1 0 0 0 Moehler M, et al. Ann Oncol 2018; 29(suppl 5): abstr O-010
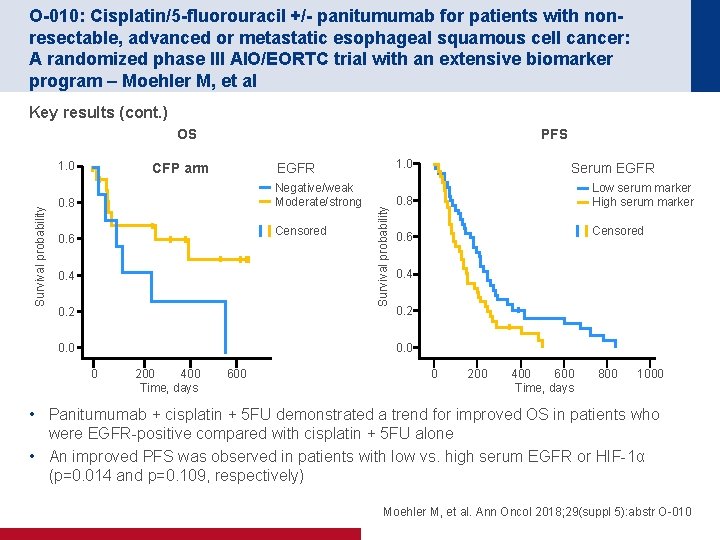
O-010: Cisplatin/5 -fluorouracil +/- panitumumab for patients with nonresectable, advanced or metastatic esophageal squamous cell cancer: A randomized phase III AIO/EORTC trial with an extensive biomarker program – Moehler M, et al Key results (cont. ) OS CFP arm 1. 0 EGFR Negative/weak Moderate/strong 0. 8 Censored 0. 6 0. 4 0. 2 Survival probability 1. 0 PFS Serum EGFR Low serum marker High serum marker 0. 8 Censored 0. 6 0. 4 0. 2 0. 0 0 200 400 Time, days 600 0 200 400 600 Time, days 800 1000 • Panitumumab + cisplatin + 5 FU demonstrated a trend for improved OS in patients who were EGFR-positive compared with cisplatin + 5 FU alone • An improved PFS was observed in patients with low vs. high serum EGFR or HIF-1α (p=0. 014 and p=0. 109, respectively) Moehler M, et al. Ann Oncol 2018; 29(suppl 5): abstr O-010

O-010: Cisplatin/5 -fluorouracil +/- panitumumab for patients with nonresectable, advanced or metastatic esophageal squamous cell cancer: A randomized phase III AIO/EORTC trial with an extensive biomarker program – Moehler M, et al Key results (cont. ) • At least one SAE was observed in 83. 3% vs. 78. 6% of patients in the panitumumab + cisplatin + 5 FU vs. cisplatin + 5 FU arms, respectively • The most common grade ≥ 3 AEs were low neutrophils (21% vs. 24%) and anaemia (13% vs. 16%) in panitumumab + cisplatin + 5 FU vs. cisplatin + 5 FU arms, respectively Conclusions • In patients with locally advanced or metastatic ESCC, the addition of panitumumab to cisplatin and 5 FU was not associated with improved OS compared with cisplatin + 5 FU alone • EGFR-1, HIF-1α and serum EGFR under EGFR-1 inhibition may be potential biomarkers in locally advanced or metastatic ESCC Moehler M, et al. Ann Oncol 2018; 29(suppl 5): abstr O-010

CANCERS OF THE PANCREAS, SMALL BOWEL AND HEPATOBILIARY TRACT

PANCREATIC CANCER

O-002: Geographic variation in systemic treatment of metastatic pancreatic adenocarcinoma (m. PAC) patients in real world across Europe – Taieb J, et al Study objective • To investigate the geographical variations in treatment selection in European patients from 9 different countries who completed 1 L treatment for metastatic pancreatic cancer Key patient inclusion criteria • Metastatic pancreatic cancer • Completed 1 L treatment* • ≥ 18 years 2 L treatment started/completed (n=1666) PD (n=2565) • A retrospective electronic chart review based on data gathered from patient records • The following information was obtained: – – General disease information and patient characteristics Disease characteristics at diagnosis Initial treatment for pancreatic cancer Details of 1 L, 2 L and 3 L treatment *Between July 2014 and January 2016 Taieb J, et al. Ann Oncol 2018; 29(suppl 5): abstr O-002
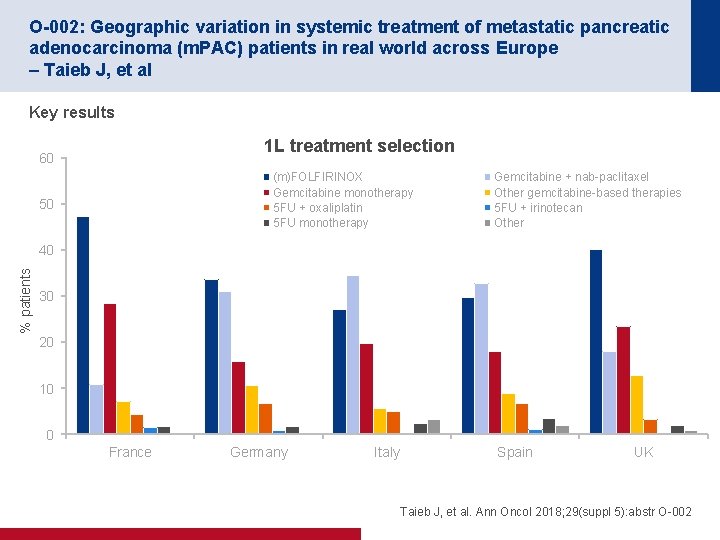
O-002: Geographic variation in systemic treatment of metastatic pancreatic adenocarcinoma (m. PAC) patients in real world across Europe – Taieb J, et al Key results 1 L treatment selection 60 (m)FOLFIRINOX Gemcitabine monotherapy 5 FU + oxaliplatin 5 FU monotherapy 50 Gemcitabine + nab-paclitaxel Other gemcitabine-based therapies 5 FU + irinotecan Other % patients 40 30 20 10 0 France Germany Italy Spain UK Taieb J, et al. Ann Oncol 2018; 29(suppl 5): abstr O-002
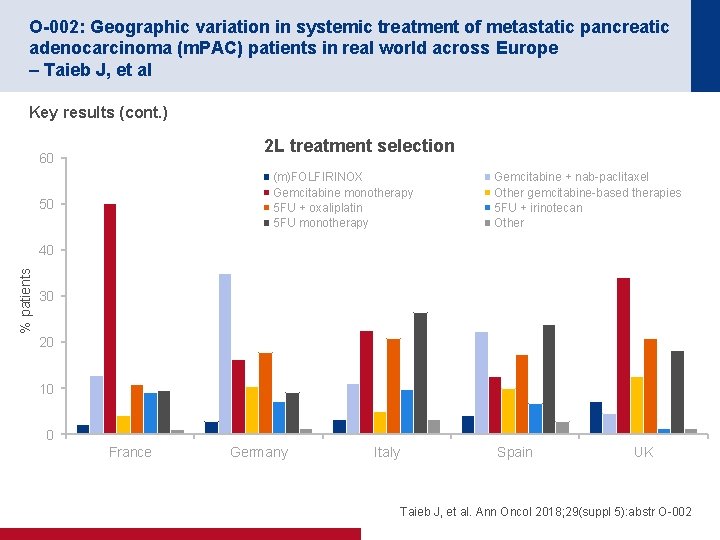
O-002: Geographic variation in systemic treatment of metastatic pancreatic adenocarcinoma (m. PAC) patients in real world across Europe – Taieb J, et al Key results (cont. ) 2 L treatment selection 60 (m)FOLFIRINOX Gemcitabine monotherapy 5 FU + oxaliplatin 5 FU monotherapy 50 Gemcitabine + nab-paclitaxel Other gemcitabine-based therapies 5 FU + irinotecan Other % patients 40 30 20 10 0 France Germany Italy Spain UK Taieb J, et al. Ann Oncol 2018; 29(suppl 5): abstr O-002

O-002: Geographic variation in systemic treatment of metastatic pancreatic adenocarcinoma (m. PAC) patients in real world across Europe – Taieb J, et al Conclusions • In European patients with metastatic pancreatic cancer the 1 L treatment selection was broadly consistent with ESMO recommendations – There was variation between countries in the relative proportion of different treatments used – 1 L treatment choice depended on local reimbursement status and the patient’s condition • 2 L treatment selection varied widely between countries – 2 L choice was dependent on 1 L treatment and local reimbursement policies • At the time of the study, there were no approved 2 L treatments for patients with metastatic pancreatic cancer Taieb J, et al. Ann Oncol 2018; 29(suppl 5): abstr O-002

