Genotoxicity Basic Aspects and Most Commonly Worldwide Employed

Genotoxicity Basic Aspects and Most Commonly Worldwide Employed and Validated In Vitro Assays Leon F. Stankowski, Jr. , Ph. D Consultant, Genetic Toxicology

Agenda • OECD Guidelines • In Vitro Assays • Mutation • Chromosome Aberration • Micronucleus

Some Terminology Mutagen • agent that induces a change in the DNA Clastogen • agent that induces chromosome breaks (structural aberration) Aneugen • agent that induces a change in chromosome number (numerical aberration)

OECD Guidelines Organisation for Economic Cooperation and Development • 471 Bacterial Reverse Mutation Test • 473 In Vitro Mammalian Chromosome Aberration Test • 474 Mammalian Erythrocyte Micronucleus Test • 475 Mammalian Bone Marrow Chromosomal Aberration Test • 476 In Vitro Mammalian Cell Gene Mutation Tests using the Hprt and xprt genes • 487 In Vitro Mammalian Cell Micronucleus Test • 488 Transgenic Rodent Somatic and Germ Cell Gene Mutation Assays • 489 In Vivo Mammalian Alkaline Comet Assay • 490 In Vitro Mammalian Cell Gene Mutation Test Using the Thymidine Kinase Gene www. oecd. org

OECD Guidelines Organisation for Economic Cooperation and Development • 471 Bacterial Reverse Mutation Test • 473 In Vitro Mammalian Chromosome Aberration Test • 474 Mammalian Erythrocyte Micronucleus Test • 475 Mammalian Bone Marrow Chromosomal Aberration Test • 476 In Vitro Mammalian Cell Gene Mutation Tests using the Hprt and xprt genes • 487 In Vitro Mammalian Cell Micronucleus Test • 488 Transgenic Rodent Somatic and Germ Cell Gene Mutation Assays • 489 In Vivo Mammalian Alkaline Comet Assay • 490 In Vitro Mammalian Cell Gene Mutation Test Using the Thymidine Kinase Gene www. oecd. org

OECD Guidelines • Apply to all testing situations • Some special modifications for human pharmaceuticals • ICH - International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use • ICH S 2(R 1) www. ich. org

Mutation Assays • 471 Bacterial Reverse Mutation Test • S. typhimurium and E. coli tester strains • Reverse mutation to his or trp independence • Basepair (bp) substitution – change in one base for another • Frameshift (fs) mutation – insertion or deletion of one or a few a bases

