Genetic Mutations in Young Children with IBD Insights

Genetic Mutations in Young Children with IBD: Insights into Pathogenesis and Definitive Therapies Scott B. Snapper, M. D. , Ph. D. CCFA Advances 2012 Hollywood, Florida December 14, 2012

Disclosures I have the following financial relationships to disclose: Hoffman La-Roche - Consultant Eisai - IBD Advisory Board * No products or services produced by these companies are relevant to my presentation.
Etiology of IBD • Complex Disease. • Combination of: – Environmental Factors – Gut Bacteria – Abnormal Immune Response – Genetics

Unique Aspects of Pediatric IBD • 10 -20% of IBD presents in children • Children often present with more severe inflammation (up to 90% pancolitis in UC) • Children with CD more often present with upper intestinal tract involvement • Young children often present with Crohn’s colitis with perianal involvement Mamula et al, AJG. 2002 Heyman et al, J. Ped. 2005

Greatest Increase in IBD Incidence Found in Very Early Onset Subgroup AGE Change in Incidence Rate 95% CI P-value* 0 -4 +5. 0% / year 0. 5% - 10. 5% 0. 032 5 -9 +7. 6% / year 4. 4% - 10. 8% <0. 0001 10 -14 +0. 63% / year -0. 9% – 2% 0. 407 15 -17 -0. 21% / year -1. 3% – 0. 9% 0. 72 * By Poisson regression analysis, controlling for sex Benchimol et al (2009) Gut 48: 1490 -97

Age Modification in the Paris Classification of Pediatric IBD: Age at Diagnosis Montreal Paris A 1: below 17 y A 2: 17 -40 y A 3: above 40 y A 1 a: < 10 A 1 b: 10 -17 A 2: 17 -40 y A 3: above 40 y Currently Very Early Onset IBD = Disease Diagnosed < 10 Years IBD J. 2011

Unique Clinical Features of VEOIBD* VEO-CD Adolescent and Adult CD • Colon involved • Colon only involved • 80% < 10 years of age • Decreases with age • Ileum involved • < 20% • Ileum involved • Up to 80% • infrequent at < 10 yrs of age • • Positive FH – 40 -50% Stricturing – 20 -46% Surgery – up to 71% Extension of disease – up to 40% • • Positive FH – 14 -20% Stricturing – 29 -40% Surgery – up to 55% Extension – up to 16% * Defined as Age < 10 by the Paris Classification Sherlock et al (unpublished); Griffiths (2004) Best Pract Res Clin; Heyman et al (2005) J Ped; Ruemmele et al (2006) JPGN; Kappelman et al (2008) IBD; Louis et al (2008) Gastro, van Limbergen et al (2008) Gastro, Vernier. Massoulle (2008) Gastro: Levine et al (2011) IBD; de Bie et al (2012) IBD

Unique Aspects of Infantile IBD (< 2 yo) • Often isolated Colonic Disease • Severe Course – refractory to multiple immunosuppressant medications, often requiring surgery, occasionally fatal • > 40 % with one or more family members with IBD • 25% first manifestation of underlying immunodeficiency Ruemmele 2006 JPGN Cannioto 2009 EJP Heyman 2005 J Ped

GWAS Studies Have Identified Over 160 IBD Susceptibility Loci: VEO-IBD Not Studied • Large meta-analyses combining all adult and pediatric data sets failed to show any polymorphisms that reliably distinguished pediatric from adult-onset IBD. (However, most children in all studies have been age > 10) Imielinski Nat Genetics 2009; Kugathasan Nat Genetics 2008; Franke Nat Genetics 2011; Anderson Nat Genetics 2011; Jostins et al. Nature Nov 1. 2012 www. neopics. org

Have Large Scale Genetic Efforts Missed Key Dominant Pathways Causing Infantile and VEO-IBD? monogenic oligogenic polygenic Environment Pediatric. Onset IBD Adult. Onset IBD Genetics Infantile and VEO-IBD Not Captured? Familial AGE Adapted from Kaser A, Zeissig S & Blumberg RS, Dig Dis 20

Primary Immunodeficiencies Often Present with Intestinal Inflammation • IPEX syndrome – Frequent enteropathy • Wiskott-Aldrich syndrome – 10% with colitis • Chronic granulomatous disease – Gastric granulomas – Crohn’s-like colitis • NEMO (NF-k. B Essential Modulator) Deficiency • enterocolitis • superficial cryptitis • Common variable immunodeficiency – Frequent enteropathy

