General Reactions Dr Ron Rusay General Chemical Reactions

















































- Slides: 60

General Reactions Dr. Ron Rusay

General Chemical Reactions ¥ Any chemical reaction can be described as a molecular or atomic change. It produces one or more observable changes. � e. g. color change, gas bubbles, heat, etc. � Reactions are generally described as: Reactant(s) Product(s)

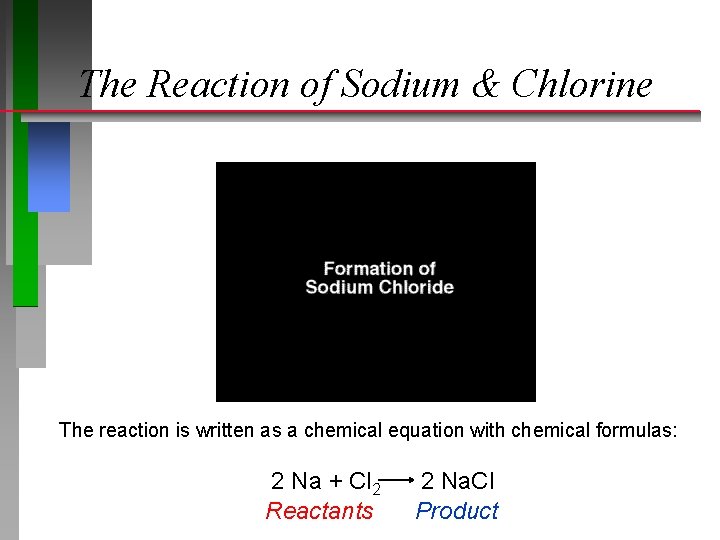
The Reaction of Sodium & Chlorine The reaction is written as a chemical equation with chemical formulas: 2 Na + Cl 2 Reactants 2 Na. Cl Product

Chemical Equations Chemical equations describe the change(s) in Reactant(s) to Product(s) including physical state(s). � Notations: (g), (l), (s), (aq)

Types of Chemical Reactions � Combination (Synthesis) � Decomposition � Single Displacement � Double Displacement � Combustion: Oxidation-Reduction _________ � Biological Reactions: Enzyme Catalysts Example: Fermentation http: //www. piney. com/Bab. Ninkasi. html)

General Chemical Reactions http: //chemconnections. org/general/movies/rxn-types. mov

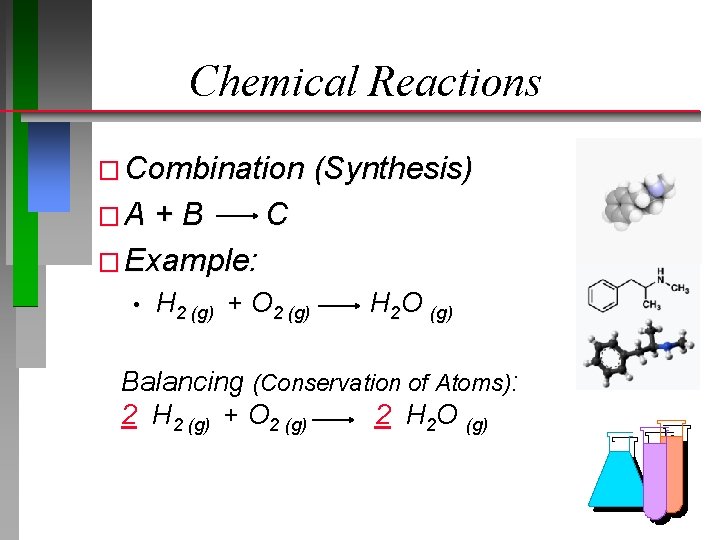
Chemical Reactions � Combination (Synthesis) �A +B C � Example: • H 2 (g) + O 2 (g) H 2 O (g) Balancing (Conservation of Atoms): 2 H 2 (g) + O 2 (g) 2 H 2 O (g)

