General Reactions Dr Ron Rusay General Chemical Reactions



















- Slides: 21

General Reactions Dr. Ron Rusay

General Chemical Reactions Any chemical reaction can be described as a molecular or atomic change. It produces one or more observable changes. e. g. color change, gas bubbles, heat, etc. Reactions are generally described as Reactant(s) Product(s) The reaction is written as a chemical equation with chemical formulas: 2 Na + Cl 2 2 Na. Cl ¥

The Reaction of Sodium & Chlorine

Chemical Equations Chemical equations describe the change(s) in Reactant(s) to Product(s) including physical state(s). Notations: (g), (l), (s), (aq) 2 Na (s) + Cl 2(g) 2 Na. Cl (s) Others: arrows for gas ( ) and solid ( ) for heat, for reversibility (equilibrium) ·

Types of Chemical Reactions Combination (Synthesis) Decomposition Single Displacement Double Displacement Combustion Others: Precipitation, Oxidation. Reduction, Neutralization

General Chemical Reactions

Chemical Reactions Combination (Synthesis) A +B C Example: • H 2 ( g) + O 2 ( g) H 2 O (g ) Balancing (Conservation of Atoms): 2 H 2 (g) + O 2 (g) 2 H 2 O (g)

Synthesis of Water Three Balloons: https: //www. youtube. com/watch? v=a 6 q. GIMq. DKw. A&index=4&list =PLA 3383 CE 72437 FC 43

An Unwanted Synthesis of Water Combustion & the Hindenburg 1937

Chemical Reactions Decomposition A B+C 2 NI 3 (s) N 2 (g) + 3 I 2 (s)

Decomposition Another • • example: Potassium chlorate (s) + heat potassium chloride (s) + oxygen (g) Balanced equation: ? 2 KCl. O 3 (s) 2 KCl (s) + 3 O 2 (g)

QUESTION Ammonium nitrate, when heated, decomposes into nitrogen gas, oxygen gas, and water vapor. What is the sum of the coefficients in the balanced equation using smallest integer coefficients? A) 3 B) 5 C) 7 D) 9

ANSWER Ammonium nitrate, when heated, decomposes into nitrogen gas, oxygen gas, and water vapor. What is the sum of the coefficients in the balanced equation using smallest integer coefficients? A) 3 B) 5 2 NH 4 NO 3 (s) C) 7 D) 9 2 N 2 (g)+ 1 O 2 (g) + 4 H 2 O (g)

Chemical Reactions ð Single Displacement ð AB + C CB + A ð Example: • • HCl (aq) + Mg (s) Mg. Cl 2 (aq) + H 2 (g) Balanced Equation: ? 2 HCl (aq) + Mg (s) Mg. Cl 2 (aq) + H 2 (g)

Single Displacement (Oxidation / Reduction) ð Write a balanced equation for the following reaction:

Chemical Reactions Double Displacement AB + CD AD + CB Example: • • A solution of sodium phosphate reacts with a solution of silver nitrate to produce aqueous sodium nitrate and a precipitate of silver phosphate. Balanced equation: ? Na 3 PO 4 (aq) + 3 Ag. NO 3 (aq) Ag 3 PO 4 (s) + 3 Na. NO 3 (aq)

Chemical Equations • Combustion: C 8 H 18(l)+ O 2(g) Oxygen reacts with octane to produce carbon dioxide and water. Reminder: CO 2(g) + H 2 O(l) the equation must balance: 2 C 8 H 18(l)+ 25 O 2(g) 16 CO 2(g)+18 H 2 O(l)

QUESTION

ANSWER D) 13 2 C 8 H 18 (l)+ 25 O 2(g) 16 CO 2(g)+18 H 2 O(l) 2 C 4 H 10 (l)+ 13 O 2(g) 8 CO 2(g)+ 10 H 2 O(l) O 2 should be balanced last since it contains only one type of element and balancing it will not cause an imbalance in another element.

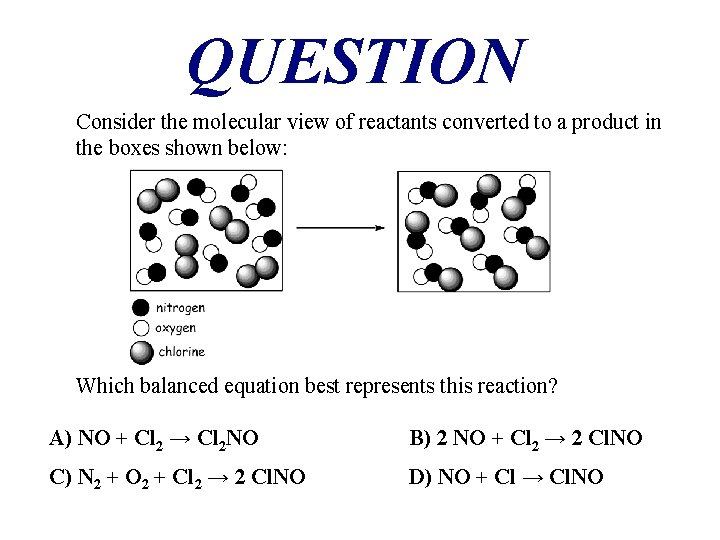
QUESTION Consider the molecular view of reactants converted to a product in the boxes shown below: Which balanced equation best represents this reaction? A) NO + Cl 2 → Cl 2 NO B) 2 NO + Cl 2 → 2 Cl. NO C) N 2 + O 2 + Cl 2 → 2 Cl. NO D) NO + Cl → Cl. NO

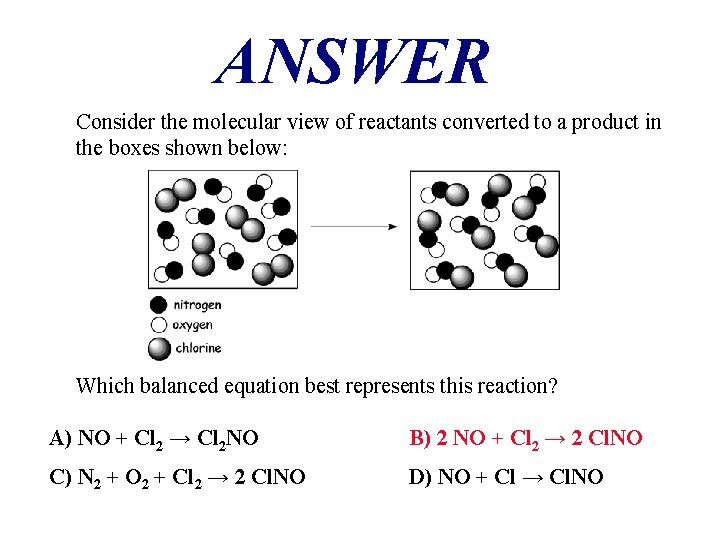
ANSWER Consider the molecular view of reactants converted to a product in the boxes shown below: Which balanced equation best represents this reaction? A) NO + Cl 2 → Cl 2 NO B) 2 NO + Cl 2 → 2 Cl. NO C) N 2 + O 2 + Cl 2 → 2 Cl. NO D) NO + Cl → Cl. NO