Georgia High School Graduation Test The Science Test










































- Slides: 60

Georgia High School Graduation Test The Science Test

What content is covered on the test? The science test is comprised of five domains: Cells & Heredity (25%) Ecology (17%) Structure and Properties of matter (26%) Energy Transformations (16%) Forces, Waves, & Electricity (16%)

Biology Review

What is the Cell Theory? • All organisms are made up of one or more cells. • The cell is the basic unit of organization of all organisms. • All cells come from other cells all ready in existence.

What are the types of cells? Eukaryotic cells • true nucleus and organelles • plants, animals, protists, and fungi are eukaryotes Prokaryotic cells • lack a nucleus and membrane bound organelles • bacteria

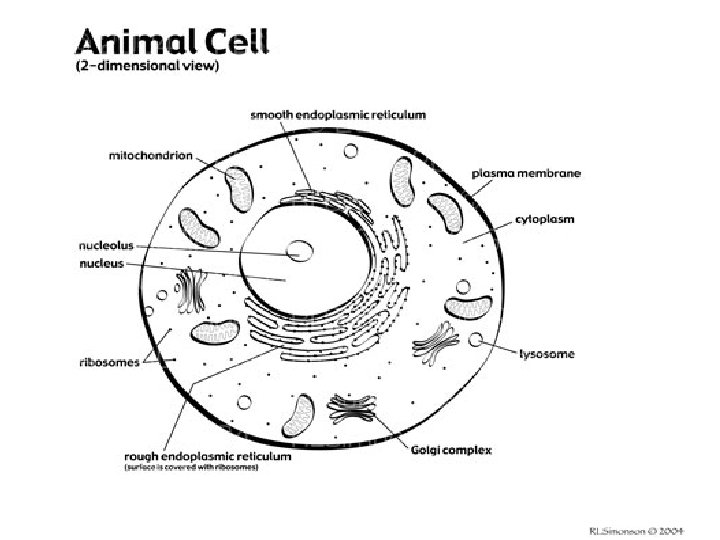
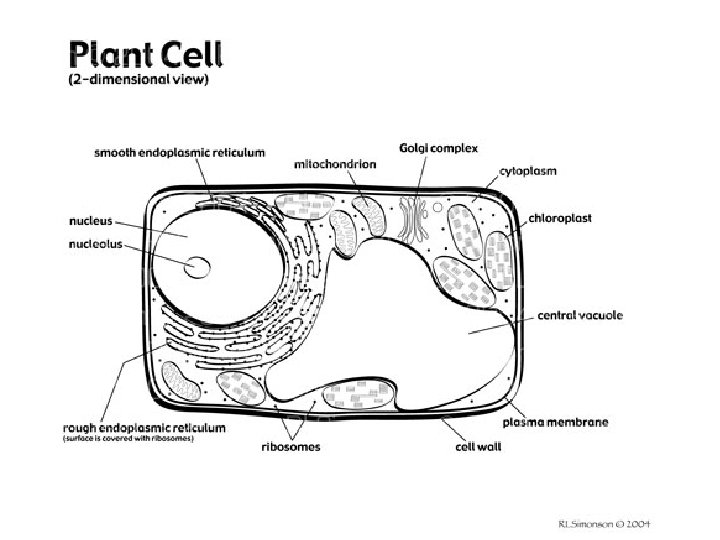
What are the parts of cells? • Cell membrane -passage of materials into and out of the cell • Nucleus - controls cell functions; DNA • Nucleolus – produces ribosomes • Mitochondria- cell energy • Ribosome- protein synthesis • Vacuole- cell storage • Lysosome- cell digestion

What are the parts of cells? (continued) • Endoplasmic reticulum- chemical synthesis • Golgi apparatus- packages proteins for secretion • Cell wall (plants only)- rigid outer wall; nonliving • Chloroplasts (plants only)- photosynthesis • Centrioles (animals only) - cell division

The Cell


How are cells organized? • Cell • Tissue – group of cells functioning together. • Organ – group of tissues functioning together. • Organ System – group of organs functioning together. • Organism – group of organ systems functioning together.

