Gestational Trophoblastic Disease YASHODHARA PRADEEP Prof OBGYN QMH

Gestational Trophoblastic Disease YASHODHARA PRADEEP Prof. , OBGYN QMH CSMMU LKO 2014

• Classification of GTT • GTD Epidemiology, Pathogenesis, Aetiology, β h. CG, • Clinical Features of Premalignant(GTD) Complete Mole(CM); Partial Mole • Management of Complete Mole, or Partial Mole • Post evacuation Follow up , Chemotherapy • Contraception • Clinical Features of Malignant (GTN) Invasive Mole, Placental site tropho blastic tumor( PSTT) Chorio CA • Investigations • GTN Chorio. CA Diagnosis, Investigation, Management • Chemotherapy • Follow up

Introduction • Gestational Trophoblastic disease is a term used to describe the heterogeneous group of interrelated lesions that arise from abnormal proliferation of placental trophoblast. GTD Lesions are histologically different and can be Benign and malignant.

WHO Classification GTD Premalignant Diseases • Complete Hydatidiform Mole ( C M ) • Partial Hyadatidiform Mole ( P M ) Malignant Diseases (Gestational Trophoblastic Neoplasia) Nonmetastatic • Invasive Mole • Placental site trophoblastic tumor ( PSTT ) • Epitheloid tumour • Metastatic • Gestational Choriocarcinoma

Epidemiology • It is common in oriental countries- Philippines, China, Indonesia, Japan, India, Central and Latin America and Africa. • Highest incidence- Philippines- 1 in 80 pregnancies • Lowest incidence- Europe- 1 in 752 pregnancies • India- 1 in 400 pregnancies • Calculated Incidence of complete mole- 1 in 1945 pregnancies partial mole- 1 in 695 pregnancies • Age C Ms most common at the extremes of reproductive age • The incidence of CM rises fm I: 1000 to 1: 76; 1: 6. 5 in subsequent pregnancies • CM is the most common antecedent to Chorio CA. But it can occur after any type of pregnancy 3% after invasive mole; 16% after CM
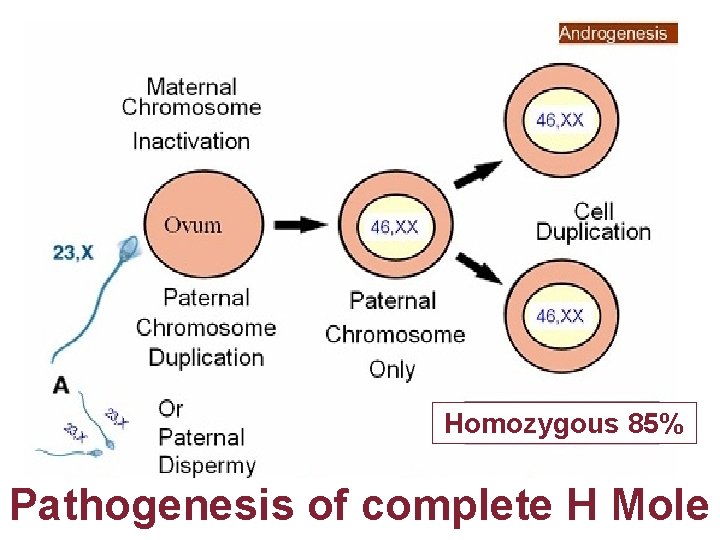
Homozygous 85% Pathogenesis of complete H Mole
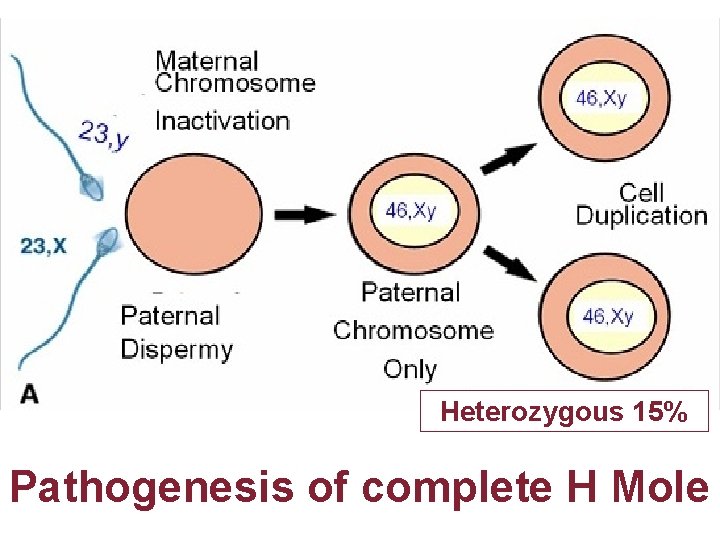
Heterozygous 15% Pathogenesis of complete H Mole
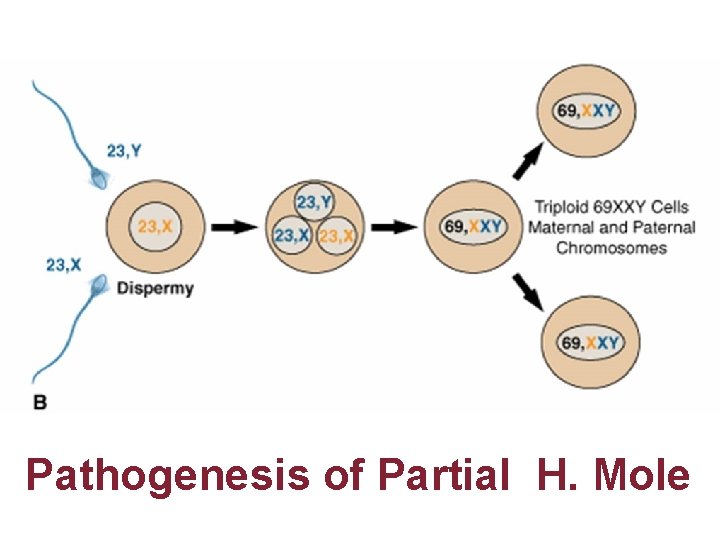
Pathogenesis of Partial H. Mole

