GESTATIONAL DIABETES Majid Valizadeh Obesity Research Center Research

GESTATIONAL DIABETES Majid Valizadeh Obesity Research Center Research Institute for Endocrine Sciences 07 Aug 2019

AGENDA: § Case presentation §Normal pregnancies/ GDM pathophysiology §Definition §Overt diabetes §GDM §Prevalence §Importance §Risk factors §Diagnosis §Two step ØManagement Nonpharmacologic Diet Exercise Pharmacologic Insulin Oral agents Perinatal care Contraception after delivery Breast feeding DM risk after delivery National guideline


GLUCOSE CHALLENGE TEST: After 50 gr glucose: BS: 132 mg/dl BS: 137 mg/dl BS: 145 mg/dl

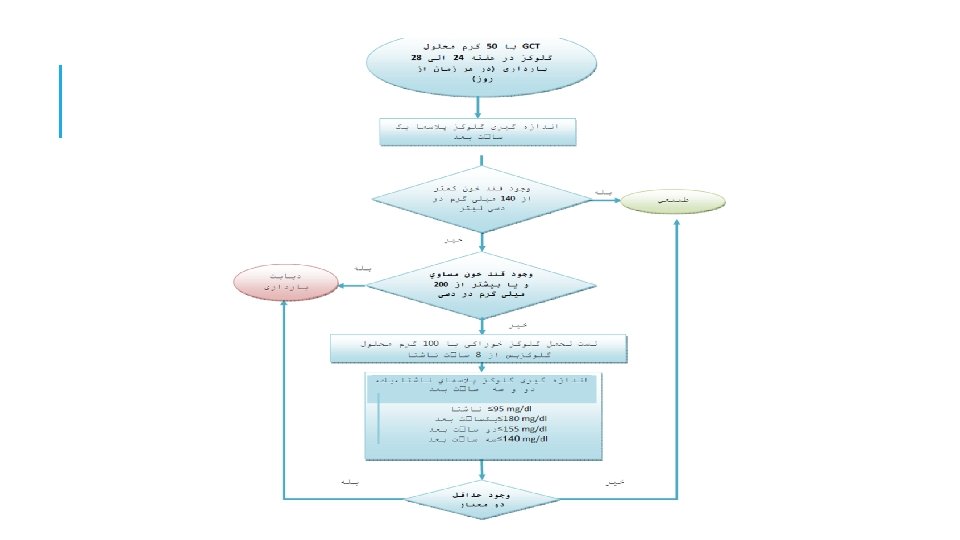
100 GRAM / 3 H 1) FBS: 89 mg/dl 1 h: 165 mg/dl 2 h: 153 mg/dl 3 h: 135 mg/dl 2) FBS: 93 mg/dl 1 h: 185 mg/dl 2 h: 153 mg/dl 3 h: 145 mg/dl 3) FBS: 93 mg/dl 1 h: 185 mg/dl 2 h: 153 mg/dl 3 h: 135 mg/dl ﺍﻗﺪﺍﻡ ﺑﻌﺪی؟

NORMAL PREGNANCY:

EARLY PREGNANCY §Increased glucose-stimulated insulin secretion §Unchanged or enhanced peripheral (muscle) insulin sensitivity §Unchanged basal hepatic glucose production §Normal or slightly improved glucose tolerance §Normal sensitivity to the blood glucose–lowering effect of exogenously administered insulin

LATE PREGNANCY §Rising concentrations of several diabetogenic hormones §Increased peripheral insulin resistance §Progressive increase in basal & postprandial insulin (up to 2 fold in third trimester) §lower insulin action in late normal pregnancy than in non pregnant women (50 -70%) §Basal endogenous hepatic glucose production increases by 16– 30%

HORMONES ASSOCIATED WITH MODIFICATIONS IN INSULIN SECRETION AND ACTION § Estrogens Insulin concentration Insulin binding § Progesterone Glucose transport Insulin binding insulin-induced hepatic gluconeogenesis

HORMONES ASSOCIATED WITH MODIFICATIONS IN INSULIN SECRETION AND ACTION…. §Cortisol Insulin resistance Phosphorylation of insulin receptor IRS-1 § h. PL, GH Insulin sensitivity Insulin secretion Insulin synthesis Utilization and glucose oxidation c. AMP metabolism -cell number -cell mass §Leptin Insulin resistance §Glucagon Insulin resistance

PATHOPHYSIOLOGY OF GDM §The development of GDM is associated with a much greater severity of insulin resistance than normal pregnant women §The degree of insulin resistance seems to be influenced by obesity & inheritance §Gestational diabetes mellitus occurs when a woman's pancreatic function is not sufficient to overcome the insulin resistance

PATHOPHYSIOLOGY OF GDM occurs as a result of a combination of insulin resistance and decreased insulin secretion The predominant pathogenic factor in GDM could be inadequate insulin secretion

DEFINITION Any degree of glucose intolerance with onset or first recognition during pregnancy New terminology: Overt diabetes Gestational diabetes


OVERT DIABETES
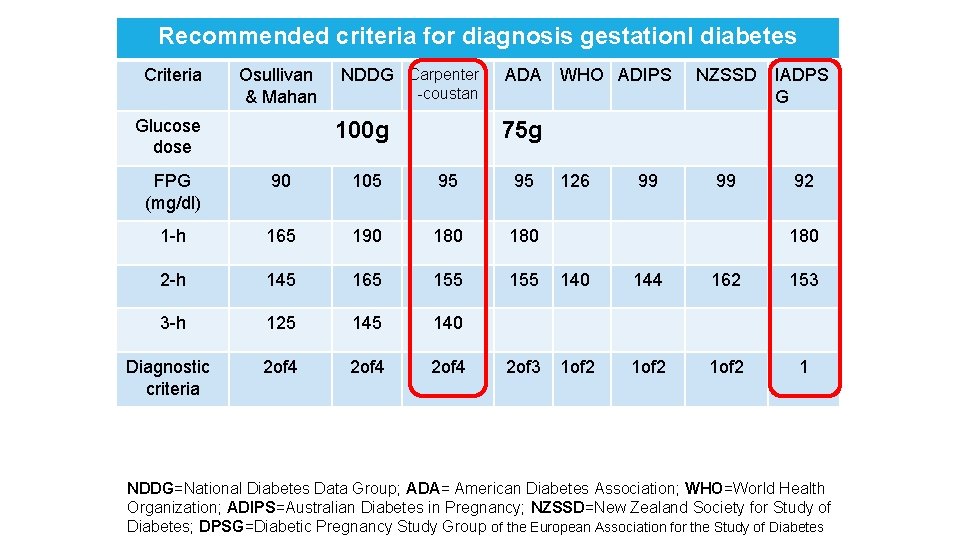
Recommended criteria for diagnosis gestationl diabetes Criteria Osullivan & Mahan Glucose dose NDDG Carpenter -coustan 100 g ADA WHO ADIPS NZSSD IADPS G 75 g FPG (mg/dl) 90 105 95 95 126 99 99 92 1 -h 165 190 180 2 -h 145 165 155 140 144 162 153 3 -h 125 140 Diagnostic criteria 2 of 4 2 of 3 1 of 2 1 180 NDDG=National Diabetes Data Group; ADA= American Diabetes Association; WHO=World Health Organization; ADIPS=Australian Diabetes in Pregnancy; NZSSD=New Zealand Society for Study of Diabetes; DPSG=Diabetic Pregnancy Study Group of the European Association for the Study of Diabetes

PREVALENCE GDM complicates approximately 1% to 14% of all pregnancies In low-risk populations, such as those found in Sweden, the prevalence in population-based studies is lower than 2% in high-risk populations, such as the Native American Cree, Northern Californian Hispanics and Northern Californian Asians, reported prevalence rates ranging from 4. 9% to 12. 8%. Curr Diab Rep (2010) 10: 224– 228

