Genetic susceptibility to head and neck cancer and

Genetic susceptibility to head and neck cancer and to lung cancer Paolo Boffetta International Agency for Research on Cancer Lyon, France
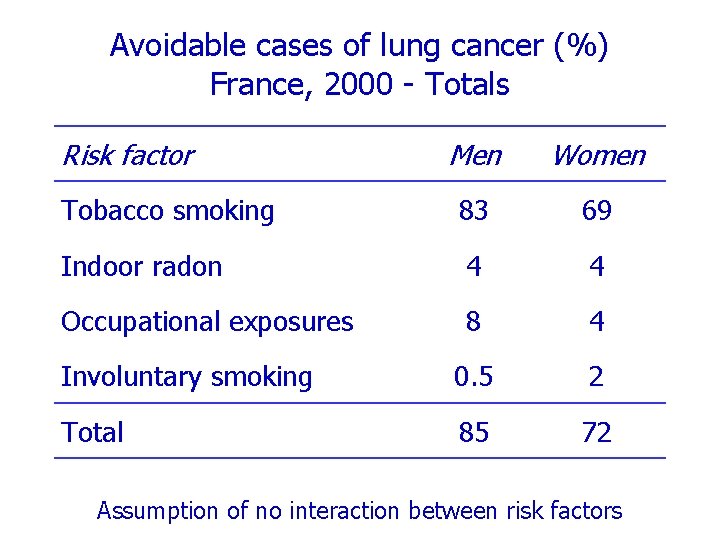
Avoidable cases of lung cancer (%) France, 2000 - Totals Risk factor Men Women Tobacco smoking 83 69 Indoor radon 4 4 Occupational exposures 8 4 Involuntary smoking 0. 5 2 Total 85 72 Assumption of no interaction between risk factors
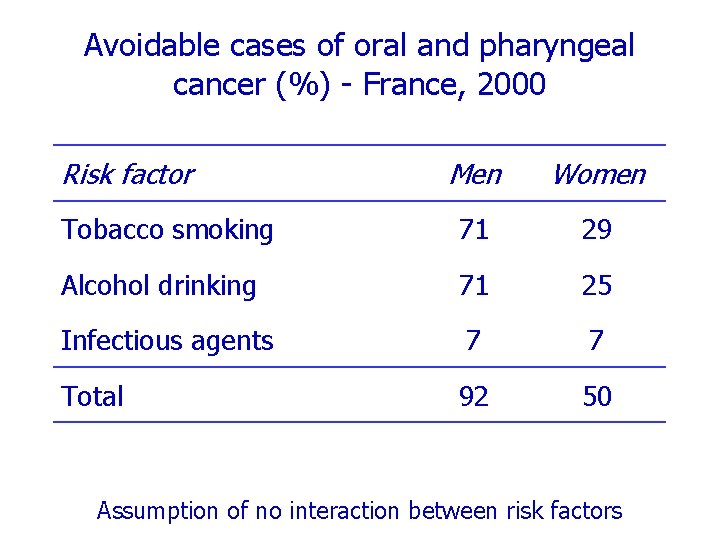
Avoidable cases of oral and pharyngeal cancer (%) - France, 2000 Risk factor Men Women Tobacco smoking 71 29 Alcohol drinking 71 25 Infectious agents 7 7 Total 92 50 Assumption of no interaction between risk factors

Avoidable cases of laryngeal cancer (%) France, 2000 Risk factor Men Women Tobacco smoking 76 65 Alcohol drinking 57 18 Occupational exposures 2 0. 2 Total 90 71 Assumption of no interaction between risk factors
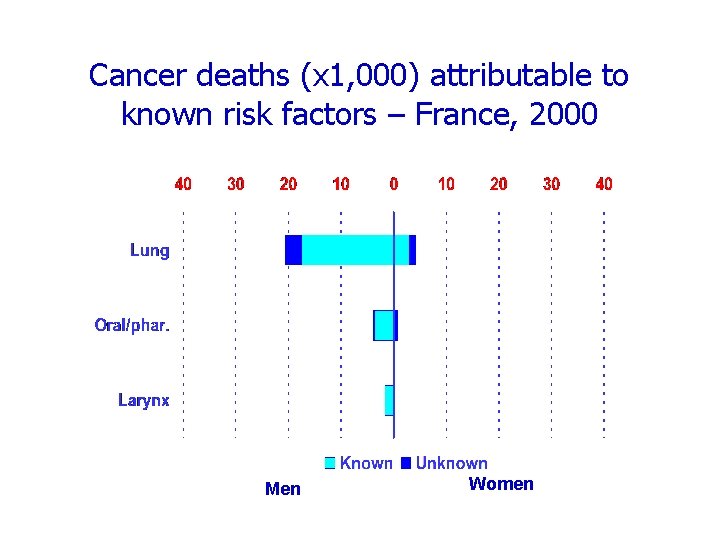
Cancer deaths (x 1, 000) attributable to known risk factors – France, 2000 Men Women

Setting the scene • Why to study genetic susceptibility to cancers whose etiology is dominated by exogenous (preventable) agents? – Quantification of individual risk, especially among those formerly exposed to the risk factors – Cancers with strong exogenous risk factors as model to study interactions between genetic and exogenous factors in carcinogenesis.

Genetic Susceptibility to Lung and H&N Cancers High-penetrance susceptibility genes do not seem to account for a substantial proportion of lung and H&N cancer cases. Family history of cancer has been reported as a risk factor
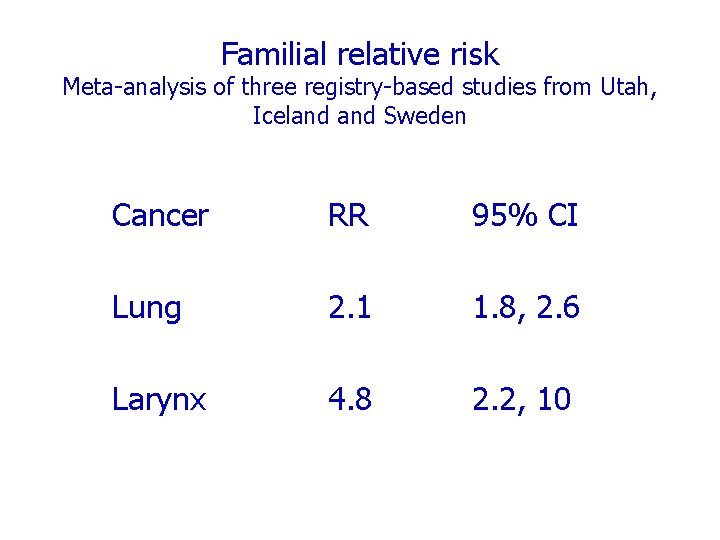
Familial relative risk Meta-analysis of three registry-based studies from Utah, Iceland Sweden Cancer RR 95% CI Lung 2. 1 1. 8, 2. 6 Larynx 4. 8 2. 2, 10

Explanations for high FRR • Shared exogenous risk factors • Shared genes with a role in pathways relevant to exogenous risk factors • Combination of the two above • Shared genes not interacting with exogenous risk factors