What is homeostasis? • Homeostasis – Steady State – Self-adjusting mechanism that helps to maintain your internal environment

How do cells transport materials? • Diffusion- movement of materials form a higher concentration to a lower concentration • Osmosis- diffusion of water through a Membrane

How do cells transport materials? • Passive transport is the movement of materials that does not require energy. – Diffusion, osmosis, facilitated diffusion • Active Transport requires the use of energy. – pumps

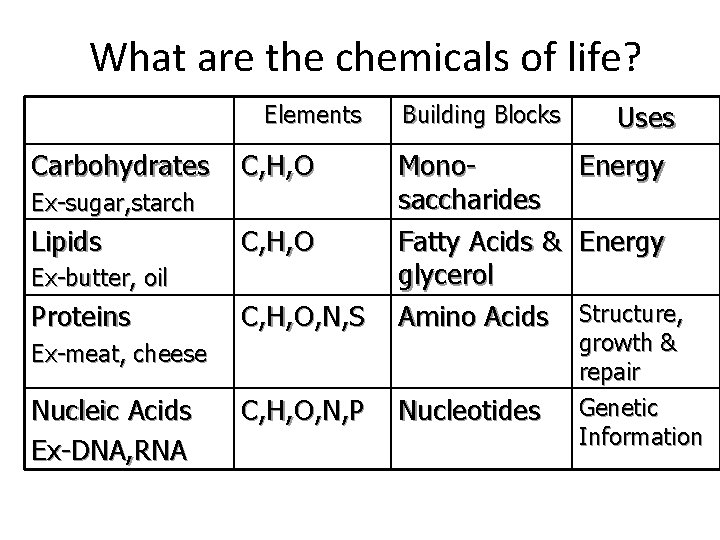
What are the chemicals of life? Elements Carbohydrates C, H, O, N, S Monosaccharides Fatty Acids & glycerol Amino Acids C, H, O, N, P Nucleotides Ex-sugar, starch Lipids C, H, O Ex-butter, oil Proteins Building Blocks Ex-meat, cheese Nucleic Acids Ex-DNA, RNA Uses Energy Structure, growth & repair Genetic Information

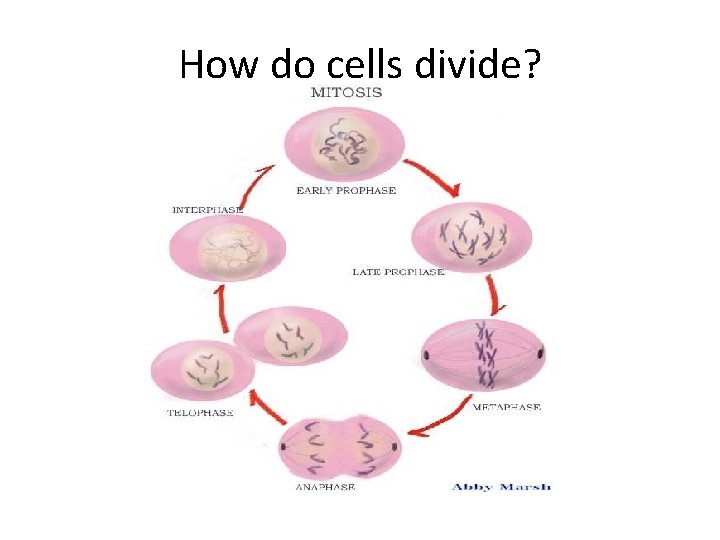
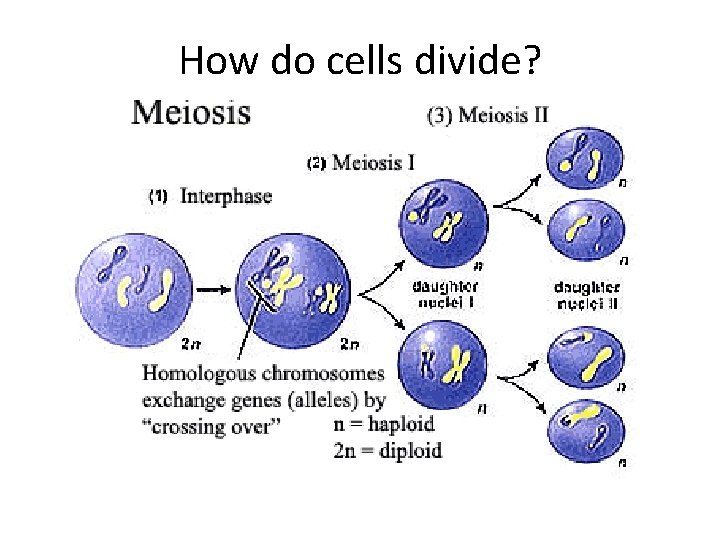
How do cells divide?