Mutation Assays • 471 Bacterial Reverse Mutation Test • Requires very specific change at G: C or A: T sites to revert to his or trp independence • Five tester strains required • • TA 1535 and TA 100 (bp) and TA 98 (fs) and TA 1537 or TA 97 a (fs) and WP 2 uvr. A or WP 2 uvr. A (p. KM 101) or TA 102 (bp) • Engineered • deficient in DNA excision repair • error-prone DNA repair function • leaky cell walls
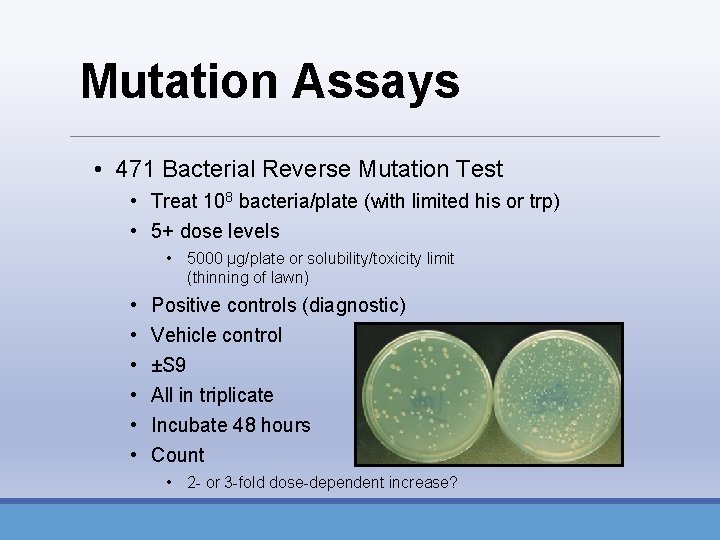
Mutation Assays • 471 Bacterial Reverse Mutation Test • Treat 108 bacteria/plate (with limited his or trp) • 5+ dose levels • 5000 µg/plate or solubility/toxicity limit (thinning of lawn) • • • Positive controls (diagnostic) Vehicle control ±S 9 All in triplicate Incubate 48 hours Count • 2 - or 3 -fold dose-dependent increase?
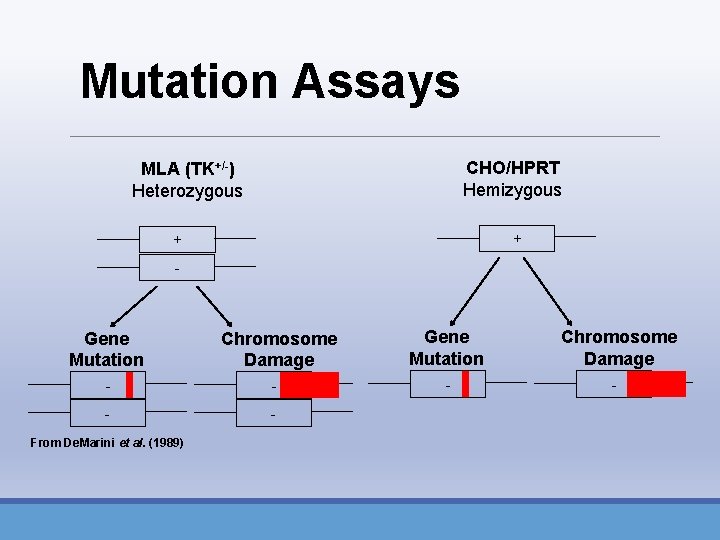
Mutation Assays CHO/HPRT Hemizygous MLA (TK+/-) Heterozygous + + - Gene Mutation Chromosome Damage - - - From De. Marini et al. (1989)
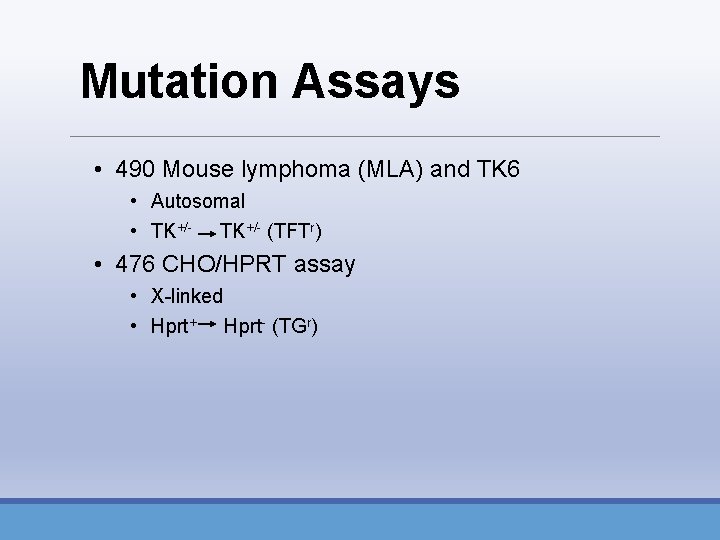
Mutation Assays • 490 Mouse lymphoma (MLA) and TK 6 • Autosomal • TK+/- (TFTr) • 476 CHO/HPRT assay • X-linked • Hprt+ Hprt- (TGr)