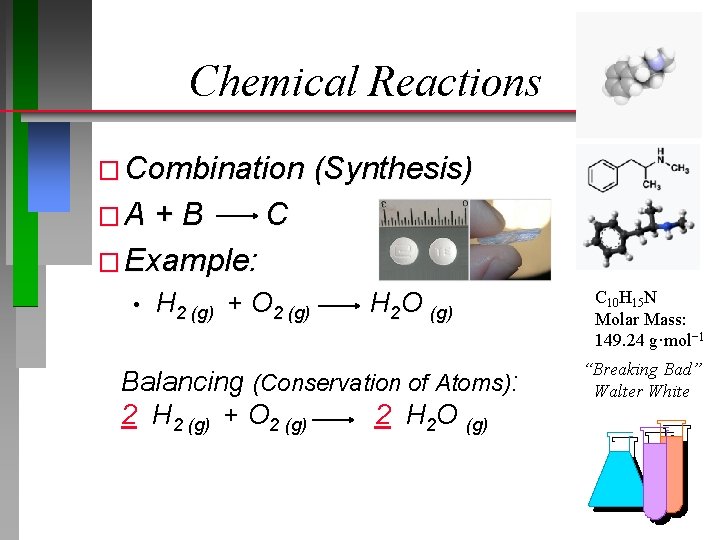
Chemical Reactions � Combination (Synthesis) �A +B C � Example: • H 2 (g) + O 2 (g) H 2 O (g) Balancing (Conservation of Atoms): 2 H 2 (g) + O 2 (g) 2 H 2 O (g) C 10 H 15 N Molar Mass: 149. 24 g·mol− 1 “Breaking Bad” Walter White

Synthesis of Water http: //chemconnections. org/general/movies/H 2 O-form. MOV Three Balloons: https: //www. youtube. com/watch? v=a 6 q. GIMq. DKw. A&index=4&list=PLA 3383 CE 72437 FC 43

An Unwanted Synthesis of Water Combustion & the Hindenburg 1937

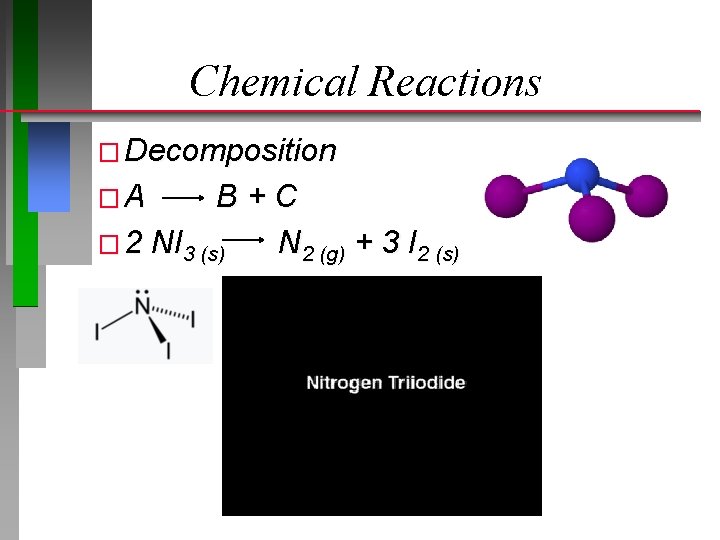
Chemical Reactions � Decomposition �A B+C � 2 NI 3 (s) N 2 (g) + 3 I 2 (s)

QUESTION Ammonium nitrate, when heated, decomposes into nitrogen gas, oxygen gas, and water vapor. What is the sum of the coefficients in the balanced equation using smallest integer coefficients? A) 3 B) 5 C) 7 D) 9

ANSWER Ammonium nitrate, when heated, decomposes into nitrogen gas, oxygen gas, and water vapor. What is the sum of the coefficients in the balanced equation using smallest integer coefficients? A) 3 2 NH 4 NO 3 (s) B) 5 C) 7 D) 9 2 N 2 (g)+ 1 O 2 (g) + 4 H 2 O (g)

Chemical Reactions ð ð ð Single Displacement AB + C CB + A Example: • • HCl (aq) + Mg (s) Mg. Cl 2 (aq) + H 2 (g) Balanced Equation: ? 2 HCl (aq) + Mg (s) Name HCl(aq) ? Mg. Cl 2 (aq) + H 2 (g) Hydrochloric acid

Single Displacement ð Write a balanced equation for the following reaction: Cu(s) + 2 Ag. NO 3(aq) 2 Ag(s) + Cu(NO 3)2 (aq)

Molecular Modeling (Individual or Collaborative) Report Form (Replacement pages for Molecular Model Lab pp. 97 -103) http: //chemconnections. org/general/chem 108/Chemistry%20108%20 Molecular%20 Modeling%20 Form%20 F all%202017. pdf Turn-in individually or one per group Due TODAY