How do cells divide?

Where Does A Cell Get Energy? • In cellular respiration, glucose is broken down to a form the cell can use. • Extra energy is stored in an ATP molecule (adenosine triphosphate).

What is photosynthesis?

What is photosynthesis? • 6 CO 2 + 6 H 2 O C 6 H 12 O 6 + 6 O 2 • Occurs in chloroplasts • Two parts: – Light reaction – Calvin cycle (dark reaction)

What is Respiration? • The process of breaking down food molecules to release energy. • Occurs in the mitochondria. • Two types: – Aerobic – requires oxygen. – Anaerobic – requires an absence of oxygen. – C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + energy

What is DNA? • DNA contains the blueprint for life. • Nucleotides are the building blocks. • Contains a deoxyribose sugar, phosphate group, and • 4 bases: – Adenine - Thymine – Cytosine - Guanine

How are DNA & RNA Different? DNA • • Deoxyribose Thymine Double stranded Found only in nucleus RNA • Ribose • Uracil • Single stranded

How does DNA control Protein Synthesis? • Transcription – m. RNA copies the message from DNA in the nucleus • Translation – Forms a protein by translating the message on RNA into an amino acid sequence

What are the levels of organization of ecology? Population – group of 1 species living in the same place Communities - group of interacting populations Ecosystem – the community and its environment Biome – group of ecosystems with the same communities Biosphere – the circle of life

Individual Population Ecosystem

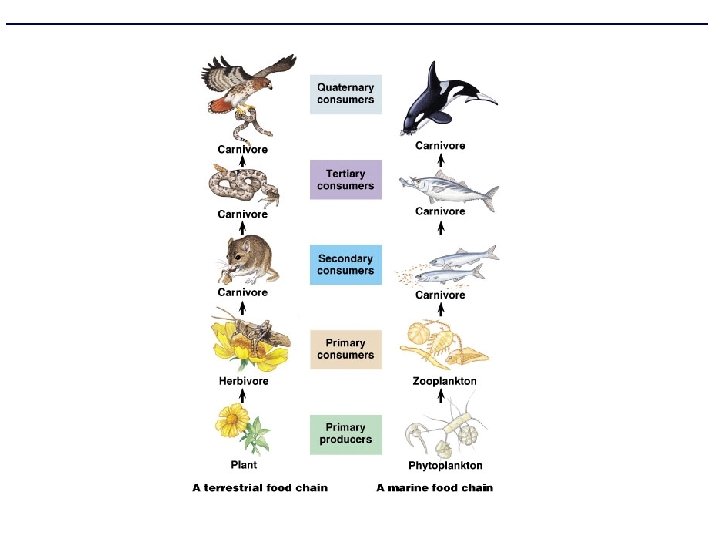
How does matter & energy flow in an ecosystem? • Food Chains – one path of energy flow • Food Webs – complex model that expresses all the possible feeding relationships in a community