HEPATOCELLULAR CARCINOMA
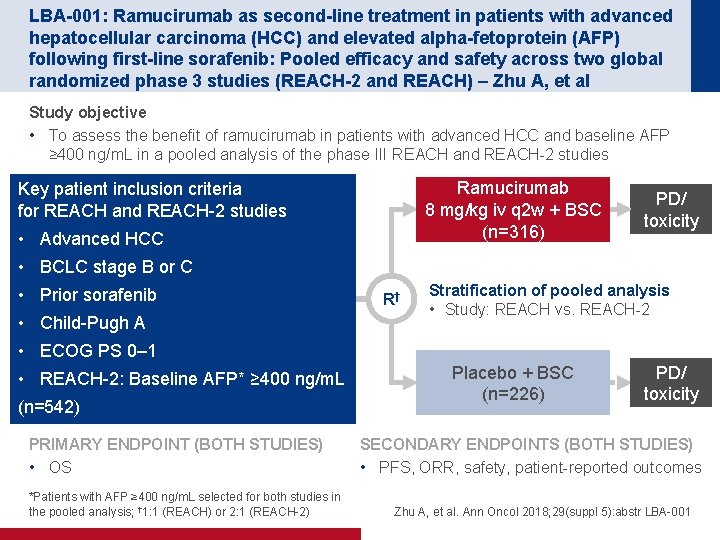
LBA-001: Ramucirumab as second-line treatment in patients with advanced hepatocellular carcinoma (HCC) and elevated alpha-fetoprotein (AFP) following first-line sorafenib: Pooled efficacy and safety across two global randomized phase 3 studies (REACH-2 and REACH) – Zhu A, et al Study objective • To assess the benefit of ramucirumab in patients with advanced HCC and baseline AFP ≥ 400 ng/m. L in a pooled analysis of the phase III REACH and REACH-2 studies Ramucirumab 8 mg/kg iv q 2 w + BSC (n=316) Key patient inclusion criteria for REACH and REACH-2 studies • Advanced HCC PD/ toxicity • BCLC stage B or C • Prior sorafenib • Child-Pugh A R† Stratification of pooled analysis • Study: REACH vs. REACH-2 • ECOG PS 0– 1 • REACH-2: Baseline AFP* ≥ 400 ng/m. L (n=542) PRIMARY ENDPOINT (BOTH STUDIES) • OS *Patients with AFP ≥ 400 ng/m. L selected for both studies in the pooled analysis; † 1: 1 (REACH) or 2: 1 (REACH-2) Placebo + BSC (n=226) PD/ toxicity SECONDARY ENDPOINTS (BOTH STUDIES) • PFS, ORR, safety, patient-reported outcomes Zhu A, et al. Ann Oncol 2018; 29(suppl 5): abstr LBA-001
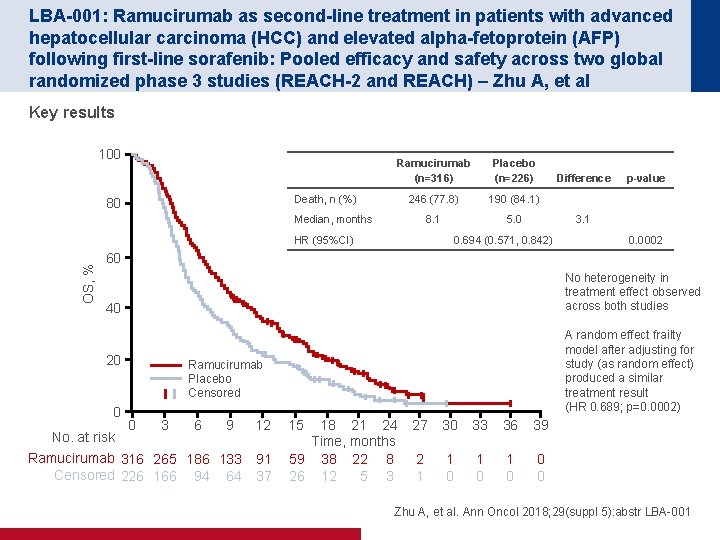
LBA-001: Ramucirumab as second-line treatment in patients with advanced hepatocellular carcinoma (HCC) and elevated alpha-fetoprotein (AFP) following first-line sorafenib: Pooled efficacy and safety across two global randomized phase 3 studies (REACH-2 and REACH) – Zhu A, et al Key results 100 Ramucirumab (n=316) Placebo (n=226) 246 (77. 8) 190 (84. 1) 8. 1 5. 0 Death, n (%) 80 Median, months OS, % HR (95%CI) Difference p-value 3. 1 0. 694 (0. 571, 0. 842) 0. 0002 60 No heterogeneity in treatment effect observed across both studies 40 20 0 No. at risk A random effect frailty model after adjusting for study (as random effect) produced a similar treatment result (HR 0. 689; p=0. 0002) Ramucirumab Placebo Censored 0 3 6 9 Ramucirumab 316 265 186 133 Censored 226 166 94 64 12 91 37 15 18 21 24 Time, months 59 38 22 8 26 12 5 3 27 30 33 36 39 2 1 1 0 1 0 0 0 Zhu A, et al. Ann Oncol 2018; 29(suppl 5): abstr LBA-001
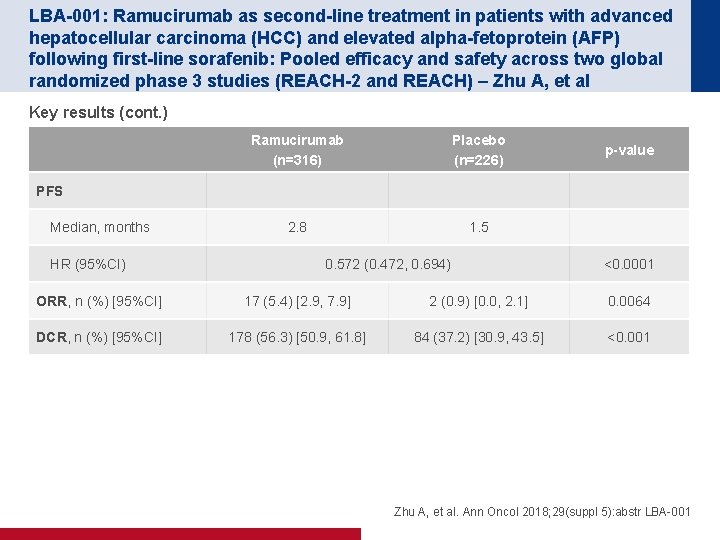
LBA-001: Ramucirumab as second-line treatment in patients with advanced hepatocellular carcinoma (HCC) and elevated alpha-fetoprotein (AFP) following first-line sorafenib: Pooled efficacy and safety across two global randomized phase 3 studies (REACH-2 and REACH) – Zhu A, et al Key results (cont. ) Ramucirumab (n=316) Placebo (n=226) 2. 8 1. 5 p-value PFS Median, months HR (95%CI) 0. 572 (0. 472, 0. 694) <0. 0001 ORR, n (%) [95%CI] 17 (5. 4) [2. 9, 7. 9] 2 (0. 9) [0. 0, 2. 1] 0. 0064 DCR, n (%) [95%CI] 178 (56. 3) [50. 9, 61. 8] 84 (37. 2) [30. 9, 43. 5] <0. 001 Zhu A, et al. Ann Oncol 2018; 29(suppl 5): abstr LBA-001
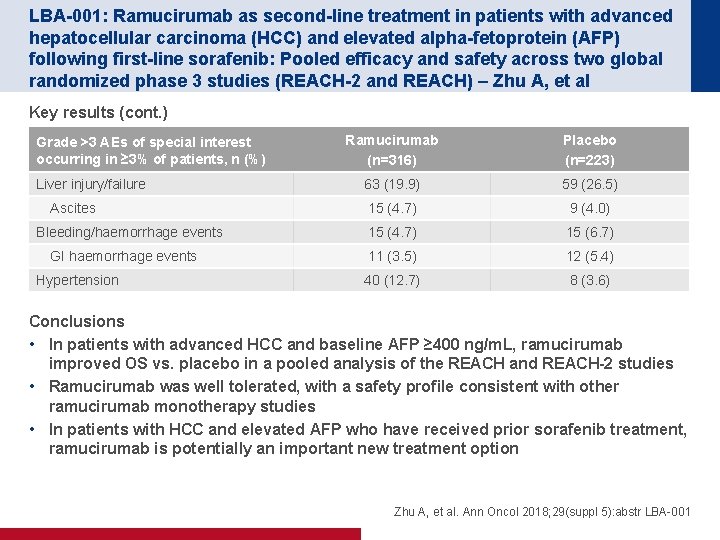
LBA-001: Ramucirumab as second-line treatment in patients with advanced hepatocellular carcinoma (HCC) and elevated alpha-fetoprotein (AFP) following first-line sorafenib: Pooled efficacy and safety across two global randomized phase 3 studies (REACH-2 and REACH) – Zhu A, et al Key results (cont. ) Grade >3 AEs of special interest occurring in ≥ 3% of patients, n (%) Liver injury/failure Ascites Bleeding/haemorrhage events GI haemorrhage events Hypertension Ramucirumab (n=316) Placebo (n=223) 63 (19. 9) 59 (26. 5) 15 (4. 7) 9 (4. 0) 15 (4. 7) 15 (6. 7) 11 (3. 5) 12 (5. 4) 40 (12. 7) 8 (3. 6) Conclusions • In patients with advanced HCC and baseline AFP ≥ 400 ng/m. L, ramucirumab improved OS vs. placebo in a pooled analysis of the REACH and REACH-2 studies • Ramucirumab was well tolerated, with a safety profile consistent with other ramucirumab monotherapy studies • In patients with HCC and elevated AFP who have received prior sorafenib treatment, ramucirumab is potentially an important new treatment option Zhu A, et al. Ann Oncol 2018; 29(suppl 5): abstr LBA-001
O-011: Assessment of tumor response, AFP response, and time to progression in the phase 3 CELESTIAL trial of cabozantinib versus placebo in advanced hepatocellular carcinoma (HCC) – Merle P, et al Study objective • To assess the tumour response, AFP response and TTP in patients with advanced HCC receiving cabozantinib vs. placebo in the CELESTIAL trial Cabozantinib 60 mg/day oral (n=470) Key patient inclusion criteria • Advanced HCC • Child-Pugh A • Prior sorafenib • ≤ 2 systemic regimens and progressed following ≥ 1 PD/ toxicity Stratification R 2: 1 • ECOG PS ≤ 1 (n=760) • Disease aetiology (HBV, HCV, other) • Region (Asia, other) • Presence of macrovascular invasion and/or extra-hepatic spread of disease (Y/N) Placebo (n=237) PD/ toxicity EXPLORATORY ENDPOINTS* • Tumour response, AFP response, TTP *Primary (OS) and secondary (PFS, ORR and safety) endpoints presented previously Merle P, et al. Ann Oncol 2018; 29(suppl 5): abstr O-011
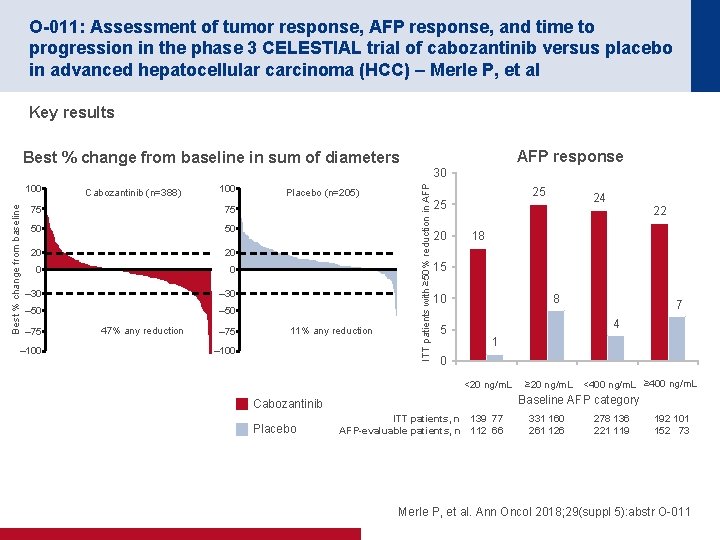
O-011: Assessment of tumor response, AFP response, and time to progression in the phase 3 CELESTIAL trial of cabozantinib versus placebo in advanced hepatocellular carcinoma (HCC) – Merle P, et al Key results AFP response Best % change from baseline in sum of diameters Best % change from baseline 100 Cabozantinib (n=388) 100 75 75 50 50 20 20 0 0 – 30 – 50 – 75 – 100 47% any reduction – 75 Placebo (n=205) 11% any reduction – 100 ITT patients with ≥ 50% reduction in AFP 30 25 25 20 22 18 15 8 10 5 7 4 1 0 <20 ng/m. L ≥ 20 ng/m. L <400 ng/m. L ≥ 400 ng/m. L Baseline AFP category Cabozantinib Placebo 24 ITT patients, n 139 77 AFP-evaluable patients, n 112 66 331 160 261 126 278 136 221 119 192 101 152 73 Merle P, et al. Ann Oncol 2018; 29(suppl 5): abstr O-011
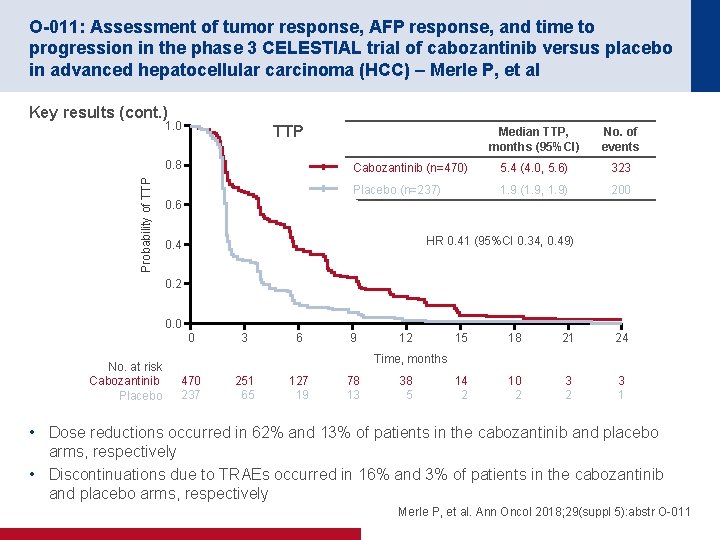
O-011: Assessment of tumor response, AFP response, and time to progression in the phase 3 CELESTIAL trial of cabozantinib versus placebo in advanced hepatocellular carcinoma (HCC) – Merle P, et al Key results (cont. ) 1. 0 TTP Probability of TTP 0. 8 Median TTP, months (95%CI) No. of events Cabozantinib (n=470) 5. 4 (4. 0, 5. 6) 323 Placebo (n=237) 1. 9 (1. 9, 1. 9) 200 0. 6 HR 0. 41 (95%CI 0. 34, 0. 49) 0. 4 0. 2 0. 0 0 No. at risk Cabozantinib Placebo 3 6 9 12 15 18 21 24 14 2 10 2 3 1 Time, months 470 237 251 65 127 19 78 13 38 5 • Dose reductions occurred in 62% and 13% of patients in the cabozantinib and placebo arms, respectively • Discontinuations due to TRAEs occurred in 16% and 3% of patients in the cabozantinib and placebo arms, respectively Merle P, et al. Ann Oncol 2018; 29(suppl 5): abstr O-011
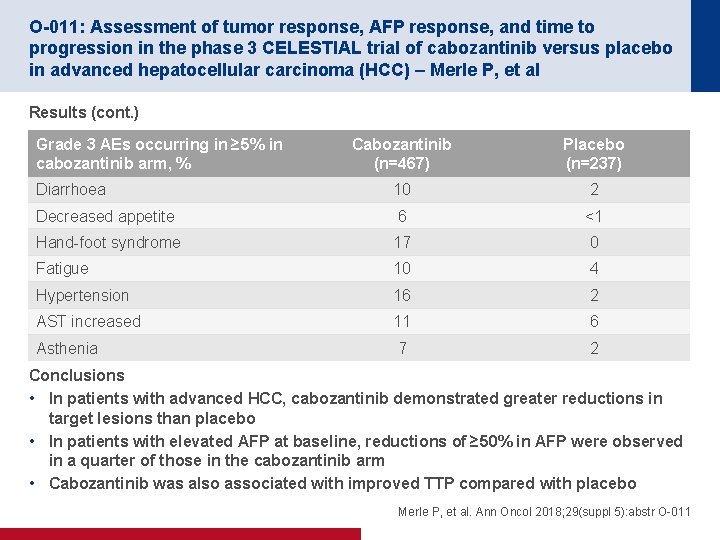
O-011: Assessment of tumor response, AFP response, and time to progression in the phase 3 CELESTIAL trial of cabozantinib versus placebo in advanced hepatocellular carcinoma (HCC) – Merle P, et al Results (cont. ) Grade 3 AEs occurring in ≥ 5% in cabozantinib arm, % Cabozantinib (n=467) Placebo (n=237) Diarrhoea 10 2 Decreased appetite 6 <1 Hand-foot syndrome 17 0 Fatigue 10 4 Hypertension 16 2 AST increased 11 6 Asthenia 7 2 Conclusions • In patients with advanced HCC, cabozantinib demonstrated greater reductions in target lesions than placebo • In patients with elevated AFP at baseline, reductions of ≥ 50% in AFP were observed in a quarter of those in the cabozantinib arm • Cabozantinib was also associated with improved TTP compared with placebo Merle P, et al. Ann Oncol 2018; 29(suppl 5): abstr O-011