Gestational Trophoblastic Disease Genetic & Pathology • • C M Always contains Paternal DNA alone so androgenic Macroscopically like bunch of grapes P M are Triploidy two paternal & one maternal sets of chromosomes hyperplasia of chorionic villi with embryo usually dies 8 -9 weeks of gestation P 57 cycline dependent kinase inhibitor paternally imprinted gene which is maternally expressed P 57 kip 2 immunostaining is negative in CM in contrast to PMs Hydropic abortions & normal placenta Invasive mole can be diagnosed by USG & rising titers of h. CG Presence of chorionic villi distinguish Invasive mole from Choriocarcinoma Chorio CA. Grossly abnormal karyotypes with diverse ploidies PSTT are rare slow growing malignant tumor; intermediate trophoblast from cytotrophoblast produce little h. CG

Aetiological factors • Genetic factor: Autosomal recessive Genomic Imprinting; Abnormal Methylation • In cases of Familial recurrent molar pregnancy; Defective locus at 19 q 13. 4 at single gene NALP 7 , it is member of CATERPILLAR family involved in inflammation & Apoptosis in genetic CM • Three other genes H 19, P 57, IGF-2 • Chorio CA. heterozygosity at specific genes deletion 7 p 12 q 11. 2; Amplification of 7 q 21 -q 31 region ; loss of 8 p 12 -p 21 • Breast & ovarian cancer also show the abnormalities in heterozygosity in amplification at 7 q 21 -q 31 , & loss of 8 p 12 p 21

Human chorionic gonadotrophin • Beta h. CG is pituitary/ placental glycoprotein • Commercially available Beta h. CG test based on sandwich assay principle & rely on two antibodies • Rare chance of false negative or false positive results with Rabit polyclonal antibodies RIA used in both serially diluted urine & blood samples. • Sensitivity of this test in serum is 1 IU/L, in urine 20 IU/L • Beta h CG is tumor marker • New Assay are in research Hyperglycosylated h. CG (h. CG-H ) specific for cases subsequently require chemotherapy (Clinical GTDs ) Cole etal 2006 a

Beta h CG as Tumor marker • Half life is 24 -36 hrs • Marker for pregnancy , GTD, Germ cell tumors & 15% of epithelial ovarian malignancies • H CG levels are also associated with tumor volume 5 IU/l corresponds to 104 -105 viable tumor size • Beta h. CG used as prognostic marker & progression of disease or response to therapy, drug resistance at earlier stage. • h. CG is best tumor marker known

Clinical features Complete Mole Most common in first trimester or early Second trimester Vaginal bleeding is the most common symptom (84 -97%) Uterus is larger than POG (28%) Presents with the features of threatened abortion; Amenorrhea, pain & bleeding P/V • Associated features of Hyperemesis Gravidarum Requires Antiemetic , IV fluids etc • Toxaemia or PET 10 -15% • hyperthyroidism 7% • •