Genetic Association Studies on Lung and H&N Cancers 1 st generation n Very small studies (<100 cases) n Usually not epidemiologic study design; 1 -2 SNPs 2 nd generation n Small studies (100 -500 cases) n More epi focus; a few SNPs 3 rd generation n Large molecular epi studies (>500 cases) n Proper epi design; pathways 4 th generation n Consortium-based pooled analyses (>2000 cases) n Gx. E analyses 5 th generation n Post-GWS studies
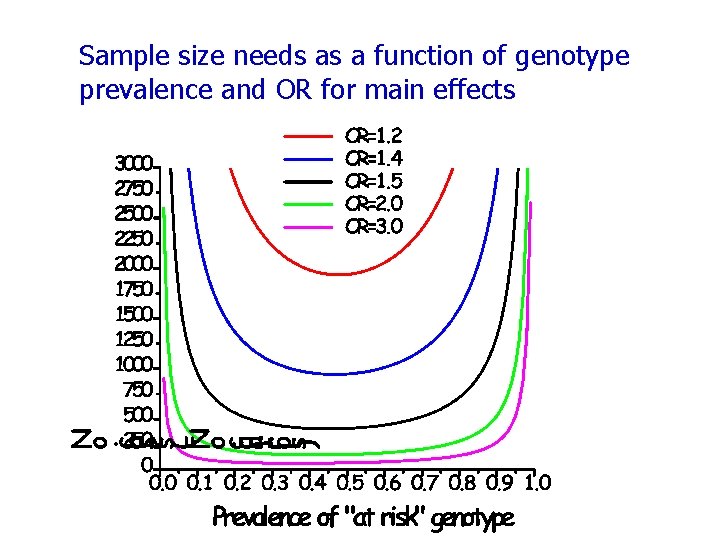
Sample size needs as a function of genotype prevalence and OR for main effects
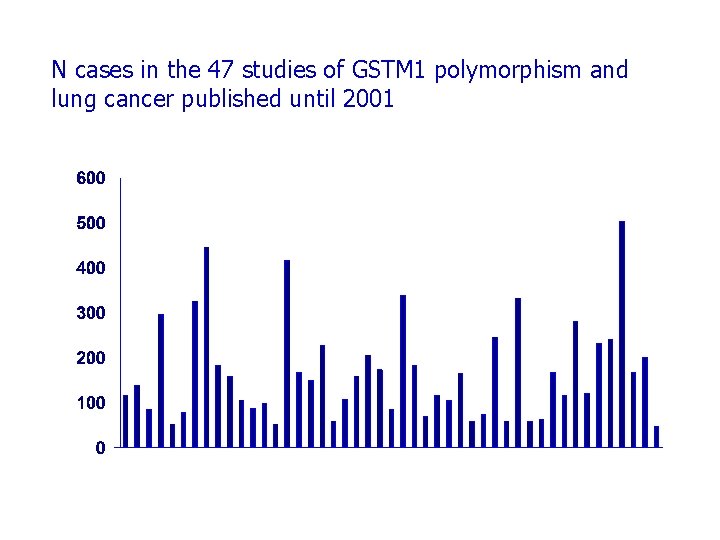
N cases in the 47 studies of GSTM 1 polymorphism and lung cancer published until 2001
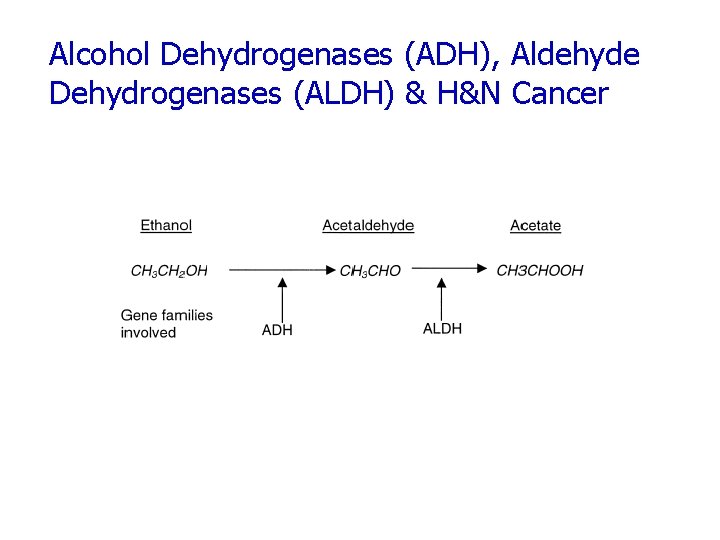
Alcohol Dehydrogenases (ADH), Aldehyde Dehydrogenases (ALDH) & H&N Cancer
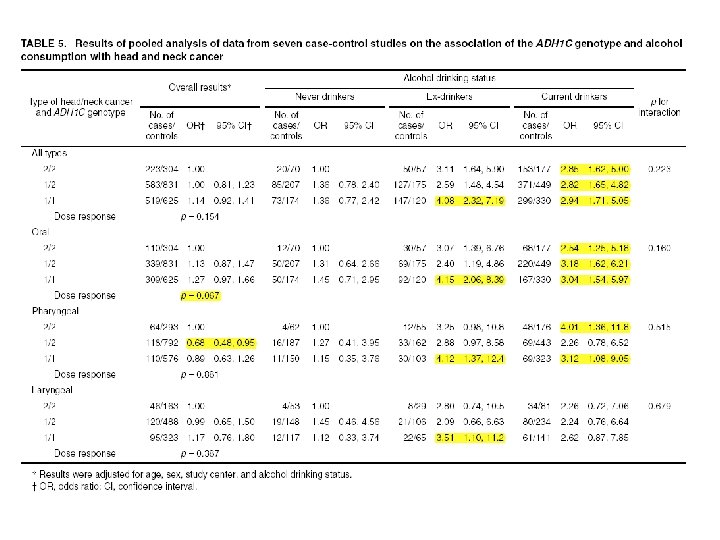
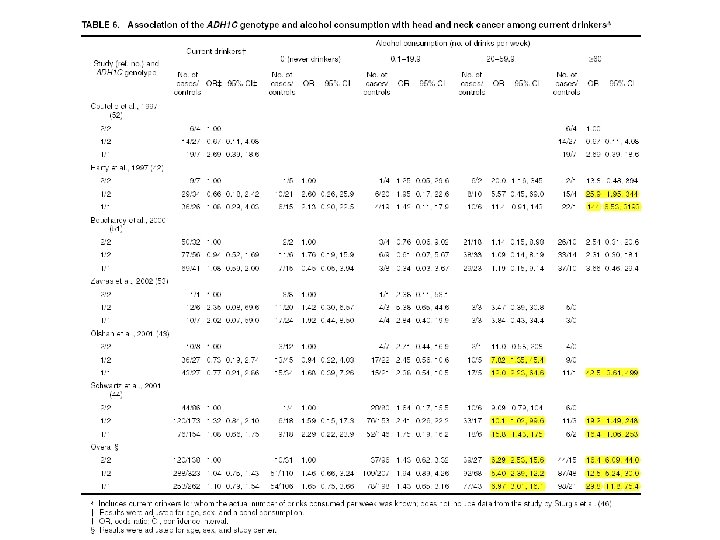
ADH 1 C and H&N Cancer ADH 1 C Ile 350 Val (rs 698) in exon 8 n n ADH 1 C*1 allele increases ethanol oxidation by 2. 5 -fold compared to *2 allele Suspected to increase risk of H&N cancer We conducted a pooled analysis of 7 published case-control studies. Brennan et al. , Pooled analysis of alcohol dehydrogenase genotypes and head and neck cancer - a Hu. GE review. Am J Epidemiol 2004.
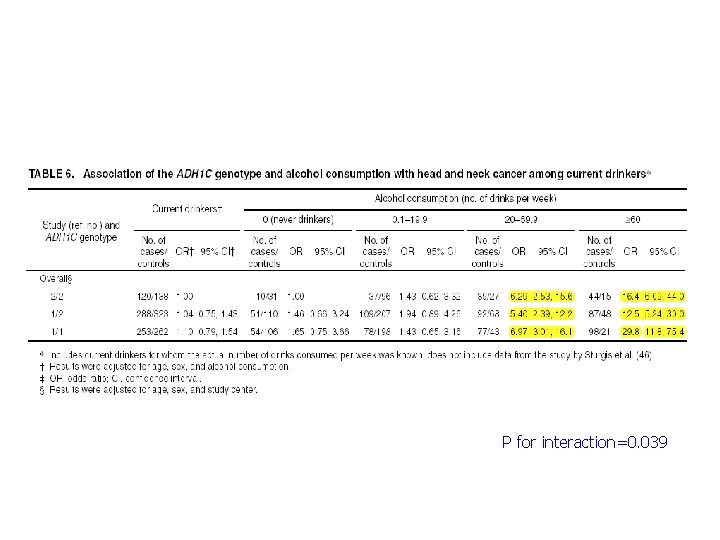
P for interaction=0. 039