REASONS FOR DIFFERENCES IN REPORTED PREVALENCE Different screening policies Different diagnostic criteria Different definitions, screening strategies and awareness of type 2 diabetes Maternal age Racial/ethnic composition of population Obstet Gynecol Clin North Am. 2007 June ; 34(2):
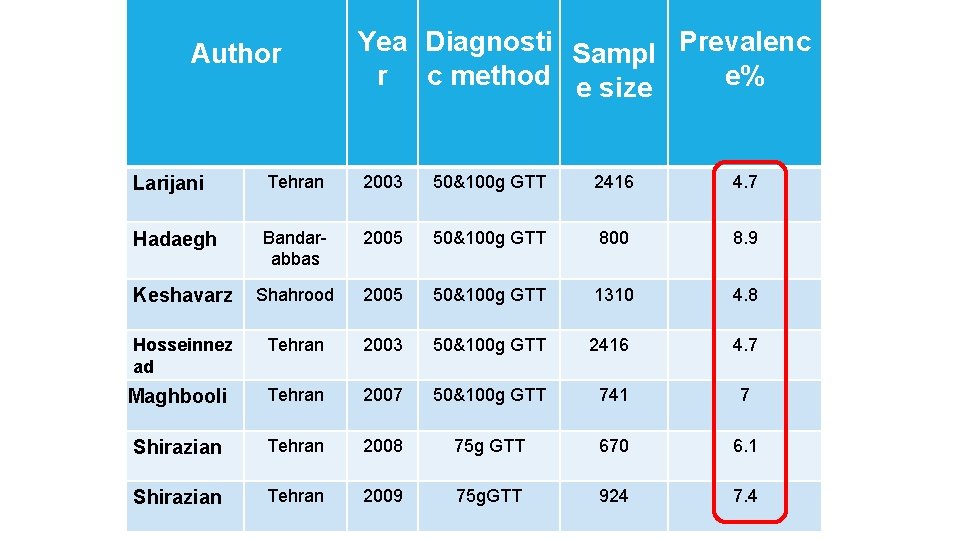
Author Yea Diagnosti Sampl Prevalenc r c method e size e% Larijani Tehran 2003 50&100 g GTT 2416 4. 7 Hadaegh Bandarabbas 2005 50&100 g GTT 800 8. 9 Keshavarz Shahrood 2005 50&100 g GTT 1310 4. 8 Hosseinnez ad Tehran 2003 50&100 g GTT 2416 4. 7 Maghbooli Tehran 2007 50&100 g GTT 741 7 Shirazian Tehran 2008 75 g GTT 670 6. 1 Shirazian Tehran 2009 75 g. GTT 924 7. 4
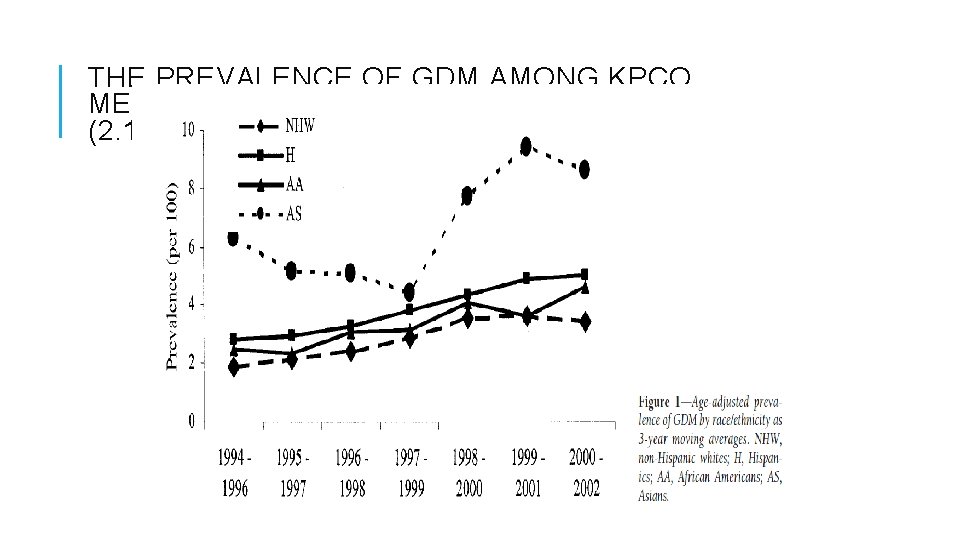
THE PREVALENCE OF GDM AMONG KPCO MEMBERS DOUBLED FROM 1994 TO 2002 (2. 1– 4. 1%, P 0. 001)

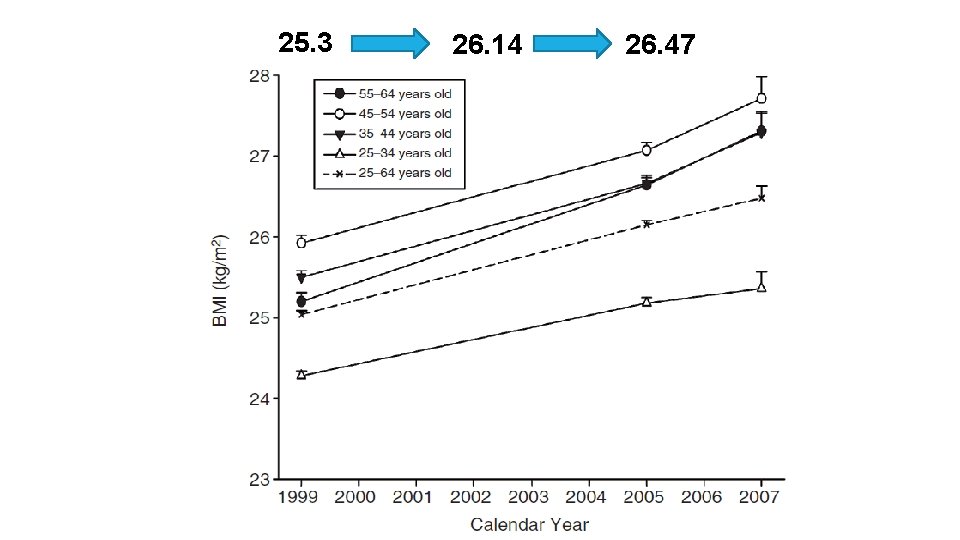
To determine secular trends of overweight and obesity among Iranian adults (25– 64 years old) within an 8 -year period (1999– 2007)
25. 3 26. 14 26. 47
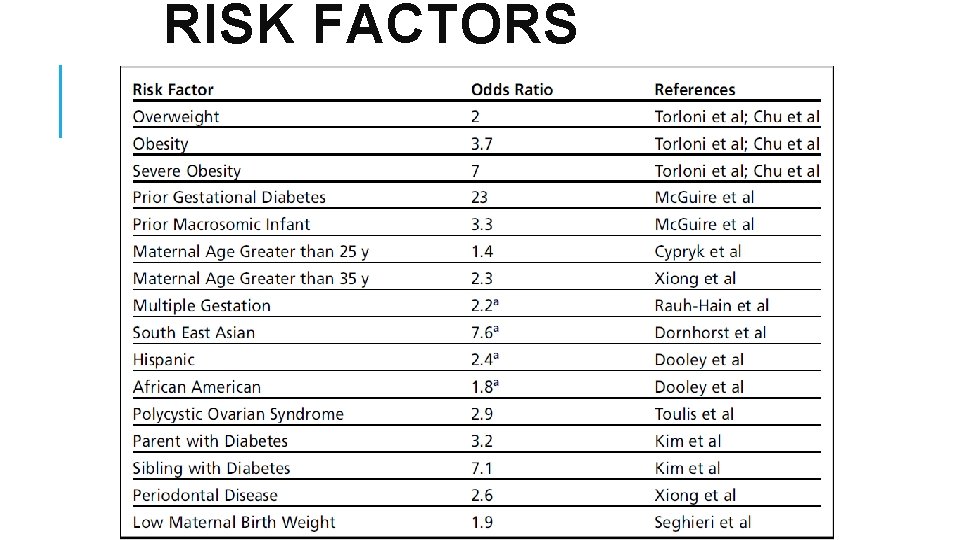
RISK FACTORS