Clinical features Theca leutin cysts Beta h CG levels > 100. 000 O/E Passage of grapes like vesicle or spontaneous expulsion of entire mole Pulmonary , cervical, Vaginal metastasis may occur disappears after evacuation of mole • The presence of metastasis does not necessarily imply invasive mole or chorio CA • Macroscopic features: Bunch of grapes due to villous swelling • Branching contain hyperplastic cyto & syncytio trophoblast with many vessels • • •

Complete Molar Pregnancy

High Risk For Developing Post molar tumor • Patients presenting with any of the following signs of marked trophoblastic proliferations • h. CG Levels > 100, 000 m. IU/L • Excessive Uterine Enlargement • Theca leutin cyst 6 cm or larger

Clinical features PARTIALMOLE • Findings not as characteristic as in CM • Most commonly presents in late Ist or second trimester • Uterus is often not enlarged more than POA • More often presents as Missed or incomplete abortion • Pre evacuation h CG levels are not more than 100, 000 IU/ L • Macoscopic : villous swelling is less intense • Embryo is present • Microscopic: Trophoblastic hyperplasia, indented outlines & round inclusions • Presence of nucleated RBS of embryo in villous structure • Due to Early demise of embryo less documentation of PM

Management Of Molar Pregnancy There are 2 important basic lines: 1. Evacuation of the mole 2. Regular follow up to detect persistent Trophoblastic disease If both basic lines are done appropriately, mortality rates can be reduced to zero.

Management of Molar Pregnancy For complete mole: Suction Curettage Procedure: 1. Emergency tray should be ready and blood should be handy. 2. Oxytocin infusion: begun before induction of anaesthesia. 3. Cervical dilation

Management 4. Suction curettage: Suction canula up to a maximum of 12 mm is usually sufficient to evacuate all complete molar pregnancies. If the uterine size is larger than 14 weeks of gestation, one hand should be placed on top of fundus and uterus should be massaged to stimulate uterine contraction and reduce the risk of perforation. 5. Sharp curettage: gentle sharp curettage is done to remove any residual molar tissue. Cervical preparation with prostaglandins or misoprostol should be avoided to reduce the risk of embolisation.

Should products of conception be examined histologically? Histological examination is indicated in: • Failed pregnancies (missed or molar): all medically or surgically managed cases • Products of conception obtained after all repeat evacuations (post abortive or post partum). There is no need after therapeutic termination: provided that foetal parts are identified on USG A urine pregnancy test should be performed 3 weeks after medical management of failed pregnancy if products of conception are not sent for histological examination.

Anti-D Prophylaxis (RCOG) NOT required in Complete Mole: because of poor vascularisation of the chorionic villi and absence of the D-antigen. Required in: -partial mole: due to presence of RBCs -complete mole: if diagnosis is not confirmed histopathologically.

Second uterine evacuation • There is NO clinical indication for the routine use of second uterine evacuation. • It may be useful for symptom control in selected patients with heavy bleeding or curative if the recurrent molar tissue is confined to the uterine cavity, particularly in those with h. CG levels <1500 IU/L. (Recent Advances)

Post Evacuation Surveillance • Done to detect persistent trophoblastic disease (GTN) • A baseline serum βh. CG is obtained within 48 hours after evacuation • Levels are monitored every 2 weeks until normalisation and urine βh. CG levels analysed monthly after this. • These levels should progressively fall to an undetectable level (<5 mu/ml). • The average time to achieve the first normal β-h. CG level after evacuation is about 9 weeks. • If symptoms are persistent, more frequent βh. CG estimation and USG examination ± second evacuation is advised.

Optimum follow up period following normalization of β h. CG If βh. CG has reverted to normal in • ≤ 56 days (8 weeks) of the pregnancy event: follow up is for 6 months from the date of uterine evacuation. • >56 days of the pregnancy event: follow up is for 6 months from normalisation of h. CG level. All women should notify at the end of any future pregnancy, whatever the outcome of the pregnancy. β h. CG levels are measured 6 -8 weeks after the end of pregnancy to exclude disease occurrence.

Contraception • Should NOT conceive until follow up is complete. • Use Barrier method until h. CG revert to normal (? OCPs may act as growth factor for trophoblastic tissue). • Once h. CG has normalised, the combined oral contraceptive pill may be used. • IUCD should not be used until h. CG levels are normal to reduce the risk of uterine perforation.

Role of hysterectomy • If the patient desires surgical sterilization, a hysterectomy may be performed with the mole in situ. • Hysterectomy does not prevent metastasis; therefore, patients will require follow up with assessment of h. CG levels. • The ovaries may be preserved at the time of surgery, even in the presence of prominent theca luiten cysts.