CANCERS OF THE COLON, RECTUM AND ANUS

LBA-004: Efficacy and safety results from IMblaze 370, a randomized phase III study comparing atezolizumab plus cobimetinib and atezolizumab monotherapy vs. regorafenib in chemotherapy-refractory metastatic colorectal cancer – Bendell J, et al Key patient inclusion criteria Atezolizumab 840 mg iv q 2 w + cobimetinib 60 mg oral 21/7 days (n=183) • Unresectable locally advanced or metastatic CRC • ≥ 2 prior regimens of cytotoxic chemotherapy R 2: 1: 1 • MSI-H capped at 5% • ECOG PS 0– 1 (n=363) Stratification Atezolizumab 1200 mg iv q 3 w (n=90) Regorafenib 160 mg oral 21/7 days (n=90) Loss of clinical benefit Study objective • To assess the efficacy and safety of atezolizumab + cobimetinib vs. atezolizumab alone vs. regorafenib in patients with chemotherapy refractory m. CRC in the IMblaze 370 study • Extended RAS mutation status (≥ 50% of patients in each arm) • Time since diagnosis of first metastasis (<18 vs. ≥ 18 months) PRIMARY ENDPOINT • OS for atezolizumab + cobimetinib or atezolizumab vs. regorafenib SECONDARY ENDPOINTS • PFS, ORR, Do. R Bendell J, et al. Ann Oncol 2018; 29(suppl 5): abstr LBA-004
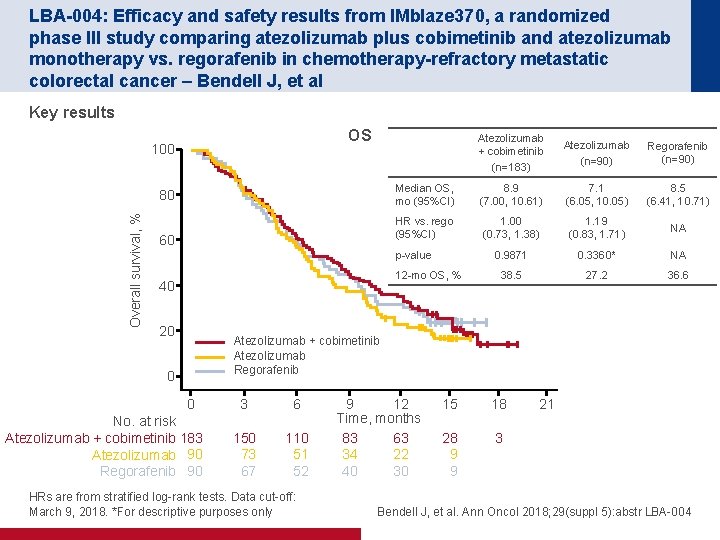
LBA-004: Efficacy and safety results from IMblaze 370, a randomized phase III study comparing atezolizumab plus cobimetinib and atezolizumab monotherapy vs. regorafenib in chemotherapy-refractory metastatic colorectal cancer – Bendell J, et al Key results OS 100 Overall survival, % 80 60 Atezolizumab + cobimetinib (n=183) Atezolizumab (n=90) Regorafenib (n=90) Median OS, mo (95%CI) 8. 9 (7. 00, 10. 61) 7. 1 (6. 05, 10. 05) 8. 5 (6. 41, 10. 71) HR vs. rego (95%CI) 1. 00 (0. 73, 1. 38) 1. 19 (0. 83, 1. 71) NA 0. 9871 0. 3360* NA 38. 5 27. 2 36. 6 p-value 12 -mo OS, % 40 20 Atezolizumab + cobimetinib Atezolizumab Regorafenib 0 0 No. at risk Atezolizumab + cobimetinib 183 Atezolizumab 90 Regorafenib 90 3 6 150 73 67 110 51 52 HRs are from stratified log-rank tests. Data cut-off: March 9, 2018. *For descriptive purposes only 9 12 Time, months 83 63 34 22 40 30 15 18 28 9 9 3 21 Bendell J, et al. Ann Oncol 2018; 29(suppl 5): abstr LBA-004
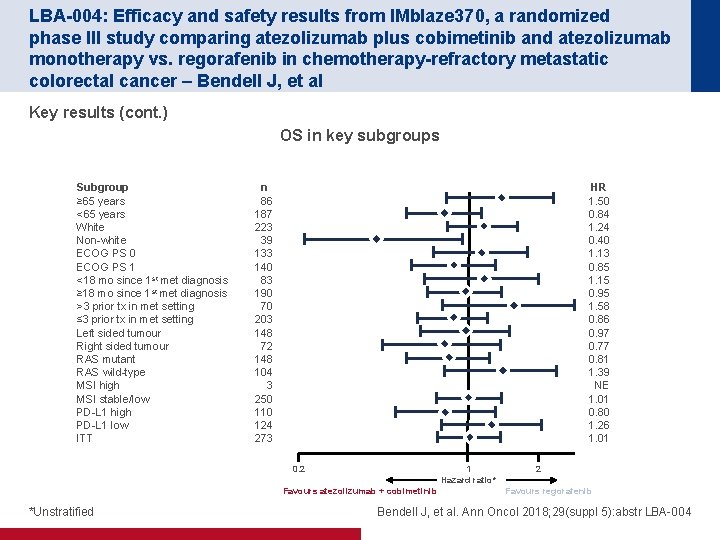
LBA-004: Efficacy and safety results from IMblaze 370, a randomized phase III study comparing atezolizumab plus cobimetinib and atezolizumab monotherapy vs. regorafenib in chemotherapy-refractory metastatic colorectal cancer – Bendell J, et al Key results (cont. ) OS in key subgroups Subgroup ≥ 65 years <65 years White Non-white ECOG PS 0 ECOG PS 1 <18 mo since 1 st met diagnosis ≥ 18 mo since 1 st met diagnosis >3 prior tx in met setting ≤ 3 prior tx in met setting Left sided tumour Right sided tumour RAS mutant RAS wild-type MSI high MSI stable/low PD-L 1 high PD-L 1 low ITT n 86 187 223 39 133 140 83 190 70 203 148 72 148 104 3 250 110 124 273 HR 1. 50 0. 84 1. 24 0. 40 1. 13 0. 85 1. 15 0. 95 1. 58 0. 86 0. 97 0. 77 0. 81 1. 39 NE 1. 01 0. 80 1. 26 1. 01 0. 2 1 Hazard ratio* Favours atezolizumab + cobimetinib *Unstratified 2 Favours regorafenib Bendell J, et al. Ann Oncol 2018; 29(suppl 5): abstr LBA-004
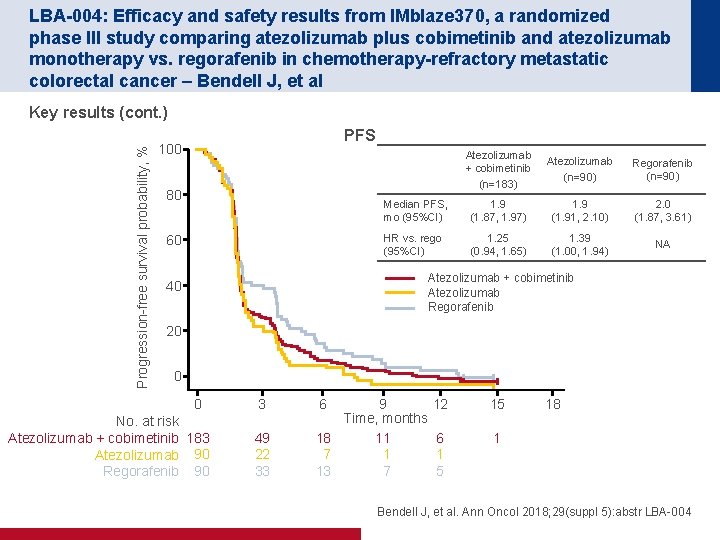
LBA-004: Efficacy and safety results from IMblaze 370, a randomized phase III study comparing atezolizumab plus cobimetinib and atezolizumab monotherapy vs. regorafenib in chemotherapy-refractory metastatic colorectal cancer – Bendell J, et al Progression-free survival probability, % Key results (cont. ) PFS 100 80 60 Atezolizumab + cobimetinib (n=183) Atezolizumab (n=90) Regorafenib (n=90) Median PFS, mo (95%CI) 1. 9 (1. 87, 1. 97) 1. 9 (1. 91, 2. 10) 2. 0 (1. 87, 3. 61) HR vs. rego (95%CI) 1. 25 (0. 94, 1. 65) 1. 39 (1. 00, 1. 94) NA Atezolizumab + cobimetinib Atezolizumab Regorafenib 40 20 0 0 No. at risk Atezolizumab + cobimetinib 183 Atezolizumab 90 Regorafenib 90 3 49 22 33 9 Time, months 11 18 1 7 7 13 6 12 15 6 1 5 1 18 Bendell J, et al. Ann Oncol 2018; 29(suppl 5): abstr LBA-004
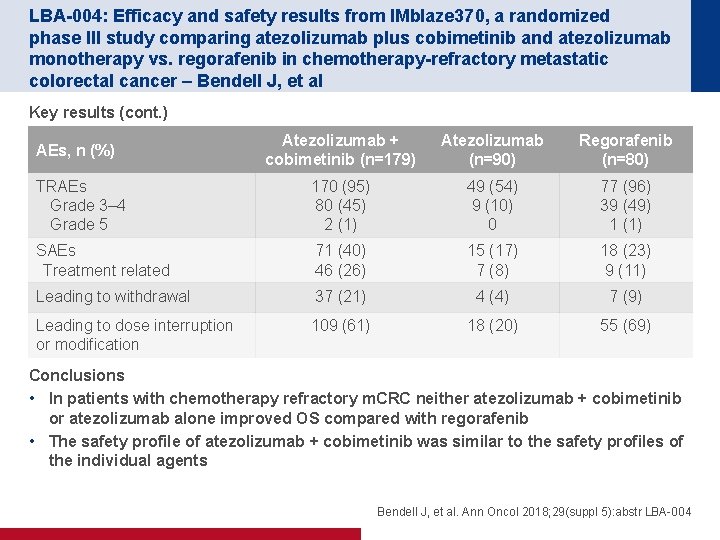
LBA-004: Efficacy and safety results from IMblaze 370, a randomized phase III study comparing atezolizumab plus cobimetinib and atezolizumab monotherapy vs. regorafenib in chemotherapy-refractory metastatic colorectal cancer – Bendell J, et al Key results (cont. ) Atezolizumab + cobimetinib (n=179) Atezolizumab (n=90) Regorafenib (n=80) TRAEs Grade 3– 4 Grade 5 170 (95) 80 (45) 2 (1) 49 (54) 9 (10) 0 77 (96) 39 (49) 1 (1) SAEs Treatment related 71 (40) 46 (26) 15 (17) 7 (8) 18 (23) 9 (11) Leading to withdrawal 37 (21) 4 (4) 7 (9) Leading to dose interruption or modification 109 (61) 18 (20) 55 (69) AEs, n (%) Conclusions • In patients with chemotherapy refractory m. CRC neither atezolizumab + cobimetinib or atezolizumab alone improved OS compared with regorafenib • The safety profile of atezolizumab + cobimetinib was similar to the safety profiles of the individual agents Bendell J, et al. Ann Oncol 2018; 29(suppl 5): abstr LBA-004

O-012: Safety and effectiveness of regorafenib in patients with metastatic colorectal cancer (m. CRC) in routine clinical practice: Final analysis from prospective, observational CORRELATE study – Ducreux M, et al Study objectives • To assess the efficacy and safety of regorafenib in patients with m. CRC in the real-world CORRELATE study Key patient inclusion criteria • m. CRC • Previously treated with other approved therapies • Physician’s decision to treat with regorafenib Regorafenib at discretion of physician according to local approved label PD (n=1037) PRIMARY ENDPOINT • Safety SECONDARY ENDPOINTS • OS, PFS Ducreux M, et al. Ann Oncol 2018; 29(suppl 5): abstr O-012
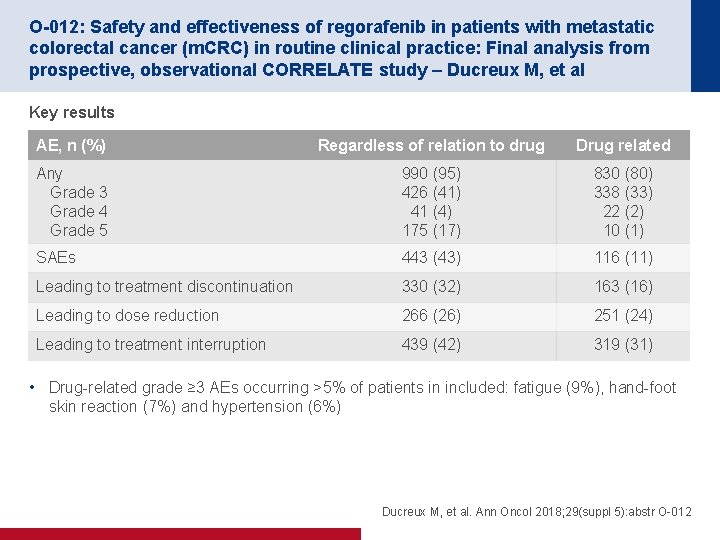
O-012: Safety and effectiveness of regorafenib in patients with metastatic colorectal cancer (m. CRC) in routine clinical practice: Final analysis from prospective, observational CORRELATE study – Ducreux M, et al Key results AE, n (%) Regardless of relation to drug Drug related Any Grade 3 Grade 4 Grade 5 990 (95) 426 (41) 41 (4) 175 (17) 830 (80) 338 (33) 22 (2) 10 (1) SAEs 443 (43) 116 (11) Leading to treatment discontinuation 330 (32) 163 (16) Leading to dose reduction 266 (26) 251 (24) Leading to treatment interruption 439 (42) 319 (31) • Drug-related grade ≥ 3 AEs occurring >5% of patients in included: fatigue (9%), hand-foot skin reaction (7%) and hypertension (6%) Ducreux M, et al. Ann Oncol 2018; 29(suppl 5): abstr O-012
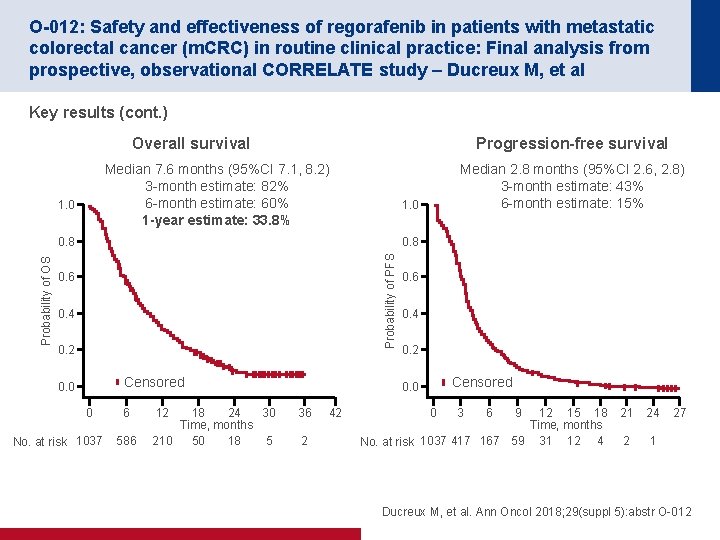
O-012: Safety and effectiveness of regorafenib in patients with metastatic colorectal cancer (m. CRC) in routine clinical practice: Final analysis from prospective, observational CORRELATE study – Ducreux M, et al Key results (cont. ) Overall survival Progression-free survival Median 7. 6 months (95%CI 7. 1, 8. 2) 3 -month estimate: 82% 6 -month estimate: 60% 1 -year estimate: 33. 8% 1. 0 0. 8 Probability of PFS 0. 8 Probability of OS Median 2. 8 months (95%CI 2. 6, 2. 8) 3 -month estimate: 43% 6 -month estimate: 15% 0. 6 0. 4 0. 2 Censored 0. 0 0 No. at risk 1037 6 586 12 18 24 30 Time, months 210 50 18 5 0. 6 0. 4 0. 2 Censored 0. 0 36 2 42 0 3 6 9 12 15 18 Time, months No. at risk 1037 417 167 59 31 12 4 21 24 2 1 27 Ducreux M, et al. Ann Oncol 2018; 29(suppl 5): abstr O-012

O-012: Safety and effectiveness of regorafenib in patients with metastatic colorectal cancer (m. CRC) in routine clinical practice: Final analysis from prospective, observational CORRELATE study – Ducreux M, et al Conclusions • In patients with m. CRC, regorafenib demonstrated a safety profile that was similar to previous findings • Nearly 50% of the patients initiated regorafenib at a lower dose than the recommended 160 mg/day • Survival rates with regorafenib were comparable to those seen in previous phase III clinical trials even with flexible dosing Ducreux M, et al. Ann Oncol 2018; 29(suppl 5): abstr O-012