Chemical Reactions � Double Displacement � AB + CD AD + CB � Example: • • A solution of sodium phosphate reacts with a solution of silver nitrate to produce aqueous sodium nitrate and a precipitate of silver phosphate. Balanced equation: ? Na 3 PO 4 (aq) + 3 Ag. NO 3 (aq) Ag 3 PO 4 (s) + 3 Na. NO 3 (aq)

Chemical Reactions Predict the products: H 2 SO 4(aq) + Na. OH(aq) → H 2 O(l) + Na 2 SO 4(aq) Balance the equation: H 2 SO 4(aq) + 2 Na. OH(aq) → 2 H 2 O(l) + Na 2 SO 4(aq)

Post lab https: //phet. colorado. edu/sims/html/balancing-chemical-equations/latest/balancing-chemicalequations_en. html

Chemical Equations • Combustion / Oxidation-Reduction: C 8 H 18(l)+ O 2(g) CO 2(g) + H 2 O(l) � Oxygen reacts with octane to produce carbon dioxide and water. � Reminder: the equation must balance: 2 C 8 H 18(l)+ 25 O 2(g) � 16 CO 2(g)+18 H 2 O(l) Carbon is oxidized: looses 4 electrons Oxygen is reduced: gains 2 electrons

QUESTION

ANSWER D) 13 2 C 8 H 18 (l)+ 25 O 2(g) 16 CO 2(g)+18 H 2 O(l) 2 C 4 H 10 (l)+ 13 O 2(g) 8 CO 2(g)+ 10 H 2 O(l) O 2 should be balanced last since it contains only one type of element and balancing it will not cause an imbalance in another element.

Combustion: Oxidation-Reduction Reactions “Redox” 1 CH 4 (g)+ 2 O 2(g) 1 CO 2(g)+ 2 H 2 O(l) 2 C 4 H 10 (g)+ 13 O 2(g) 8 CO 2(g)+ 10 H 2 O(l) 2 C 8 H 18 (l)+ 25 O 2(g) 16 CO 2(g)+18 H 2 O(l) Carbon is oxidized: loosing 4 electrons Oxygen is reduced: gaining 2 electrons

Combustion Products Formulas & Multiple Proportions http: //chemconnections. org/general/movies/multiple-proportions. MOV Carbon can also be oxidized by loosing 2 electrons Oxygen remains reduced by gaining 2 electrons 2 CH 4 (g)+ 3 O 2(g) 1 CH 4 (g)+ 2 O 2(g) 2 COg)+ 4 H 2 O(l) 1 CO 2(g)+ 2 H 2 O(l)

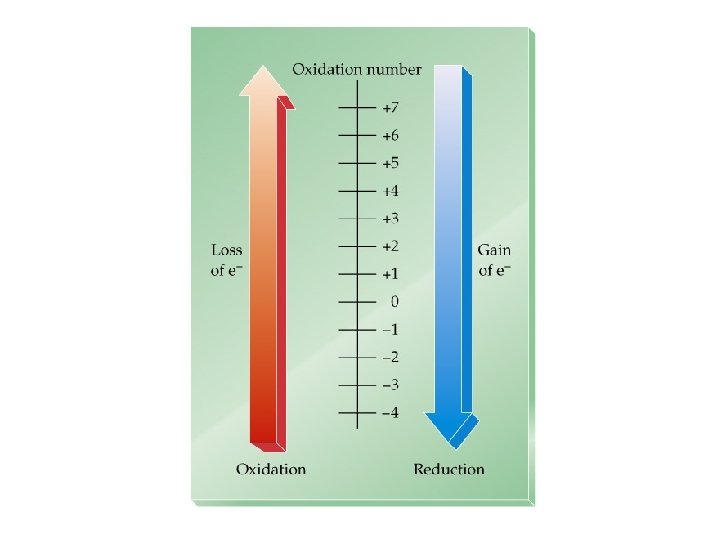
Oxidation-Reduction Reactions “Redox”: Changes in Oxidation State ð ð Oxidation is the loss of electrons. Reduction is the gain of electrons. The reactions occur together. One does not occur without the other. The terms are used relative to the change in the oxidation state or oxidation number of the reactant(s).