Role of prophylactic chemotherapy • The long term prognosis for women with a H. Mole is not improved with prophylactic chemotherapy. Because toxicity- including death- may be significant, it is not recommended routinely (ACOG 2004). • It may be useful in the high-risk cases when follow up are unavailable or unreliable. HIGH RISK FACTORS: § h. CG level >100, 000 m. IU/ml § Excessive uterine enlargement § Theca lutien cysts 6 cm in diameter

Chances of malignant transformation • Complete H. Mole 16% • Partial H. Mole 0. 5%

Gestational trophoplastic Neoplasia Nonmetastatic Invasive Mole: Clinical features • Clinical diagnosis by persistence or rising titers of Beta h CG in the weeks after molar evacuation & USG • Persistent bleeding p/v • Lower abdominal pain due to invasion in myometrium, vulva, vagina or intra abdominal metastasis • It may spread to adjacent pervic structures bladder , rectum; hematuria, bleeding P/ R • Pulmonary metastasis • Mostly due to initial diagnosis of CM is missed & not on Beta h CG follow up

Clinical features Placental Site Trophoblastic Tumor (PSTT): • it is rare slow growing tumor • Women mainly presents with menstrual irregularities & lower abdominal pain, galactorrhea due to hyperprolactinemia increased h PL • Little or no h CG is produced ( Free B h. CG fragment ) • Rarely presents as nephrotic syndrome, hematuria or DIC • Spread is late local Infiltration & metastasis is through lymphatic • Microscopically: In the normal placenta it is distinct from villous trophoblast & infiltrates the decidua, myometrium, & spiral arteries. • Mainly from intermediate trophoblast derived fm cytotrophoblast

Metastatic Gestational Trophoblastic Neoplasia Ø Ø Ø Second most common GTN 1 in 30, 000 pregnancies Early haematogenous spread is characterstic of choriocarcinoma 40% after molar pregnancy: Easily Diagnosed 60% non-molar pregnancy: Difficult to Diagnose

Metastatic Gestational Trophoblastic Neoplasia Ø GTN arises when the normal regulatory mechanisms controlling the proliferation and invasiveness of trophoblastic tissue are lost. Ø Diagnosis of the GTN is made on Ø Clinical presentation Ø Elevated -HCG Ø Evidence of metastasis Ø Imaging Ø Tissue for Histology- Seldom obtained (Definitive Histological Diagnosis of type of GTN is not made in most cases).

Metastatic Gestational Trophoblastic Neoplasia CHORIOCARCINOMA: Clinical features • Occurs mainly following any form of pregnancy, mainly after CM • Difficult to differentiate from Invasive mole as H/P is not possible • Clinical features of bleeding p/v , lower abdominal pain, or in 1/3 of cases no pelvic symptoms but symptoms of distant metastasis lungs , brain , liver, skin, cauda equina & the heart may present • Highly malignant , appears as soft purple largely hemorrhagic mass

Clinical features of chorio CA • Microscopic: implanting blastocyst with central cores of mononuclear cytotropho surrounded by rim of multinucleated syncytiotrophoblast & distinct absence of chorionic villi extensive areas of necrosis & haemorrage &frequent evidence of tumor in the sinuses • The hypervascularity & absence of connective tissue support are the reason for its highly malignant behaviour • DIAGNOSIS IS ALWAYS BY BETA h CG

investigations • • • Quantitative beta h. CG X Ray Chest Pelvic Doppler USG Abdominal doppler USG to rule out liver & renal metastasis CT chest , abdomen MRI brain Beta h CG in cerebrospinal fluid PET FD PET Genetic studies

Which women should be investigated for Persistent GTD after a non molar pregnancy? (RCOG Guideline) • Any women who develops persistent vaginal bleeding after a pregnancy event. • A urine pregnancy test should be performed in all cases of persistent or irregular vaginal bleeding after a pregnancy event. • Symptoms from metastatic disease, such as dyspnoea or abnormal neurology, can occur very rarely.