O-013: Safety and efficacy of trifluridine/tipiracil in previously treated metastatic colorectal cancer (m. CRC): Preliminary results from the phase IIIb, international, open-label, early-access PRECONNECT study – Falcone A, et al Study objective • To assess the efficacy and safety of trifluridine/tipiracil in previously treated patients with m. CRC in the open-label, early access PRECONNECT study (preliminary data reported) Key patient inclusion criteria • m. CRC • ≥ 2 prior chemotherapy regimens • Refractory, intolerant or unsuitable for fluoropyrimidine, irinotecan, oxaliplatin, anti-VEGF or anti-EGFR for RAS WT Trifluridine/tipiracil 35 mg/m 2 oral bid D 1– 5, 8– 12 of 28 -day cycle PD/ toxicity • ECOG PS 0– 1 (n=462) ENDPOINTS • Safety, PFS, ORR, DCR, time to deterioration to ECOG PS ≥ 2, Qo. L Falcone A, et al. Ann Oncol 2018; 29(suppl 5): abstr O-013
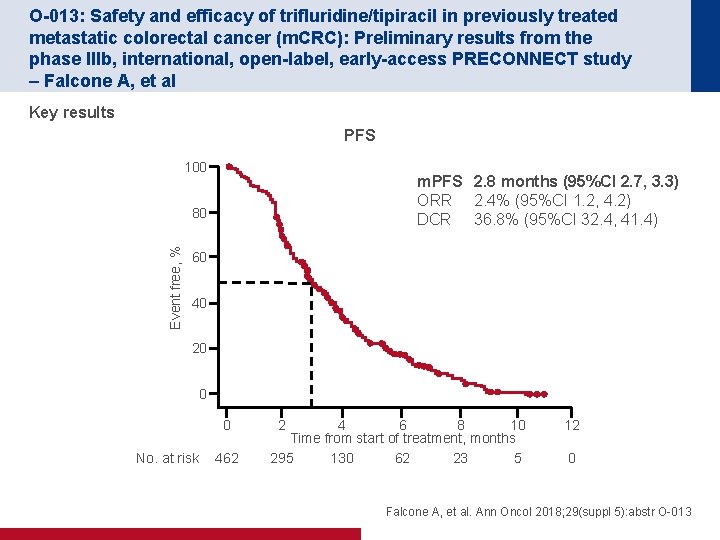
O-013: Safety and efficacy of trifluridine/tipiracil in previously treated metastatic colorectal cancer (m. CRC): Preliminary results from the phase IIIb, international, open-label, early-access PRECONNECT study – Falcone A, et al Key results PFS 100 m. PFS 2. 8 months (95%CI 2. 7, 3. 3) ORR 2. 4% (95%CI 1. 2, 4. 2) DCR 36. 8% (95%CI 32. 4, 41. 4) Event free, % 80 60 40 20 0 0 No. at risk 462 8 10 4 6 Time from start of treatment, months 23 5 295 130 62 2 12 0 Falcone A, et al. Ann Oncol 2018; 29(suppl 5): abstr O-013
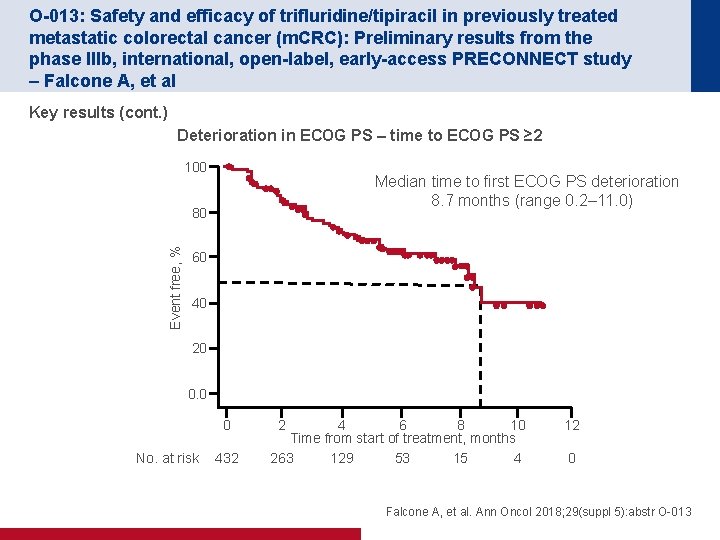
O-013: Safety and efficacy of trifluridine/tipiracil in previously treated metastatic colorectal cancer (m. CRC): Preliminary results from the phase IIIb, international, open-label, early-access PRECONNECT study – Falcone A, et al Key results (cont. ) Deterioration in ECOG PS – time to ECOG PS ≥ 2 100 Median time to first ECOG PS deterioration 8. 7 months (range 0. 2– 11. 0) Event free, % 80 60 40 20 0. 0 0 No. at risk 432 8 10 4 6 Time from start of treatment, months 15 4 263 129 53 2 12 0 Falcone A, et al. Ann Oncol 2018; 29(suppl 5): abstr O-013
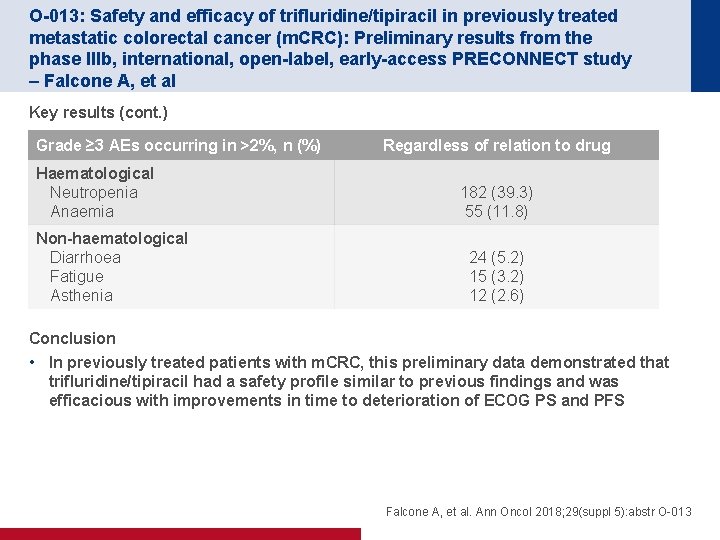
O-013: Safety and efficacy of trifluridine/tipiracil in previously treated metastatic colorectal cancer (m. CRC): Preliminary results from the phase IIIb, international, open-label, early-access PRECONNECT study – Falcone A, et al Key results (cont. ) Grade ≥ 3 AEs occurring in >2%, n (%) Haematological Neutropenia Anaemia Non-haematological Diarrhoea Fatigue Asthenia Regardless of relation to drug 182 (39. 3) 55 (11. 8) 24 (5. 2) 15 (3. 2) 12 (2. 6) Conclusion • In previously treated patients with m. CRC, this preliminary data demonstrated that trifluridine/tipiracil had a safety profile similar to previous findings and was efficacious with improvements in time to deterioration of ECOG PS and PFS Falcone A, et al. Ann Oncol 2018; 29(suppl 5): abstr O-013