Mutation Assays • HPRT and TK • Treat 2 - 3 x 106 cells • 4+ dose levels • 2000 µg/m. L or 10 m. M or solubility/toxicity limit (10 – 20% RS or RTG) • Positive and vehicle controls • ±S 9 • All in duplicate
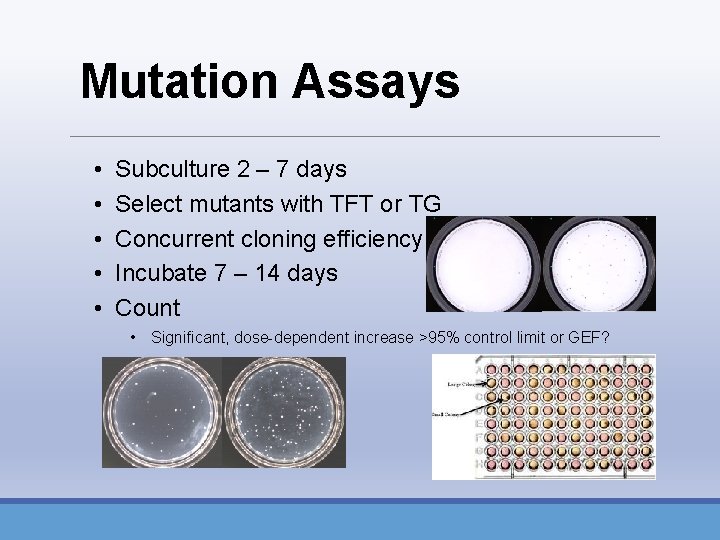
Mutation Assays • • • Subculture 2 – 7 days Select mutants with TFT or TG Concurrent cloning efficiency Incubate 7 – 14 days Count • Significant, dose-dependent increase >95% control limit or GEF?

Cytogenetics Assays • 473 In Vitro Mammalian Chromosomal Aberration Test • HPBL, RPBL, CHO, CHL cells • Structural aberrations • breaks/rearrangements (clastogens) • Numerical aberrations • polyploidy/endoreduplication (aneugens)

Cytogenetics Assays • 473 In Vitro Mammalian Chromosomal Aberration Test • Treat exponentially growing cells • stimulate primary cultures with PHA • 3+ dose levels • 2000 µg/m. L or 10 m. M or solubility/toxicity limit (40 – 50% RPD, RICC, MI) • • Positive and vehicle controls Short treatment ±S 9 Extended treatment –S 9 only Single or duplicate
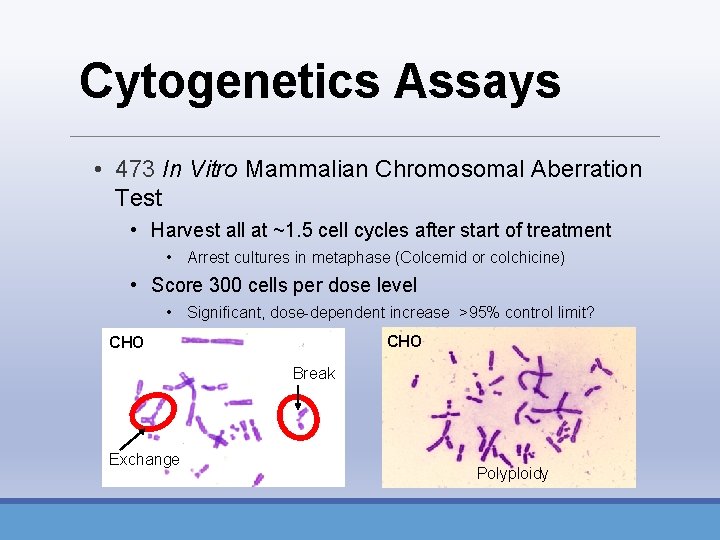
Cytogenetics Assays • 473 In Vitro Mammalian Chromosomal Aberration Test • Harvest all at ~1. 5 cell cycles after start of treatment • Arrest cultures in metaphase (Colcemid or colchicine) • Score 300 cells per dose level • Significant, dose-dependent increase >95% control limit? CHO Break Exchange Polyploidy

Cytogenetics Assays • 473 In Vitro Mammalian Chromosomal Aberration Test HPBL dicentric HPBL break