Inconsistent results may be due to: Moderate prior probabilities that each SNP individually alters gene function or confers substantial increase in risk Difficulty in detecting associations with modest risk sequence variants in studies of small sample size False positives Publication bias Meta-Analysis limitations Publication Bias Unable to adjust for potential confounders Low power in stratified analysis Unable to examine gene-gene or gene-environment interactions Pooled-Analysis limitation Publication and participation bias
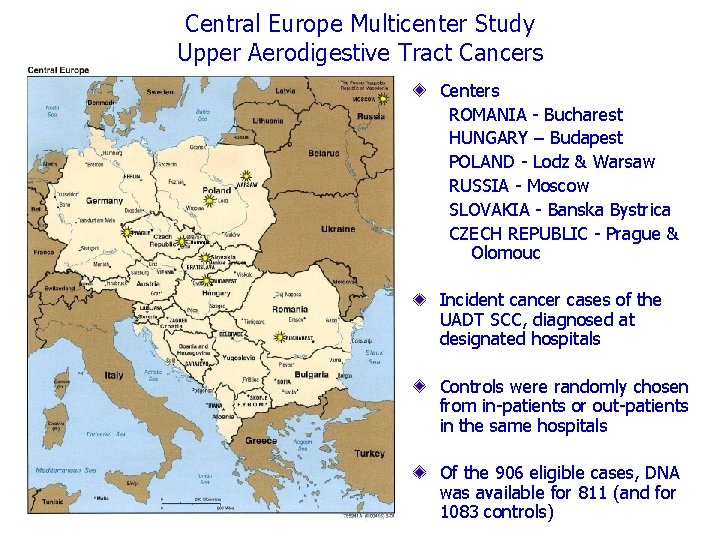
Central Europe Multicenter Study Upper Aerodigestive Tract Cancers Centers ROMANIA - Bucharest HUNGARY – Budapest POLAND - Lodz & Warsaw RUSSIA - Moscow SLOVAKIA - Banska Bystrica CZECH REPUBLIC - Prague & Olomouc Incident cancer cases of the UADT SCC, diagnosed at designated hospitals Controls were randomly chosen from in-patients or out-patients in the same hospitals Of the 906 eligible cases, DNA was available for 811 (and for 1083 controls)

Genotyping was by Taqman Designs of genotyping assays for SNP were taken from the SNP 500 website ~100 SNP were selected based on a combination of prior knowledge (functional data, reports of associations, important pathways) and allele frequency observed in Europeans. Results available on ~50 SNP n DNA repair n Cell cycle n Carcinogen metabolism

SNP in Alcohol Dehydrogenases (ADH) & Aldehyde Dehydrogenases (ALDH) ADH 1 B Arg 48 His (rs 1229984) in exon 3 n His = *2 allele = ‘fast’ allele ADH 1 C Ile 350 Val (rs 698) in exon 8 n Val = *1 allele = ‘fast’ allele ADH 1 C Arg 272 Gln (rs 1693482) in exon 6 ALDH 2 355 A>G (rs 886205) in the 5’ UTR ALDH 2 348 T>C (rs 440) in intron 6 ALDH 2 483 T>C (rs 441) in intron 6 Linkage disequilibrium demonstrated for ADH 1 B*2 (fast) and ADH 1 C*1 (fast) in Asians
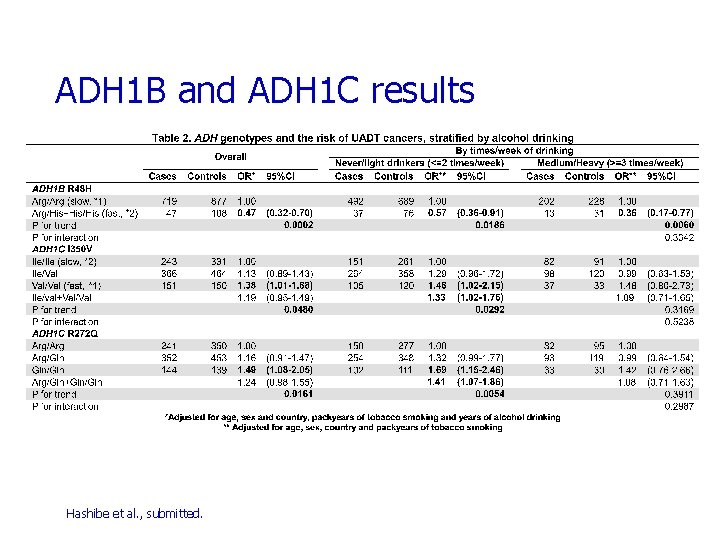
ADH 1 B and ADH 1 C results Hashibe et al. , submitted.
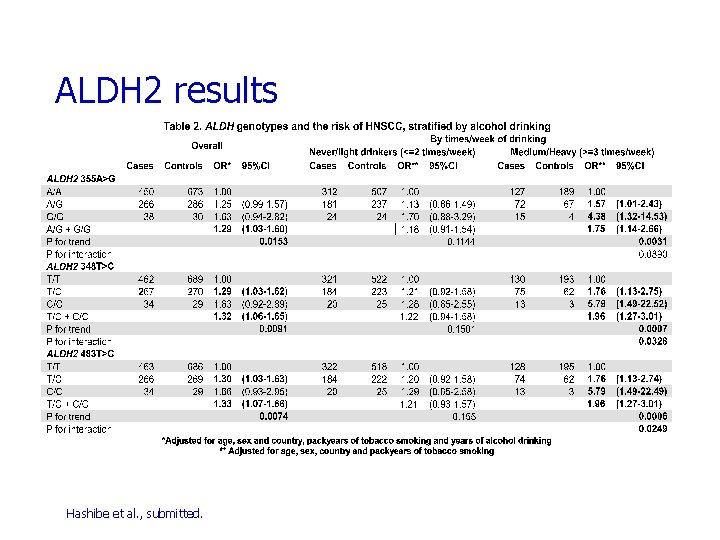
ALDH 2 results Hashibe et al. , submitted.