Mutation in Fox. P 3, Transcription Factor Critical for Regulatory T cells Development, Results in IPEX Syndrome – Immune Dysfunction, Polyendocrinopathy, Enteropathy, X-linked. – Affected patients develop autoimmune enteropathy (AIE). – Failure of Regulatory T cell Development Powell et al, J. Pediatr. 1982 Wildin et al, Nat. Genet. 2001 Bennett et al, Nat. Genet. 2001 Khalidi et al, Hum. Pathol. 2006 Ruemmele et al, Curr Op. Gastro 2008 Villous atrophy Crypt elongation Loss of Paneth cells, increase in IELs

The Wiskott Aldrich Syndrome: An Immunodeficiency Associated with Autoimmunity, Colitis and Defects in Regulatory T cells • Mutations in the gene encoding the cytoskeletal regulator Wiskott-Aldrich Syndrome Protein (WASP) • Up to 50% of patients have two or more autoimmune complications - AIHA, Arthritis, Vasculitis, Purpura, Uveitis, Myositis • Inflammatory bowel disease (IBD) in 5 -10% of the patients • Regulatory T cell Defects Puck et al, NEJM Folwaczny et al, Endoscopy 2002 Puck et al, NEJM 2006; 355(17): 1759 -61 Nguyen Gastro 2007; Maillard JEM, 2007; Rawlings et al, JCI 2007; Roncarolo et al, JEM 2007; Schwartzberg et al 2008; Westerberg et al. Blood 2008

- Harvesting Genetic Fruit and Understanding IBD – Low Hanging Fruit Immunodeficiencies and Infantile IBD: Another Low Hanging Fruit? www. neopics. org Muise, Snapper, Kugathasan: Gastro 2012

Hypothesis: Major insight can come with studying younger children with IBD Until Recently No Studies have Focused on IBD in Young Children Study of Infantile and VEO-IBD Patients Provides a More Homogenous Population To Understand IBD An International Effort is Required to Advance our Understanding

NEOPICS: inter. National Early Onset Pediatric IBD Cohort Study Principal Investigators: �Aleixo Muise – Sick Kids, UT �Christoph Klein – University Children’s Hospital Munich �Scott Snapper – Boston Children’s, HMS NEOPICS – expanded to 63 Centers on 5 continents with access to over 1000 VEO-IBD patients

Case History • Presented in 1 st year of life with severe colitis • Persian ancestry • Enteric fistulae, recurrent folliculitis Severe Colitis Perianal disease with multiple fistulas Joint effusions Folliculitis Glocker, EO. Klein, C. NEJM 2009

Case History Distance from the upper telomere of Chr. 21 in Mb I-1 I-2 II-1 II-2 II-3 II-4 20. 6 D 21 S 1437 130 138 130 134 130 138 130 130 130 23. 7 D 21 S 1257 169 167 169 169 169 24. 5 D 21 S 1914 212 214 208 212 214 214 214 27. 7 D 21 S 2052 136 144 136 128 144 136 136 136 27. 7 D 21 S 1258 153 139 153 151 139 153 153 153 30. 6 D 21 S 1270 193 193 189 ND ND 193 193 31. 7 D 21 S 2049 118 120 118 132 120 118 118 118 32. 7 D 21 S 262 148 144 148 148 148 33. 5 D 21 S 1898 236 228 236 234 228 236 236 236 33. 9 D 21 S 1910 247 219 245 213 247 213 219 245 247 245 IL 10 RBTrp 159 T A C T G G A A IL 10 RBTrp 159 X A T A C T G A A