Oxidation Number (State) http: //chemconnections. org/general/movies/Oxid%20 States. MOV


Tutorial https: //www. youtube. com/watch? v=w 0 Rf. MRDy 34 w

Tutorial https: //www. youtube. com/watch? v=w 0 Rf. MRDy 34 w

QUESTION

ANSWER B) NO 2 Oxygen has an oxidation state of – 2 when part of a compound.

QUESTION (More Challenging) What is the oxidation number of chromium in ammonium dichromate? A) +3 Formula: ? B) +4 C) +5 (NH 4)2 Cr 2 O 7 D) +6

ANSWER What is the oxidation number of chromium in ammonium dichromate? A) +3 B) +4 C) +5 2 x + 7(-2) = -2 Cr 2 O 722 x = +14 -2 x = 12/2 D) +6

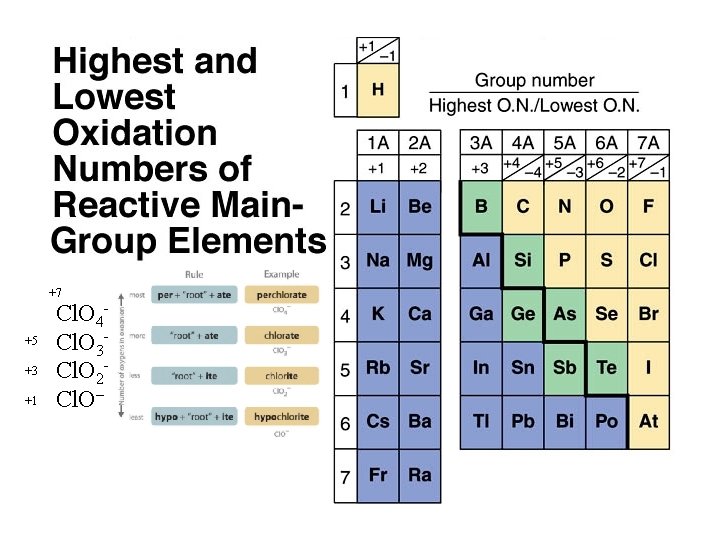
+7 +5 +3 +1 Cl. O 4– Cl. O 3– Cl. O 2– Cl. O–

QUESTION Hypochlorous acid is related to the anion found in common household bleach. Identify the formula of the polyatomic anion. A. B. C. D. Cl. O 4– Cl. O 3– Cl. O 2– Cl. O– https: //chem. libretexts. org/

Answer Hypochlorous acid is related to the anion found in common household bleach. Identify the formula of the polyatomic anion. A. B. C. D. Cl. O 4– Cl. O 3– Cl. O 2– Cl. O– What are the different oxidation numbers for chlorine in choices A. thru D. ? https: //chem. libretexts. org/


QUESTION In a redox reaction, oxidation and reduction must both occur. Which statement is an accurate statement? A. The substance (atom) that is oxidized has a lower oxidation number in the product. B. The substance that is oxidized gains electrons. C. The substance that is oxidized must have a higher oxidation number afterwards. D. The substance that is oxidized must combine with oxygen.

ANSWER In a redox reaction, oxidation and reduction must both occur. Which statement is an accurate statement? A. The substance (atom) that is oxidized has a lower oxidation number in the product. B. The substance that is oxidized gains electrons. C. The substance that is oxidized must have a higher oxidation number afterwards. D. The substance that is oxidized must combine with oxygen.

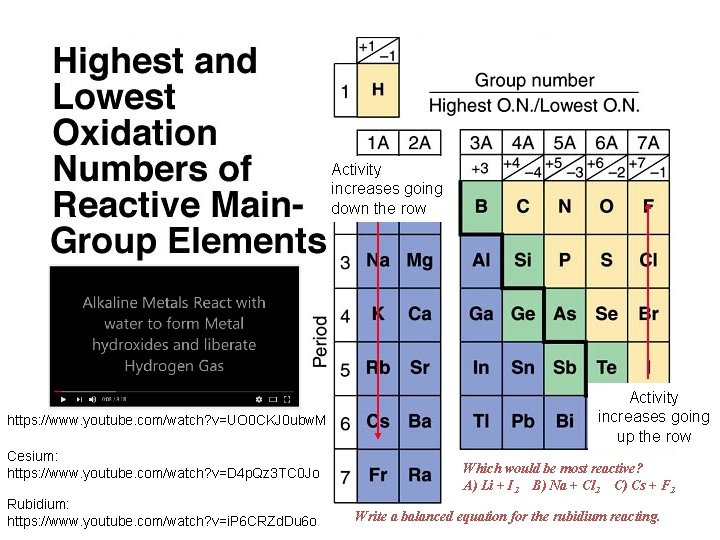
Activity increases going down the row https: //www. youtube. com/watch? v=UO 0 CKJ 0 ubw. M Cesium: https: //www. youtube. com/watch? v=D 4 p. Qz 3 TC 0 Jo Rubidium: https: //www. youtube. com/watch? v=i. P 6 CRZd. Du 6 o Activity increases going up the row Which would be most reactive? A) Li + I 2 B) Na + Cl 2 C) Cs + F 2 Write a balanced equation for the rubidium reacting.