ADH and ALDH Results for esophageal cancer
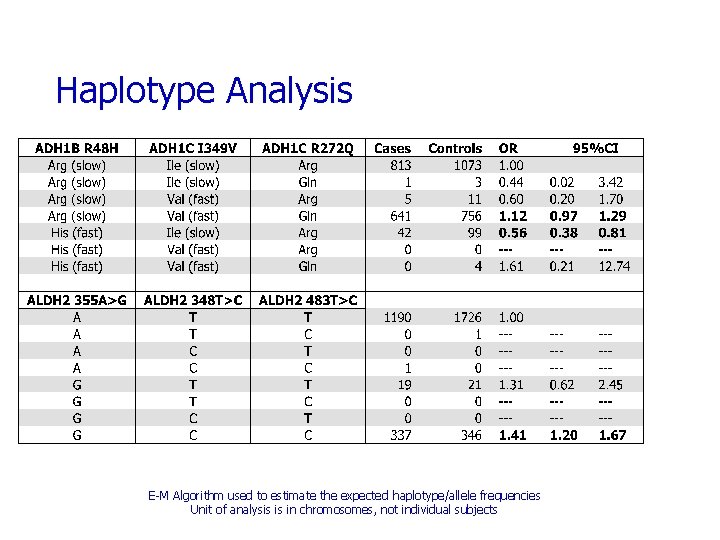
Haplotype Analysis E-M Algorithm used to estimate the expected haplotype/allele frequencies Unit of analysis is in chromosomes, not individual subjects
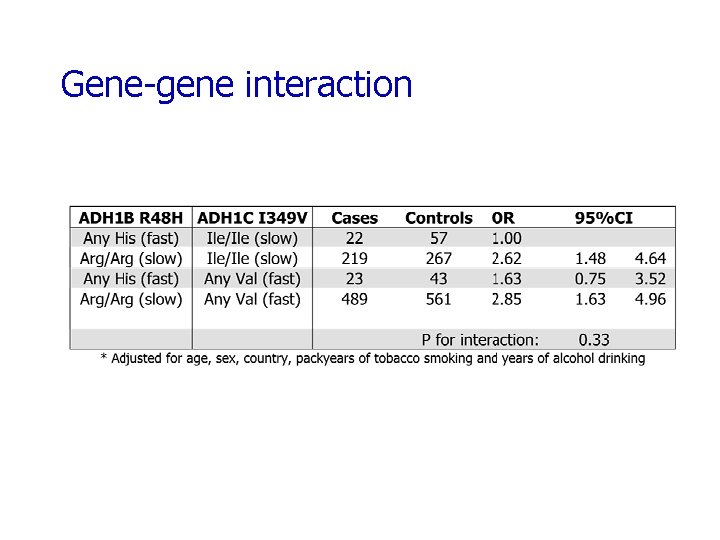
Gene-gene interaction
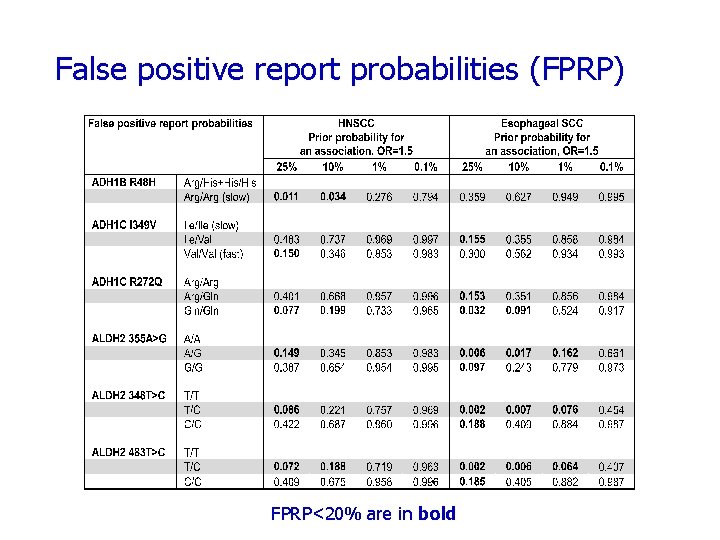
False positive report probabilities (FPRP) FPRP<20% are in bold

ADH/ALDH Summary of Results ADH 1 B R 48 H variant appeared to be protective against UADT cancers 2 ADH 1 C variants moderately increased the risk of UADT cancers These associations were most apparent for esophageal cancer Gene-alcohol interaction observed for ADH 1 B and ALDH 2 variants Gene-gene interactions were assessed but none were apparent Replication of these results is necessary Subsequent studies should focus not only on these variants, but other variants including haplotype tagging SNPs from Hapmap