MATERNAL AND FETAL COMPLICATIONS: Maternal: fetal v Preeclampsia v. Macrosomia v Cesarean delivery v. Neonatal hypoglycemia v Developing diabetes v. Hyperbilirubinemia v. Shoulder dystocia v. Birth trauma v. Still birth v……………. . ØChildhood obesity and diabetes

SCREENING § Early pregnancy § 24 -28 weeks

SHOULD WE SCREEN FOR GESTATIONAL DIABETES? The disease should be prevalent The disease should be causally associated with selected adverse outcomes. The disease should have an asymptomatic phase during which detection is possible. Treatment should be available to ameliorate the effects of the disease.
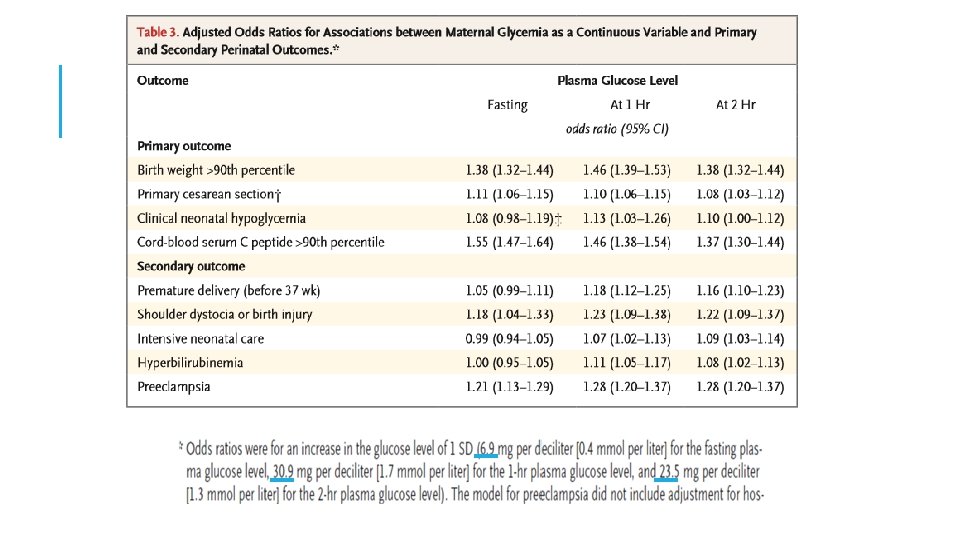
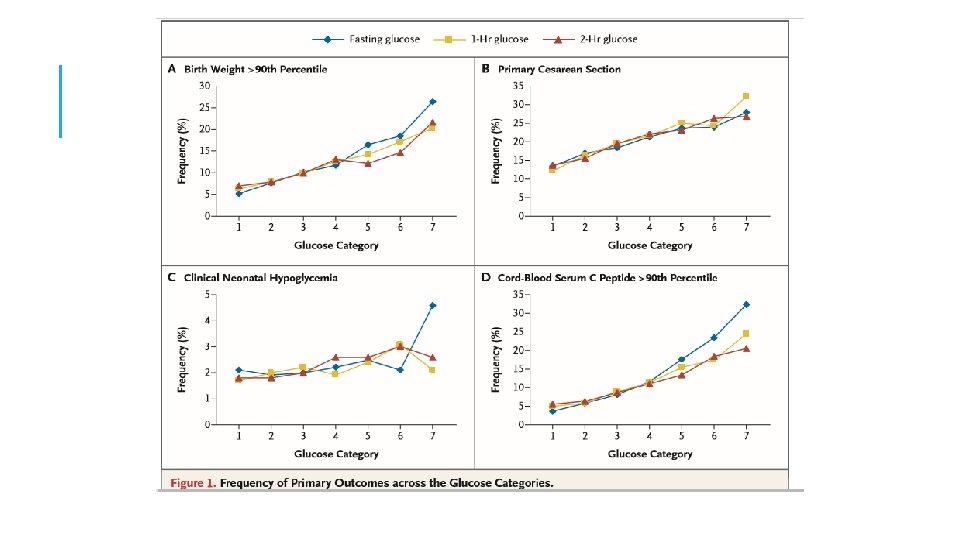

ADA 2018
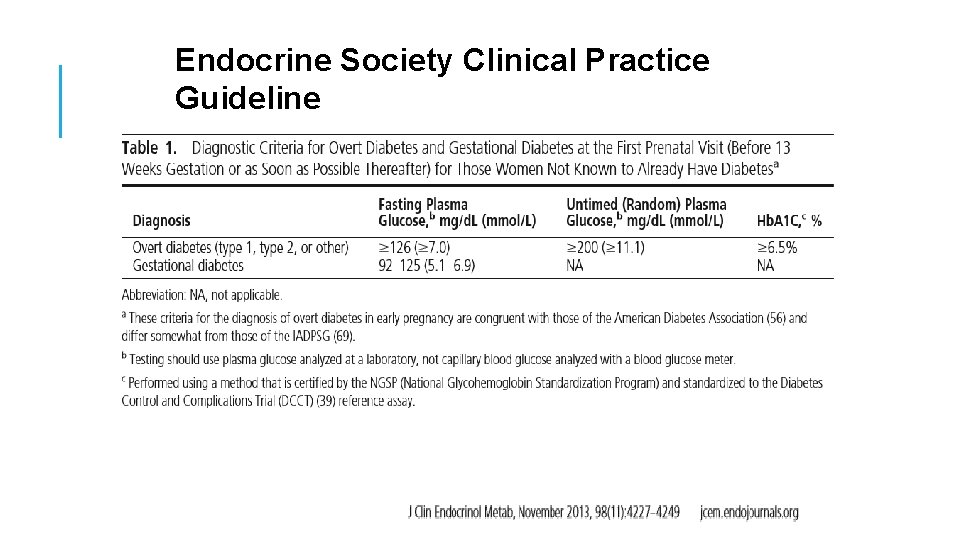
Endocrine Society Clinical Practice Guideline

ADA 2018 ……. The International Association of the Diabetes and Pregnancy Study Groups (IADPSG) GDM diagnostic criteria for the 75 -g. OGTT were not derived from data in the first half of pregnancy, so the diagnosis of GDM in early pregnancy by either FPG or OGTT values is not evidence based…….

Diabetes Care Volume 39, January 2016

EARLY PREGNANCY SCREENING: For detection of undiagnosed T 2 DM Who: ? Overweight or Obese women with other risk factors The best test: ? (FBS, 2 h post 75 gr glucose, or start with 50 gr glucose, Hb A 1 c? With less sensitivity)
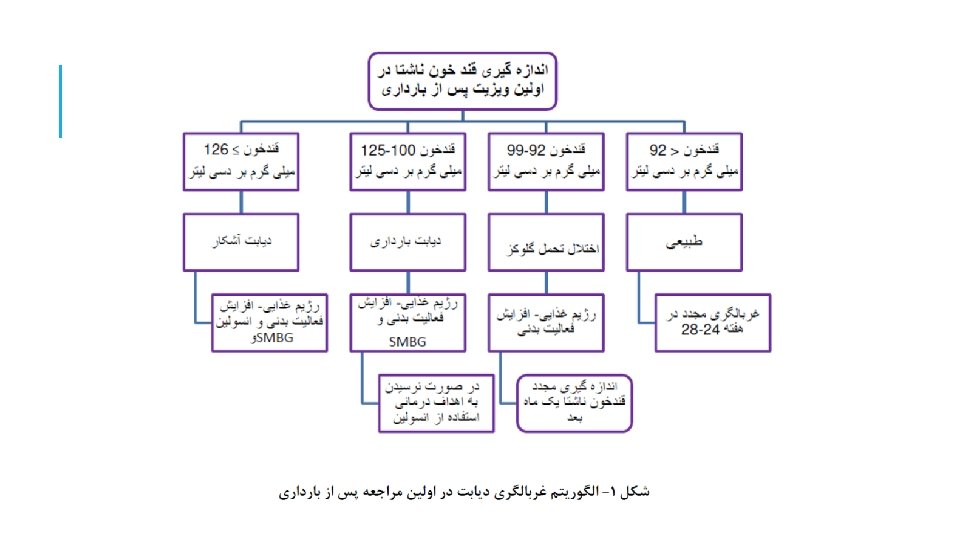


