Genetic Causes of Developmental Disability Craig A Erickson

Genetic Causes of Developmental Disability Craig A. Erickson, MD Director, Fragile X Research and Treatment Center Director, Developmental Disabilities Research and Treatment in Psychiatry Medical Director, P 3 Southwest Neurodevelopmental Disorders Inpatient Unit Associate Professor of Psychiatry Cincinnati Children’s Hospital Medical Center Craig. erickson@cchmc. org
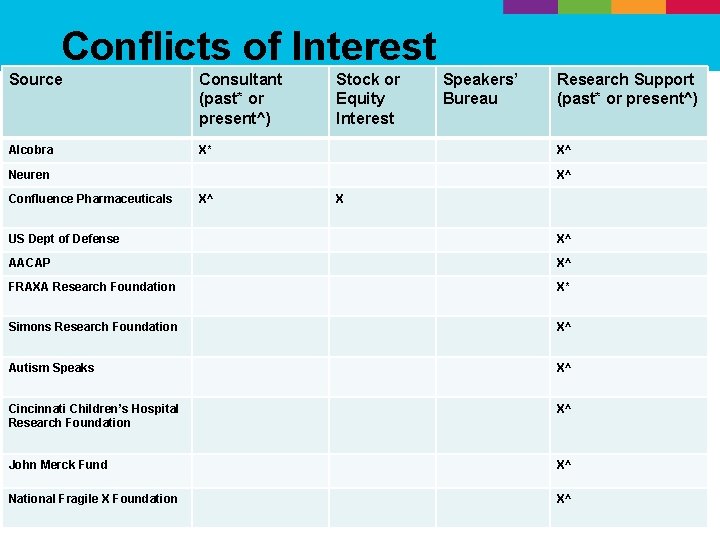
Conflicts of Interest Source Consultant (past* or present^) Alcobra X* Stock or Equity Interest Research Support (past* or present^) X^ Neuren Confluence Pharmaceuticals Speakers’ Bureau X^ X^ X US Dept of Defense X^ AACAP X^ FRAXA Research Foundation X* Simons Research Foundation X^ Autism Speaks X^ Cincinnati Children’s Hospital Research Foundation X^ John Merck Fund X^ National Fragile X Foundation X^

Roadmap: Non Inclusive, Ever Growing • • • Angelman Syndrome Prader Willi Syndrome Down Syndrome Tuberous Sclerosis 22 q 11. 2 Deletion Syndrome (AKA Di. George or Velocardiofacial Syndromes) Phelan Mc. Dermid Syndrome Smith Magenis Syndrome Williams Syndrome Fragile X Syndrome

Angelman Syndrome • • History Genetics Prevalence Clinical Features of AS – – Sleep Seizures Behavior Challenges Autism in Angelman?

History of AS • In 1965, Dr. Harry Angelman described three children with characteristic features now known as AS in a paper entitled “Puppet Children” • Dr. Angelman reports the name “Puppet Children” came to him after viewing a painting called “Boy with a Puppet” • The name of the disorder was changed to Angelman Syndrome in the 1980 s
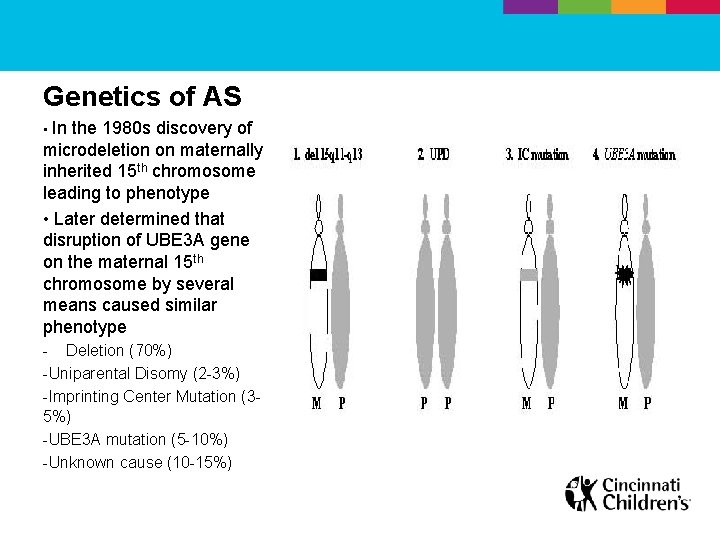
Genetics of AS • In the 1980 s discovery of microdeletion on maternally inherited 15 th chromosome leading to phenotype • Later determined that disruption of UBE 3 A gene on the maternal 15 th chromosome by several means caused similar phenotype - Deletion (70%) -Uniparental Disomy (2 -3%) -Imprinting Center Mutation (35%) -UBE 3 A mutation (5 -10%) -Unknown cause (10 -15%)
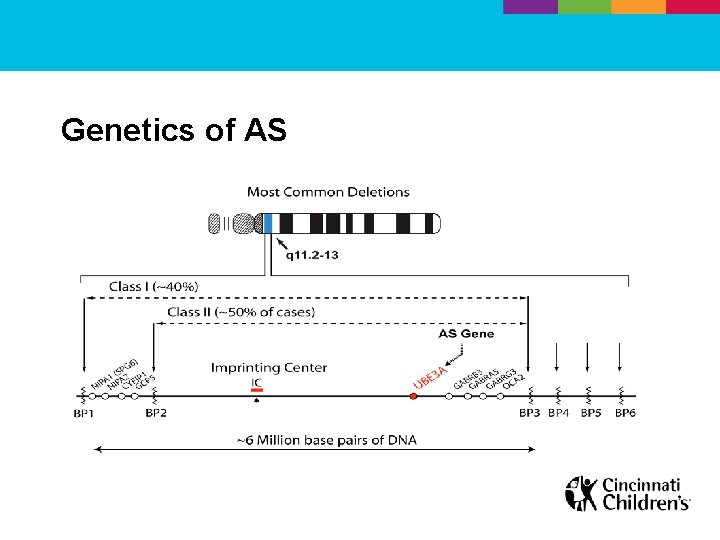
Genetics of AS