QUESTION

ANSWER C) 2 0 0 Cu(s) + 2 Ag. NO 3(aq) 2 Ag(s) + Cu(NO 3)2(aq) 0 0 N 2(g) + 3 H 2(g) 2 NH 3(g) If an element is a reactant or product, it is a redox reaction. The oxidation number = 0.

QUESTION Cu(s) + 2 Ag. NO 3(aq) 2 Ag(s) + Cu(NO 3)2(aq) TRUE (A) / FALSE (B) Copper is oxidized in the reaction and silver is reduced.

0 +1 Answer 0 +2 Cu(s) + 2 Ag. NO 3(aq) 2 Ag(s) + Cu(NO 3)2(aq) TRUE (A) / FALSE (B) Copper is oxidized in the reaction and silver is reduced.

Question 0 +1 0 +2 Cu(s) + 2 Ag. NO 3(aq) 2 Ag(s) + Cu(NO 3)2(aq) TRUE (A) / FALSE (B) Copper looses 2 electrons in the reaction and each silver atom gains 1 electron.

Answer 0 +1 0 +2 Cu(s) + 2 Ag. NO 3(aq) 2 Ag(s) + Cu(NO 3)2(aq) TRUE (A) / FALSE (B) Copper looses 2 electrons in the reaction and each silver atom gains 1 electron.

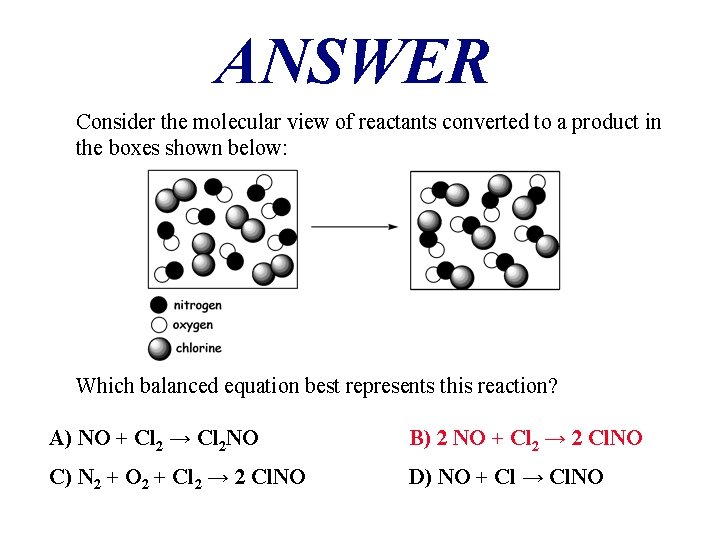
QUESTION Consider the molecular view of reactants converted to a product in the boxes shown below: Which balanced equation best represents this reaction? A) NO + Cl 2 → Cl 2 NO B) 2 NO + Cl 2 → 2 Cl. NO C) N 2 + O 2 + Cl 2 → 2 Cl. NO D) NO + Cl → Cl. NO

ANSWER Consider the molecular view of reactants converted to a product in the boxes shown below: Which balanced equation best represents this reaction? A) NO + Cl 2 → Cl 2 NO B) 2 NO + Cl 2 → 2 Cl. NO C) N 2 + O 2 + Cl 2 → 2 Cl. NO D) NO + Cl → Cl. NO


Molecular Modeling (Individual or Collaborative) Report Form (Replacement pages for Molecular Model Lab pp. 97 -103) http: //chemconnections. org/general/chem 108/Chemistry%20108%20 Molecular%20 Modeling%20 Form%20 F all%202017. pdf Computers & Internet available in PS 110, if needed http: //molview. org Turn-in individually or one per group Due Today