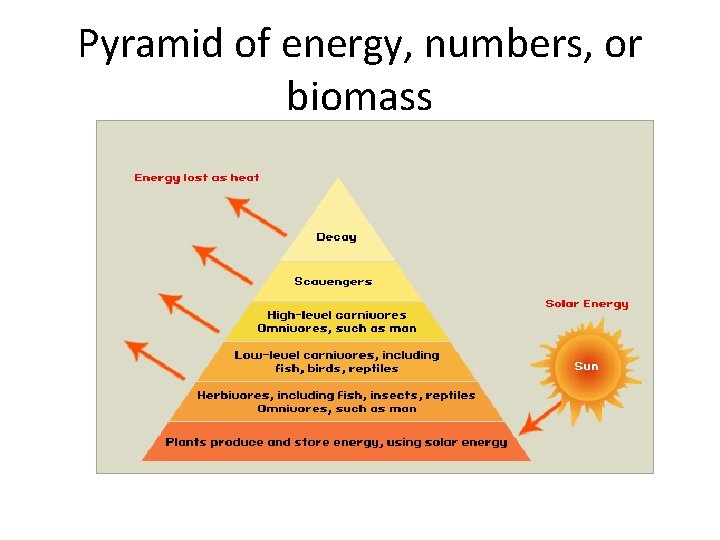
Pyramid of energy, numbers, or biomass

Ga. HSGT Chemistry Review

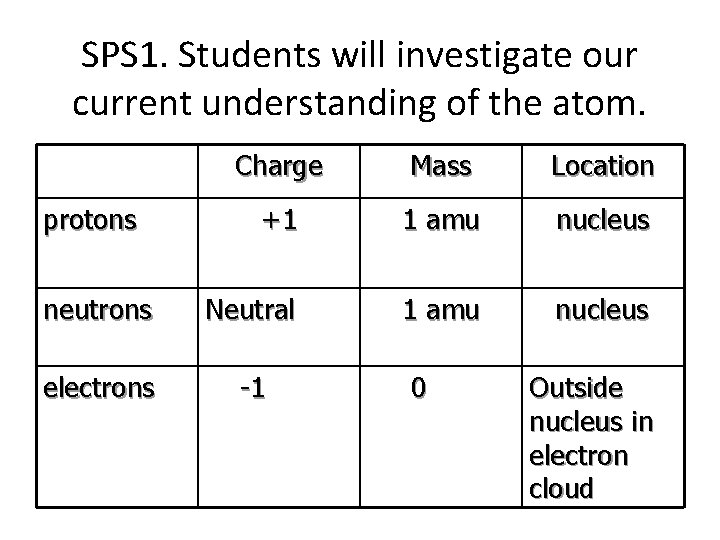
SPS 1. Students will investigate our current understanding of the atom. Charge Mass Location +1 1 amu nucleus neutrons Neutral 1 amu nucleus electrons -1 protons 0 Outside nucleus in electron cloud

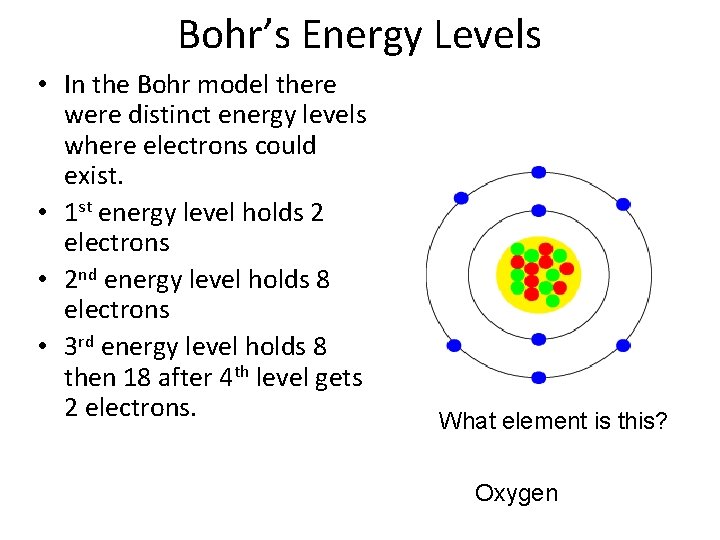
Bohr’s Energy Levels • In the Bohr model there were distinct energy levels where electrons could exist. • 1 st energy level holds 2 electrons • 2 nd energy level holds 8 electrons • 3 rd energy level holds 8 then 18 after 4 th level gets 2 electrons. What element is this? Oxygen

Modern View of the Atom § The atom is mostly empty space. § Two regions • Nucleus- protons and neutrons. • Electron cloudregion where you might find an electron.

Atomic Number The number of protons in an atom of a particular element is its atomic number. Each element has a different atomic number.

Atomic Mass v. Protons and neutrons are responsible for most of the atomic mass of an atom. v. The number of protons and neutrons in an atom are the mass number of that atom.

Remember to use the Periodic Table provided on the Test.