Aim: Impact of new IADPSG criteria on diagnosis of GDM compared with ADA criteria Diabetes Care 33: 2018– 2020, 2010

METHOD Sample Size: 1283 pregnant women Universal screening with 75 g. OGTT at 24 -28 weeks of gestation ADA and new IADPSG criteria were applied

RESULTS ADA criteria identified 12. 9%women with GDM ADPSG criteria identified 37. 7% women with GDM

KEY MESSAGE The IADPSG criteria increased GDM prevalence nearly threefold
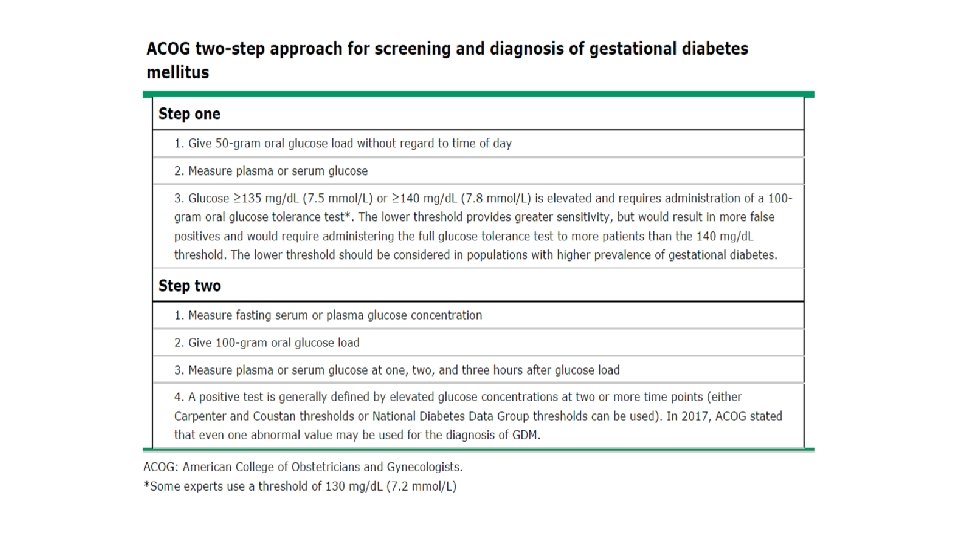
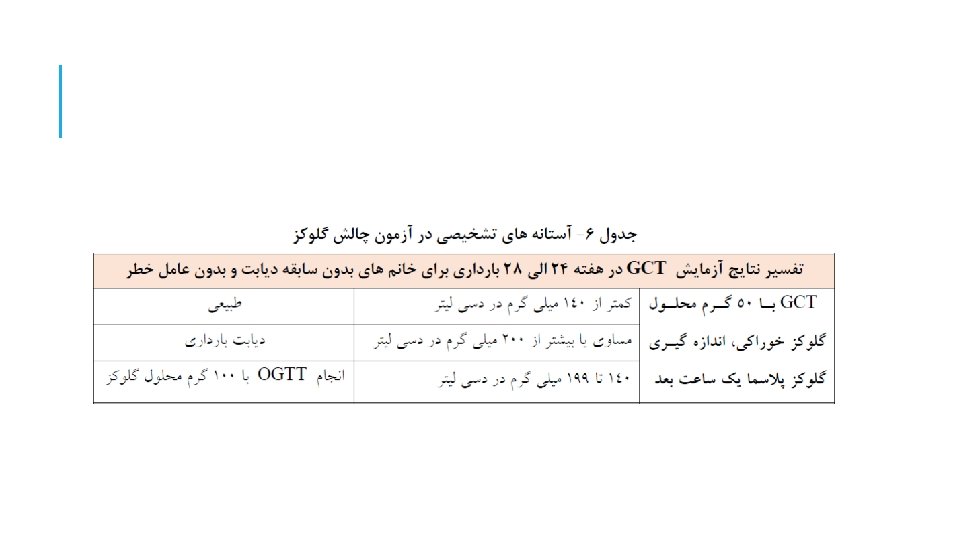
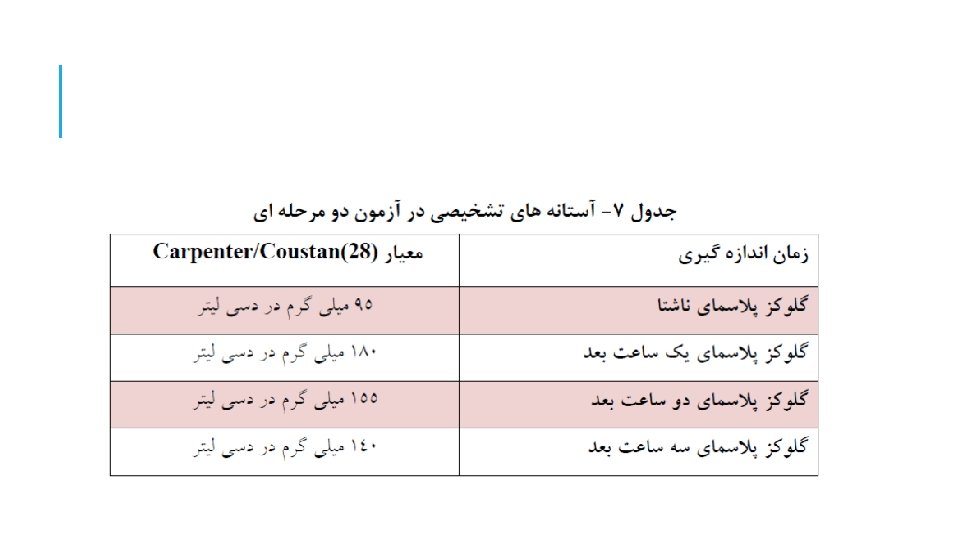

MANAGEMENT:

§All included studies compared diet modification, glucose monitoring, and insulin as needed with no treatment. §Treating GDM results in less preeclampsia (RR 0. 62, 95% CI 0. 43 -0. 89), shoulder dystocia (RR 0. 42, 95% CI 0. 23 -0. 77), and macrosomia (RR 0. 50, 95% CI 0. 35 - 0. 71). Moderate evidence §Current evidence does not show an effect on neonatal hypoglycemia or future poor metabolic outcomes. Low or insufficient evidence §There is little evidence of short-term harm of treating GDM other than an increased demand for services. Ann Intern Med. 2013; 159: 123 -129. 44

GLYCEMIC TARGETS PRECONCEPTIONALLY FOR WOMEN WITH OVERT DIABETES AND DURING PREGNANCY FOR WOMEN WITH EITHER OVERT DIABETES OR GESTATIONAL DIABETES Target preprandial blood glucose is 90 mg/d. L if this can be safely achieved without undue hypoglycemia. Hb. A 1 C 7% (ideally 45 J Clin Endocrinol Metab 98: 4227– 4249, 2013 6. 5%)

GLYCEMIC GOAL FOR PREGNANT WOMEN WITH PRE EXISTING TYPE 1 OR TYPE 2 DM (ADA 2015) Premeal, bedtime, and overnight glucose 60– 99 mg/d. L Peak postprandial glucose 100– 129 mg/d. L Hb. A 1 C < 6. 0%

SELF MONITORING OF BLOOD GLUCOSE We recommend self-monitoring of blood glucose in all pregnant women with gestational or overt diabetes(1++++) We suggest testing before and either 1 or 2 hours after the start of each meal and, as indicated, at bedtime and during the night(2++O 0) 47 J Clin Endocrinol Metab 98: 4227– 4249, 2013