Genetics of AS • UBE 3 A encodes E 6 -AP protein, important in the ubiquitin-proteasome pathway – Pathway allows ubiquitin to be attached to other proteins, which allows them to be degraded – UBE 3 A is associated with neuronal synaptic functioning

Prevalence of AS • Occurs in approximately 1/15, 000 individuals • The exact incidence of AS is unknown – Best available data from studies of school age children living in Sweden and Denmark – Diagnosis of AS children in medical clinics was collected over an 8 year period of about 45, 000 births – Swedish study showed an AS prevalence of about 1/12, 000 – Danish study showed a minimum AS prevalence of about 1/10, 000

Clinical Features of AS • • Developmental Delay Speech Impairment Movement or Balance Disorder Unique behavioral features – Frequent laughter/smiling – Easily excitable – Hypermotoric behavior Photo from Maggie’s Family Website: http: //www. sheldonhickey. com/angelman/maggie_medical. html

Clinical Features of AS • Other frequently associated features – – – – Seizures Abnormal EEG even without seizures Abnormal sleep/wake cycles Light hair and eyes Fascination with water and crinkly items Mouthing/chewing behaviors Drooling Constipation

Sleep in AS • Sleep problems are frequent in AS – – – – 20 -80% of individuals with sleep issues Trouble settling Frequent night wakening Contributes to caregiver stress Can be challenging to treat Melatonin (more on this later) Safe sleep spaces

Seizures in AS • • • Onset often prior to age 3 Can be treatment refractory Abnormal EEG Medication management Dietary management – Low glycemic index treatment Photo posted by Kiano’s mommy: http: //www. angelmanforum. org/viewtopic. php? t=2373&sid=6 a 8255 c 64 cfb 2 d 4 f 30912 dd 272 cf 551 f

Behavior Challenges in AS • Hyperactivity/Impulsivity/Inattention – – – Occurs in essentially all children with AS Ceaseless activity, constant movement Limited attention span in childhood Improves with age May respond to behavioral therapy Medications may be used at times

Behavior Challenges in AS • Disruptive/Aggressive Behavior – May arise more commonly in adolescents with AS – Multiple causes • Impulsivity • Communication deficits – Treatment primarily behavioral – Medication management in severe case – Look for upcoming module addressing this topic on ASF website

Autism in AS? • AS sometimes described as a genetic form of autism • Overlapping features including hand-flapping, stereotypic behaviors, language delays • To date no consensus on prevalence in AS • Evidence that duplication of chromosome 15 q 11 -13 is associated with autism • Likely that autism effects a small percentage of individuals with AS

Prader Willi Syndrome • Disruption, most commonly deletion, of the paternal copy of the same region of chromosome 15 involved in Angelman Syndrome – Spontaneous event/ non inherited • Associated with mild to moderate intellectual disability