O-014: Regorafenib dose optimization study (re. DOS): Randomized phase II trial to evaluate escalating dosing strategy and pre-emptive topical steroids for regorafenib in refractory metastatic colorectal cancer (m. CRC) – An ACCRU network study – Bekaii-Saab T, et al Study objective • To determine the optimal dose of regorafenib to enable maintenance of benefits and improve tolerability in patients with refractory m. CRC in the Re. DOS study Arm A 1: Regorafenib start low* + pre-emptive strategy for PPES Key patient inclusion criteria • Refractory m. CRC • Failure of all standard iv regimens including appropriate biologics R 1: 1: 1: 1 • No prior regorafenib • ECOG PS 0– 1 (n=363) PRIMARY ENDPOINT • Proportion of patients who completed 2 cycles and cold initiate cycle 3 *Cycle 1 week 1 80 mg, week 2 120 mg and week 3 160 mg Arm A 2: Regorafenib start low* + reactive strategy for PPES Arm B 1: Regorafenib 160 mg/day oral 21 days + pre-emptive strategy for PPES Arm B 2: Regorafenib 160 mg/day oral 21 days + reactive strategy for PPES SECONDARY ENDPOINTS • OS, PFS, TTP Bekaii-Saab T, et al. Ann Oncol 2018; 29(suppl 5): abstr O-014
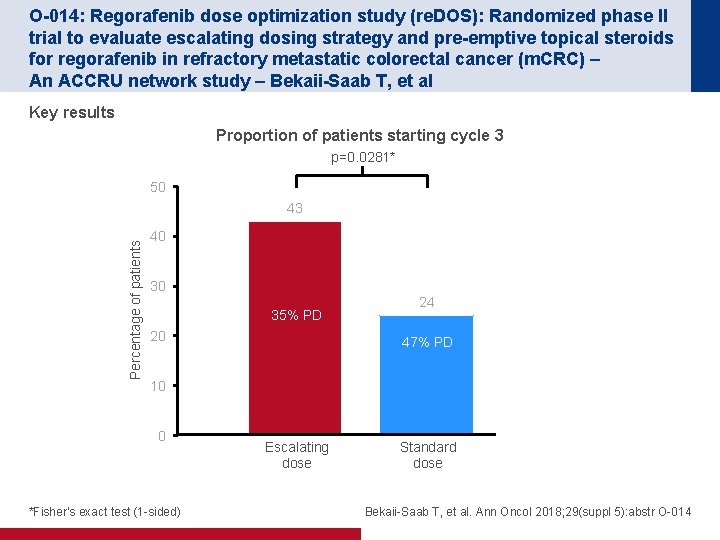
O-014: Regorafenib dose optimization study (re. DOS): Randomized phase II trial to evaluate escalating dosing strategy and pre-emptive topical steroids for regorafenib in refractory metastatic colorectal cancer (m. CRC) – An ACCRU network study – Bekaii-Saab T, et al Key results Proportion of patients starting cycle 3 p=0. 0281* 50 Percentage of patients 43 40 30 35% PD 20 24 47% PD 10 0 *Fisher’s exact test (1 -sided) Escalating dose Standard dose Bekaii-Saab T, et al. Ann Oncol 2018; 29(suppl 5): abstr O-014
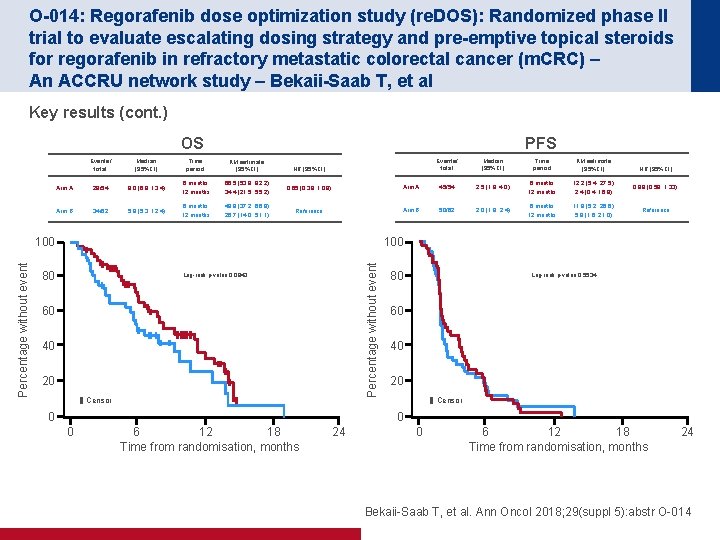
O-014: Regorafenib dose optimization study (re. DOS): Randomized phase II trial to evaluate escalating dosing strategy and pre-emptive topical steroids for regorafenib in refractory metastatic colorectal cancer (m. CRC) – An ACCRU network study – Bekaii-Saab T, et al Key results (cont. ) OS PFS Events/ total Median (95%CI) Time period KM estimate (95%CI) Arm A 29/54 9. 0 (6. 8, 13. 4) 6 months 12 months 66. 5 (53. 8, 82. 2) 34. 4 (21. 5, 55. 2) 0. 65 (0. 39, 1. 08) Arm B 34/62 5. 9 (5. 3, 12. 4) 6 months 12 months 49. 8 (37. 2, 66. 8) 26. 7 (14. 0, 51. 1) Reference Median (95%CI) Time period KM estimate (95%CI) Arm A 45/54 2. 5 (1. 9, 4. 0) 6 months 12. 2 (5. 4, 27. 5) 2. 4 (0. 4, 16. 9) 0. 89 (0. 59, 1. 33) Arm B 50/62 2. 0 (1. 8, 2. 4) 6 months 12 months 11. 8 (5. 2, 26. 6) 5. 9 (1. 6, 21. 0) Reference HR (95%CI) 100 80 Percentage without event 100 Percentage without event Events/ total Log-rank p-value 0. 0943 60 40 20 Censor 0 80 Log-rank p-value 0. 5534 60 40 20 Censor 0 0 6 12 18 Time from randomisation, months 24 Bekaii-Saab T, et al. Ann Oncol 2018; 29(suppl 5): abstr O-014
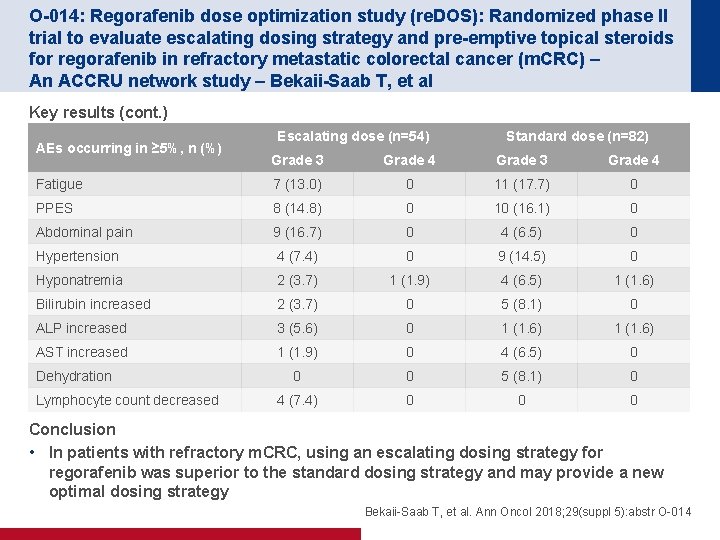
O-014: Regorafenib dose optimization study (re. DOS): Randomized phase II trial to evaluate escalating dosing strategy and pre-emptive topical steroids for regorafenib in refractory metastatic colorectal cancer (m. CRC) – An ACCRU network study – Bekaii-Saab T, et al Key results (cont. ) AEs occurring in ≥ 5%, n (%) Escalating dose (n=54) Standard dose (n=82) Grade 3 Grade 4 Fatigue 7 (13. 0) 0 11 (17. 7) 0 PPES 8 (14. 8) 0 10 (16. 1) 0 Abdominal pain 9 (16. 7) 0 4 (6. 5) 0 Hypertension 4 (7. 4) 0 9 (14. 5) 0 Hyponatremia 2 (3. 7) 1 (1. 9) 4 (6. 5) 1 (1. 6) Bilirubin increased 2 (3. 7) 0 5 (8. 1) 0 ALP increased 3 (5. 6) 0 1 (1. 6) AST increased 1 (1. 9) 0 4 (6. 5) 0 0 0 5 (8. 1) 0 4 (7. 4) 0 0 0 Dehydration Lymphocyte count decreased Conclusion • In patients with refractory m. CRC, using an escalating dosing strategy for regorafenib was superior to the standard dosing strategy and may provide a new optimal dosing strategy Bekaii-Saab T, et al. Ann Oncol 2018; 29(suppl 5): abstr O-014
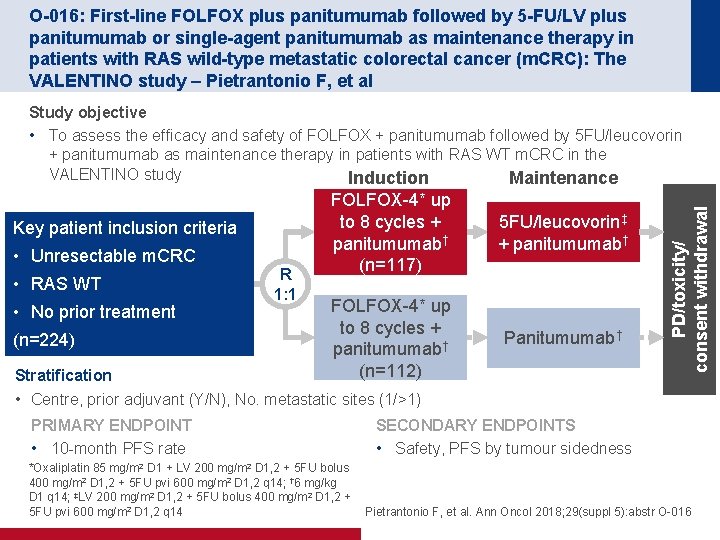
O-016: First-line FOLFOX plus panitumumab followed by 5 -FU/LV plus panitumumab or single-agent panitumumab as maintenance therapy in patients with RAS wild-type metastatic colorectal cancer (m. CRC): The VALENTINO study – Pietrantonio F, et al Key patient inclusion criteria • Unresectable m. CRC • RAS WT • No prior treatment (n=224) R 1: 1 FOLFOX-4* up to 8 cycles + panitumumab† (n=117) 5 FU/leucovorin‡ + panitumumab† FOLFOX-4* up to 8 cycles + panitumumab† (n=112) Panitumumab† Stratification • Centre, prior adjuvant (Y/N), No. metastatic sites (1/>1) PRIMARY ENDPOINT • 10 -month PFS rate PD/toxicity/ consent withdrawal Study objective • To assess the efficacy and safety of FOLFOX + panitumumab followed by 5 FU/leucovorin + panitumumab as maintenance therapy in patients with RAS WT m. CRC in the VALENTINO study Induction Maintenance SECONDARY ENDPOINTS • Safety, PFS by tumour sidedness *Oxaliplatin 85 mg/m 2 D 1 + LV 200 mg/m 2 D 1, 2 + 5 FU bolus 400 mg/m 2 D 1, 2 + 5 FU pvi 600 mg/m 2 D 1, 2 q 14; † 6 mg/kg D 1 q 14; ‡LV 200 mg/m 2 D 1, 2 + 5 FU bolus 400 mg/m 2 D 1, 2 + 5 FU pvi 600 mg/m 2 D 1, 2 q 14 Pietrantonio F, et al. Ann Oncol 2018; 29(suppl 5): abstr O-016
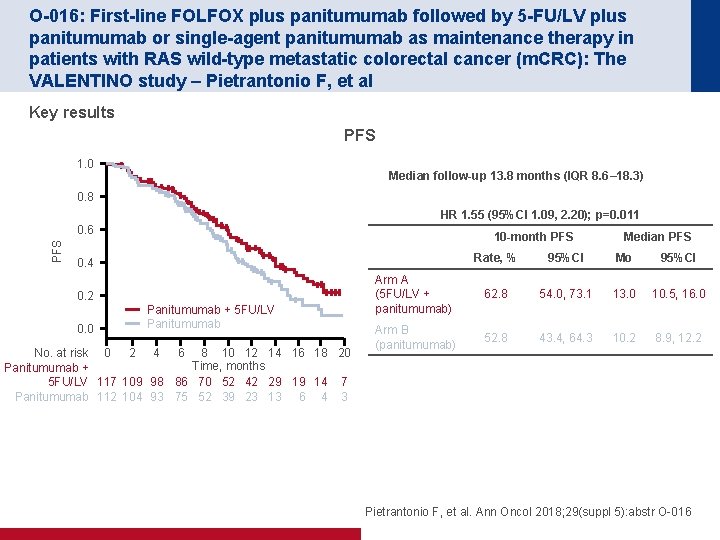
O-016: First-line FOLFOX plus panitumumab followed by 5 -FU/LV plus panitumumab or single-agent panitumumab as maintenance therapy in patients with RAS wild-type metastatic colorectal cancer (m. CRC): The VALENTINO study – Pietrantonio F, et al Key results PFS 1. 0 Median follow-up 13. 8 months (IQR 8. 6– 18. 3) 0. 8 HR 1. 55 (95%CI 1. 09, 2. 20); p=0. 011 PFS 0. 6 10 -month PFS Rate, % 95%CI Mo 95%CI Arm A (5 FU/LV + panitumumab) 62. 8 54. 0, 73. 1 13. 0 10. 5, 16. 0 Arm B (panitumumab) 52. 8 43. 4, 64. 3 10. 2 8. 9, 12. 2 0. 4 0. 2 0. 0 Panitumumab + 5 FU/LV Panitumumab No. at risk 0 2 4 6 8 10 12 14 16 18 20 Time, months Panitumumab + 5 FU/LV 117 109 98 86 70 52 42 29 19 14 7 Panitumumab 112 104 93 75 52 39 23 13 6 4 3 Median PFS Pietrantonio F, et al. Ann Oncol 2018; 29(suppl 5): abstr O-016
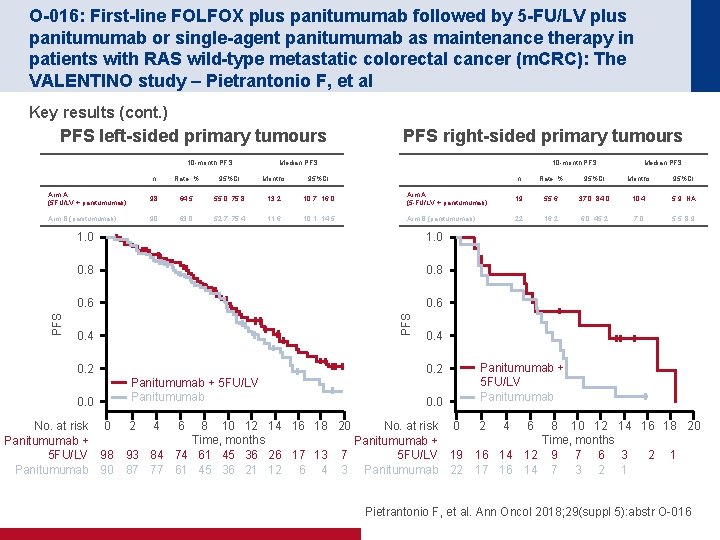
O-016: First-line FOLFOX plus panitumumab followed by 5 -FU/LV plus panitumumab or single-agent panitumumab as maintenance therapy in patients with RAS wild-type metastatic colorectal cancer (m. CRC): The VALENTINO study – Pietrantonio F, et al Key results (cont. ) PFS left-sided primary tumours 10 -month PFS Median PFS n Rate, % 95%CI Months 95%CI Arm A (5 FU/LV + panitumumab) 98 64. 5 55. 0, 75. 8 13. 2 10. 7, 16. 0 Arm B (panitumumab) 90 63. 0 52. 7, 75. 4 11. 6 10. 1, 14. 5 10 -month PFS Rate, % 95%CI Months 95%CI Arm A (5 -FU/LV + panitumumab) 19 55. 6 37. 0, 84. 0 10. 4 5. 9, NA Arm B (panitumumab) 22 16. 2 6. 0, 45. 2 7. 0 5. 5, 8. 9 1. 0 0. 8 0. 6 0. 4 0. 2 0. 0 No. at risk 0 Panitumumab + 5 FU/LV 98 Panitumumab 90 0. 4 0. 2 Panitumumab + 5 FU/LV Panitumumab 0. 0 No. at risk 0 8 10 12 14 16 18 20 Time, months Panitumumab + 5 FU/LV 19 93 84 74 61 45 36 26 17 13 7 87 77 61 45 36 21 12 6 4 3 Panitumumab 22 2 4 6 Median PFS PFS right-sided primary tumours Panitumumab + 5 FU/LV Panitumumab 2 4 6 8 10 12 14 16 18 20 Time, months 16 14 12 9 7 6 3 2 1 17 16 14 7 3 2 1 Pietrantonio F, et al. Ann Oncol 2018; 29(suppl 5): abstr O-016
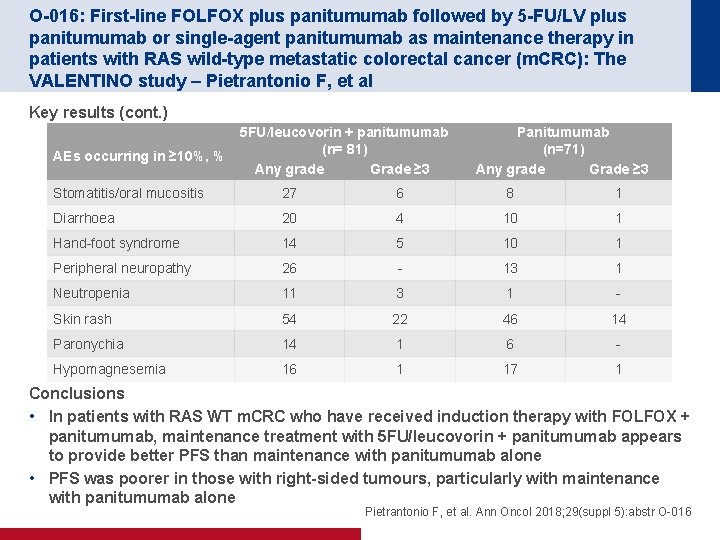
O-016: First-line FOLFOX plus panitumumab followed by 5 -FU/LV plus panitumumab or single-agent panitumumab as maintenance therapy in patients with RAS wild-type metastatic colorectal cancer (m. CRC): The VALENTINO study – Pietrantonio F, et al Key results (cont. ) AEs occurring in ≥ 10%, % 5 FU/leucovorin + panitumumab (n= 81) Any grade Grade ≥ 3 Panitumumab (n=71) Any grade Grade ≥ 3 Stomatitis/oral mucositis 27 6 8 1 Diarrhoea 20 4 10 1 Hand-foot syndrome 14 5 10 1 Peripheral neuropathy 26 - 13 1 Neutropenia 11 3 1 - Skin rash 54 22 46 14 Paronychia 14 1 6 - Hypomagnesemia 16 1 17 1 Conclusions • In patients with RAS WT m. CRC who have received induction therapy with FOLFOX + panitumumab, maintenance treatment with 5 FU/leucovorin + panitumumab appears to provide better PFS than maintenance with panitumumab alone • PFS was poorer in those with right-sided tumours, particularly with maintenance with panitumumab alone Pietrantonio F, et al. Ann Oncol 2018; 29(suppl 5): abstr O-016
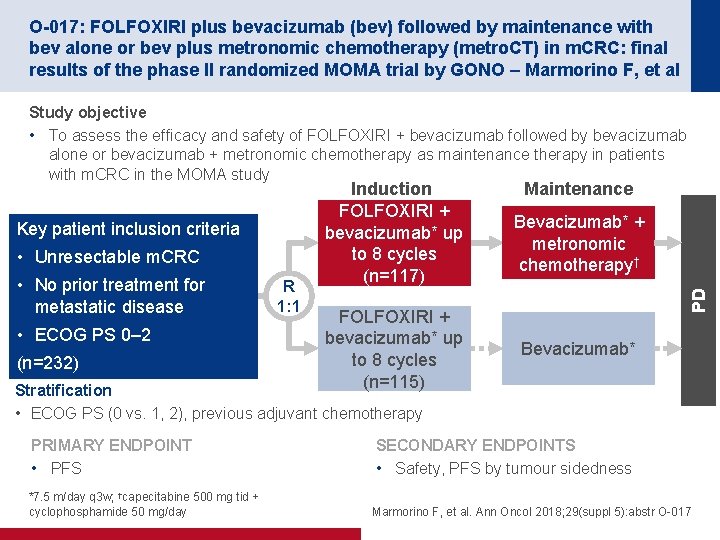
O-017: FOLFOXIRI plus bevacizumab (bev) followed by maintenance with bev alone or bev plus metronomic chemotherapy (metro. CT) in m. CRC: final results of the phase II randomized MOMA trial by GONO – Marmorino F, et al Study objective • To assess the efficacy and safety of FOLFOXIRI + bevacizumab followed by bevacizumab alone or bevacizumab + metronomic chemotherapy as maintenance therapy in patients with m. CRC in the MOMA study • Unresectable m. CRC • No prior treatment for metastatic disease • ECOG PS 0– 2 (n=232) R 1: 1 FOLFOXIRI + bevacizumab* up to 8 cycles (n=115) Maintenance Bevacizumab* + metronomic chemotherapy† PD Key patient inclusion criteria Induction FOLFOXIRI + bevacizumab* up to 8 cycles (n=117) Bevacizumab* Stratification • ECOG PS (0 vs. 1, 2), previous adjuvant chemotherapy PRIMARY ENDPOINT • PFS *7. 5 m/day q 3 w; †capecitabine 500 mg tid + cyclophosphamide 50 mg/day SECONDARY ENDPOINTS • Safety, PFS by tumour sidedness Marmorino F, et al. Ann Oncol 2018; 29(suppl 5): abstr O-017
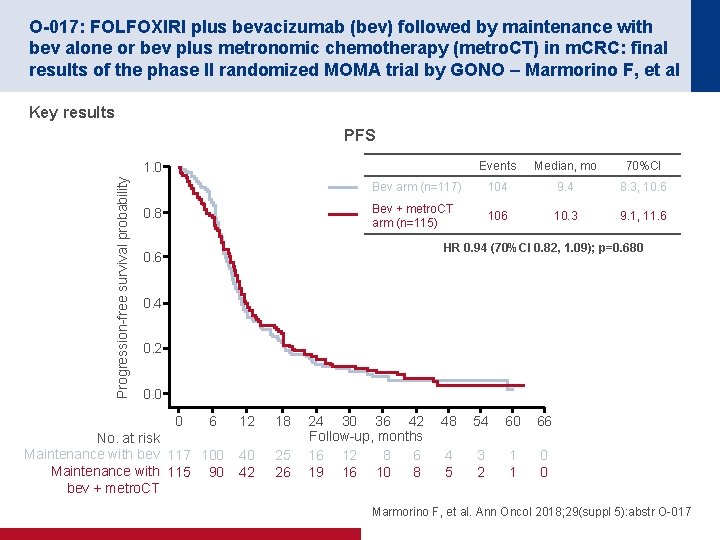
O-017: FOLFOXIRI plus bevacizumab (bev) followed by maintenance with bev alone or bev plus metronomic chemotherapy (metro. CT) in m. CRC: final results of the phase II randomized MOMA trial by GONO – Marmorino F, et al Key results PFS Events Median, mo 70%CI Bev arm (n=117) 104 9. 4 8. 3, 10. 6 Bev + metro. CT arm (n=115) 106 10. 3 9. 1, 11. 6 Progression-free survival probability 1. 0 0. 8 HR 0. 94 (70%CI 0. 82, 1. 09); p=0. 680 0. 6 0. 4 0. 2 0. 0 0 6 No. at risk Maintenance with bev 117 100 Maintenance with 115 90 bev + metro. CT 12 18 40 42 25 26 24 30 36 42 Follow-up, months 16 12 8 6 19 16 10 8 48 54 60 66 4 5 3 2 1 1 0 0 Marmorino F, et al. Ann Oncol 2018; 29(suppl 5): abstr O-017
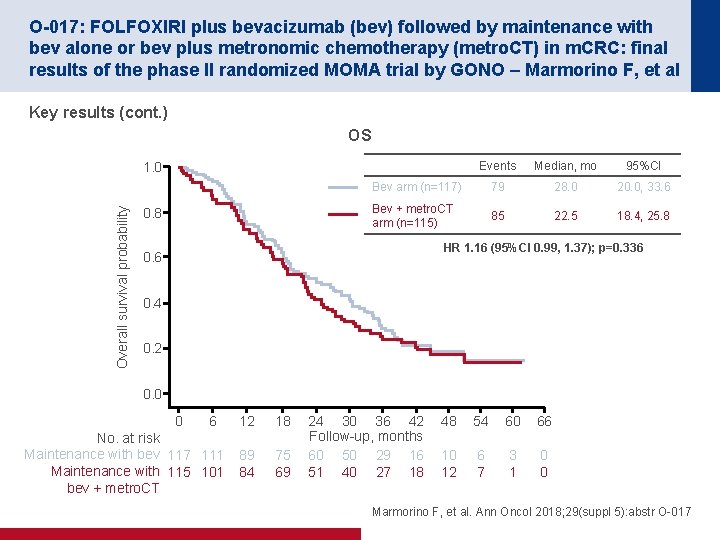
O-017: FOLFOXIRI plus bevacizumab (bev) followed by maintenance with bev alone or bev plus metronomic chemotherapy (metro. CT) in m. CRC: final results of the phase II randomized MOMA trial by GONO – Marmorino F, et al Key results (cont. ) OS Events Median, mo 95%CI Bev arm (n=117) 79 28. 0 20. 0, 33. 6 Bev + metro. CT arm (n=115) 85 22. 5 18. 4, 25. 8 Overall survival probability 1. 0 0. 8 HR 1. 16 (95%CI 0. 99, 1. 37); p=0. 336 0. 4 0. 2 0. 0 0 6 No. at risk Maintenance with bev 117 111 Maintenance with 115 101 bev + metro. CT 12 18 89 84 75 69 24 30 36 42 Follow-up, months 60 50 29 16 51 40 27 18 48 54 60 66 10 12 6 7 3 1 0 0 Marmorino F, et al. Ann Oncol 2018; 29(suppl 5): abstr O-017
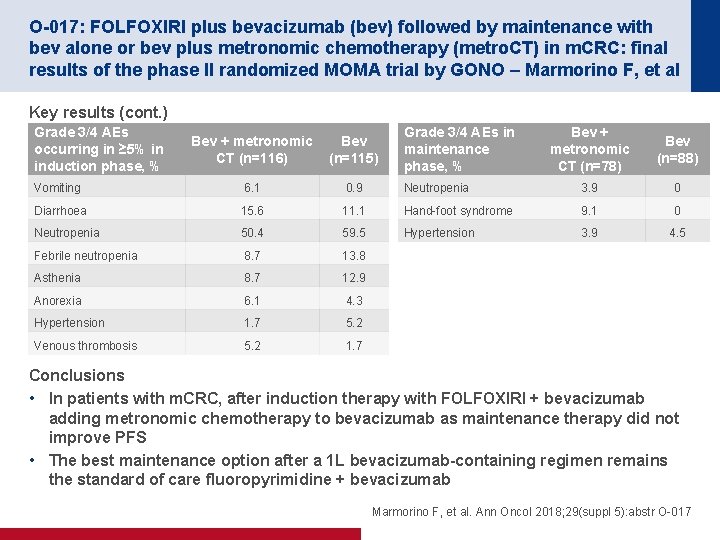
O-017: FOLFOXIRI plus bevacizumab (bev) followed by maintenance with bev alone or bev plus metronomic chemotherapy (metro. CT) in m. CRC: final results of the phase II randomized MOMA trial by GONO – Marmorino F, et al Key results (cont. ) Grade 3/4 AEs occurring in ≥ 5% in induction phase, % Bev + metronomic CT (n=78) Bev (n=88) Neutropenia 3. 9 0 11. 1 Hand-foot syndrome 9. 1 0 50. 4 59. 5 Hypertension 3. 9 4. 5 Febrile neutropenia 8. 7 13. 8 Asthenia 8. 7 12. 9 Anorexia 6. 1 4. 3 Hypertension 1. 7 5. 2 Venous thrombosis 5. 2 1. 7 Bev + metronomic CT (n=116) Bev (n=115) Vomiting 6. 1 0. 9 Diarrhoea 15. 6 Neutropenia Grade 3/4 AEs in maintenance phase, % Conclusions • In patients with m. CRC, after induction therapy with FOLFOXIRI + bevacizumab adding metronomic chemotherapy to bevacizumab as maintenance therapy did not improve PFS • The best maintenance option after a 1 L bevacizumab-containing regimen remains the standard of care fluoropyrimidine + bevacizumab Marmorino F, et al. Ann Oncol 2018; 29(suppl 5): abstr O-017
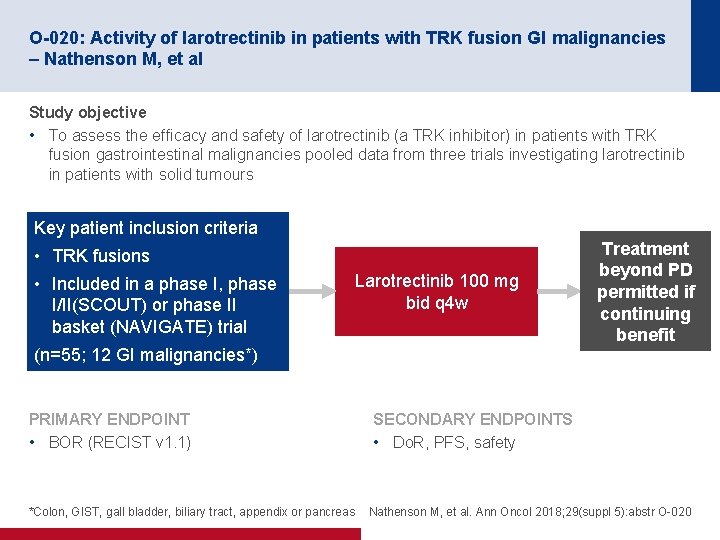
O-020: Activity of larotrectinib in patients with TRK fusion GI malignancies – Nathenson M, et al Study objective • To assess the efficacy and safety of larotrectinib (a TRK inhibitor) in patients with TRK fusion gastrointestinal malignancies pooled data from three trials investigating larotrectinib in patients with solid tumours Key patient inclusion criteria • TRK fusions • Included in a phase I, phase I/II(SCOUT) or phase II basket (NAVIGATE) trial Larotrectinib 100 mg bid q 4 w (n=55; 12 GI malignancies*) PRIMARY ENDPOINT • BOR (RECIST v 1. 1) *Colon, GIST, gall bladder, biliary tract, appendix or pancreas Treatment beyond PD permitted if continuing benefit SECONDARY ENDPOINTS • Do. R, PFS, safety Nathenson M, et al. Ann Oncol 2018; 29(suppl 5): abstr O-020
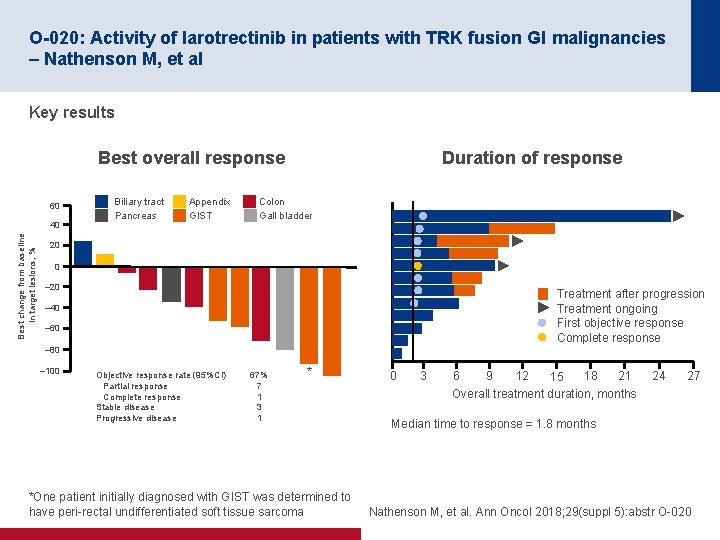
O-020: Activity of larotrectinib in patients with TRK fusion GI malignancies – Nathenson M, et al Key results Best overall response 60 Best change from baseline in target lesions, % 40 Biliary tract Pancreas Appendix GIST Duration of response Colon Gall bladder 20 0 – 20 Treatment after progression Treatment ongoing First objective response Complete response – 40 – 60 – 80 – 100 Objective response rate (95%CI) Partial response Complete response Stable disease Progressive disease 67% 7 1 3 1 * *One patient initially diagnosed with GIST was determined to have peri-rectal undifferentiated soft tissue sarcoma 0 3 6 9 12 18 21 15 Overall treatment duration, months 24 27 Median time to response = 1. 8 months Nathenson M, et al. Ann Oncol 2018; 29(suppl 5): abstr O-020