Tyk 2 Jak 1 IL-10 RA IL-10 RB IL-10 RA • IL-10 R is heterotetramer – IL 10 RA directly binds IL-10 – IL 10 RB interacts with IL 10 RA and stabilizes structure • Receptor assembly recruits Jak 1 and Tyk 2 • Phosphorylation, dimerization and translocation of STAT 3 – IL-10 -mediated transcriptional program that results in immunosuppression IL-10 RB Interleukin-10 Signaling Jak 1 Tyk 2 STAT 3 p. STAT 3 SOCS 3 Immunosuppression
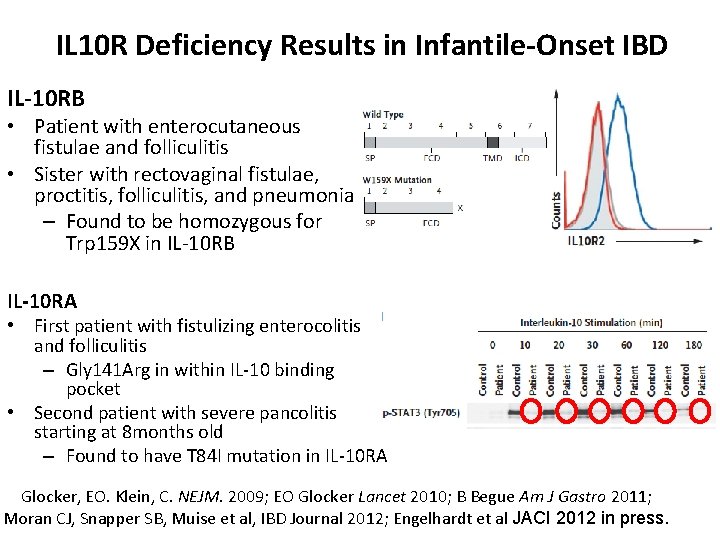
IL 10 R Deficiency Results in Infantile-Onset IBD IL-10 RB • Patient with enterocutaneous fistulae and folliculitis • Sister with rectovaginal fistulae, proctitis, folliculitis, and pneumonia – Found to be homozygous for Trp 159 X in IL-10 RB IL-10 RA • First patient with fistulizing enterocolitis and folliculitis – Gly 141 Arg in within IL-10 binding pocket • Second patient with severe pancolitis starting at 8 months old – Found to have T 84 I mutation in IL-10 RA Glocker, EO. Klein, C. NEJM. 2009; EO Glocker Lancet 2010; B Begue Am J Gastro 2011; Moran CJ, Snapper SB, Muise et al, IBD Journal 2012; Engelhardt et al JACI 2012 in press.
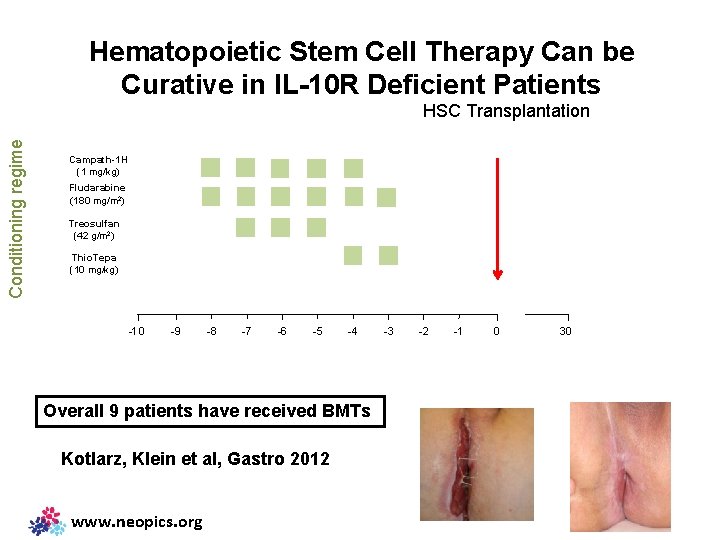
Hematopoietic Stem Cell Therapy Can be Curative in IL-10 R Deficient Patients Conditioning regime HSC Transplantation Campath-1 H (1 mg/kg) Fludarabine (180 mg/m 2) Treosulfan (42 g/m 2) Thio. Tepa (10 mg/kg) -10 -9 -8 -7 -6 -5 -4 Overall 9 patients have received BMTs Kotlarz, Klein et al, Gastro 2012 www. neopics. org -3 -2 -1 0 30

Do IL 10 RA/IL 10 RB Polymorphisms Alter Risk for Very Early-Onset IBD? • Exon sequencing of 188 patients (from Sick. Kids and Wisconsin) with early-onset IBD (EO-IBD < 18 yo) and 188 healthy controls • Found at least 25 rare/novel polymorphisms in the EO-IBD and especially VEO-IBD population • Two SNPs associated with VEO-UC – replicated in two independent cohorts (rs 2228054 and rs 2228055 - OR 3. 08, P= 1. 42 x 10 -4; and OR 2. 93, P= 1. 4 x 10 -4 , respectively) • Points to a broader role for the IL 10 R Pathway in the pathogenesis of VEO-IBD: Diagnosed 5 yo and younger www. neopics. org Moran CJ, Snapper SB, Muise et al, IBD Journal 2012