NONPHARMACOLOGIC TREATMENTS v Dietary modifications v. Goals: v. Achieve normal blood glucose v. Prevent ketosis v. Provide adequate weight gain v. Contribute to appropriate fetal growth and development v Exercise v Glucose monitoring

DIET §A structured diet: §Caloric allotment, / Carbohydrate intake, / Caloric distribution §CHO 35 -45%, Protein 20 -25%, fat 30 -40% § - Use of food with low glycaemic index to avoid post-prandial hyperglycaemia § - Distribute the amount of CHO in three main meals and two to three snacks, taking into account BG, insulin regimen and physical activity
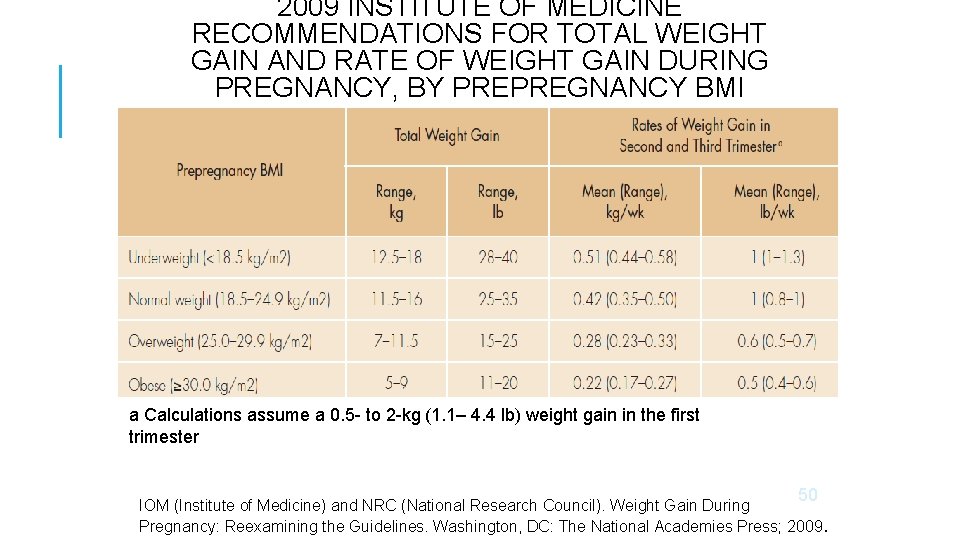
2009 INSTITUTE OF MEDICINE RECOMMENDATIONS FOR TOTAL WEIGHT GAIN AND RATE OF WEIGHT GAIN DURING PREGNANCY, BY PREPREGNANCY BMI a Calculations assume a 0. 5 - to 2 -kg (1. 1– 4. 4 lb) weight gain in the first trimester 50 IOM (Institute of Medicine) and NRC (National Research Council). Weight Gain During Pregnancy: Reexamining the Guidelines. Washington, DC: The National Academies Press; 2009.

EXERCISE § Most of RCTs have small sample size § Improvement in glucose levels § Moderate exercise program ( 30 min/d moderate intensity aerobic exercise at least 5 days of week or 150 min/W ) § Simple exercise such as walking 10 -15 min after each meal

GDM CLASSIFICATION Diet controlled GDM: Class A 1 GDM Require medication to achieve euglycemia: Class A 2 GDM

PHARMACOLOGIC TREATMENT: §Insulin §Human §Analogs §Oral agents

POSITION OF INSULIN IN GDM TREATMENT RECOMMENDATIONS FROM DIFFERENT SOCIETIES §American Diabetes Association-2018: § Insulin is the preferred agent for management of diabetes in pregnancy. (E) §American College of Obstetricians and Gynecologists-2018: § When pharmacologic treatment of GDM is indicated, insulin is considered the preferred treatment for diabetes in pregnancy. (A) §Iranian societies (Iran Endocrine Society/NAIGO/Iranian Scientific Society of Perinatology)-2017: § Insulin is the first-line medical therapy in GDM. 54

§ 53 relevant studies (103 publications), reporting data for 7381 women were included. § 46 of these studies reported data for 6435 infants but our analyses were based on fewer number of studies/participants. § 40 of the 53 included trials were not blinded. §Overall, the quality of the evidence ranged from moderate to very low quality. Cochrane Database of Systematic Reviews 2017, Issue 11. Art. No. : CD 012037. 55

MAIN FINDINGS FOR THE MOTHER: §Insulin versus oral anti-diabetic pharmacological therapy: § Insulin was associated with an increased risk for hypertensive disorders of pregnancy [risk ratio (RR) 1. 89, 95% confidence interval (CI) 1. 14 to 3. 12; four studies, 1214 women; moderate-quality evidence]. § No clear evidence of a difference for the risk of: § pre-eclampsia § cesarean section § postnatal weight retention § developing type 2 diabetes § The outcomes of perineal trauma/tearing or postnatal depression were not reported in the included studies. Cochrane Database of Systematic Reviews 2017, Issue 11. Art. No. : CD 012037. 56

MAIN FINDINGS FOR THE INFANT: §Insulin versus oral anti-diabetic pharmacological therapy: § No clear evidence of a difference for the risk of: § large-for-gestational age § perinatal (fetal and neonatal death) mortality § death or serious morbidity composite § neonatal hypoglycemia § neonatal adiposity at birth § childhood adiposity § neurosensory disabilities in later childhood § Later infant mortality, and childhood diabetes were not reported as outcomes in the included studies. Cochrane Database of Systematic Reviews 2017, Issue 11. Art. No. : CD 012037. 57

MAIN FINDINGS: (CONT…) §With regards to comparisons for regular human insulin versus other insulin analogues, insulin versus diet/standard care, insulin versus exercise and comparisons of insulin regimens, there was insufficient evidence to determine any differences for many of the key health outcomes. Cochrane Database of Systematic Reviews 2017, Issue 11. Art. No. : CD 012037. 58

CONCLUSION §The available evidence suggests that there are very few differences in short-term outcomes for the mother and baby between treatment with injected insulin and treatment with oral medication. There is not enough evidence yet for the long-term outcomes. §Considering other comparisons, evidence is insufficient. Cochrane Database of Systematic Reviews 2017, Issue 11. Art. No. : CD 012037. 59

USE OF INSULIN ANALOGS IN PREGNANCY We suggest that the long-acting insulin analog detemir may be initiated during pregnancy for those women who require basal insulin and for whom (NPH) insulin, in appropriate doses, has previously resulted in, or for whom it is thought NPH insulin may result in, problematic hypoglycemia Insulin detemir may be continued in those women with diabetes already successfully taking insulin detemir before pregnancy (2 ) J Clin Endocrinol Metab 98: 4227– 4249, 2013

USE OF INSULIN ANALOGS IN PREGNANCY We suggest that women with diabetes successfully using the longacting insulin analogs insulin glargine preconceptionally may continue with this therapy before and then during pregnancy. J Clin Endocrinol Metab 98: 4227– 4249, 2013

INSULIN DOSE §The majority of studies have reported a total insulin dose ranging from 0. 7 to 2 units per kg (present pregnant weight) to achieve glucose control. §The dose and type of insulin used is calculated based upon the specific abnormality of fasting or postprandial blood glucose noted during monitoring. §The starting insulin dose should be considered just that, a starting point. §Adjustments in insulin dosage in response to high glucose values are typically in the range of 10 -20%. §Twin gestations complicated by GDM may require an approximate doubling of the insulin requirement throughout pregnancy. Durnwald C. www. uptodate. com © 2018 Up. To. Date, Inc. Last updated 31. 10. 2018. 62