FIGO REQUIREMENT FOR MAKING DIAGNOSIS OF GTN 1. Four values or more of plateaue h. CG over at least 3 weeks: days 1, 7, 14 and 21. 2. A rise of h. CG of 10% or greater for 3 values or more over at least 2 weeks: days 1, 7, and 14. 3. Histologic diagnosis of choriocarcinoma. 4. Persistence of h. CG beyond 6 months after mole evacuation.
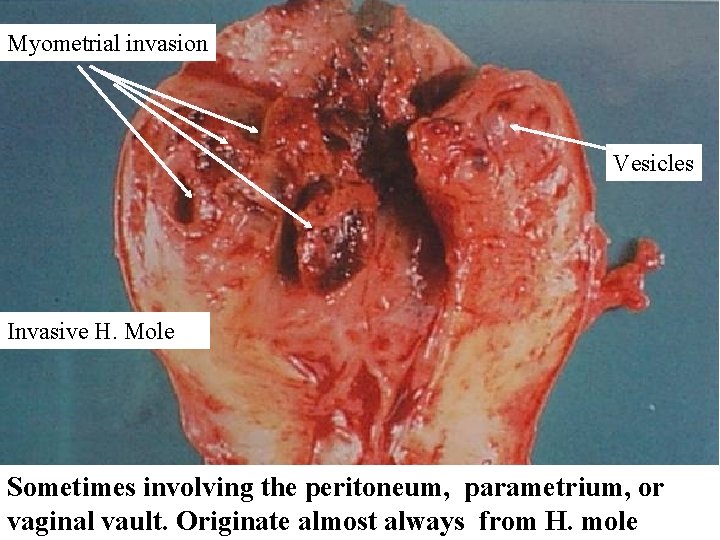
Myometrial invasion Vesicles Invasive H. Mole Sometimes involving the peritoneum, parametrium, or vaginal vault. Originate almost always from H. mole

Metastatic disease Sites: Symptoms: a. Pulmonary- 80% Chest pain, dyspnoea b. Vagina- 30% Vagina bleeding , purulent discharge and nodule. c. Liver- 10% Epigastric pain, jaundice, hepatic rupture leading to intraperitoneal haemorrhage. d. Brain- 10% Focal neurological deficit.

Metastatic Disease If the pelvic examination & chest X-ray are negative, it is very uncommon to have metastatic involvement of other sites.

Chest X ray: widespread metastatic lesions.
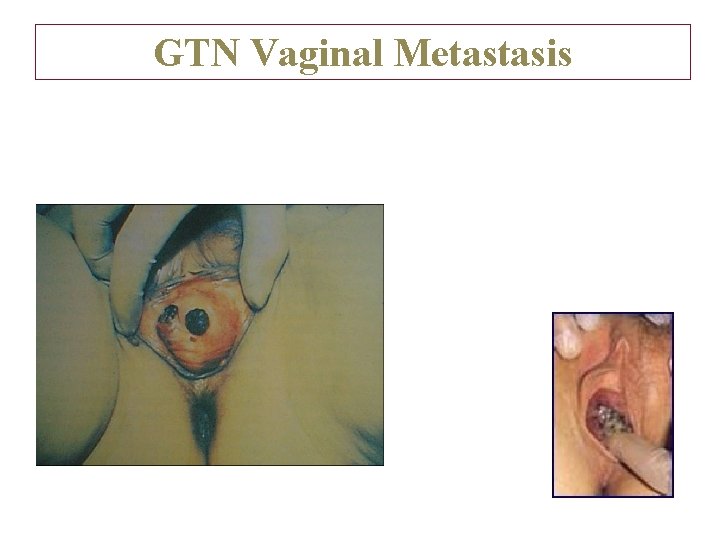
GTN Vaginal Metastasis
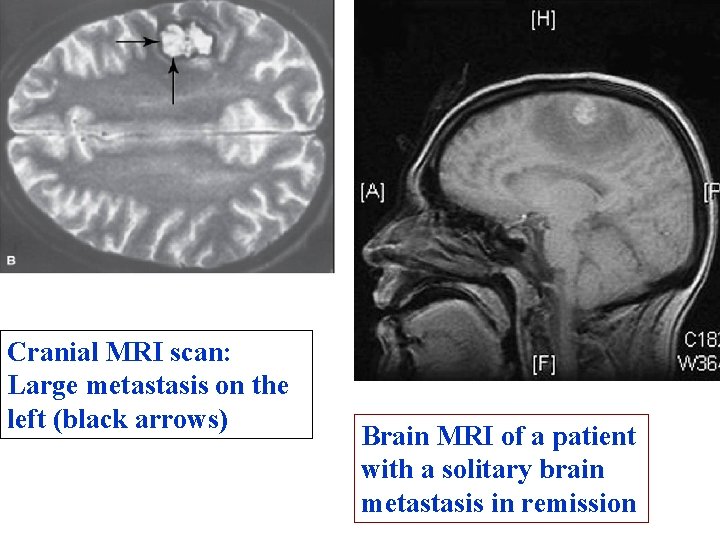
Cranial MRI scan: Large metastasis on the left (black arrows) Brain MRI of a patient with a solitary brain metastasis in remission
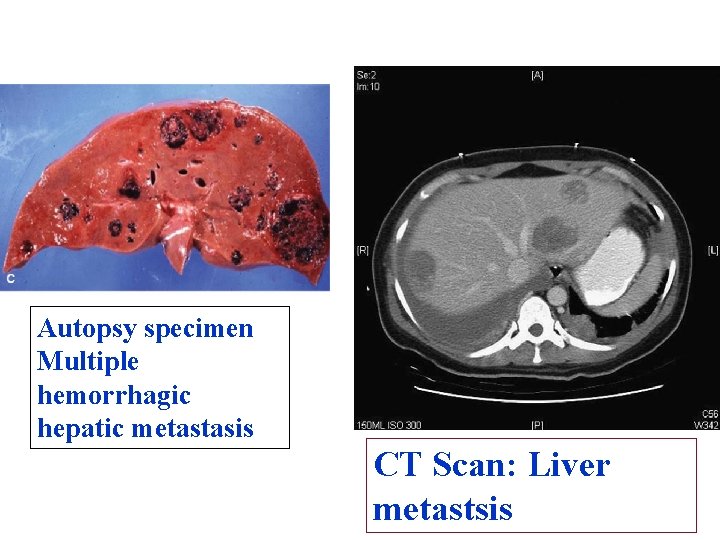
Autopsy specimen Multiple hemorrhagic hepatic metastasis CT Scan: Liver metastsis