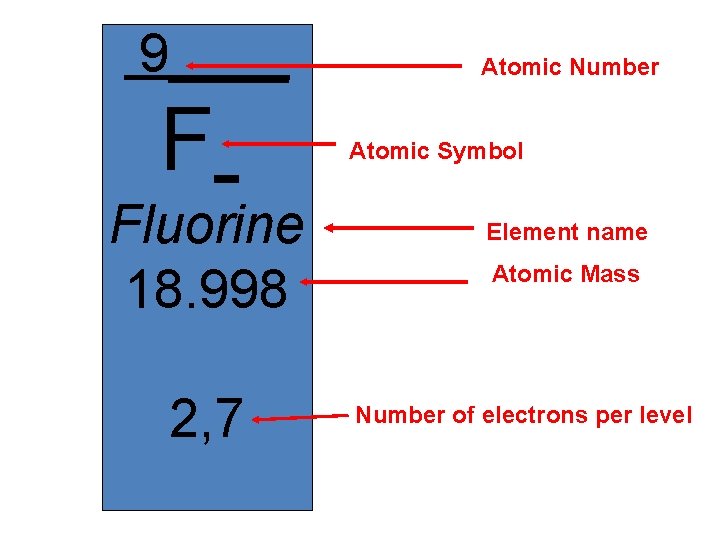
9____ F Fluorine 18. 998 2, 7 Atomic Number Atomic Symbol Element name Atomic Mass Number of electrons per level

Isotopes For any given element, all nuclei have the same number of protons, but the number of neutrons may vary. These atoms of the same element with different atomic masses are called isotopes.

SPS 6. Students will investigate the properties of solutions. • A solution is a homogeneous mixture composed of a solute and a solvent. – A solute is the dissolved substance in a solution. There is less of it. salt in salt water sugar in soda carbon dioxide in soda – A solvent is the dissolving medium. You have more of it. water in salt water in sodas

Dilute vs Concentrated

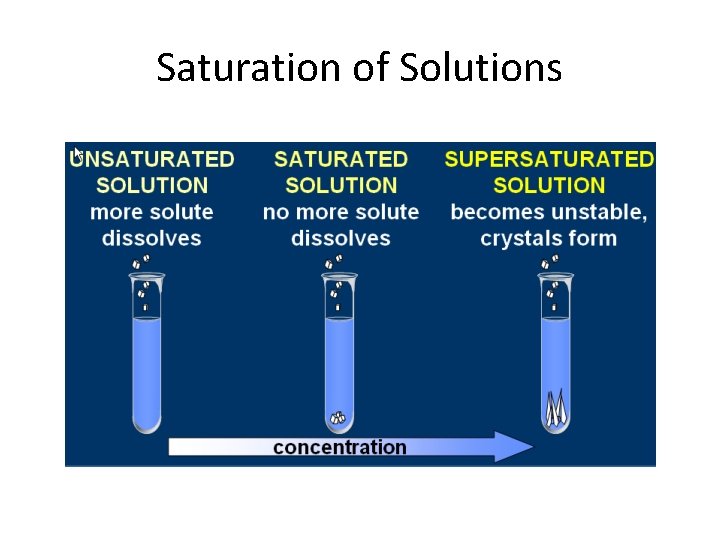
Saturation of Solutions

Conductivity of Solutions • An electrolyte is a substance whose water solution conducts electricity. – – ionic compounds such as Na. Cl (sodium chloride) acids and bases such as HCl (hydrochloric acid) and Na. OH (sodium hydroxide)

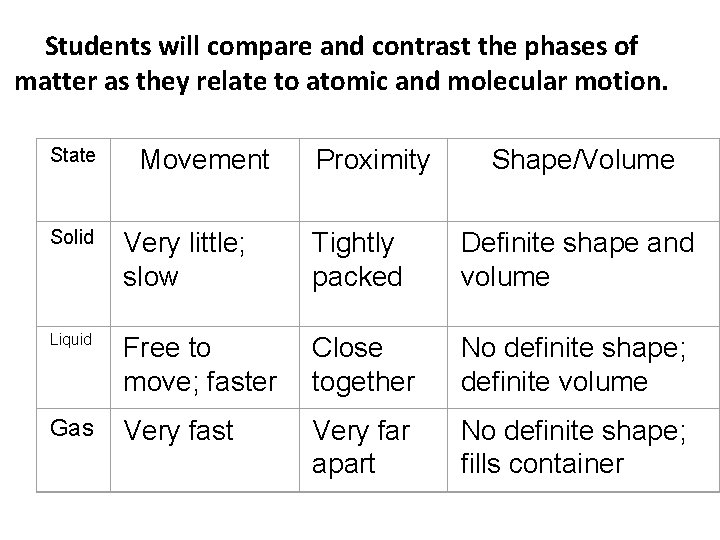
Students will compare and contrast the phases of matter as they relate to atomic and molecular motion. State Movement Proximity Shape/Volume Solid Very little; slow Tightly packed Definite shape and volume Liquid Free to move; faster Close together No definite shape; definite volume Gas Very fast Very far apart No definite shape; fills container

Solid Liquid Gas

Plasma- the 4 th state of matter • Plasmas consist of freely moving charged particles, i. e. , electrons and ions. • Lightning creates a jagged column of plasma. • When an electric current is passed through neon gas, it produces both plasma and light.