O-020: Activity of larotrectinib in patients with TRK fusion GI malignancies – Nathenson M, et al Key results (cont. ) Grade 3 TRAEs, % Increased ALT/AST 5 Dizziness 2 Nausea 2 Anaemia 2 Decreased neutrophil count 2 Conclusion • In patients with TRK fusion gastrointestinal malignancies, larotrectinib provided durable and clinically meaningful responses and was associated with minimal toxicity with prolonged treatment Nathenson M, et al. Ann Oncol 2018; 29(suppl 5): abstr O-020

O-021: Safety and antitumor activity of pembrolizumab in patients with advanced microsatellite instability–high (MSI-H) colorectal cancer: KEYNOTE -164 – Le D, et al Study objective • To assess the efficacy and safety of pembrolizumab in patients with advanced MSI-H CRC in the KEYNOTE-164 study Key patient inclusion criteria • Locally advanced, unresectable or metastatic CRC • d. MMR/MSI-H CRC by IHC/PCR • ≥ 1 prior line of therapy Pembrolizumab 200 mg q 3 w • ECOG PS 0– 1 (n=63) PRIMARY ENDPOINT • ORR Treatment for ~2 years (35 cycles) or until PD/toxicity/ withdrawal SECONDARY ENDPOINTS • Do. R, PFS, OS, safety Le D, et al. Ann Oncol 2018; 29(suppl 5): abstr O-021
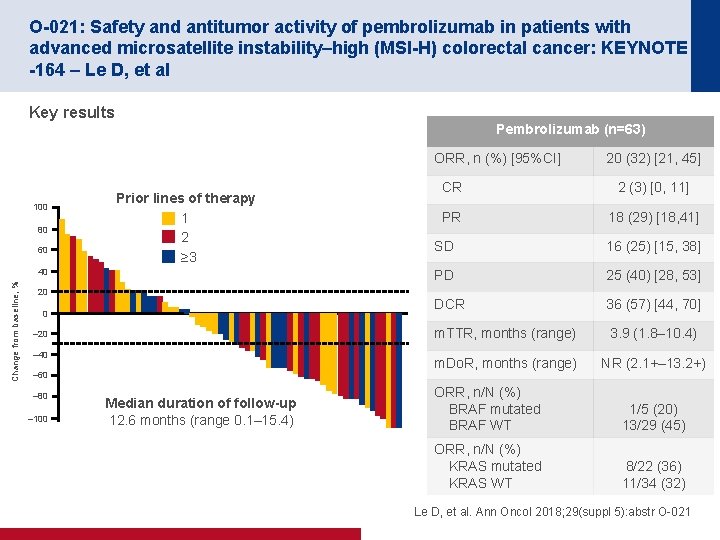
O-021: Safety and antitumor activity of pembrolizumab in patients with advanced microsatellite instability–high (MSI-H) colorectal cancer: KEYNOTE -164 – Le D, et al Key results Pembrolizumab (n=63) ORR, n (%) [95%CI] 100 80 60 Prior lines of therapy 1 2 ≥ 3 Change from baseline, % 40 20 0 – 20 – 40 – 60 – 80 – 100 Median duration of follow-up 12. 6 months (range 0. 1– 15. 4) 20 (32) [21, 45] CR 2 (3) [0, 11] PR 18 (29) [18, 41] SD 16 (25) [15, 38] PD 25 (40) [28, 53] DCR 36 (57) [44, 70] m. TTR, months (range) 3. 9 (1. 8– 10. 4) m. Do. R, months (range) NR (2. 1+– 13. 2+) ORR, n/N (%) BRAF mutated BRAF WT 1/5 (20) 13/29 (45) ORR, n/N (%) KRAS mutated KRAS WT 8/22 (36) 11/34 (32) Le D, et al. Ann Oncol 2018; 29(suppl 5): abstr O-021
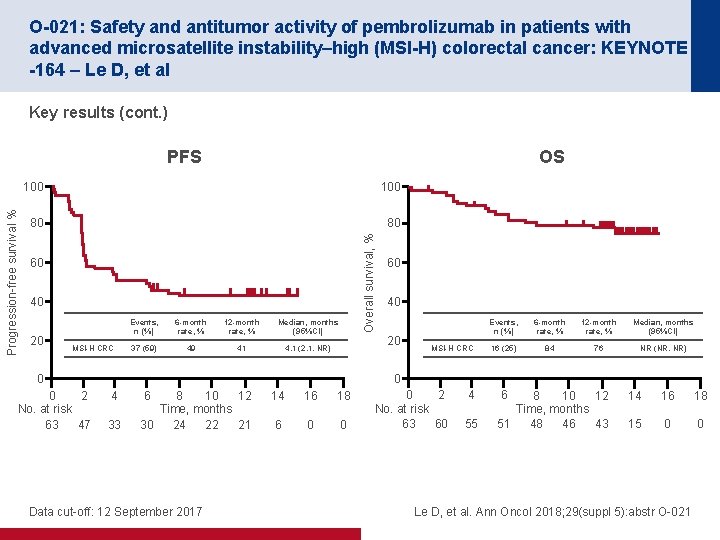
O-021: Safety and antitumor activity of pembrolizumab in patients with advanced microsatellite instability–high (MSI-H) colorectal cancer: KEYNOTE -164 – Le D, et al Key results (cont. ) OS 100 80 80 Overall survival, % Progression-free survival % PFS 60 40 20 MSI-H CRC Events, n (%) 6 -month rate, % 12 -month rate, % Median, months (95%CI) 37 (59) 49 41 4. 1 (2. 1, NR) 40 20 0 0 2 No. at risk 63 47 60 MSI-H CRC Events, n (%) 6 -month rate, % 12 -month rate, % Median, months (95%CI) 16 (25) 84 76 NR (NR, NR) 0 4 33 6 8 10 12 Time, months 30 24 22 21 Data cut-off: 12 September 2017 14 16 18 6 0 0 0 2 No. at risk 63 60 4 55 6 8 10 12 Time, months 51 48 46 43 14 16 18 15 0 0 Le D, et al. Ann Oncol 2018; 29(suppl 5): abstr O-021
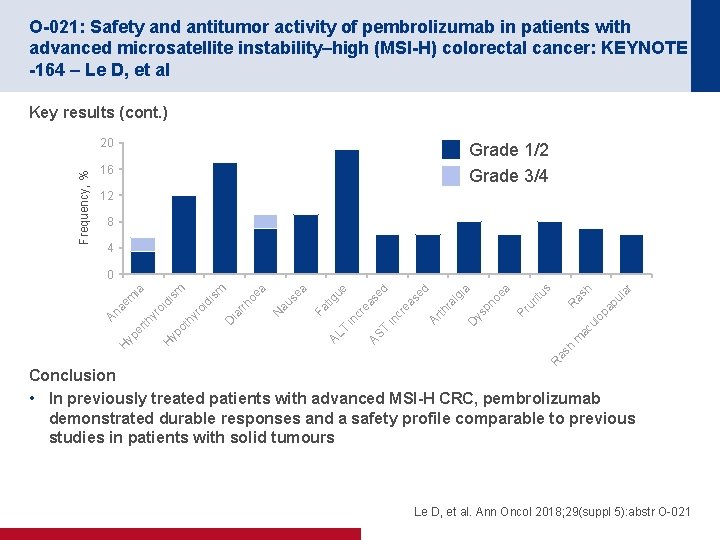
O-021: Safety and antitumor activity of pembrolizumab in patients with advanced microsatellite instability–high (MSI-H) colorectal cancer: KEYNOTE -164 – Le D, et al Key results (cont. ) Frequency, % 20 Grade 1/2 Grade 3/4 16 12 8 4 ar h ac ul op a R pu l as us rit Pr u a pn oe D ys ra lg ia d Ar th se re a T in c ea se d R as h m AS AL T in cr Fa tig ue ea au s N oe a D ia rrh sm oi di ot h yr oi di sm yp er th yr H H yp An a em ia 0 Conclusion • In previously treated patients with advanced MSI-H CRC, pembrolizumab demonstrated durable responses and a safety profile comparable to previous studies in patients with solid tumours Le D, et al. Ann Oncol 2018; 29(suppl 5): abstr O-021