Management of GTN Ø Ø Prognostic score is central to the management of GTN. A single universally accepted, anatomical staging and prognostic scoring system for GTN was developed by FIGO in 2000.

ANATOMIC FIGO STAGING SYSTEM FOR GTN (2000) Stage Criteria I. Disease confined to the uterus II. Disease outside of uterus but is limited to the genital structures III. Disease extends to the lungs with or without known genital tract involvement IV. All other metastatic sites
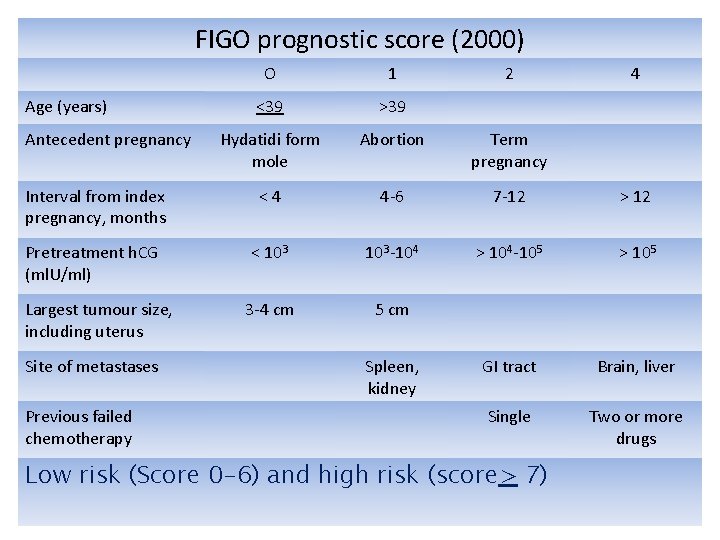
FIGO prognostic score (2000) O 1 <39 >39 Hydatidi form mole Abortion Term pregnancy Interval from index pregnancy, months <4 4 -6 7 -12 > 12 Pretreatment h. CG (ml. U/ml) < 103 -104 > 104 -105 > 105 3 -4 cm 5 cm GI tract Brain, liver Single Two or more drugs Age (years) Antecedent pregnancy Largest tumour size, including uterus Site of metastases Previous failed chemotherapy Spleen, kidney 2 Low risk (Score 0 -6) and high risk (score> 7) 4

WORK UP OF GTN Ø History & physical examination Ø Serial weekly h. CG measurement in serum. Ø Hemogram, LFT, KFT Ø Chest X-ray Ø USG whole abdomen pelvis to rule out hepatic and renal metastasis Ø CT or MRI brain Ø CSF analysis – in metastatic disease. Ø PET Ø FDG PET Ø Curette to be done if there is uterine bleeding. There is grave dangerous of hemorrhage at the biopsy site.