Prader Willi Syndrome • Behavioral Features – Temper outbursts/stubbornness – Compulsive behavior- skin picking • Physical/Medical Features – Sleep Disturbances – Facial Features • Narrow forehead • Almond-shaped Eyes • Triangular Mouth – Fair skin/light-colored hair – Underdeveloped genitals • Delayed/incomplete puberty, infertile

Prader Willi Syndrome • Obesity – Childhood onset insatiable appetite • Chronic over eating – Many develop Type 2 Diabetes • Prevalence: 1 in about 10, 000 worldwide • Practical Issues – Weight management- lock the fridge – Repetitive behavior – Some irritability

Down Syndrome • Occurs when an individual has a full or partial extra copy of chromosome 21 • Most common genetic form of developmental disability – 1 in 691 babies in the USA is born with Down Syndrome – More than 400, 000 persons with Down Syndrome in the USA

Down Syndrome • Increased risk of Down Syndrome with increasing maternal age – Due to higher fertility rates in younger women 85% of children with Down Syndrome are born to women under age 35 years • Most common genetic form of developmental disability – 1 in 691 babies in the USA is born with Down Syndrome – More than 400, 000 persons with Down Syndrome in the USA

Down Syndrome • Increased risk of the following medical conditions: – – – Congenital heart defects Respiratory and hearing deficits Alzheimer’s disease Childhood leukemia Hypothyroidism

Down Syndrome • Physical Features – – Low Muscle Tone Small Stature Upward slant of the eyes Single deep palmar crease

Down Syndrome • Generally mild to moderate cognitive delay • Increased risk for autism in Down Syndrome even when controlling for IQ – Seems counter intuitive given the highly social nature of many persons with Down Syndrome • Life expectancy has increased from 25 in 1983 to 60 years of age today • In mental health, we primarily treat associated anxiety, depression and agitation

Tuberous Sclerosis • Caused by mutations in the TSC 1 or TSC 2 genes – Code for the proteins hamartin and tuberin respectively • Proteins help regulate cellular growth • Act as tumor suppressors • Inheritance: – 2/3 cases spontaneous mutations – 1/3 cases inherited from parent: autosomal dominant inheritance

Tuberous Sclerosis • Prevalence: 1 in 6, 000 people • Developmental Issues: borderline IQ to mild/moderate ID, risk for autism – Variable presentation • Medical Issues – Numerous benign tumors • Skin, brain kidney, other organs • Requires frequent monitoring • Facial angiofibromas

Tuberous Sclerosis • Frequent Seizures • Behavioral concerns: hyperactivity and aggression • Targeted treatment development – Sirolimus, m. Tor inhibitor • May reduce tumor growth • Can improve seizure outcome

22 q 11. 2 Deletion Syndrome • Also known as Di. George or Velocardiofacial Syndrome • Variable presentations with deletions in this chromosomal region • Many children have developmental delays, speech delay, and growth delays • Increased likelihood of ADHD and possible autism • Impacts 1 in 4, 000 persons

22 q 11. 2 Deletion Syndrome • Autosomal dominant disorder – Most cases, though, from random events – 10% cases are inherited • In adult life, increased risks of depression, anxiety, bipolar disorder and schizophrenia

22 q 11. 2 Deletion Syndrome • Common medical features – – – – – Heart abnormalities Cleft palate Recurrent infections/immune system dysfunction Low levels of calcium in blood (can cause seizures) Autoimmune disorders such as Graves disease or rheumatoid arthritis Breathing difficulties Kidney abnormalities Hearing Loss GI problems and feeding challenges Skeletal abnormalities/short stature

Phelan Mc. Dermid Syndrome • Due to chromosome 22 q 13. 3 deletion – Disrupts the SHANK 3 gene • Behavioral and Cognitive Phenotype: – – High risk for autism Moderate to profound intellectual disability Delayed speech Pica

Phelan Mc. Dermid Syndrome • Physical/Medical Phenotype – Reduced sensitivity to pain – Possible reduced ability to sweat- prone to overheating – Risk of cyclic vomiting – Risk of gastroesophageal reflux (GERD) – Long narrow head, prominent ears, pointed chin, ptosis, deep-set eyes – Large hands and feet – Some have rapid growth – Fusion of second and third toes – Small toenails

Phelan Mc. Dermid Syndrome • Exact prevalence unknown – Considering an increasingly well known genetic cause of autism spectrum disorder – Most cases are not inherited and occur spontaneously • Subject of targeted clinical drug trials – – – Utilizing Insulin Growth Factor 1 (IGF 1) treatment High risk for autism Moderate to profound intellectual disability Delayed speech Pica