International Head and Neck Cancer Epidemiology Consortium Collaboration of research groups leading large molecular epidemiology studies of H&N cancer. Established in 2004 http: //inhance. iarc. fr
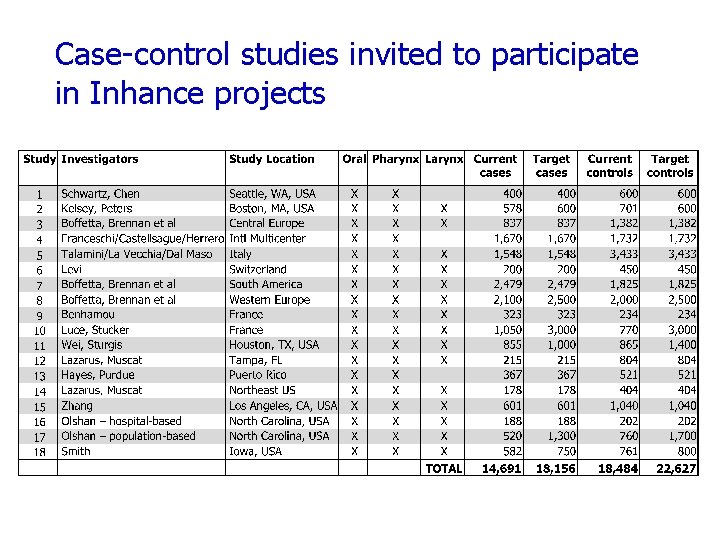
Case-control studies invited to participate in Inhance projects
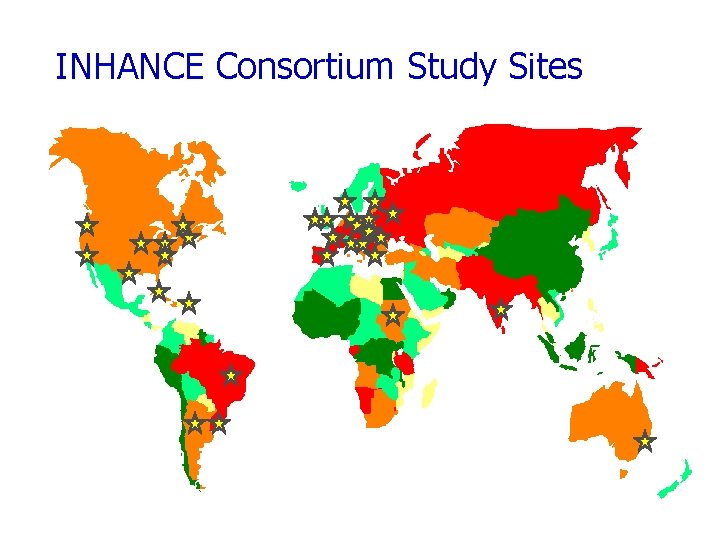
INHANCE Consortium Study Sites
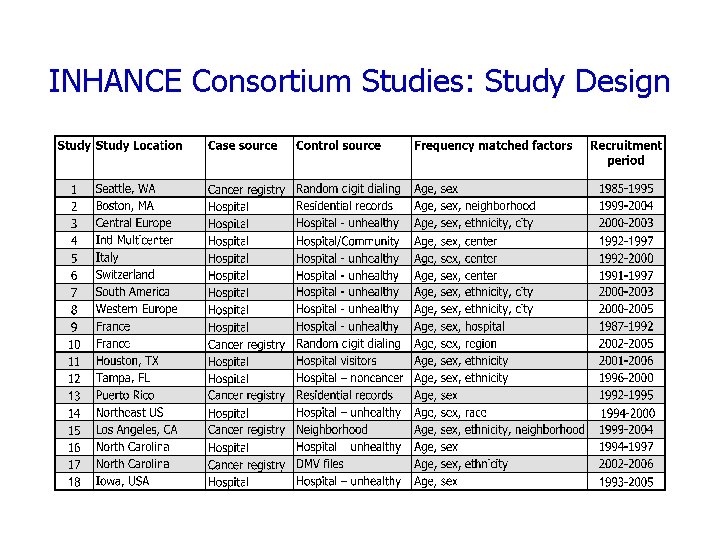
INHANCE Consortium Studies: Study Design
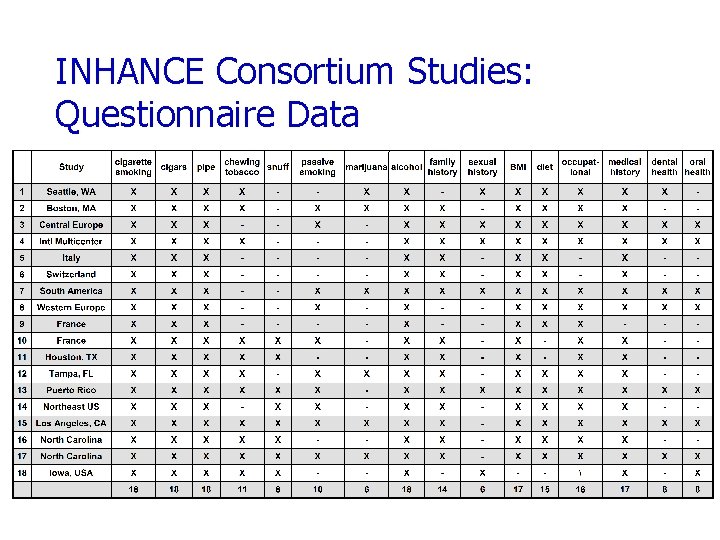
INHANCE Consortium Studies: Questionnaire Data
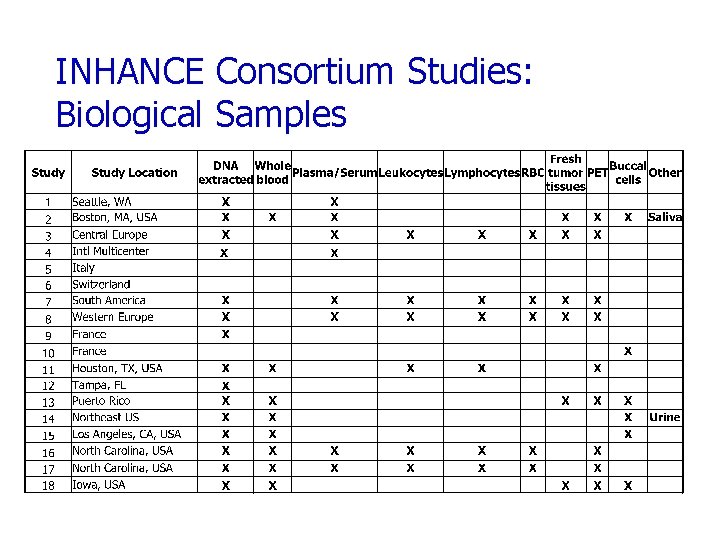
INHANCE Consortium Studies: Biological Samples
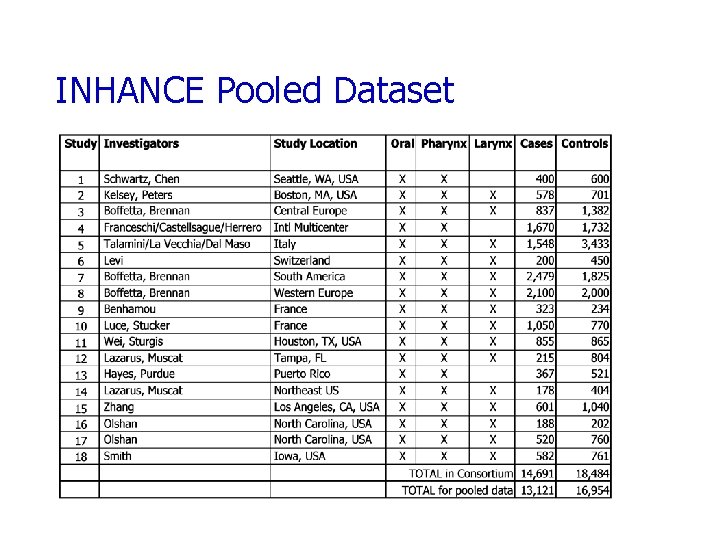
INHANCE Pooled Dataset
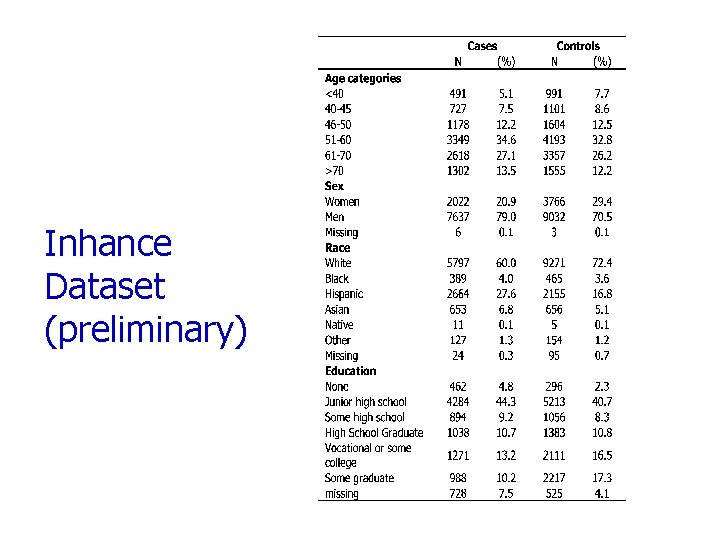
Inhance Dataset (preliminary)

SNP data pooling project Studies on various SNPs and head and neck cancer have not been consistent due to limitations in sample size, publication bias, false positives Greater statistical power in pooled analyses may help to identify modest risk SNPs Assessed SNPs genotyped in common across the Inhance studies n 18 SNPs genotyped in at least 3 studies Supported by the NIDCR R 03 grant
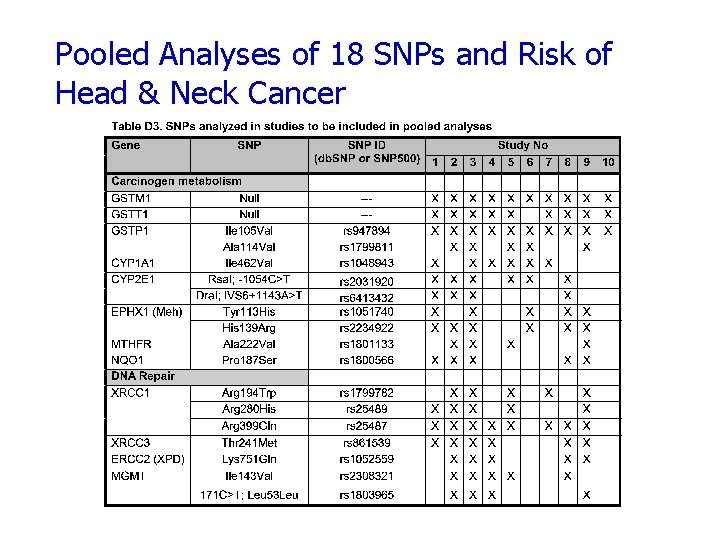
Pooled Analyses of 18 SNPs and Risk of Head & Neck Cancer

Basics of whole genome scan • • Genotype all cases and controls for panel of 300 k+ SNPs Identify most ‘interesting’ and replicate in second independent sample (and third etc) Advantages – No prior hypothesis Disadvantages – Sample size – Funding – Analysis