GTN FIGO scoring Low risk (<6) High risk (> 6) Single-agent chemotherapy Combination chemotherapy Serial h. CG levels Resolution Life-long h. CG follow-up Relapsed/resistant disease Second-line chemotherapy+ surgical debulking

Low Risk GTN FIGO score 6 or less. Ø FIGO stage I, II & III. Ø Drugs schedules: single agent chemotherapy Most commonly used regimen. Methotrexate: 1 mg/kg 1 M on days 1, 3, 5, 7 Folinic acid 0. 1 mg/kg 1 M on days, 2, 4, 6 and 8. Side Effects: Stomatitis, conjunctivitis, abdominal and chest pain. Actinomycin- D: (primary therapy in case of abnormal liver function) Ø

Role of hysterectomy in non metastatic disease i. e. Stage-I Ø Ø Ø If patient does not wish to preserve fertility hysterectomy with adjuvant single agent chemotherapy given. In case of stage-I PSTT. Role of adjuvant chemotherapy. 1. 2. 3. Decrease dissemination of viable tumor cells at surgery. Maintain cytotoxic level of chem. To treat any occult metastasis.

Follow up in Low risk GTN Ø Ø Weekly h. CG titre until normal for 3 consecutive weeks. Monthly HCG level until normal for 12 consecutive months. Effective contraception during the follow up (OCP) FIGO recommends additional 2 courses of chemotherapy after initial negative h. CG.

High Risk GTN . Ø Ø Ø § § Stage I, III With FIGO score 7 or greater or Stage IV. Primary intensive combination chemotherapy and selective use of radiation and surgery. Regimes given : MAC. Modified Bagshawe (CHAMOCA) EMA-CO EMA-EP.

MAC-III Regime Mtx 1 mg/kg IM/1 V Days 1, 3, 5, 7 Folinic acid 0. 1 mg/kg IM Days 2, 4, 6, 8 Actinomycin-D 12 g/kg IV Days 1 -5 Cyclophosphamide 3 mg/kg IV Days 1 -5 To be repeated every 15 days if toxicity permits.

EMA-CO Regime Etoposide, methotrexate, actinomycin D, alternating weekly with cyclophosphamide and oncovin. Day 1 Actinomycin D 500 micrograms IV push new IV. Etoposide 100 mg/m 2 over 30 -50 min. Methotrexate 100 mg/m 2 IV infusion over 1 hr and then Methotrexate 200 mg/m 2 IV infusion over 12 hrs Day 2 Actinomycin D 500 micrograms IV push new IV Etoposide 100 mg/m 2 over 30 -50 min. Folinic acid 15 mg IV push Q 6 hrs for 8 doses beginning 24 hrs after methotrexate bolus. Day 8 Vincristine (Oncovin) 1 mg/m 2 IV Cyclophosphamide 600 mg/m 2 IV SIDE EFFECT: - Myelosuppresion , Mucositis , Neuropathy , Reversible alopecia

. EMA-EP In patient resistant to EMA-CO On day 8 Ø Etoposide- 10 mg/m 2 iv. Ø Cisplatin- 80 mg/m 2 iv Ø treatment – until 3 consecutive weekly titres normal. Ø 2 -4 cycles given further after initial normal H. C. G. Ø

Chemotherapy Should Not Be Repeated Unless ØWBC > 3000/cu mm ØPolymorph > 1500/ cu mm ØPlatelets > 100, 000/ cu mm ØBUN, SGOT, SGPT are normal ØNo febrile course ØNo oral or GIT ulceration

Follow up OF High risk GTN Ø Ø Ø Weekly HCG until normal for 3 consecutive weeks. Monthly HCG for 24 consecutive months. Contraception in follow up period.

Management of sites of metastasis VAGINAL &PELVIS -30% Ø Single agent chemotherapy –Low risk Ø Combination chemotherapy –High risk Ø If bleeding occurs. Vaginal packing Wide local excision Arteriographic embolisation of hypogastric arteries

PULMONARY-80% Present as – Chest pain, cough, hemoptysis dyspnoea. Ø X-ray features- Snowstorm Ø Discrete round densities. Ø Pleural effusion Ø Embolic pattern Ø Rx Single agent chemotherapy-low risk. Combination – high risk. Thoracotomy-viable pulmonary Metastasis following combination chemotherapy. Ø

HEPATIC-10% . Ø Ø Worse prognosis. Usually associated with widespread metastasis Intraperitoneal bleeding may occur. Rx Ø Ø Chemo & concurrent radiation (2000 -3000 c. Gy). Hepatic resection to excise resistant foci. Cerebral-10% Ø Ø Acute focal neurological deficits. Combination chemo + WBRT (2000 -3000). Patients with metastasis on initial evaluation respond better than who develop Lesion during therapy. During period of WBRT- Mtx in EMA-CO be increased to 1 g/m 2 with Folinic. Acid rescue.