Case 1 – IL 10 R Pathway Lessons • IL 10 R deficiency should be considered in all infants with severe colitis, perianal disease, folliculitis, +/- joint disease • All patients so far (> 30) have had symptoms < 3 months of age • Bone Marrow Transplant should be considered if IL 10/IL 10 R defects are identified • A subset of VEO-IBD patients may have subtle defects in the IL 10 R pathway – candidates for treatment with IL 10? www. neopics. org Muise, Snapper, Kugathasan Gastroenterol 2012
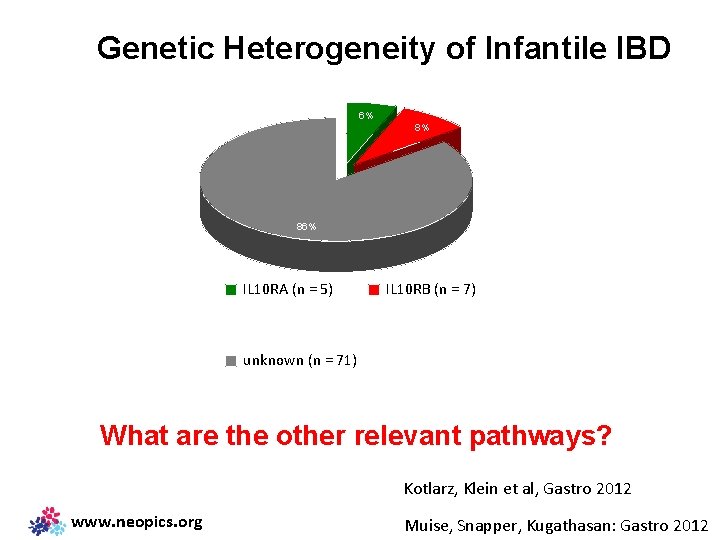
Genetic Heterogeneity of Infantile IBD 6% 8% 86% IL 10 RA (n = 5) IL 10 RB (n = 7) unknown (n = 71) What are the other relevant pathways? Kotlarz, Klein et al, Gastro 2012 www. neopics. org Muise, Snapper, Kugathasan: Gastro 2012

Case 2 • Patient ET – Presented at 2 months of age: – Blood in stool – Diagnosed with cow’s milk protein allergy • Diagnosed < 1 yo with Crohn’s colitis. • Developed perianal and small bowel disease < 2 years of age. • No evidence of chronic infections or immunodeficiency. • No family history of IBD, parents not consanguineous. • Has abnormal low normal reactive oxygen species (ROS) production (3 x). Muise, et al Gut, 2011 All images are used with the permission of the patient and family

Defects in NADPH Oxidase Genes Results in Reduced “Reactive Oxygen Species (ROS)” and CGD • Chronic Granulomatous disease (CGD) is associated with defects in NADPH oxidase complex genes • Patients with CGD develop: – recurrent and life-threatening infections – granulomatous inflammation of the intestine – 40% of CGD Patients have Crohn’s Disease-like Colitis Am J Gastroenterol. 2009 Jan; 104(1): 117 -24.

NADPH Oxidase Genes and CGD Gene Inheritance Frequency CYBB: gp 91 phox X-Linked Recessive ~65% CYBA: p 22 phox Autosomal Recessive <5% NCF 1: p 47 phox Autosomal Recessive ~25% NCF 2: p 67 phox Autosomal Recessive ~5% NCF 4: p 40 phox Autosomal Recessive < 1% www. neopics. org Lam et al. , 2010

Hypothesis: Defects in the NADPH oxidase genes that do not cause overt Chronic Granulomatous Disease (CGD) are associated with susceptibility to IBD. Muise, et al Gut, 2011