Smith-Magenis Syndrome • Caused by a genetic deletion on chromosome 17 disrupting the RAI 1 gene • Not inherited • Impacts 1 in 15, 000 persons • Physical features – Prominent lower jar, deep-set eyes, flattened nasal bridge, downward turning mouth – Dental abnormalities and large tongue – Short Stature and scoliosis – Reduced sensitivity to pain and temperature – Hearing loss – Near sightedness

Smith-Magenis Syndrome • Behavioral, developmental and other features – Disturbed sleep pattern: day/night reversed – Often affectionate and engaging – Prone to tantrums, aggression, anxiety, self-injury, impulsivity, and inattention – Possible repetitive hugging and/or licking of fingers and flipping of pages – Mild to profound intellectual disability

William Syndrome • Caused by deletion on chromosome 7 in the 26 to 28 region – Disruption of several genes in this region • Most cases are not inherited – Impacts 1 in 7, 500 persons • Impacts 1 in 15, 000 persons • Mild to moderate intellectual disability • Outgoing engaging personalities with an extreme interest in other people

William Syndrome • Other behavioral/cognitive features – Difficulties with visual spatial tasks – ADHD, anxiety and phobias are common • Facial Features – – Broad Forehead Short nose with broad tip Full cheeks Wide mouth with full lips

William Syndrome • Other medical features – Dental problems • Small, widely spaced and at times missing teeth – – – Aortic stenosis: narrowing of large blood vessal High blood pressure Joint problems and soft, loose skin Increased calcium levels in blood Coordination difficulties Short stature

Fragile X Syndrome: History • 1943: Originally reported by Martin and Bell as “mental deficiency showing sex-linkage” • 1969: Discovery on X chromosome of “Fragile” site that fractured in folate-deficient medium • 1991: Cloning and characterization of the fragile X mental retardation gene (FMR 1) – Gene sequencing indicated that a portion of the gene was dramatically expanded • 2004: Metabotropic glutamate receptor theory of fragile X – Theory has led to large wave of ongoing translational treatment development efforts 39

Triplet Repeats • Cytosine, guanine (CGG) repeat expansion noted near promoter of FMR 1 gene – Net effect of expansion is gene methylation with little FMR 1 RNA produced or protein (Fragile X Mental Retardation Protein (FMRP)) translated – Gene is effectively silenced 40
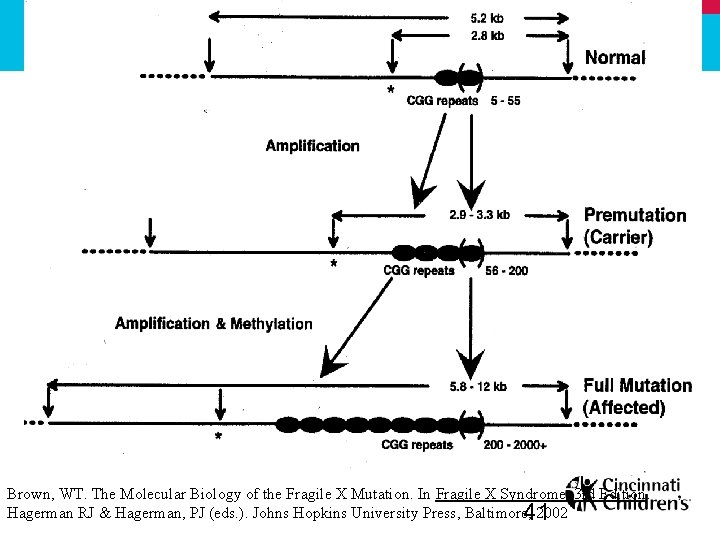
Brown, WT. The Molecular Biology of the Fragile X Mutation. In Fragile X Syndrome, 3 rd Edition. Hagerman RJ & Hagerman, PJ (eds. ). Johns Hopkins University Press, Baltimore, 2002 41

Full Mutation Fragile X Syndrome (FXS): Epidemiology • Males: Approximately 1 in 4000 – 50% likely remain undiagnosed • Females: Approximately 1 in 6000 -8000 – Variable phenotype given random X inactivation patterns ACOG committee opinion. No. 338: Screening for fragile X syndrome. " Obstet Gynecol 107(6): 14831485. 42

Full Mutation FXS: Epidemiology • Most common inherited cause of intellectual disability (ID) – 2 -6% of persons with intellectual disability have FXS; ID universal among males • Most common known single gene cause of autistic disorder – 2 -3% cases of autism associated with FXS – 2 in 3 males with FXS meet additional criteria for an autism spectrum disorder Hatton, D. D. , J. Sideris, et al. (2006). "Autistic behavior in children with fragile X syndrome: prevalence, stability, and the impact of FMRP. " Am J Med Genet A 140 A(17): 1804 -1813. 43