. AFTER THERAPY OF GTN SURVEILLANCE DURING AND (DISAIA Gynaecolog oncology 2007) Monitor serum quantitative h. CG levels every week during chemotherapy: 1. Response: >10% decline in h. CG during one cycle 2. Plateau: ± 10% change in h. CG during one cycle 3. Resistance: >10% rise in h. CG during one cycle or plateau for two cycles of chemotheraphy - evaluate for new metastasis - consider alternative chemotherapy - consider extirpation of drug-resistant sites of disease

REMISSION: 3 consecutive normal weekly h. CG values 1. Maintenance chemotherapy Surveillance of remission: 1. h. CG values every 2 weeks x 3 months 2. h. CG values every months to complete one year of follow-up 3. h. CG values every 6 -12 months indefinitely; at least 3 -5 years
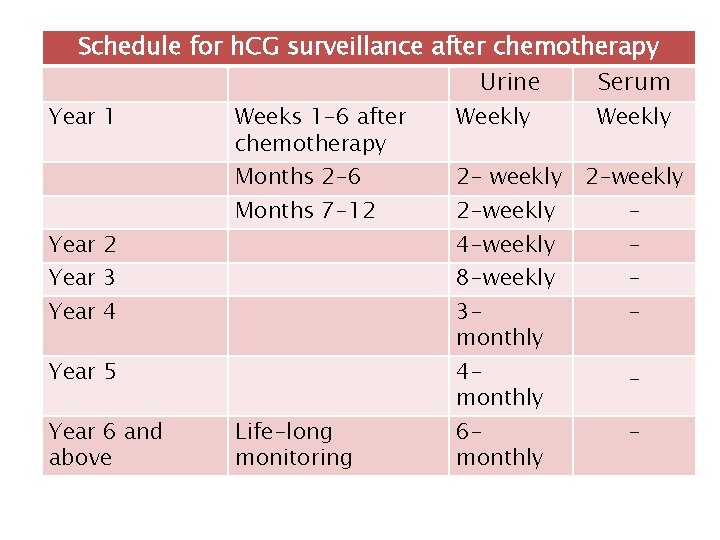
Schedule for h. CG surveillance after chemotherapy Urine Serum Year 1 Weeks 1 -6 after chemotherapy Weekly Months 7 -12 2 -weekly Months 2 -6 Year 2 Year 3 2 -weekly 4 -weekly - 3 monthly Year 5 Year 6 and above 2 - weekly 8 -weekly Year 4 Life-long monitoring Weekly 4 monthly 6 monthly _ -

. CONTRACEPTION DISAIA Gynaecology oncology 2007 During follow up period I. OCP’s Ø Safe Ø Started soon after evacuation of H. mole. Ø Given during the entire period of follow up. Ø No increased risk of postmolar GTN or rate of decline of h. CG titre with OCPs use. II. Barrier methods Ø Safe alternative to OCP’s in patients whom OCP are contraindicated. III. IUCD Ø Contraindicated due to an increased risk of uterine perforation.

Future Child Bearing . DISAIA Gynaecology oncology 2007 Ø Ø Ø Pregnancy outcomes in patient with history of molar gestation are similar to patient with no such history with respect to live birth 1 st and 2 nd trimester abortion, anomalies, Stillbirth, pre-maturity and Caesarean rate. Pregnancy outcome is similar irrespective of complete or partial mole. For subsequent pregnancy. Ø Ist trimester TVS to confirm normal pregnancy. Ø HCG 6 wks after termination of pregnancy. Ø After effective treatment for non-malignant GTN molar pregnancy occur in 1 -2% subsequent pregnancies.

Ø Ø § § Treatment of malignant GTN with Chemotherapy is compatible with preservation of fertility and not assess with increased risk of congenital malformations. Women undergoing I. U. I. after previous molar gestations, occasionally develops repeated moles. This is due to an oocyte defect predisposing to abnormal fertilization. Therapeutic alterations are: o ICSI with pre-implementation genetic diagnosis o Donor oocyte IVF

Co-Existence of normal pregnancy and GTN Ø Ø Ø Ø Described in both spontaneous & IVF gestations. Estimated incidence of twin pregnancy with 1 mole and 1 fetus 1 per 221, 000 -1, 000 or > 125 cases reported. Diagnosis difficult based on imaging alone. In many cases diagnosis made only after termination of pregnancy. Increased risk for haemorrhage persistent GTN and medical complications. Pregnancy coexistent with H. mole need be termination. Usually preterm delivery is often required because of complications like bleeding or preeclampsia.

Thank You
- Slides: 70