Get out your reading guide but do not

Get out your reading guide but do not turn it in Agenda AQOD • Go over reading guide • Notes on classifying matter • HW: Concept Review Worksheet 2. 1 • What is the difference between a compound an element?

Matter • Anything that has mass and takes up space.

Both elements and compounds have a definite makeup and definite properties. Elements Compounds substance only one kind with definite of atom; atomsmakeup are bonded it and the element properties, molecules Packard, Jacobs, Marshall, Chemistry Pearson AGS Globe, page (Figure 2. 4. 1) Mixtures two or more kinds more substances of atoms kinds of that are atom physically mixed bonded

Pure Substances Element – composed of identical atoms – EX: copper wire, aluminum foil Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Pure Substances Compound – composed of 2 or more elements in a fixed ratio – properties differ from those of individual elements – EX: table salt (Na. Cl) Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Pure Substances Law of Definite Composition – A given compound always contains the same, fixed ratio of elements. Law of Multiple Proportions – Elements can combine in different ratios to form different compounds. Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Pure Substances For example… Carbon, C Oxygen, O Carbon monoxide, CO Carbon dioxide, CO 2 Two different compounds, each has a definite composition. Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Mixtures Variable combination of two or more pure substances that can be separated into its individual parts Heterogeneous Homogeneous Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Mixtures Solution – homogeneous – very small particles – no Tyndall effect Tyndall Effect – particles don’t settle – EX: rubbing alcohol Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Mixtures Suspension – heterogeneous – large particles – Tyndall effect – particles settle – EX: fresh-squeezed lemonade Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Mixtures Colloid – heterogeneous – medium-sized particles – Tyndall effect – particles don’t settle – EX: milk Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Matter Flowchart Examples: – graphite element – pepper hetero. mixture – sugar (sucrose) compound – paint hetero. mixture – soda solution homo. mixture Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Mixtures Examples: – mayonnaise colloid – muddy water suspension – fog colloid – saltwater solution – Italian salad dressing suspension Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
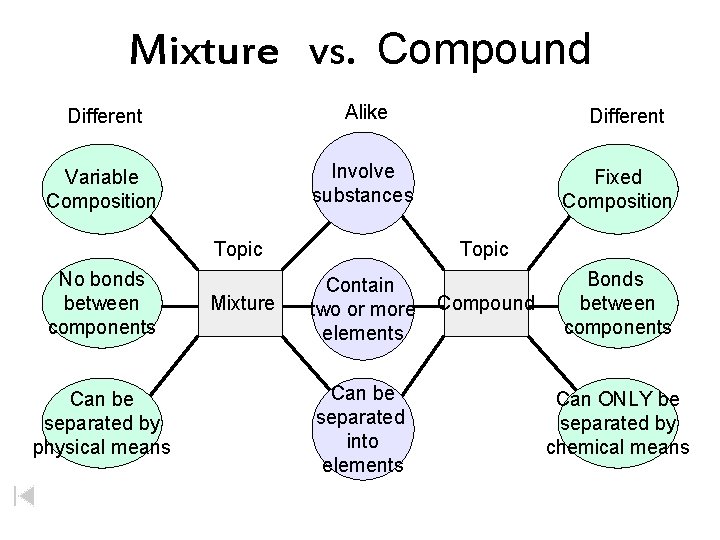
Mixture vs. Compound Different Alike Variable Composition Involve substances Topic No bonds between components Can be separated by physical means Mixture Different Fixed Composition Topic Contain two or more elements Can be separated into elements Compound Bonds between components Can ONLY be separated by chemical means
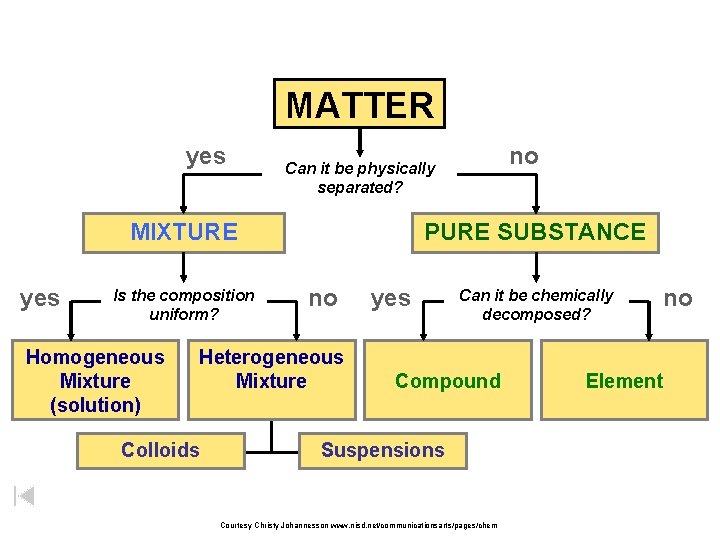
MATTER yes MIXTURE yes Is the composition uniform? Homogeneous Mixture (solution) PURE SUBSTANCE no Heterogeneous Mixture Colloids no Can it be physically separated? yes Can it be chemically decomposed? Compound Suspensions Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem no Element
- Slides: 15