Sequencing of NADPH Oxidase Genes in Infantile and VEO-IBD Patients Identifies Deleterious Mutations • Identified a novel NCF 2 variant - (c. 113 G/A) resulting in a mutation in p 67 phox R 38 Q • Variant results in aberrant Rac 2 binding • Examined this mutation – 2 independent VEO-IBD cohorts – 4% of VEO-IBD patients (11/268) – 0. 3% of older IBD patients (1/330) – 0. 2% of healthy controls (1/480) Muise, et al Gut, 2011 www. neopics. org
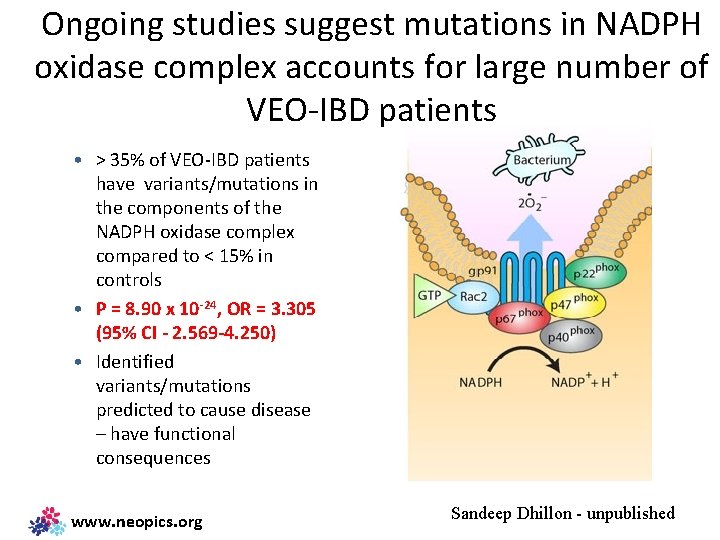
Ongoing studies suggest mutations in NADPH oxidase complex accounts for large number of VEO-IBD patients • > 35% of VEO-IBD patients have variants/mutations in the components of the NADPH oxidase complex compared to < 15% in controls • P = 8. 90 x 10 -24, OR = 3. 305 (95% CI - 2. 569 -4. 250) • Identified variants/mutations predicted to cause disease – have functional consequences www. neopics. org Sandeep Dhillon - unpublished

Conclusions • Mutated NCF 2 results in reduced RAC 2 binding and decreased ROS production leading to susceptibility to CD in infantile and VEO-IBD patients. • Functional mutations in other NADPH oxidase complex members are associated with VEO-IBD risk. • SNPs in other NADPH oxidase components (e. g. , NCF 4) have also been shown to be important in older children with IBD. • These findings may suggest the use of alternate therapies effective in CGD patients (e. g. , antibiotics) or shown to block pathways that drive the noninfectious inflammation in CGD patients (e. g. , anakinra – IL 1 R antagonist) www. neopics. org Patient now 4 yo – treated with 5 -ASA and antibiotics during flares – maintained good growth and symptom control.

How do we assess the heritability of the remaining fraction? 6% 8% 86% IL 10 RA (n = 5) IL 10 RB (n = 7) unknown (n = 71) 1. Deep sequencing of GWAS loci for identification of rare variants 2. Immunochip analysis of selected genes in similar cohort 3. Whole Exome Sequencing/Whole Genome Sequencing WES in VEO-IBD patient leads to identification of mutation in XIAP (associated with X-linked lymphoproliferative syndrome – XLPS 2) EA Worthey Genetics in Medicine 2011 www. neopics. org

Whole Exome Sequencing Efforts • Completing sequencing on 7 consanguineous families (trios) with plan to complete 20 families this year (have already identified two genes) • WES on complex NEOPICS families with multiple affected siblings • None have IL 10/R, XIAP, NAPDH oxidase or other known mutations • Completed RNA seq on the Trios – discover pathway affected by genetic defects www. neopics. org Lucas Mastropaolo - unpublished .

