Genomewide RNAi screening in Caenorhabditis elegans Ravi S

Genome-wide RNAi screening in Caenorhabditis elegans Ravi S. Kamath & Julie Ahringer
What is RNAi? A cellular mechanism to regulate the expression of genes, mutant gene products and the replication of viruses

(some) History of RNAi 19 84 19 90 19 95 19 20 120 98 0 03 • 1984: Stout & Caskey show antisense RNA can be used to silence gene expression in Mammalian tissue cultures • 1990: Fire & Moerman show antisense RNA can disrupt myofilament protein encoding genes • 1995: Guo & Kemphues accidentally discover that sense RNA can is as effective as antisense RNA in gene silencing • 1998: Mello & Fire illustrate that ds. RNA is the agent that leads to potent and specific genetic interference…not ss. RNA • 2001: Fraser et al. complete RNAi screen of 90% of chromosome I • 2003: Ahringer & Kamath unveil the results of a genome-wide RNAi screen

How does this stuff work? The cool movie revisited
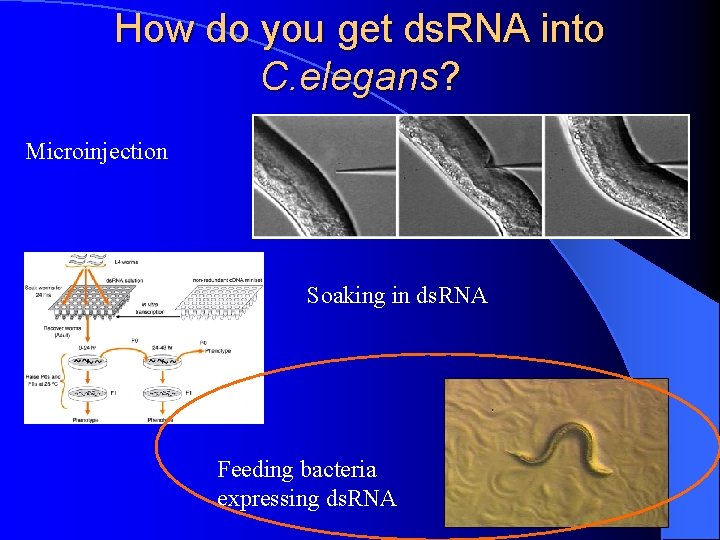
How do you get ds. RNA into C. elegans? Microinjection Soaking in ds. RNA Feeding bacteria expressing ds. RNA

Advantages of feeding for highthroughput RNAi screening • Fast • Cheap • Less labor intensive Disadvantage: Lots of molecular biology work to clone a fragment of a gene into a feeding vector and then transform it into an appropriate bacterial strain

Aim of this paper Ø provide research community with a rapid screening tool Ø describe methods for bacterial feeding library construction Ø Identify new gene functions

Methods Cloning: Conveniently, Genepairs primers commercially available - optimized for max. overlap with coding region - amplify 1000 -1500 bp fragments at 5’ end of gene Problem: How do you rapidly get PCR products into vector for high-throughput analysis? How do you screen >19, 000 genes rapidly and efficiently for RNAi phenotypes?

Construction of feeding library: Making the construct Need ds. RNA to yield effective RNAi phenotypes: Used L 4440 (p. PD 129. 36) MCS

Construction of feeding library: Making the construct • Cut L 4440 once with Eco. RV and religated • Cut L 4440 with Eco. RV to create blunt ends for 3’ dd. TTP addition by Td. T • Recircularized to eliminate non-tailed products • Ligated PCR A-tailed PCR products directly into MCS of vector