Moles / Molar Mass and Molecular Formulas i-clicker QUIZ QUESTIONS http: //chemconnections. org/general/chem 108/Moles-Molar Mass Quiz. html Submit individually on-line Refer to Calendar & Resources pages for links

Molecular Shapes Quiz) http: //chemconnections. org/general/chem 108/Molecular%20 Shapes%20 Quiz. htm l Submit individually on-line DUE Friday

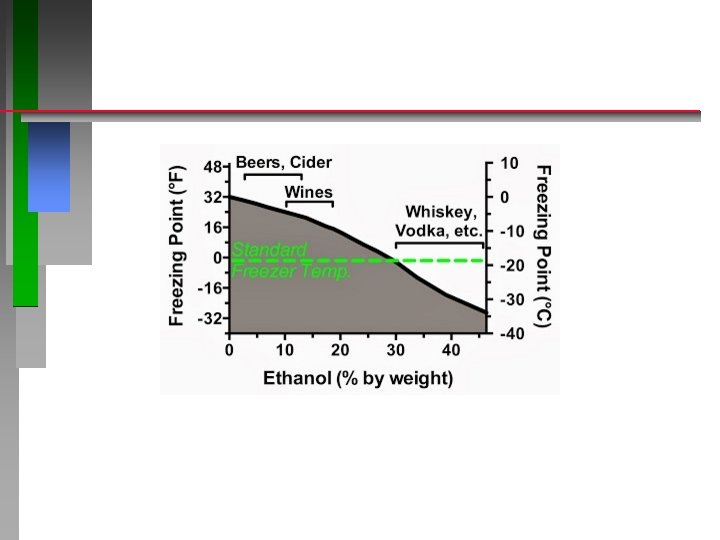
Biological Reactions eg. Fermentation https: //www. youtube. com/watch? v=v. W 99 JEa. DAp. Y http: //www. piney. com/Bab. Ninkasi. html

https: //www. youtube. com/watch? v=OKRJ 73 Ro 5 jw https: //www. youtube. com/watch? v=r. L 14 u. Lb. SLLc

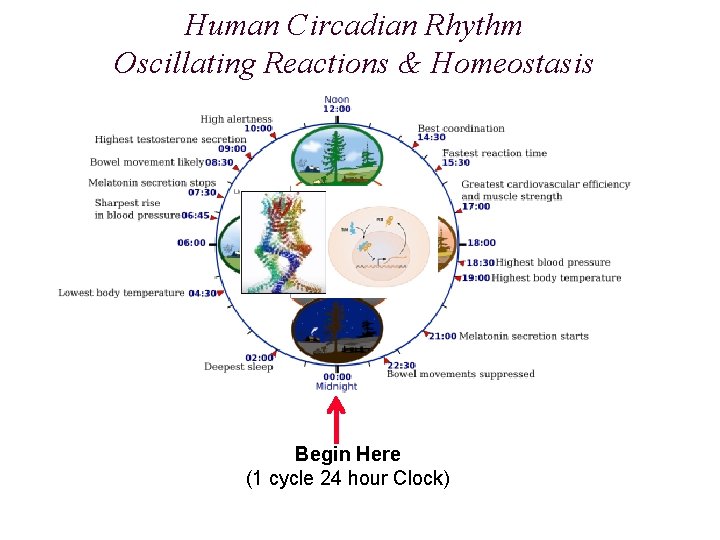
Human Circadian Rhythm Oscillating Reactions & Homeostasis Begin Here (1 cycle 24 hour Clock)


General Chemical Reactions ¥ � � � Any chemical reaction can be described as a molecular or atomic change. It produces one or more observable changes. e. g. color change, gas bubbles, heat, etc. Reactions are generally described as Reactant(s) Product(s) The reaction is written as a chemical equation with chemical formulas: 2 Na + Cl 2 2 Na. Cl

The Reaction of Sodium & Chlorine

Chemical Equations Chemical equations describe the change(s) in Reactant(s) to Product(s) including physical state(s). � Notations: (g), (l), (s), (aq) � 2 Na (s) + Cl 2(g) 2 Na. Cl (s) � Others: arrows for gas ( ) and solid ( ) for heat, for reversibility (equilibrium)

QUESTION