Two meta analysis in 2015

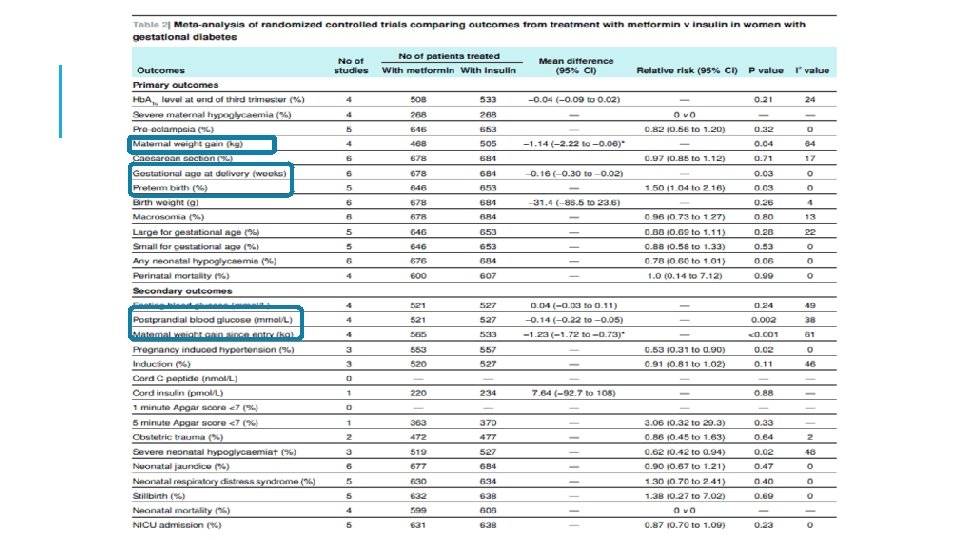
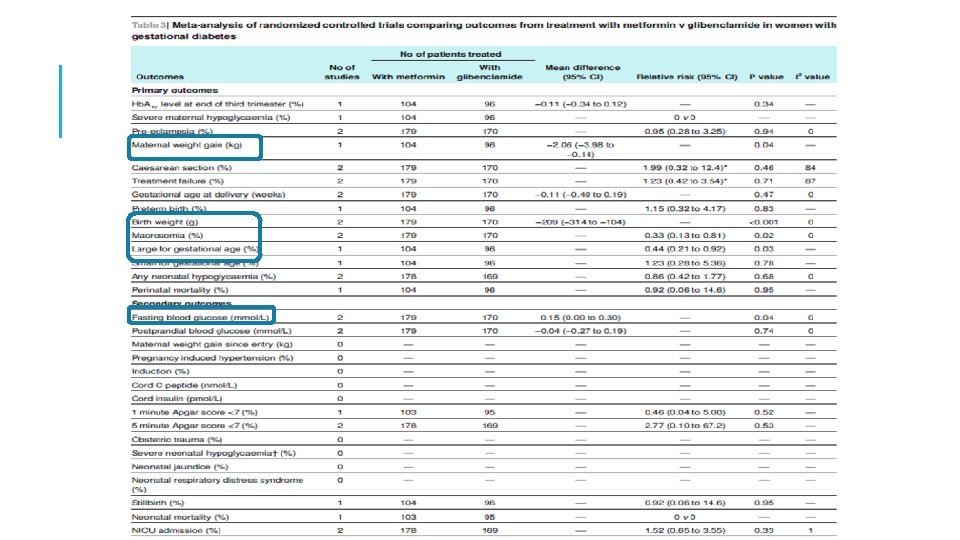
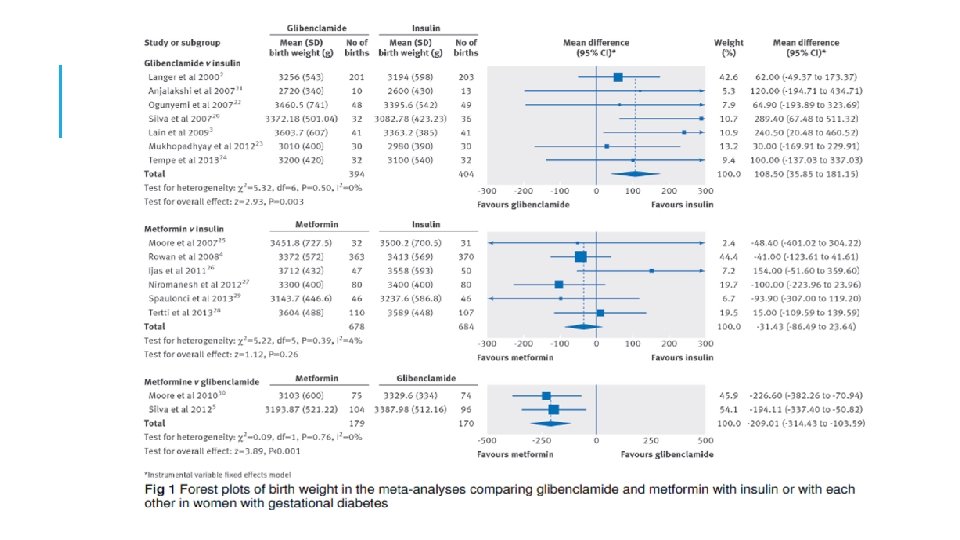
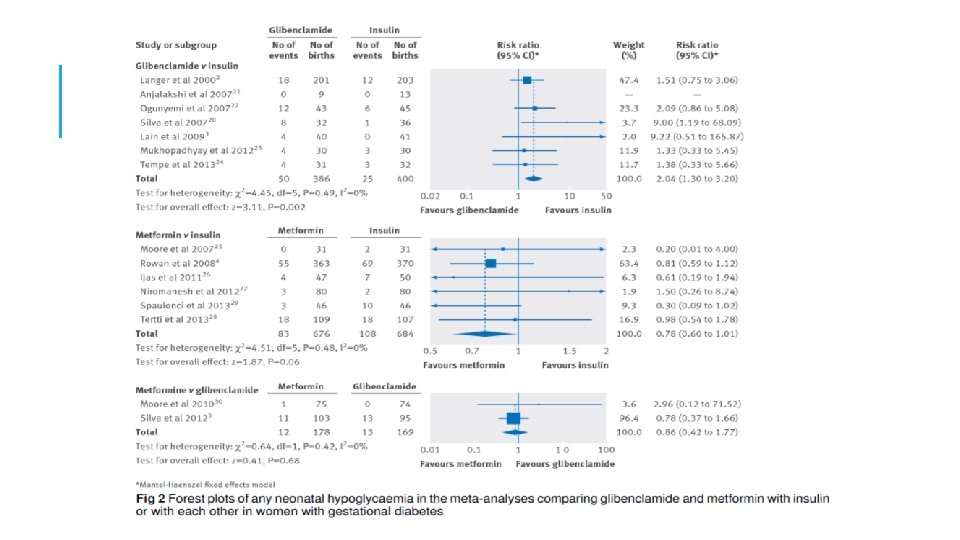
META-ANALYSIS: GLIBENCLAMIDE VS INSULIN §Increased birth weight (109 g) §Increased risk of macrosomia RR=2. 62 (1. 35 -5. 08) §Increased risk of neonatal hypoglycemia RR=2. 04 (1. 30 -3. 20) §Glib. should not be used in the treatment of GDM women if insulin or metformin is available Balsells M. BMJ 2015

META-ANALYSIS: METFORMIN VS INSULIN §Decreased maternal weight gain, (-1. 14 kg) §Decreased pregnancy induced hypertension, RR=0. 53 (0. 310. 90) §Increased risk of preterm birth, RR=1. 50 (1. 04 -2. 16) Balsells M. BMJ 2015

META-ANALYSIS: METFORMIN VS GLIBENCLAMIDE §Decreased maternal weight gain, (-2. 06 kg) §Decreased birth weight (-209 g) §Decreased risk of macrosomia, RR=0. 33 (0. 13 -0. 81) Balsells M. BMJ 2015

Yun-fa Jiang, JCEM 2105

META-ANALYSIS: METFORMIN VS INSULIN Lower maternal weight gain(WMD: -1. 49 kg, 95%CI: -2. 26 to -0. 31) Shorter gestational age (WMD: -0. 16 weeks, 95%CI: -0. 30 to -0. 03) Increased incidence of premature(OR: 1. 63, 95%CI: 1. 07 to 2. 48), Yun-fa Jiang, JCEM 2105