Construction of feeding library: Suitable Bacterial strain Transformed RNAi constructs into HT 115(DE 3) • RNase III-deficient strain • Tetracycline resistant • Increased transformation efficiency using TSS
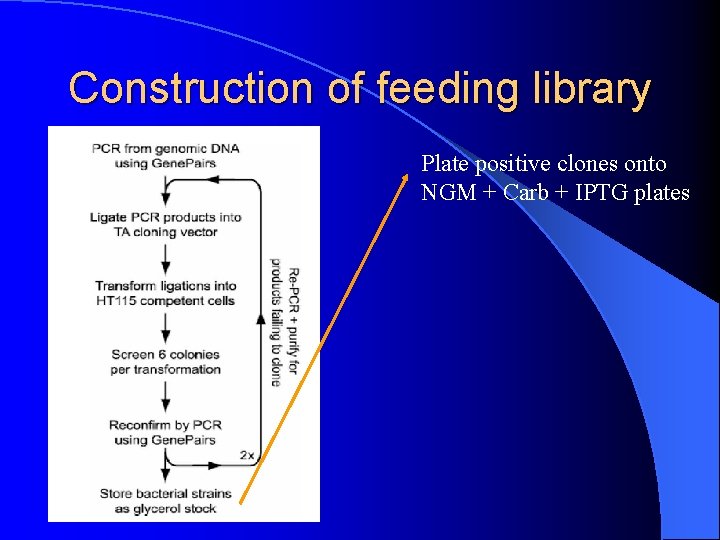
Construction of feeding library Plate positive clones onto NGM + Carb + IPTG plates

High-throughput phenotype screening 7 -10 worms Clone 3 adult worms to 3 separate wells

High-throughput phenotype screening: Timeline

High-throughput phenotype screening: Analysis of phenotypes

Interlude

Drawbacks Some genes hard to target 1. Genes whose protein product has a long ½ life 2. Nervous system genes difficult to target 3. Variability in phenotypes 4. Inconsistency between animals 5. Phenotypes can resemble hypomorph rather than amorph 6. Silencing of related genes 7. Genes with close homologs can often be abated in addition to target

How rapid is this screen? • Once the operation is a well-oiled machine you can screen 200 genes/day with 3 people • Can screen entire genome in 3 months • Most labor is in manipulating worms & scoring How could you speed up this assay?

Results of genome-wide screen & library construction 1. Identified novel gene functions for ~10% of the ~19000 genes screened using N 2 worms 2. Created a functional, rapid means to perform a large-scale RNAi screen 3. Now a mutant analysis tool is available to the whole worm community…. at a cost $$$

Follow-up Screen • Simmer et al. (2003) used rrf-3 , an RNAi-hypersensitive strain to re-assay the RNAi feeding library 1. Found additional loss-of-function phenotypes for 393 genes 2. In replicates of experiments, found consistent falsepositives
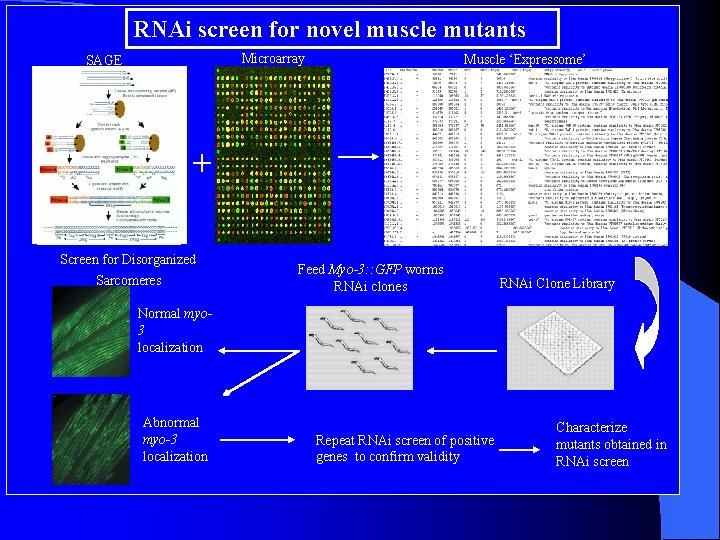
RNAi screen for novel muscle mutants Microarray SAGE Muscle ‘Expressome’ + Screen for Disorganized Sarcomeres Feed Myo-3: : GFP worms RNAi clones RNAi Clone Library Normal myo 3 localization Abnormal myo-3 localization Repeat RNAi screen of positive genes to confirm validity Characterize mutants obtained in RNAi screen

- Slides: 22