O-022: Phase II study evaluating trifluridine/tipiracil+bevacizumab and capecitabine+bevacizumab in first-line unresectable metastatic colorectal cancer (m. CRC) patients who are non-eligible for intensive therapy (TASCO 1): results of the primary analysis – Van Cutsem E, et al Study objective • To assess the efficacy and safety of trifluridine/tipiracil + bevacizumab and capecitabine + bevacizumab as a 1 L therapy for patients with unresectable m. CRC who are not eligible for intensive therapy in the TASCO 1 study Trifluridine/tipiracil 35 mg bid D 1– 5, 8– 12 + bevacizumab 5 mg/kg D 1, 15 q 4 w (n=77) Key patient inclusion criteria • m. CRC • No prior treatment for metastatic disease • Not eligible for intensive therapy according to investigator’s judgement • ECOG PS 0– 2 (n=153) PRIMARY ENDPOINT • PFS R PD/ toxicity/ patient decision Stratification • RAS status, ECOG PS, country Capecitabine 1250 or 1000 mg/m 2 bid D 1– 14 + bevacizumab 7. 5 mg/kg iv D 1 q 3 w (n=76) PD/ toxicity/ patient decision SECONDARY ENDPOINTS • OS, ORR, DCR, safety, Qo. L Van Cutsem E, et al. Ann Oncol 2018; 29(suppl 5): abstr O-022
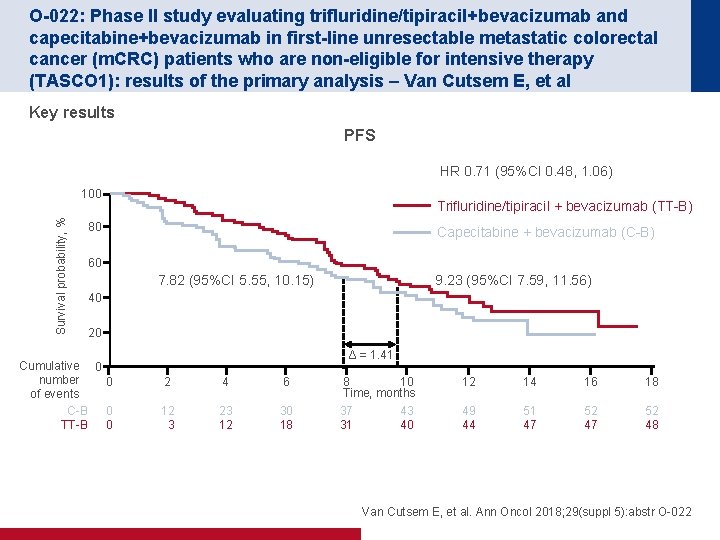
O-022: Phase II study evaluating trifluridine/tipiracil+bevacizumab and capecitabine+bevacizumab in first-line unresectable metastatic colorectal cancer (m. CRC) patients who are non-eligible for intensive therapy (TASCO 1): results of the primary analysis – Van Cutsem E, et al Key results PFS HR 0. 71 (95%CI 0. 48, 1. 06) Survival probability, % 100 Trifluridine/tipiracil + bevacizumab (TT-B) 80 Capecitabine + bevacizumab (C-B) 60 7. 82 (95%CI 5. 55, 10. 15) 9. 23 (95%CI 7. 59, 11. 56) 40 20 Cumulative 0 number 0 of events 0 C-B 0 TT-B Δ = 1. 41 2 4 6 12 3 23 12 30 18 8 10 Time, months 37 43 31 40 12 14 16 18 49 44 51 47 52 48 Van Cutsem E, et al. Ann Oncol 2018; 29(suppl 5): abstr O-022
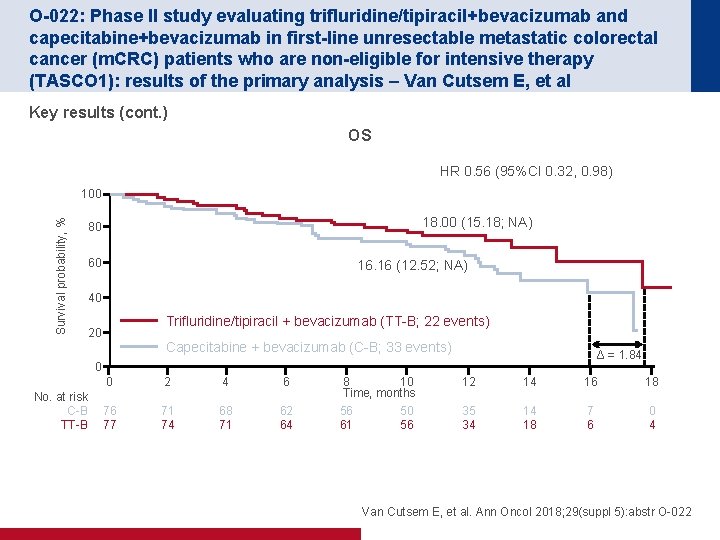
O-022: Phase II study evaluating trifluridine/tipiracil+bevacizumab and capecitabine+bevacizumab in first-line unresectable metastatic colorectal cancer (m. CRC) patients who are non-eligible for intensive therapy (TASCO 1): results of the primary analysis – Van Cutsem E, et al Key results (cont. ) OS HR 0. 56 (95%CI 0. 32, 0. 98) Survival probability, % 100 18. 00 (15. 18; NA) 80 60 16. 16 (12. 52; NA) 40 Trifluridine/tipiracil + bevacizumab (TT-B; 22 events) 20 Capecitabine + bevacizumab (C-B; 33 events) Δ = 1. 84 0 No. at risk C-B TT-B 0 2 4 6 76 77 71 74 68 71 62 64 8 10 Time, months 56 50 61 56 12 14 16 18 35 34 14 18 7 6 0 4 Van Cutsem E, et al. Ann Oncol 2018; 29(suppl 5): abstr O-022
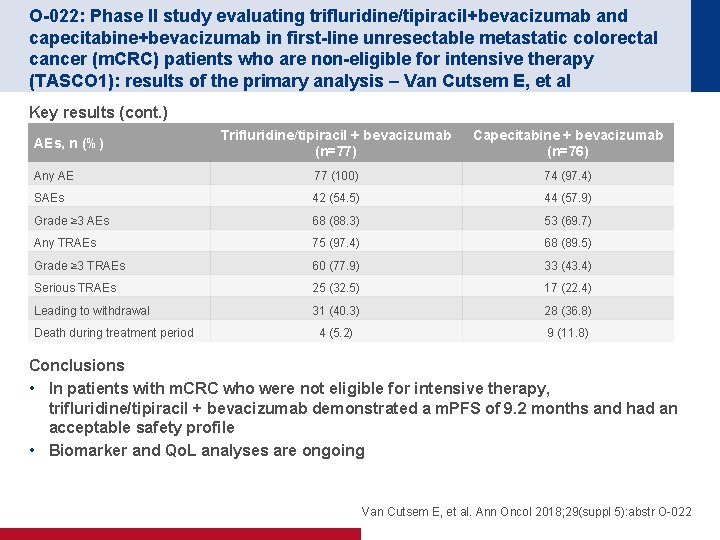
O-022: Phase II study evaluating trifluridine/tipiracil+bevacizumab and capecitabine+bevacizumab in first-line unresectable metastatic colorectal cancer (m. CRC) patients who are non-eligible for intensive therapy (TASCO 1): results of the primary analysis – Van Cutsem E, et al Key results (cont. ) Trifluridine/tipiracil + bevacizumab (n=77) Capecitabine + bevacizumab (n=76) Any AE 77 (100) 74 (97. 4) SAEs 42 (54. 5) 44 (57. 9) Grade ≥ 3 AEs 68 (88. 3) 53 (69. 7) Any TRAEs 75 (97. 4) 68 (89. 5) Grade ≥ 3 TRAEs 60 (77. 9) 33 (43. 4) Serious TRAEs 25 (32. 5) 17 (22. 4) Leading to withdrawal 31 (40. 3) 28 (36. 8) 4 (5. 2) 9 (11. 8) AEs, n (%) Death during treatment period Conclusions • In patients with m. CRC who were not eligible for intensive therapy, trifluridine/tipiracil + bevacizumab demonstrated a m. PFS of 9. 2 months and had an acceptable safety profile • Biomarker and Qo. L analyses are ongoing Van Cutsem E, et al. Ann Oncol 2018; 29(suppl 5): abstr O-022