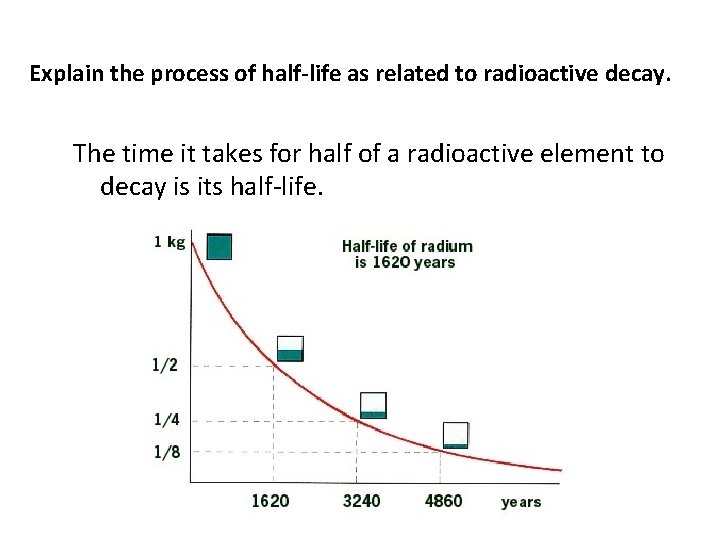
Explain the process of half-life as related to radioactive decay. The time it takes for half of a radioactive element to decay is its half-life.

Physics Review

Velocity • Distance covered in a certain amount of time • Velocity = distance time

Acceleration • Rate at which velocity changes • Acceleration = final velocity – initial velocity time

Force • A push or pull • Measured in Newtons • An object at rest and an object moving at a constant velocity is being acted upon by a net force of zero

Newton’s 1 st Law of Motion An object in motion will stay in motion and an object at rest will stay at rest unless acted upon by an unbalanced force Inertia

Acceleration due to Gravity • On Earth, all objects fall with a constant acceleration of 9. 80 m/s 2 in the absence of air resistance. • In other words, a falling object’s velocity increases by 9. 8 m/s each second it falls!

Mass vs. Weight • Mass—the amount of matter in an object • Weight—the force on a body due to the gravitational attraction of another body • Weight changes based on location, mass does not!

Work and Power • Work = Force x Distance – Measured in Newton-meters or Joules • Power = Work Time – Measured in Watts

Waves A disturbance that transmits energy through a medium or empty space Frequency—the number of events (waves, vibrations, oscillations) per time

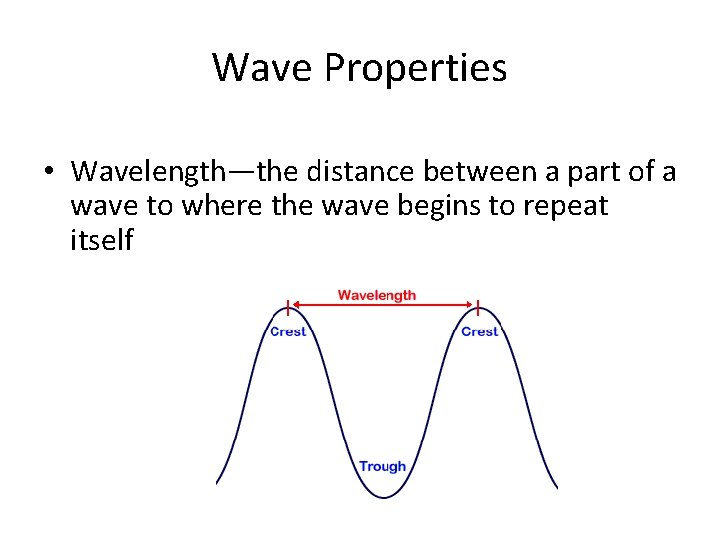
Wave Properties • Wavelength—the distance between a part of a wave to where the wave begins to repeat itself