Proposal of GWS of H&N cancer • Phase 1 – 300 k SNP – Central European study • 1000 cases and 2000 controls (already genotyped as part of a parallel GWS of lung cancer) • Phase 2 – 5 k SNP – ARCAGE study (Western Europe) • 1300 case-control pairs – Latin American study • 1500 case-control pairs • Phase 3 – 30 -50 SNP – INHANCE • 5 -10 K case-control pairs

Conclusions 1 What have we learned about H&N cancer susceptibility? • Role of genes encoding for enzymes involved in alcohol metabolism and DNA repair – Improve marker selection • Select SNPs that are likely to alter gene function • Tag SNPs • Detailed analysis of genes having strong evidence of association • Quantify risk from genetic variants and interaction with alcohol drinking • Need to expand to other pathways

Conclusions 2 What have we learned about search for cancer susceptibility factors? • Strengths of large-scale molecular epidemiological studies and consortia – role of local collaborators and junior investigators – need for novel funding mechanisms • Need for novel approaches – Examine multiple markers • multigenic model • combine genotype and phenotype data for a pathway driven approach – Statistical analyses • hierarchical modeling • How to put the evidence together?

http: //www. cdc. gov/genomics/hugenet/

Acknowledgements - 1 IARC Mia Hashibe Paul Brennan Janet Hall Federico Canzian Rayjean Hung Norman Moullan Amélie Chabrier Valérie Gaborieau Julien Berthiller CNG Mark Lathrop Central European Study David Zaridze Neonila Szeszenia-Dabrowska Dana Mates Vladimir Janout Eleonóra Fabiánová Peter Rudnai Vladimir Bencko Witold Zatonski No. N Muin Khoury John Ioannidis Julian Little

Acknowledgements - 2 INHANCE members - Chu Chen, Stephen Schwartz, Karl Kelsey, Silvia Franceschi, Simone Benhamou, Daniel Luce, Isabelle Stucker, Erich Sturgis, Qingyi Wei, Richard Hayes, Mark Purdue, Philip Lazarus, Joshua Muscat, Zuo-Feng Zhang, Andrew Olshan, Elaine Smith, Edward S. Peters, Ana Menezes, Alexander W. Daudt, Maria Paula Curado, Sergio Koifman, Victor Wünsch Filho, Jose Eluf Neto, Elena Matos, Vladimir Bencko, Eleonora Fabianova, Vladimir Janout, Witold Zatonski, Rolando Herrero, Xavier Castellsague, Renato Talamini, Luigino Dal Maso, Fabio Levi, Carlo La Vecchia, Pagona Lagiou, Antonio Agudo, Wolfgang Ahrens, Bernard E. Mc. Cartan, David Conway, Andres Metspalu, Gary J. Macfarlane, Ray Lowry, Kristina Kjaerheim, Lorenzo Simonato, Ivana Holcátová, Franco Merletti, Ariana Znaor


Possible explanations for protective OR observed for ADH 1 B*1 ‘fast’ allele Linkage Disequilibrium n The SNP may be in LD with another SNP in another ADH that is protective n Ensemble of ADH activity may be lower even though this ADH 1 B allele is associated with higher activity Residual confounding by drinking behavior n Though we did not observe an association between genotypes & drinking behavior in our data, perhaps subjects with the ADH 1 B slow genotype had different drinking behaviors such as binge drinking. Complication by other substrates n ADHs can metabolize retinol. Protective effect may be due to dietary intake of vitamin A that is being metabolized more by ‘fast’ allele? Functional studies not consistent n Perhaps ‘fast’ allele is not really fast n In vitro studies showed enzyme activity differences, but the ADH 1 B genotype did not affect blood acetaldehyde levels in individuals Acetaldehyde Clearance n Enzyme activity of ADH 1 B*2 variant is 100 -200 times higher. Perhaps a fast initial metabolism leads to a peak in acetaldehyde that induces clearance mechanisms n Enzyme activity of ADH 1 C*1 fast allele is 2. 5 times, so it may not induce clearance?

How to move forward in the study of gene-environment interactions • Improve marker selection – – Select SNPs that are likely to alter gene function Tag SNPs Detailed analysis of genes having strong evidence of association Expand focus to other pathways • Examine multiple markers – Multigenic model – Combine genotype & phenotype data for a pathway driven approach • Employ novel statistical analyses – FPRP – Hierarchical modeling • Enhance statistical power – Limitations of previous studies include small sample size, publication bias, false positives – Meta-analysis, pooled analysis, consortia