Cytogenetics Assays • 487 In Vitro Mammalian Cell Micronucleus Test • HPBL, RPBL, CHO, CHL, TK 6 cells • Structural aberrations • breaks/rearrangements (clastogens) • Numerical aberrations • polyploidy/endoreduplication (aneugens)
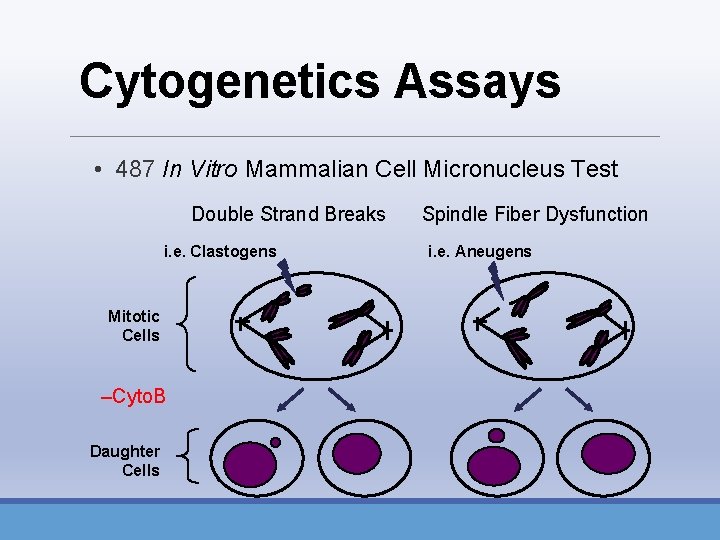
Cytogenetics Assays • 487 In Vitro Mammalian Cell Micronucleus Test Double Strand Breaks i. e. Clastogens Mitotic Cells –Cyto. B Daughter Cells Spindle Fiber Dysfunction i. e. Aneugens
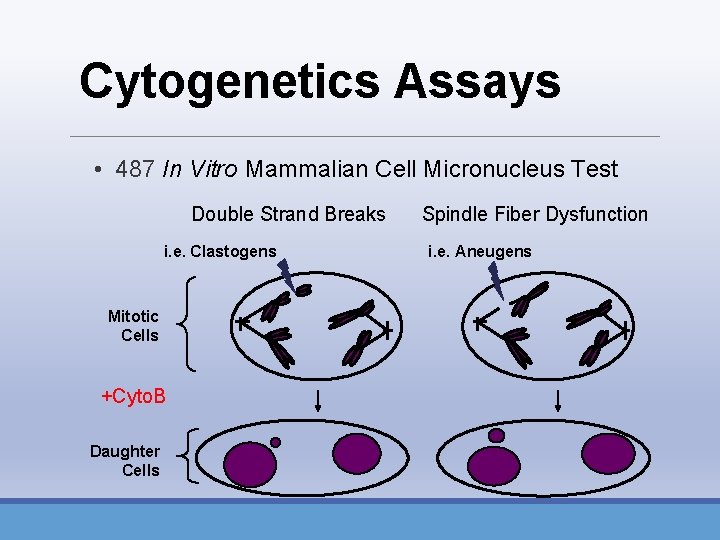
Cytogenetics Assays • 487 In Vitro Mammalian Cell Micronucleus Test Double Strand Breaks i. e. Clastogens Mitotic Cells +Cyto. B Daughter Cells Spindle Fiber Dysfunction i. e. Aneugens

Cytogenetics Assays • 487 In Vitro Mammalian Cell Micronucleus Test • Treat exponentially growing cells • stimulate primary cultures with PHA • 3+ dose levels • 2000 µg/m. L or 10 m. M or solubility/toxicity limit (50 – 60% RPD or RICC for cell lines, or CPBI or RI in primary cells and when using Cyto. B) • • Positive and vehicle controls Short treatment ±S 9 Extended treatment –S 9 only Single or duplicate
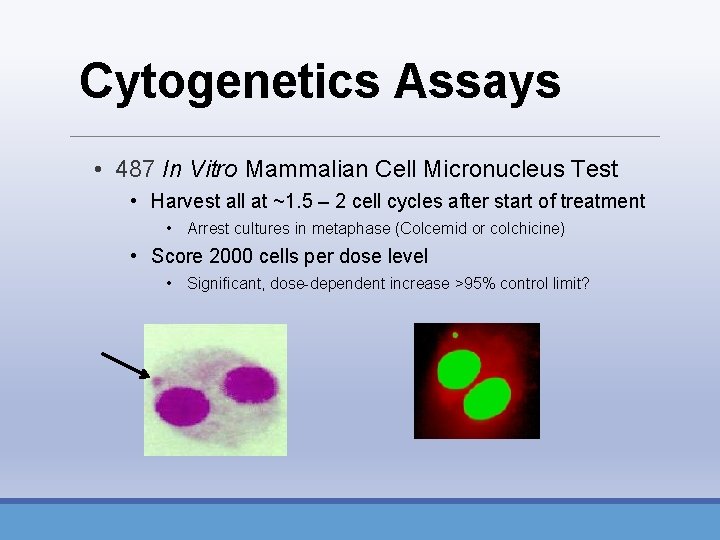
Cytogenetics Assays • 487 In Vitro Mammalian Cell Micronucleus Test • Harvest all at ~1. 5 – 2 cell cycles after start of treatment • Arrest cultures in metaphase (Colcemid or colchicine) • Score 2000 cells per dose level • Significant, dose-dependent increase >95% control limit?

