Geochemical Cycles Geochemical Cycles Carbon Cycle Early atmosphere

Geochemical Cycles

Geochemical Cycles Carbon Cycle § Early atmosphere of Earth 95% CO 2. Photosynthetic plants removed some of the CO 2 and added O 2. Today’s atmosphere is 0. 04% CO 2! Carbon is found in the § Reactions of photosynthesis and cellular atmosphere primarily as CO 2 respiration couldn’t take place without carbon. These two reactions form a continuous cycle. § Two important sources of Carbon are the ocean (since CO 2 dissolves easily in H 20)Respiration and rocks : Photosynthesis : Cellular (such as coal, oreofand. Organisms limestonetake formed fromand in Plants taking CO 2 out that sugar dead organisms) the atmosphere and the process of burning energy using it to produce sugar. release CO 2 back into the atmosphere.
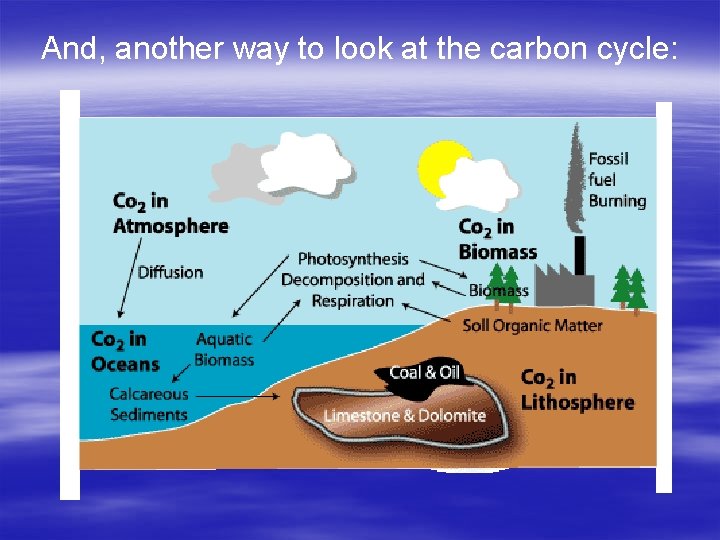
And, another way to look at the carbon cycle:

Humans affect the Carbon Cycle § Burning of fossil fuels, (oil, coal and natural gas). § Fossil fuels were formed very long ago and is “fixed”: essentially locked out of the carbon cycle. § By burning fossil fuels the carbon is released back into the cycle.

Humans affect the Carbon Cycle § We presently release more carbon into the air than can be reabsorbed by photosynthetic organisms, thereby we have a net INCREASE of carbon in the cycle. § This atmospheric carbon has a role to play in the warming of the atmosphere.

Geochemical Cycles § § Nitrogen Cycle Organisms require Nitrogen to form amino acids for the building of proteins. Lots of N 2 in our atmosphere Unfortunately, most organisms CANNOT use atmospheric nitrogen. Nitrogen-fixing bacteria CAN use N 2 from the atmosphere. Nitrogen-fixing bacteria convert atmospheric N 2 into ammonia (NH 4) which is a form of nitrogen that plants CAN use.

Nitrogen Cycle Continued … § Nitrogen-fixing bacteria live in the soil and in roots of legumes. § These bacteria also form nitrites (NO 2) and nitrates (NO 3); which are compounds containing N and O. § Nitrate is the most common source of N for plants. § Animals get N from the proteins they eat. § Decomposers return N to the soil in the form of ammonia and the cycle repeats. § So, oftentimes, the nitrogen cycle does not require the N to be returned to atmospheric form!

Nitrogen Cycle Summary § All living organisms require nitrogen – to form amino acids to build proteins. § Proteins are important for locomotion, reproduction, defense, and structure. § Nitrogen makes up 78% of atmosphere as N 2 § Nitrogen-fixing bacteria are very important N 2 needs to be “fixed” before it can be used by most living things.
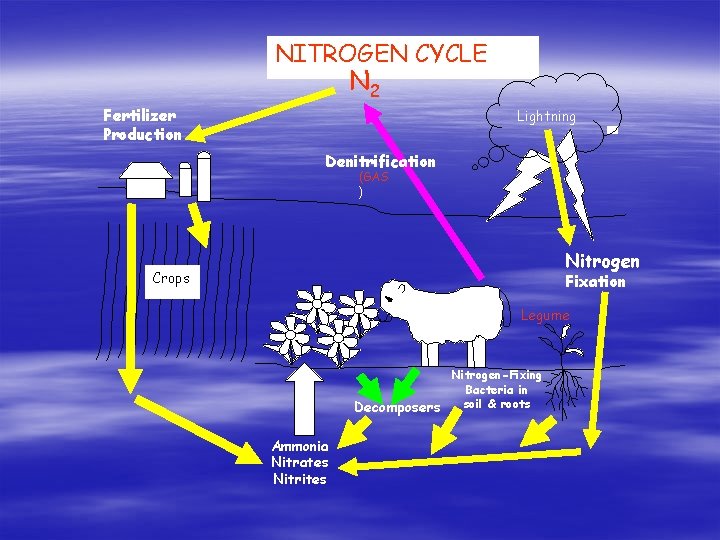
NITROGEN CYCLE N 2 Fertilizer Production Lightning Denitrification (GAS ) Nitrogen Crops Fixation Sheep Legume Nitrogen-Fixing Bacteria in soil & roots Decomposers Ammonia Nitrates Nitrites
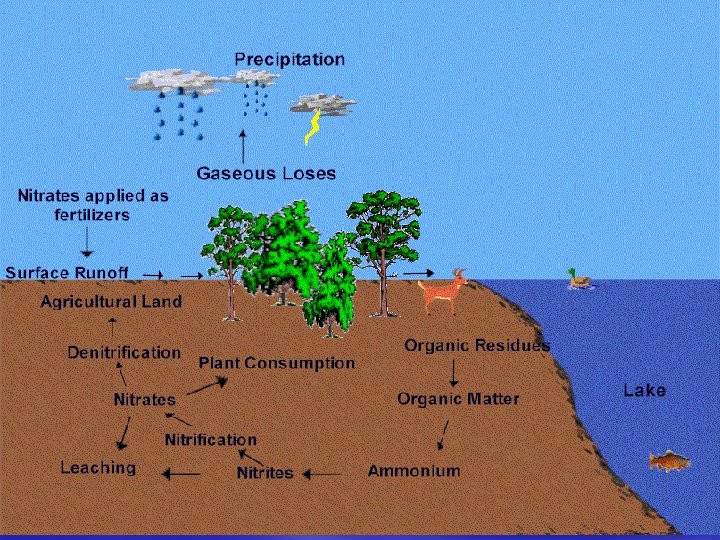

Humans affect the Nitrogen Cycle § From the production and use of nitrogen fertilizers to the burning of fossil fuels in automobiles, power plants, and industries, humans impact this cycle. § Nitrogen is essential to living organisms and its availability plays a crucial role in the world's ecosystems. § Excessive nitrogen additions can pollute ecosystems

Humans affect the Nitrogen Cycle § Increased global concentrations of nitrous oxide (N 2 O), a potent greenhouse gas, in the atmosphere § Increased concentrations of nitric oxide, (NO) that drive the formation of smog along with N 2 O § Losses of soil nutrients such as calcium and potassium that are essential for long-term soil fertility

Humans affect the Nitrogen Cycle § Acidification of soils and of the waters of streams and lakes § Greatly increased transport of nitrogen by rivers into estuaries and coastal waters where it is a major pollutant.
- Slides: 13