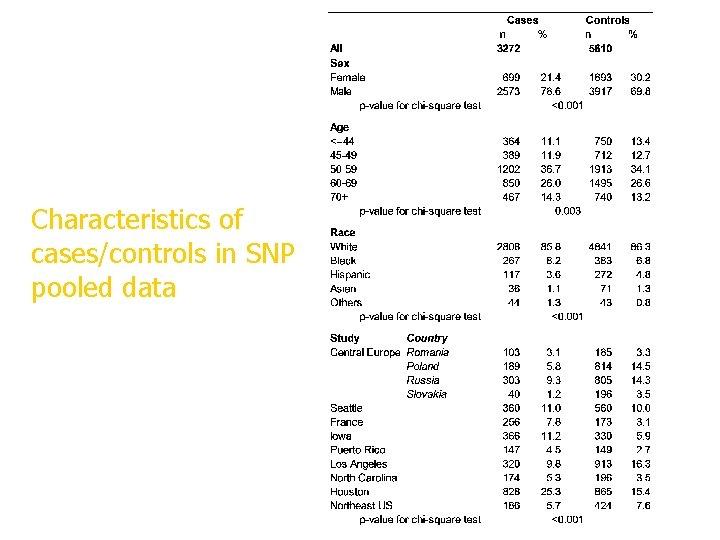
Characteristics of cases/controls in SNP pooled data
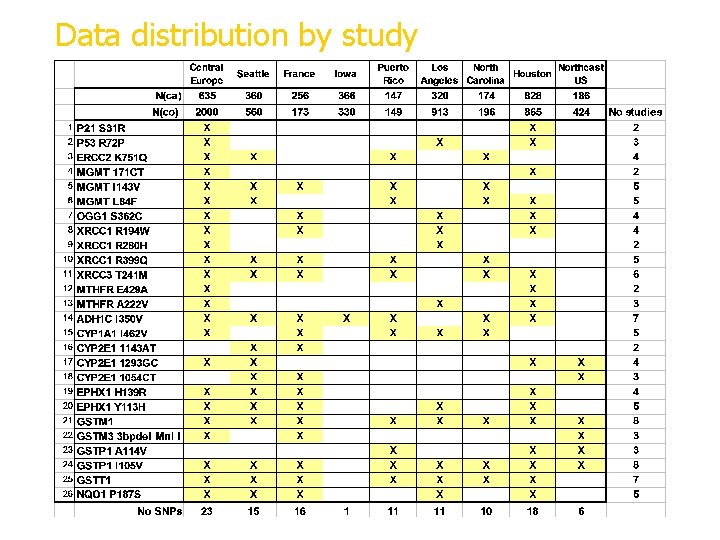
Data distribution by study X=data available
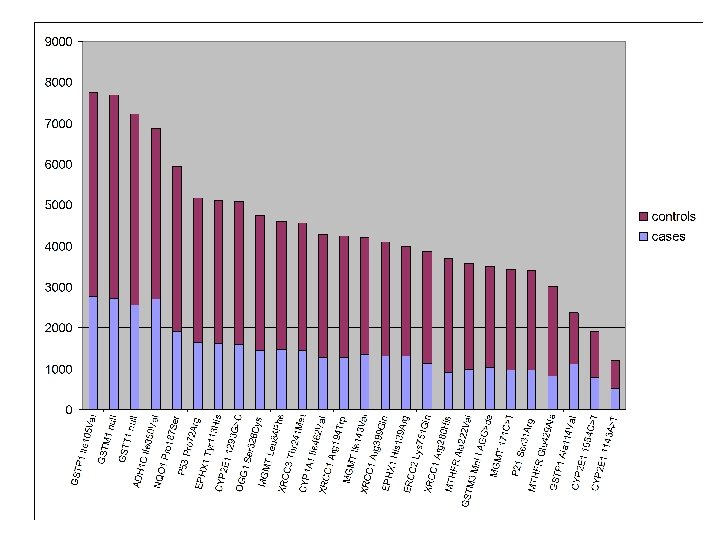
Sample size
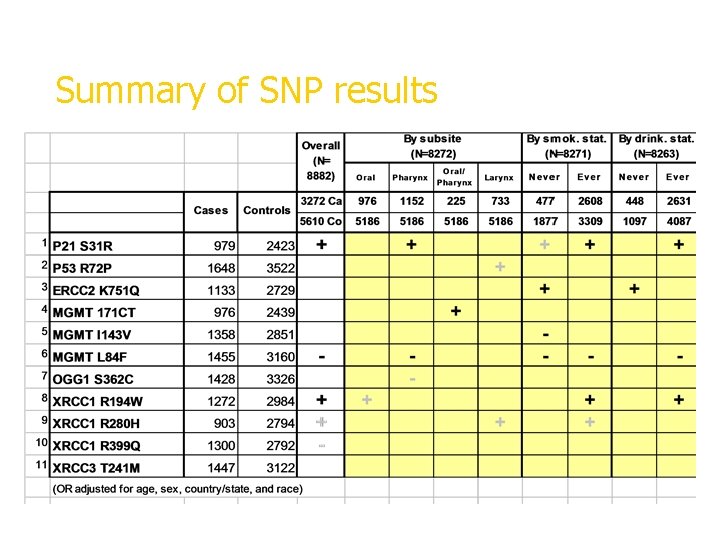
Summary of SNP results + significant increased risk, - significant decreased risk, gray=borderline
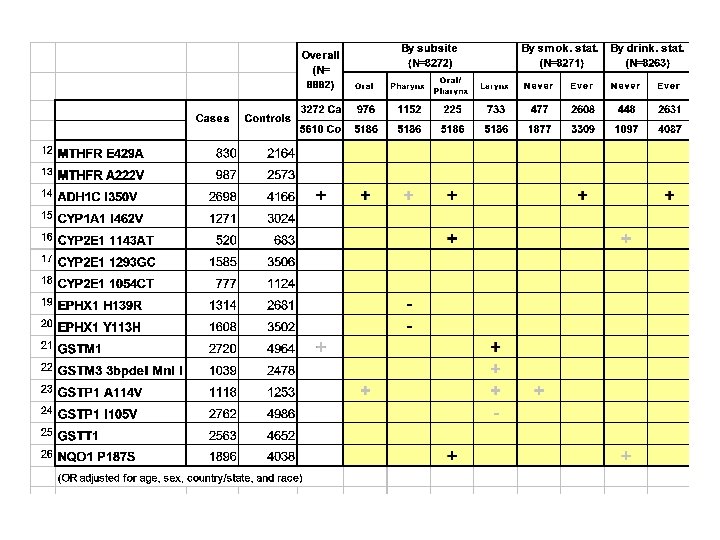
+ significant increased risk, - significant decreased risk, gray=borderline
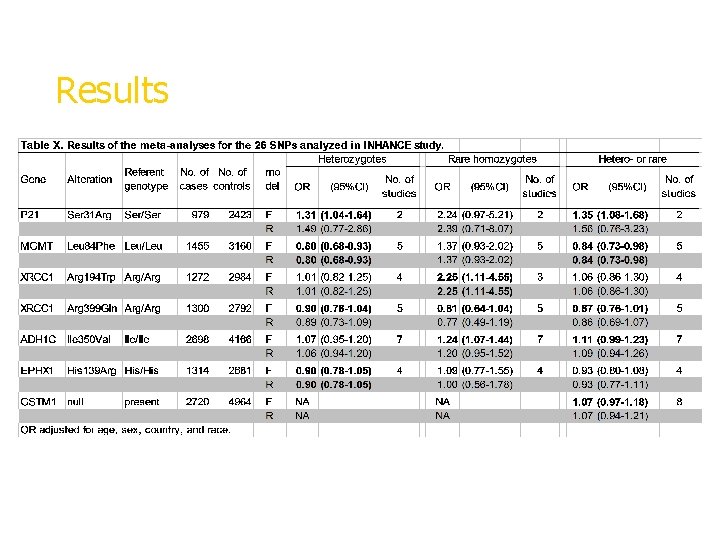
Results
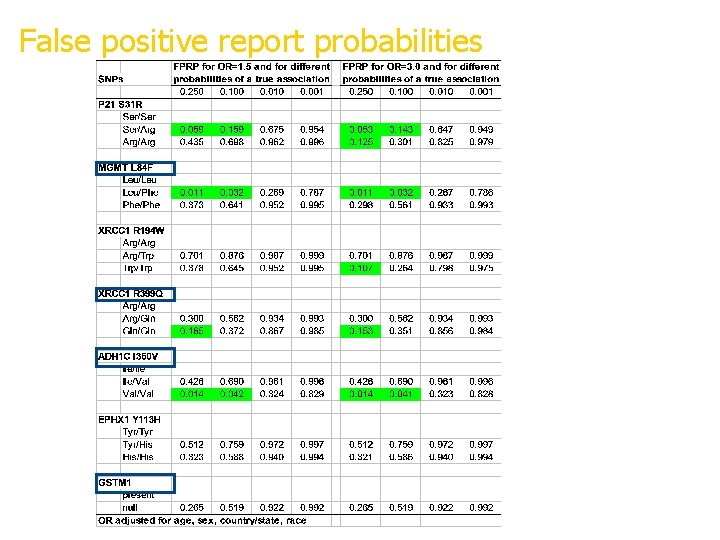
False positive report probabilities FPRP<20% highlighted Some overlap with previously published pooled analyses

Summary Our results support the association of several SNPs in DNA repair genes and ADH 1 C Next steps: n Assess interactions with tobacco/alcohol n Multigenic models / hierarchical models w Difficulties because of missing data

Future directions for Inhance The INHANCE consortium provides a flexible and dynamic means for pursuing new ideas and hypotheses of interest to its members. Proposals under consideration n n Diet & gene-environment interactions HPV status & tobacco/alcohol Coordinate genotyping from a priority list of SNPs Genome-wide association study on a subset of subjects We envision that the consortium will: n n help to overcome the limitations of previous studies be a major step toward improving our understanding of the causes and mechanisms of head and neck cancer, and a long-standing cooperation among the research groups.
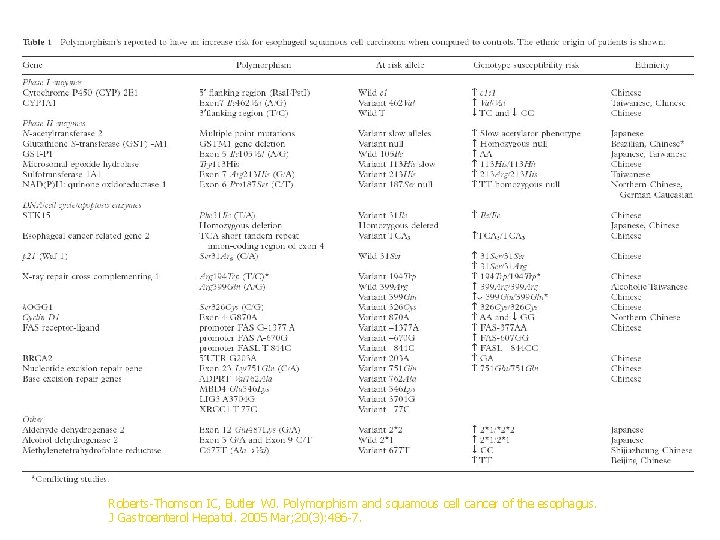
Roberts-Thomson IC, Butler WJ. Polymorphism and squamous cell cancer of the esophagus. J Gastroenterol Hepatol. 2005 Mar; 20(3): 486 -7.