Cytogenetics Assays • 487 In Vitro Mammalian Cell Micronucleus Test • Mechanism of Action • Fluorescent In Situ Hybridization (FISH) • Antikinetochore staining (CREST)
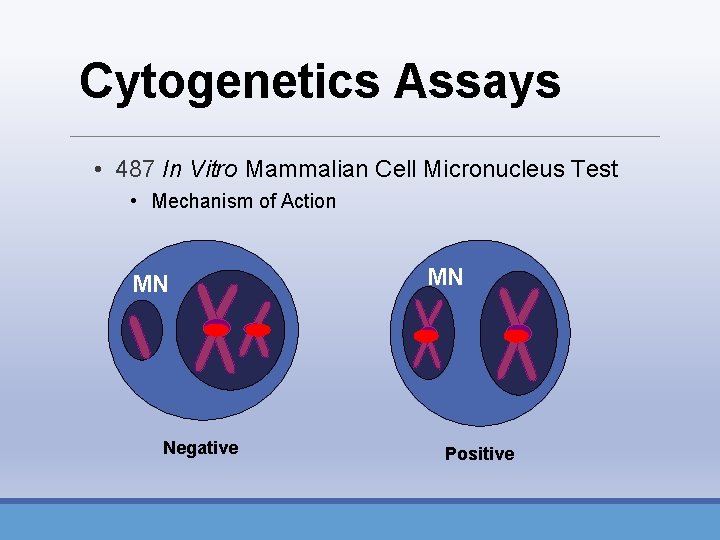
Cytogenetics Assays • 487 In Vitro Mammalian Cell Micronucleus Test • Mechanism of Action MN Negative MN Positive
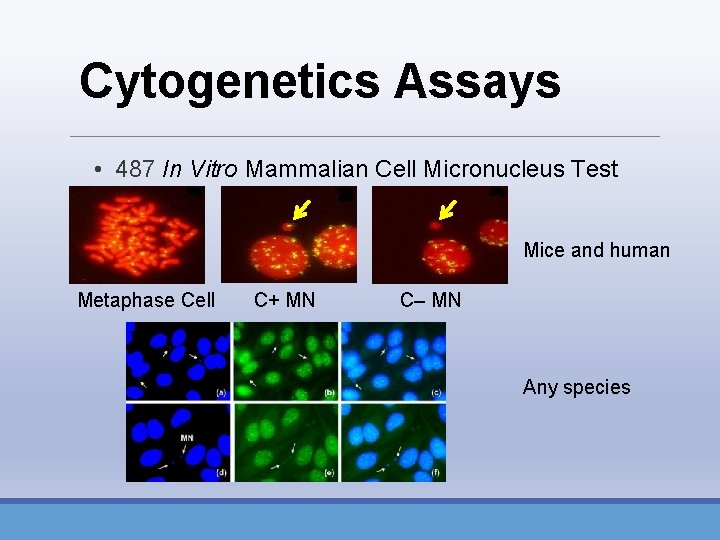
Cytogenetics Assays • 487 In Vitro Mammalian Cell Micronucleus Test Mice and human Metaphase Cell C+ MN C– MN Any species

Thank You! Questions?

Thank You! Questions? leon. stankowski@wilresearch. com (31 August 2015)
- Slides: 27