META-ANALYSIS: GLIBENCLAMIDE VS INSULIN Higher neonatal birth weight (WMD: 130. 68 g, 95%CI: 55. 98 to 205. 38), Increased incidence of neonatal hypoglycemia(OR: 2. 64, 95%CI: 1. 59 to 4. 38), Increased incidence of macrosomia (OR: 3. 09, 95%CI: 1. 59 to 6. 04). Yun-fa Jiang, JCEM 2105

META-ANALYSIS: GLIBENCLAMIDE VS METFORMIN lower maternal weight gain, WMD: 1. 34 (-3. 05, 0. 38) lower neonatal birth weight, -187. 7 (-270. 2, 106. 5) Increased incidence of neonatal hypoglycemia, 2. 59 (1. 25, 5. 01) Increased incidence of macrosomia, 7. 46 (2. 20, 23. 31) Yun-fa Jiang, JCEM 2105

ORAL AGENTS (AFTER 24 WEEKS/FBS <110 MG/DL) metformin Glibenclamide Intial dose: 500 mg after dinner Intial dose: 2. 5 mg before breakfast Maximal dose: 2500 -3000 mg Maximal dose: 25 -30 mg Failure:
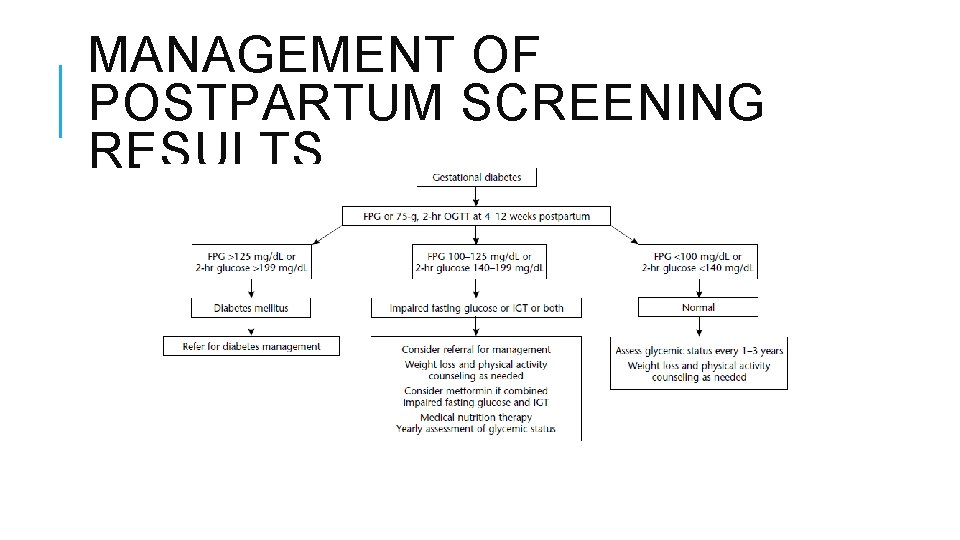
MANAGEMENT OF POSTPARTUM SCREENING RESULTS

POSTPARTUM CONTRACEPTION We recommend that the choice of a contraceptive method for a woman with overt DM or a history of GDM should not be influenced by virtue of having overt DM or a history of GDM J Clin Endocrinol Metab 98: 4227– 4249, 2013

POSTPARTUM CONTRACEPTION Combined OCP use by women with type 1 DM does not affect their glycemic control or increase their risk of end-organ injury. Combined OCP use by women with history of GDM does not increase the risk of later developing type 2 DM. Use of a contraceptive patch or vaginal ring exerts a similar metabolic effect to that of oral contraceptives J Clin Endocrinol Metab 98: 4227– 4249, 2013

TAKE HOME MESSAGE Early pregnancy screening The best test? GDM?


THANK YOU FOR YOUR ATTENTION

Thank you for your attention

HOW IS GDM DIAGNOSED? How is GDM diagnosed? What are the benefits of treating GDM? How should blood glucose be monitored in a woman with GDM? What nonpharmacologic treatments are effective in managing GDM? What pharmacologic treatments are effective in managing GDM? Is fetal assessment indicated in pregnancies complicated by GDM? What are delivery consideration in pregnancies complicated by GDM? How should women with a history of GDM be screened and counseled postpartum?

A 27 -year-old woman presents for consultation regarding newly diagnosed gestational diabetes mellitus in her first pregnancy. She is at 30 weeks' gestation, and her pregnancy has otherwise been uncomplicated. She has no history of hyperglycemia or diabetes despite a diagnosis of polycystic ovary syndrome. Her father and sister have diabetes mellitus. She has gained 40 lb (18. 2 kg) during the pregnancy and has lately been experiencing marked polyuria, polydipsia, and nocturia. Results from a 50 -g oral glucose tolerance test, performed at 28 weeks‘ gestation, are abnormal with a blood glucose value of 220 mg/d. L (12. 2 mmol/L). Lifestyle modification is initiated in response to this abnormal test result. Fetal ultrasonography documents a large-for-gestational-age fetus.