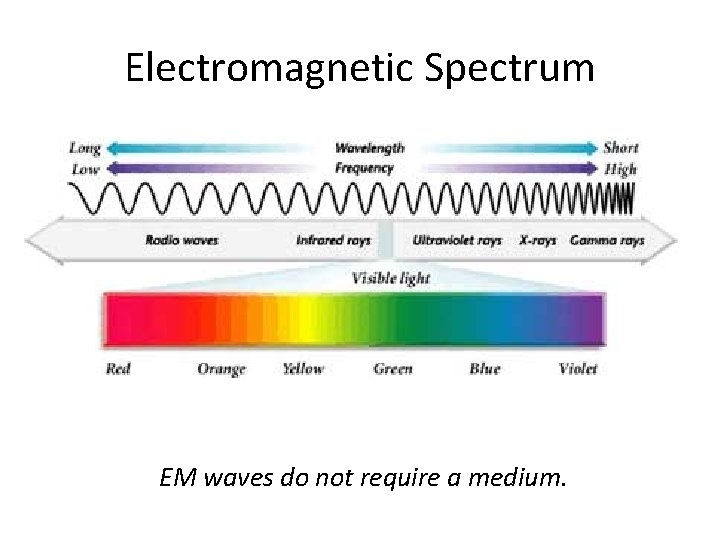
Electromagnetic Spectrum EM waves do not require a medium.

Mechanical Waves • Require a medium • Sound is an example of a mechanical wave

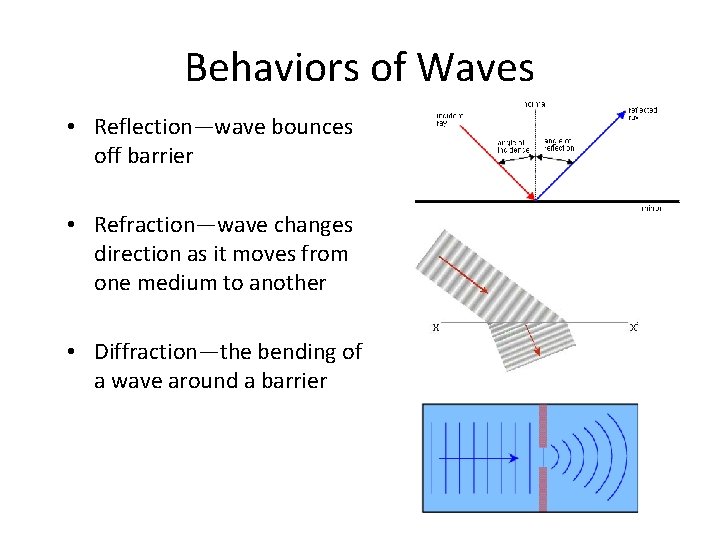
Behaviors of Waves • Reflection—wave bounces off barrier • Refraction—wave changes direction as it moves from one medium to another • Diffraction—the bending of a wave around a barrier

To increase the strength of an electromagnet by a. Increase the number of coils b. Increase the number of batteries

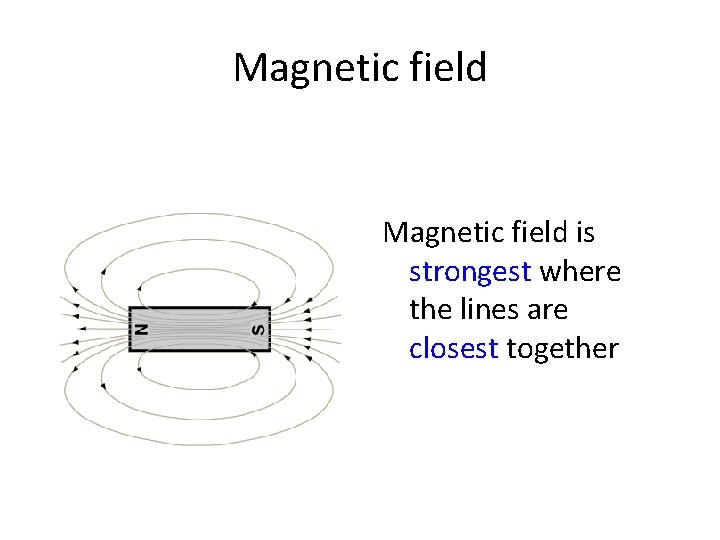
Magnetic field is strongest where the lines are closest together