Full Mutation: Common Physical Features/Medical Comorbidities • Long face • Prominent Ears • High-arched palate • Hyperextensible joints • Heart murmur/mitral valve prolapse • Strabismus • Flat feet • Chronic otitis media (38 -63%) • Macroorchidism in males (83 -92%) • Seizure Disorder (10 -20%) 44
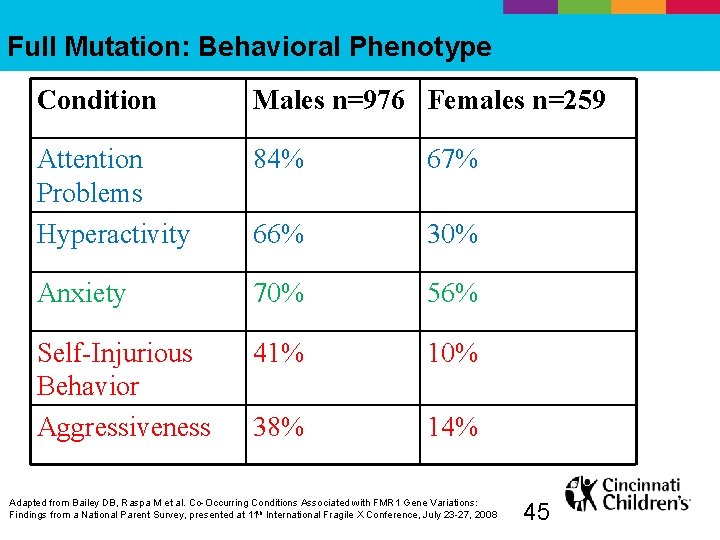
Full Mutation: Behavioral Phenotype Condition Males n=976 Females n=259 Attention Problems Hyperactivity 84% 67% 66% 30% Anxiety 70% 56% Self-Injurious Behavior Aggressiveness 41% 10% 38% 14% Adapted from Bailey DB, Raspa M et al. Co-Occurring Conditions Associated with FMR 1 Gene Variations: Findings from a National Parent Survey, presented at 11 th International Fragile X Conference, July 23 -27, 2008 45

Full Mutation: Behavioral Phenotype Classic Gaze Aversion of Fragile X www. fragilex. org 46

Fragile X: Indications for Testing • Persons with intellectual and developmental disabilities/delays and/or autistic behaviors • Features consistent with premutation carrier pathology (beyond scope of this talk) – Adult women with features of early menopause – Older adults with gait ataxia, intention tremor or both – 1 in 209 females is a FXS premutation carrier Hagerman RJ & Hagerman PJ, Testing for fragile X gene mutations throughout the life span. JAMA. 300: 20, 2419 -2421 Tassone, F. , K. P. Iong, et al. (2012). "FMR 1 CGG allele size and prevalence ascertained through newborn screening in the United States. " Genome Med 4(12): 100. 47

Fragile X Testing: The Future • Universal Newborn Screening – Need to have inexpensive testing: likely less than $1 per test – Now testing is available using the “blood spot card” • Still somewhat costly • Two large-scale newborn screening studies ongoing in United States • Push driven by prospect of disease modifying drugs and need for early genetic counseling Tassone, F. , K. P. Iong, et al. (2012). "FMR 1 CGG allele size and prevalence ascertained through newborn screening in the United States. " Genome Med 4(12): 100. 48

Genetic Testing • Everyone with developmental disability and/or autism needs the following – – Chromosomal microarray testing General chromosomes Testing for Fragile X Syndrome THE FUTURE: • Whole Exome Sequencing – Discovering the many, many disorders not yet discovered – Finding many rare syndromes and disorders where diagnosis was previously not feasible 49

Genetic Testing • Challenges – Insurance, in particular medicaid, coverage – What is “medically necessary”? – Is finding out your child has a disorder important to their “medical treatment” – An ongoing battle 50

Future Understanding of Developmental Disability • In large part may hinge on enhanced genetic understanding – Finding more and more likely rare phenomena that cause DD and often MI – Key element is enhanced availability of state of the art genetic testing – Enhanced genetic knowledge spurs on targeted treatment development • • Fragile X Phelan Mc. Dermid Syndrome Rett Syndrome Others 51

Questions • Craig. erickson@cchmc. org
- Slides: 52