O-024: m. FOLFOXIRI + panitumumab versus FOLFOXIRI as first-line treatment in patients with RAS wild- type metastatic colorectal cancer m(CRC): A randomized phase II VOLFI trial of the AIO (AIO-KRK 0109) – Geissler M, et al Study objective • To assess the efficacy and safety of panitumumab + m. FOLFOXIRI vs. FOLFOXIRI as 1 L treatment in patients with m. CRC Panitumumab 6 mg/kg + m. FOLFOXIRI q 2 w (n=63) Key patient inclusion criteria • Unresectable m. CRC • WT RAS • 1 L (1 cycle of FOLFIRINOX permitted prior to randomisation) Stratification R 2: 1 • Cohort 1: Histologically confirmed and definitively inoperable/unresectable • Cohort 2: Chance of secondary resection with curative intent (pre-treatment liver/tumour biopsy) • ECOG PS 0– 1 (n=96) PRIMARY ENDPOINT • ORR *If resectable: surgery then protocol treatment for up to 12 cycles; if CR/PR/SD after 12 cycles: re-induction (same combination) recommended on PD PD/ resectability/ maximum 12 cycles* FOLFOXIRI q 2 w (n=33) PD/ resectability/ maximum 12 cycles* SECONDARY ENDPOINTS • TRR, time-to-relapse, PFS, OS, safety, Qo. L Geissler M, et al. Ann Oncol 2018; 29(suppl 5): abstr O-024
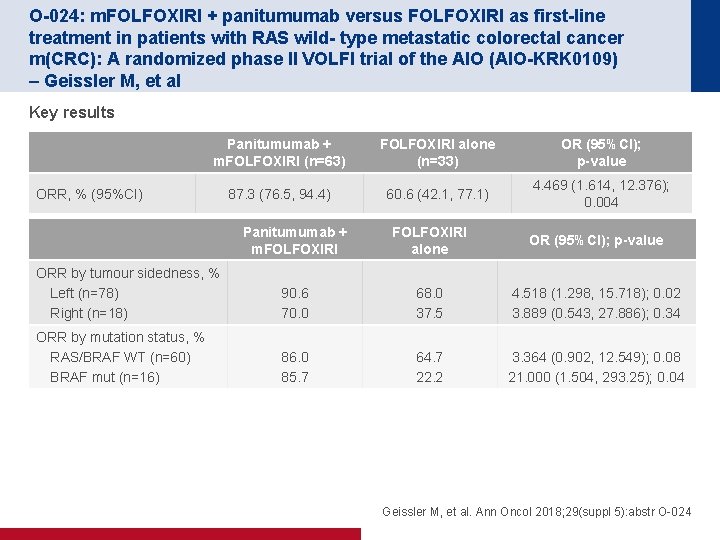
O-024: m. FOLFOXIRI + panitumumab versus FOLFOXIRI as first-line treatment in patients with RAS wild- type metastatic colorectal cancer m(CRC): A randomized phase II VOLFI trial of the AIO (AIO-KRK 0109) – Geissler M, et al Key results Panitumumab + m. FOLFOXIRI (n=63) FOLFOXIRI alone (n=33) OR (95%CI); p-value 87. 3 (76. 5, 94. 4) 60. 6 (42. 1, 77. 1) 4. 469 (1. 614, 12. 376); 0. 004 ORR, % (95%CI) Panitumumab + m. FOLFOXIRI alone OR (95%CI); p-value ORR by tumour sidedness, % Left (n=78) Right (n=18) 90. 6 70. 0 68. 0 37. 5 4. 518 (1. 298, 15. 718); 0. 02 3. 889 (0. 543, 27. 886); 0. 34 ORR by mutation status, % RAS/BRAF WT (n=60) BRAF mut (n=16) 86. 0 85. 7 64. 7 22. 2 3. 364 (0. 902, 12. 549); 0. 08 21. 000 (1. 504, 293. 25); 0. 04 Geissler M, et al. Ann Oncol 2018; 29(suppl 5): abstr O-024
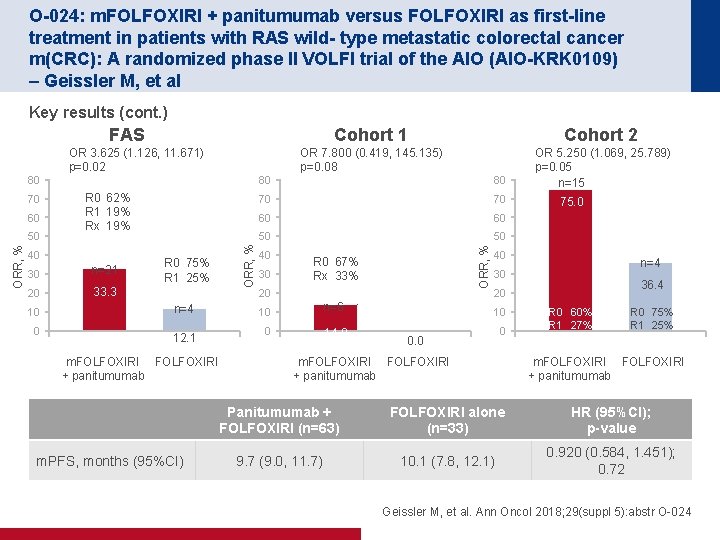
O-024: m. FOLFOXIRI + panitumumab versus FOLFOXIRI as first-line treatment in patients with RAS wild- type metastatic colorectal cancer m(CRC): A randomized phase II VOLFI trial of the AIO (AIO-KRK 0109) – Geissler M, et al Key results (cont. ) FAS 60 ORR, % 50 R 0 62% R 1 19% Rx 19% 40 30 n=21 20 33. 3 10 0 R 0 75% R 1 25% n=4 12. 1 m. FOLFOXIRI + panitumumab m. PFS, months (95%CI) Cohort 2 OR 7. 800 (0. 419, 145. 135) p=0. 08 OR 5. 250 (1. 069, 25. 789) p=0. 05 n=15 80 70 70 60 60 50 50 40 30 ORR, % 70 80 ORR, % 80 OR 3. 625 (1. 126, 11. 671) p=0. 02 Cohort 1 R 0 67% Rx 33% 20 10 n=6 0 14. 0 75. 0 40 n=4 30 36. 4 20 10 0. 0 0 m. FOLFOXIRI + panitumumab R 0 60% R 1 27% Rx 13% R 0 75% R 1 25% m. FOLFOXIRI + panitumumab Panitumumab + FOLFOXIRI (n=63) FOLFOXIRI alone (n=33) HR (95%CI); p-value 9. 7 (9. 0, 11. 7) 10. 1 (7. 8, 12. 1) 0. 920 (0. 584, 1. 451); 0. 72 Geissler M, et al. Ann Oncol 2018; 29(suppl 5): abstr O-024
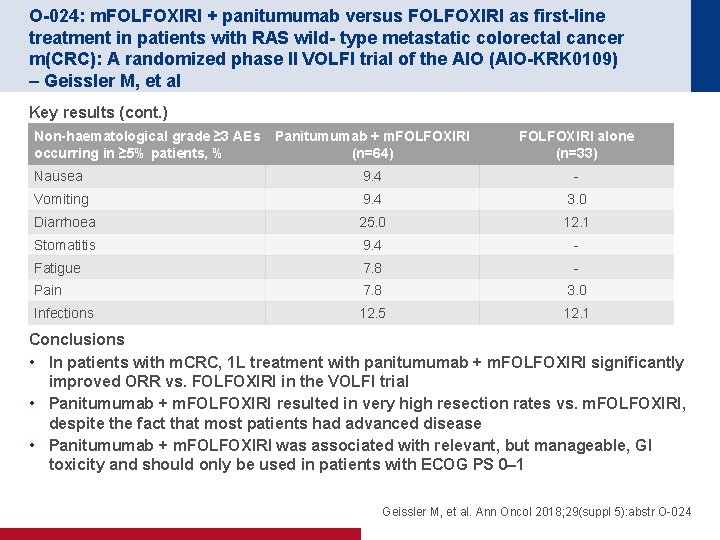
O-024: m. FOLFOXIRI + panitumumab versus FOLFOXIRI as first-line treatment in patients with RAS wild- type metastatic colorectal cancer m(CRC): A randomized phase II VOLFI trial of the AIO (AIO-KRK 0109) – Geissler M, et al Key results (cont. ) Panitumumab + m. FOLFOXIRI (n=64) FOLFOXIRI alone (n=33) Nausea 9. 4 - Vomiting 9. 4 3. 0 Diarrhoea 25. 0 12. 1 Stomatitis 9. 4 - Fatigue 7. 8 - Pain 7. 8 3. 0 Infections 12. 5 12. 1 Non-haematological grade ≥ 3 AEs occurring in ≥ 5% patients, % Conclusions • In patients with m. CRC, 1 L treatment with panitumumab + m. FOLFOXIRI significantly improved ORR vs. FOLFOXIRI in the VOLFI trial • Panitumumab + m. FOLFOXIRI resulted in very high resection rates vs. m. FOLFOXIRI, despite the fact that most patients had advanced disease • Panitumumab + m. FOLFOXIRI was associated with relevant, but manageable, GI toxicity and should only be used in patients with ECOG PS 0– 1 Geissler M, et al. Ann Oncol 2018; 29(suppl 5): abstr O-024

O-027: BEACON CRC study safety lead-in: Assessment of the BRAF inhibitor encorafenib + MEK inhibitor binimetinib + EGFR inhibitor cetuximab for BRAFV 600 E m. CRC – Van Cutsem E, et al Study objective • To assess the efficacy and safety of binimetinib + encorafenib + cetuximab in patients with BRAF V 600 E mutant m. CRC following completion of a safety lead-in* Key patient inclusion criteria • Progressed after 1 or 2 previous regimens • No prior treatment with RAF, MEK, EGFR inhibitors or irinotecan R 1: 1: 1 Encorafenib + cetuximab (n=205) Stratification FOLFIRI + cetuximab or irinotecan + cetuximab (n=205) • Eligible for cetuximab • ECOG PS 0– 1 (n=615) PRIMARY ENDPOINT • ORR *Safety lead-in (n=30): binimetinib 45 mg bid; encorafenib 300 mg/day; cetuximab 400 mg/m 2 (initial) then 250 mg/m 2 qw PD Binimetinib + encorafenib + cetuximab (n=205) • BRAF V 600 E mutant m. CRC • BRAF V 600 E mutation status, ECOG PS, no. of prior regimens (1/2) SECONDARY ENDPOINTS • OS, PFS, safety Van Cutsem E, et al. Ann Oncol 2018; 29(suppl 5): abstr O-027
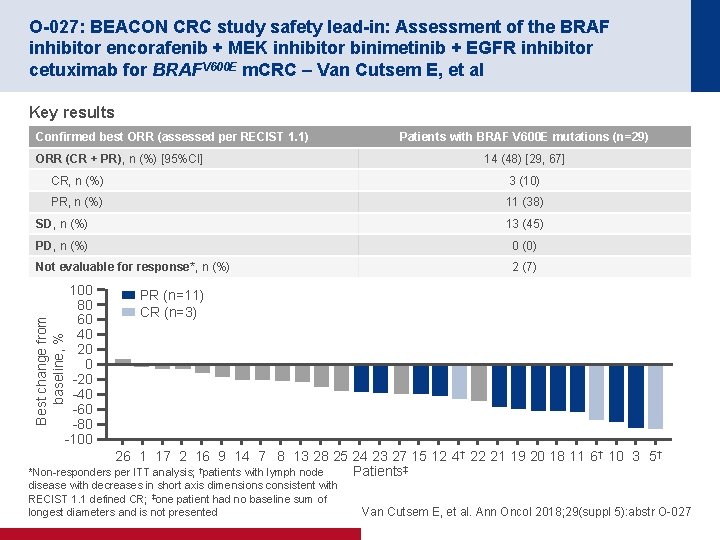
O-027: BEACON CRC study safety lead-in: Assessment of the BRAF inhibitor encorafenib + MEK inhibitor binimetinib + EGFR inhibitor cetuximab for BRAFV 600 E m. CRC – Van Cutsem E, et al Key results Confirmed best ORR (assessed per RECIST 1. 1) ORR (CR + PR), n (%) [95%CI] Patients with BRAF V 600 E mutations (n=29) 14 (48) [29, 67] CR, n (%) 3 (10) PR, n (%) 11 (38) SD, n (%) 13 (45) PD, n (%) 0 (0) Not evaluable for response*, n (%) 2 (7) Best change from baseline, % 100 80 60 40 20 0 -20 -40 -60 -80 -100 PR (n=11) CR (n=3) 26 1 17 2 16 9 14 7 8 13 28 25 24 23 27 15 12 4† 22 21 19 20 18 11 6† 10 3 5† Patients‡ *Non-responders per ITT analysis; †patients with lymph node disease with decreases in short axis dimensions consistent with RECIST 1. 1 defined CR; ‡one patient had no baseline sum of longest diameters and is not presented Van Cutsem E, et al. Ann Oncol 2018; 29(suppl 5): abstr O-027
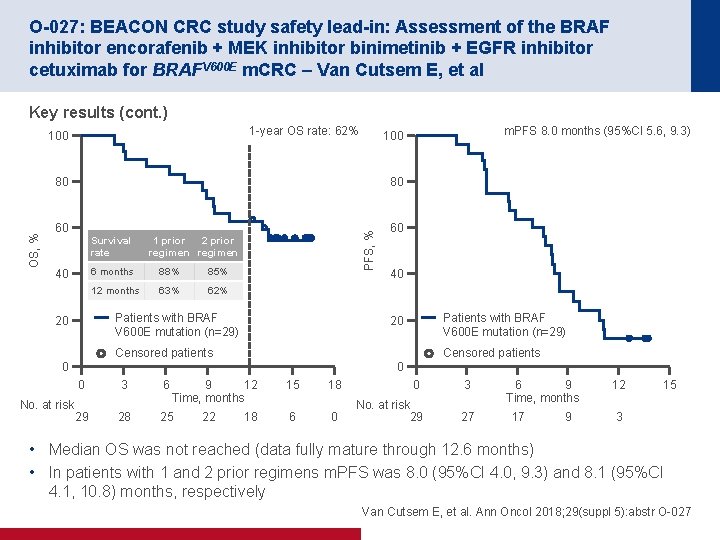
O-027: BEACON CRC study safety lead-in: Assessment of the BRAF inhibitor encorafenib + MEK inhibitor binimetinib + EGFR inhibitor cetuximab for BRAFV 600 E m. CRC – Van Cutsem E, et al Key results (cont. ) 1 -year OS rate: 62% 100 80 60 Survival rate 40 PFS, % 80 OS, % m. PFS 8. 0 months (95%CI 5. 6, 9. 3) 100 1 prior 2 prior regimen 6 months 88% 85% 12 months 63% 62% Patients with BRAF V 600 E mutation (n=29) 20 60 40 Patients with BRAF V 600 E mutation (n=29) 20 Censored patients 0 No. at risk 0 0 3 6 9 12 Time, months 29 28 25 22 18 15 18 0 3 6 0 No. at risk 29 27 6 9 Time, months 17 9 12 15 3 • Median OS was not reached (data fully mature through 12. 6 months) • In patients with 1 and 2 prior regimens m. PFS was 8. 0 (95%CI 4. 0, 9. 3) and 8. 1 (95%CI 4. 1, 10. 8) months, respectively Van Cutsem E, et al. Ann Oncol 2018; 29(suppl 5): abstr O-027
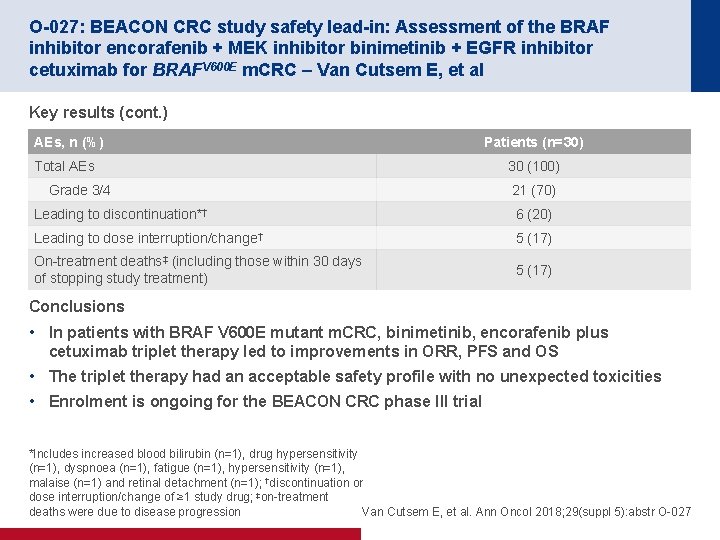
O-027: BEACON CRC study safety lead-in: Assessment of the BRAF inhibitor encorafenib + MEK inhibitor binimetinib + EGFR inhibitor cetuximab for BRAFV 600 E m. CRC – Van Cutsem E, et al Key results (cont. ) AEs, n (%) Total AEs Grade 3/4 Patients (n=30) 30 (100) 21 (70) Leading to discontinuation*† 6 (20) Leading to dose interruption/change† 5 (17) On-treatment deaths‡ (including those within 30 days of stopping study treatment) 5 (17) Conclusions • In patients with BRAF V 600 E mutant m. CRC, binimetinib, encorafenib plus cetuximab triplet therapy led to improvements in ORR, PFS and OS • The triplet therapy had an acceptable safety profile with no unexpected toxicities • Enrolment is ongoing for the BEACON CRC phase III trial *Includes increased blood bilirubin (n=1), drug hypersensitivity (n=1), dyspnoea (n=1), fatigue (n=1), hypersensitivity (n=1), malaise (n=1) and retinal detachment (n=1); †discontinuation or dose interruption/change of ≥ 1 study drug; ‡on-treatment deaths were due to disease progression Van Cutsem E, et al. Ann Oncol 2018; 29(suppl 5): abstr O-027
- Slides: 74