On physical examination, she is a pregnant woman with an appropriate affect. Her blood pressure is 131/72 mm Hg, and pulse rate is 78 beats/min. Height is 63 in (160 em), and weight is 160 lb (72. 7 kg) (BMI == 28. 3 kg/m 2). Mildfacial hirsutism is present. She has no cushingoid features. She has acanthosis nigricans on the posterior neck. Her abdomen is gravid. Her hemoglobin A 1 c level is 7. 7% ( 61 mmol/mol). Her glucometer download shows fasting blood glucose values that are consistently in the range of 141 to 160 mg/d. L (7. 8 to 8. 9 mmol/L) and 2 -hour postprandial blood glucose values that are consistently above 200 mg/d. L (> 11. 1 mmol/L). In addition to nutritional counseling and lifestyle modification, the most ~ppropriate treatment regimen for this patient would consist of which of the following? A. Metformin B. A sulfonylurea C. Insulin D. A thiazolidinedione E. A dipeptidyl-peptidase 4 inhibitor
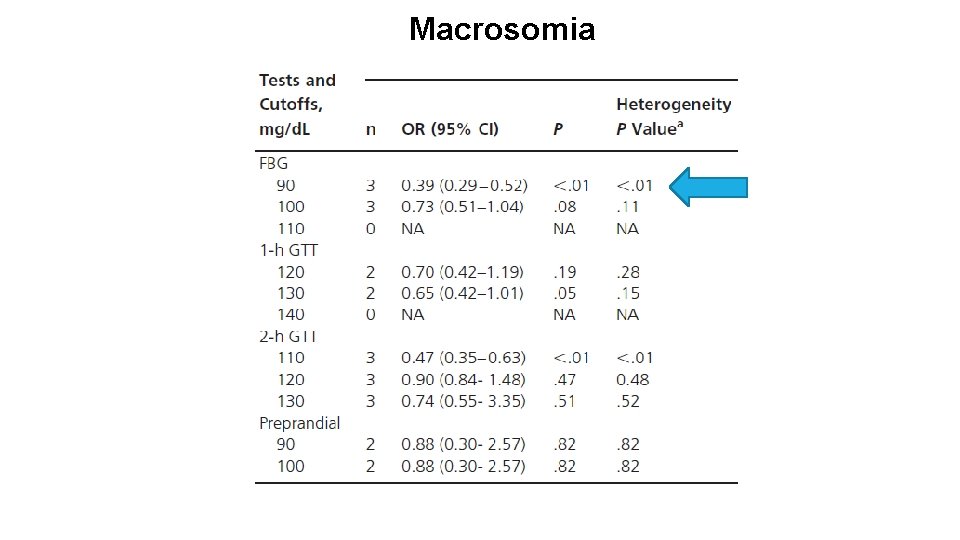
Macrosomia
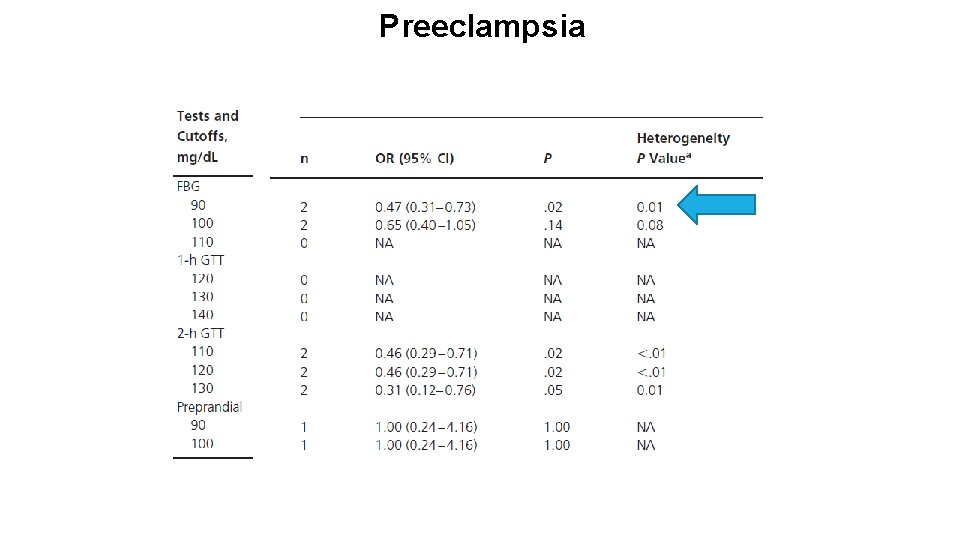
Preeclampsia
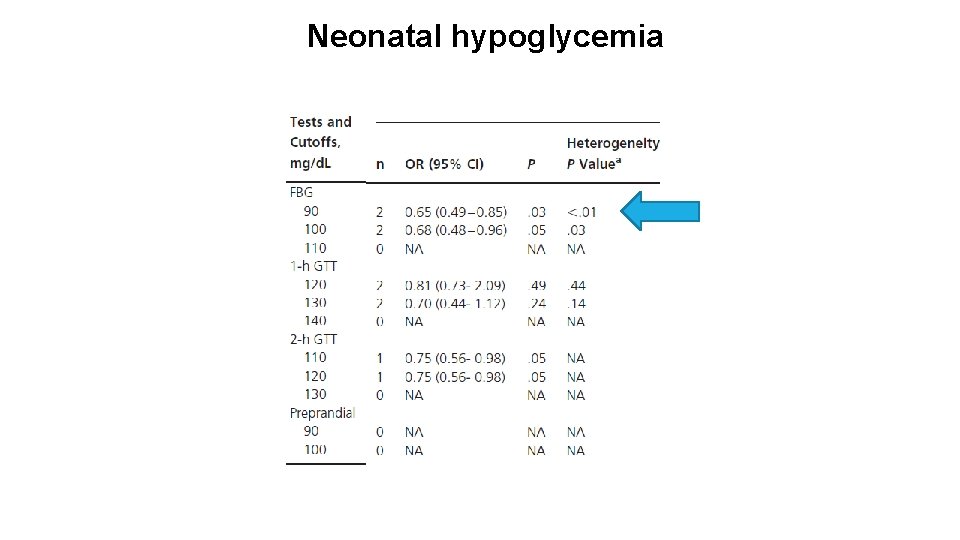
Neonatal hypoglycemia
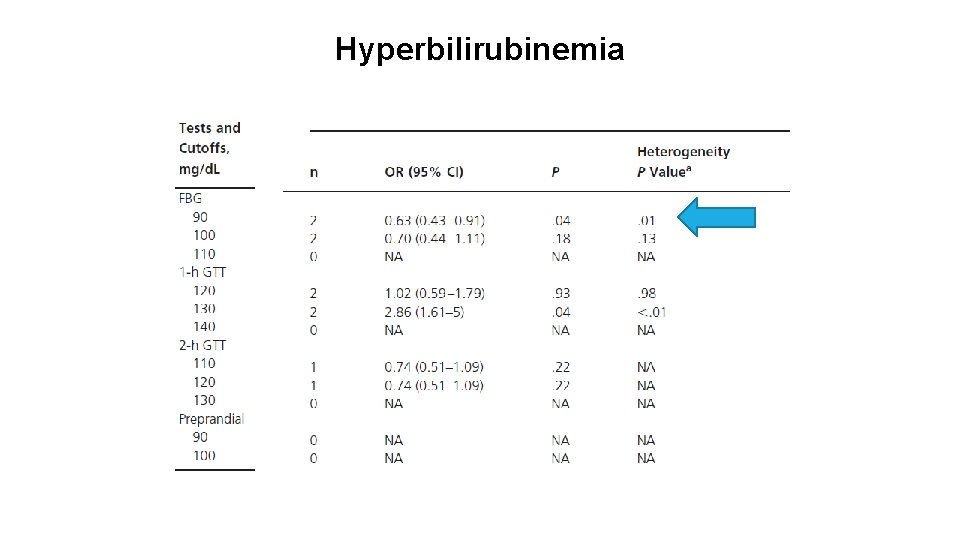
Hyperbilirubinemia
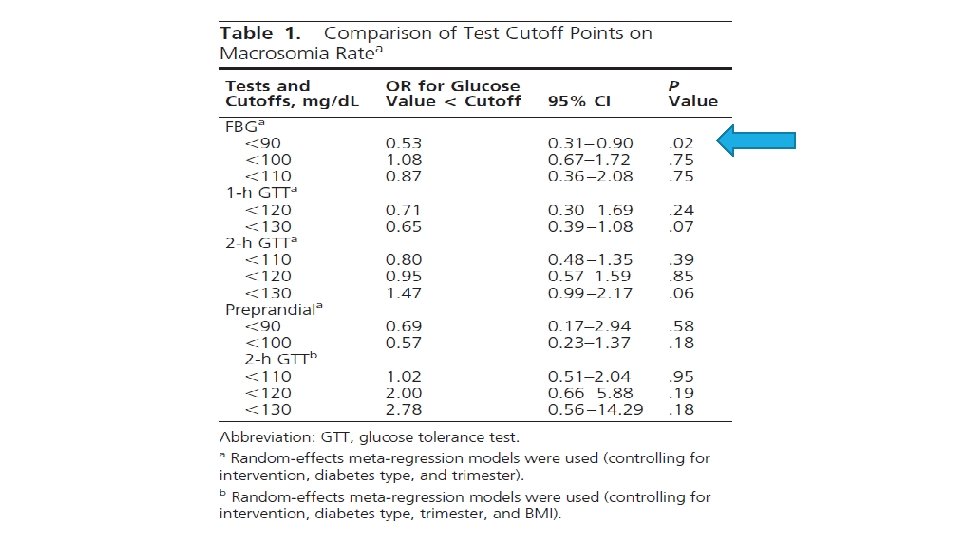

MAIN FINDINGS Evidence showed that FBG 90 mg/d. L is associated with reduction in the risk of macrosomia, neonatal hypoglycemia, and maternal preeclampsia in GDM during the third trimester (Low quality evidence) For women with T 1 DM or T 2 DM before pregnancy, the evidence was inconclusive Data regarding pre- or postprandial blood glucose targets were sparse, and the quality of such evidence is very low

MAIN FINDINGS. . Whether these targets can be extrapolated to women with diabetes before pregnancy or whether targets above or below this threshold offer a better benefit/ risk balance remains unclear.

MAJOR GAP! There are no well-conducted large RCTs of women with GDM or with diabetes before pregnancy comparing different glucose targets and their impact on fetal-maternal benefits and harms of importance to patients.

Thirty four studies enrolling 9433 women were included(15 RCT, 18 Cohort, 1 case control) Twenty two studies of GDM, 5 of T 1 DM, and 1 of T 2 DM; and 6 studies evaluated mixed types of diabetes The studies had moderate to high risk of bias due to evidence of reporting bias and insufficient adjustment for important covariates, particularly maternal body mass index. J Clin Endocrinol Metab 98: 4319– 4324, 2013













- Slides: 108