Conclusion – VEO-IBD • GWAS have shown that genetics in adult and adolescent pediatric IBD overlap considerably. • Immunodeficiencies account for a significant percentage of patients presenting with infantile IBD • Unique genetic abnormalities may be more dominant in VEO-IBD (e. g. , IL-10 R; NCF 2); however, data is limited • Whole exome sequencing (and ultimately whole genome sequencing) will greatly expand our ability to detect rare variants in individual patients An International Effort is Required to Advance our www. neopics. org Understanding
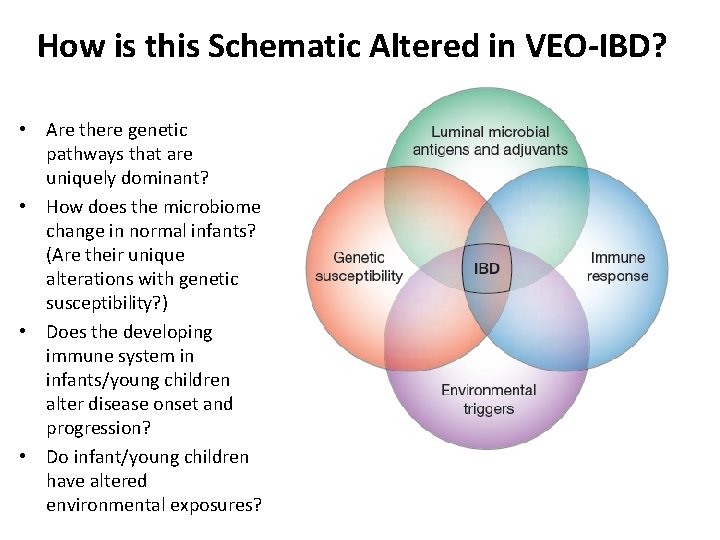
How is this Schematic Altered in VEO-IBD? • Are there genetic pathways that are uniquely dominant? • How does the microbiome change in normal infants? (Are their unique alterations with genetic susceptibility? ) • Does the developing immune system in infants/young children alter disease onset and progression? • Do infant/young children have altered environmental exposures?

Very Early Onset IBD: Mapping the Research Agenda January 10 -12 th, 2013 Royal Netherland Academy of Arts and Science: Principal Investigators Scott B. Snapper, MD, PHD Wolpow Family Chair in IBD Treatment and Research Director, IBD Center, Boston Children’s Hospital Harvard Medical School Aleixo Muise, MD, PHD Christoph Klein, MD, PHD Staff Scientist, Hospital for Sick Children, Chair, Department of Pediatrics, , Ludwig. University of Toronto Maximilians-University Munich, Germany Working Group Members • Judy Cho Director, Inflammatory Bowel Disease Center, Yale University • Hans Clevers Director of the Hubrecht Institute, Utrecht, The Netherlands • Lynn Goldman Dean, School of Public Health and Health Services, George Washington University, DC • Dermot Mc. Govern • Josef Penninger Senior Scientist and Scientific Director, Institute of Molecular Biotechnology, Austria • Fiona Powrie Chair in Gastroenterology, Head of Nuffield Department of Medicine – Experimental Medicine, University of Oxford • David Relman Professor, Department of Microbiology & Immunology, Stanford University School of Medicine, CA Director, Translational Medicine, Inflammatory Bowel Disease Center and Immunobiology Research Institute, Cedars Sinai Hospital, LA • Eric Schadt Professor of Immunobiology; Investigator, Howard Hughes Medical Institute, Yale University • Dan Turner • Ruslan Medzhitov Keynote: Chair of the Department of Genetics and Genomics Sciences, Director of the Institute for Genomics and Multiscale Biology, Mount Sinai School of Medicine, NYC Head of the Pediatric Gastroenterology and Nutrition Unit at Shaare Zedek Hospital, Jerusalem, Israel. Richard Flavell ( Investigator, HHMI; Department Chair, Immunobiology)

NEOPICS Mission Statement: The inter. National Early Onset Paediatric IBD Cohort Study (NEOPICS) brings together international Pediatric Gastroenterologists and Scientists from academic centers across the globe to work together to identify and investigate the causes, and develop new treatments for very young children and infants with IBD. www. neopics. org 37

Acknowledgements Boston Children’s Hospital Athos Bousvaros Chris Moran (MGH) Jeremy Goettel John Garber (MGH) Dror Shouval Jodie Ouahed Katelyn Mc. Cann Jill Plevinsky Lori Ashworth Sarah Weber Ben Atkinson Deanna Nguyen (MGH) Dr. von Hauner Children's Hospital; Ludwig Maximilians University Munich Christoph Klein, M. D. , Ph. D. Daniel Kotlarz Hospital for Sick Children Aleixo Muise, M. D. , Ph. D. Thomas Walters Anne Griffiths Karoline Fiedler Sandeep Dhillon Abdul Elkadri Lucas Mastropaolo University of Toronto Mark Silverberg Emory University Subra Kugathasan Cedar Sinai Dermot Mc. Govern

Thank You Please Visit Our Website: www. NEOPICS. org 39
- Slides: 39