International Lung Cancer Consortium (ILCCO) Mc. Laughlin Wichmann Schwarts Wild Boffetta Harris Goodman Risch Kiyohara Brennan Benhamou Wiencke Christiani Zhang Stucker Yang Spitz Berwick Tajima Landi Hong Vineis Lan Chen Lazarus Thun Le Marchand 3 cohort studies 17 population based case-control studies 13 hospital based case-control studies 2 studies with mixed controls 1 cross-sectional study

ILCCO • Participants – 30 teams – 36 studies – 17, 000 cases (target 28, 000 cases) • Executive committee – IARC, MD Anderson, HSPH, NCI • Working groups • Levels of collaborations – – Pooled analysis (questionnaire data; biomarker results) Replications of novel findings Coordinated generation of new data Design a new study to address the novel hypothesis based on the network

On-going ILCCO projects • Pooled analysis on DNA repair genes (NCI R 03) • Pooled analysis of family history (in preparation) • Pooled analysis of occupational exposures(in preparation) • Pooled analysis on Indoor Air pollution(in preparation)

Acknowledgments • IARC – P. Brennan, R. Hung, P. Hainaut, S. Tavtigian • European pooled analysis – L. Simonato, S. Darby, F. Merletti, H. E. Wichmann • Central European study – J. Lissowska, P. Rudnai, D. Mates, I. Holcatova, D. Zardize • Janus study – A. Andersen, E. Weiderpass, R. Peto • Indian study – V. Gajalakshmi, A. Mathew • ILCCO – M. Spitz, D. Christiani, N. Caporaso • Financial support – EC DG Research – EC SANCO – NCI

Example of Gx. E interaction CHEK 2 and lung cancer • Risk of 1100 del. C in breast cancer • I 157 T polymorphism has a moderate effect on risk of 6 cancer types • Replication of decreased risk of lung cancer in a large series Nature Genetics, 2002, 31: 55 -59 Am. J. Hum. Genet. , 2004; 75: 1131 -35
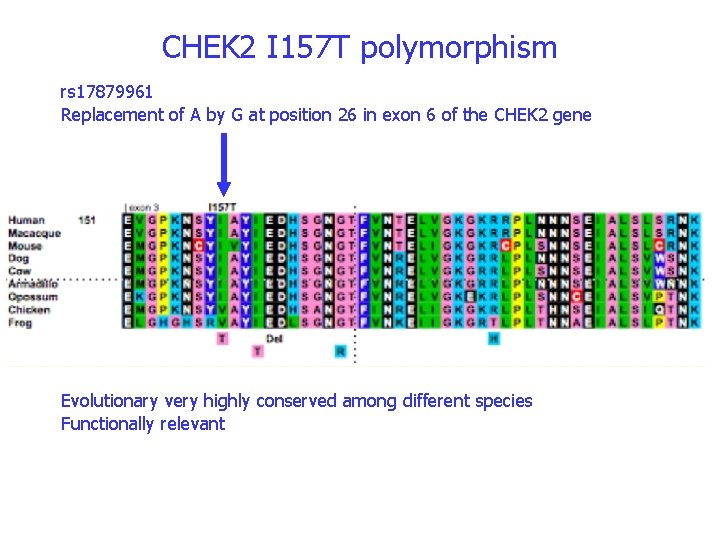
CHEK 2 I 157 T polymorphism rs 17879961 Replacement of A by G at position 26 in exon 6 of the CHEK 2 gene Evolutionary very highly conserved among different species Functionally relevant
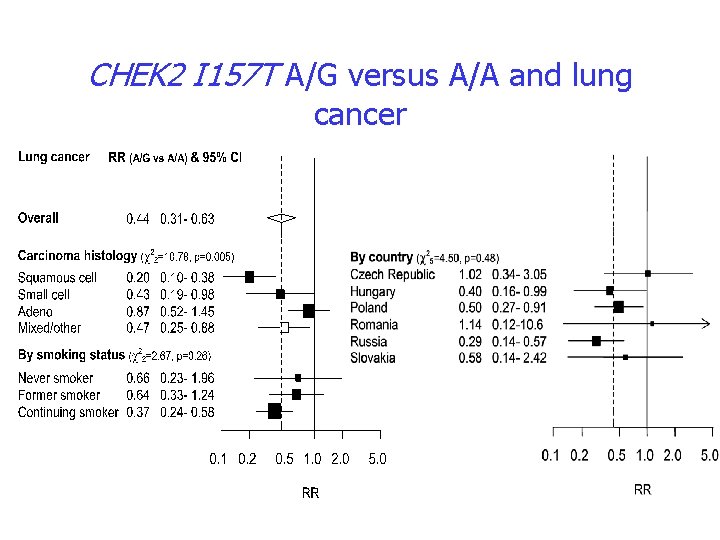
CHEK 2 I 157 T A/G versus A/A and lung cancer

Gene-environment interaction in lung carcinogenesis ITC intake and GST polymorphims • Green cruciferous vegetables – Protective effect against lung cancer strongly suggested – Rich in isothiocyanates (ITCs) • Experimental evidence of lung cancer chemoprevention • A causal association between ITC and decreased risk of any cancer cannot be established due to potential confounding by other nutrients (IARC, 2004) • Glutathione S-transferase (GSTs) – Promote elimination of ITCs – GSTM 1 or T 1 null: does not have enzyme activity increased ITC levels – The distribution of GSTM 1 and T 1 genotypes is unlikely to be associated with other dietary sources
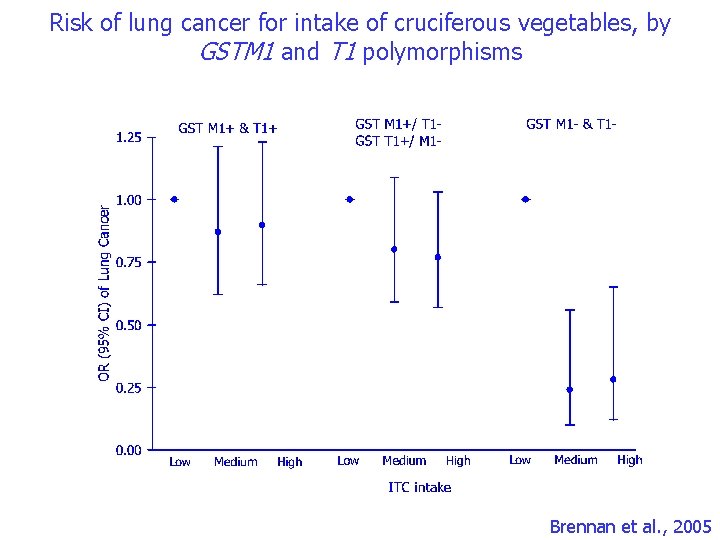
Risk of lung cancer for intake of cruciferous vegetables, by GSTM 1 and T 1 polymorphisms Brennan et al. , 2005
- Slides: 69