General properties ACIDS Taste sour Turn litmus blue


General properties ACIDS • Taste sour • Turn litmus blue to red • Reacts with active metals – Fe, Zn, Mg • • • React with bases to form salts and water • Hydronium Ion present (H 3 O)+ BASES Taste bitter Turn litmus red to blue Feel soapy or slippery (react with fats to make soap) React with acids to form salts and water Hydroxide Ion present (OH)-

Acid/Base Definitions Arrehenius only in water • Acids – produce H+ • Bases - produce OH- • Acids – donate H+ Bronsted-Lowry + • Bases – accept H any solvent Lewis • Acids – accept e- pair • Bases – donate e- pair used in organic chemistry, wider range of substances

Dissociation of Strong Acids & Bases Strong acids & bases are assumed to dissociate (breakdown into ionic parts) completely in solution. http: //www. chembio. uoguelph. ca/educmat/chm 19104/chemtoons 1. htm
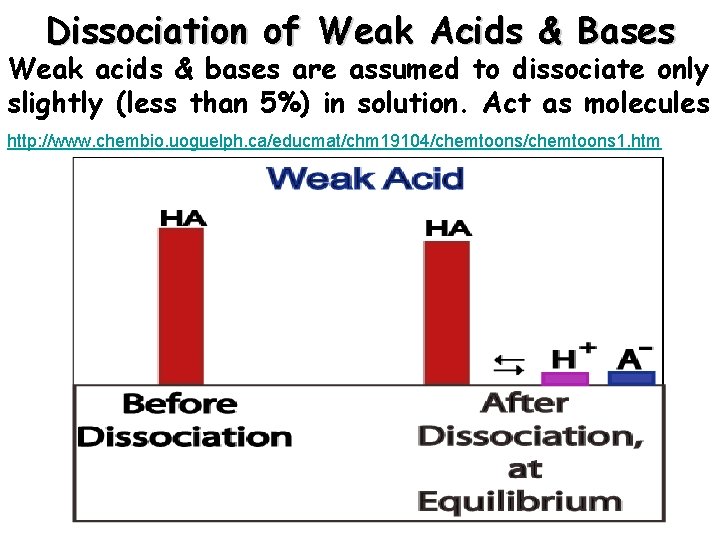
Dissociation of Weak Acids & Bases Weak acids & bases are assumed to dissociate only slightly (less than 5%) in solution. Act as molecules http: //www. chembio. uoguelph. ca/educmat/chm 19104/chemtoons 1. htm

Hydrochloric Acid (Strong) Hydrobromic Acid (Strong) Carbonic Acid (Strong) Sulfuric Acid (Strong) Nitric Acid (Strong) Acetic Acid (Weak) Phosphoric Acid (Weak) Citric Acid (Weak) Lactic Acid (Weak) HCl HBr Muriatic Acid - common name; Toilet bowl cleaners, masonry cleaners, gastric juice H 2 CO 3 Carbonated water; club soda - soft drinks, blood buffer H 2 SO 4 Battery Acid; lead battery, air pollutant mist, acid rain HNO 3 HC 2 H 3 O 2 H 3 PO 4 Remove warts, causes yellow stains on skin Vinegar; cooking, salads Acidify soft drinks, in ATP and DNA H 3 C 6 H 8 O 6 Fruit juices HC 3 H 5 O 3 Builds up in muscles during exercise

Sodium Hydroxide (Strong) Barium Hydroxide (Strong) Na. OH Ba(OH)2 Lye, Caustic); make soap and textiles, oven cleaner, Drano, Liquid plumber make soap and textiles Potassium Hydroxide (Strong) KOH Magnesium Hydroxide (Strong) Mg(OH)2 Milk of magnesia; laxative and antacid Ca(OH)2 Lime Water; astringent -causes contraction of skin pores NH 4 OH Ammonia in water; window cleaner, other cleaning solutions Calcium Hydroxide (Strong) Ammonium Hydroxide (Weak) Ammonia (Weak) NH 3 Gas; inhalant to revive an unconscious person, anhydrous or liquid ammonia is injected into soil as a fertilizer Sodium Carbonate (Weak) Na 2 CO 3 Soda Ash; Detergents Sodium Phosphate (Weak) Na 3 PO 4 Sodium triphosphate; Detergents

1. Acids that are made of only two elements (Binary) A. Prefix is always hydro B. Name the second element with the suffix -ic C. HCl = Hydrochloric Acid 2. Acids that are made of more than two elements; usually contain a polyatomic ion (Ternary) A. Polyatomic ions ending with –ate, you will change the suffix to -ic. example: H 2 SO 4 Sulfate Ion = Sulfuric Acid B. Polyatomics ending in –ite become –ous. For the acid containing the polyatomic with one less oxygen than the -ic, use the suffix -ous. example: H 2 SO 3 Sulfite Ion = Sulfurous Acid 2 Friends = ate-ic and ite-ous

1. Simply use the normal rules for naming compounds; ionic or covalent depending on the elements in the compound. 2. Most bases end with the polyatomic hydroxide ion – OH. 3. example: Na. OH = Sodium Hydroxide 4. Mg(OH)2 = Magnesium Hydroxide 5. 3. For salts, use your oxidation table and follow normal ionic and covalent naming properties. 6. example: KCl = Potassium Chloride

What is acid rain? Dissolved carbon dioxide lowers the p. H CO 2 (g) + H 2 O H 2 CO 3 H+ + HCO 3 Atmospheric pollutants from combustion NO, NO 2 + H 2 O … HNO 3 SO 2, SO 3 + H 2 O … H 2 SO 4 p. H < 5. 3 both strong acids
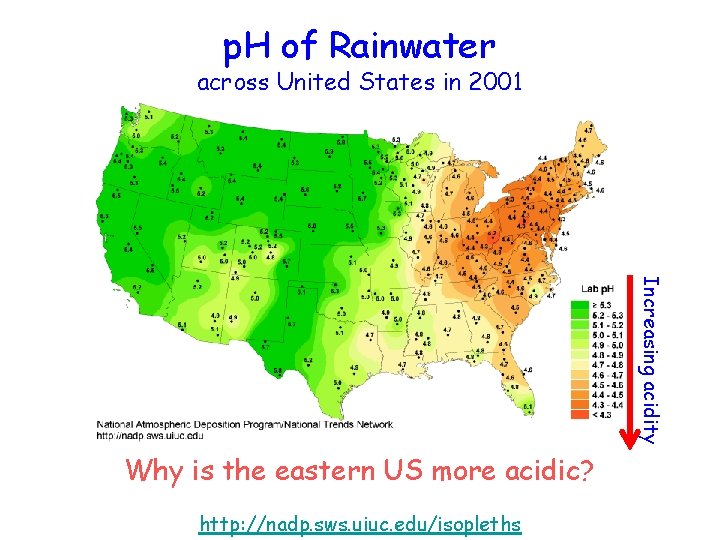
p. H of Rainwater across United States in 2001 Increasing acidity Why is the eastern US more acidic? http: //nadp. sws. uiuc. edu/isopleths

When acids and bases with equal amounts of hydrogen ion H+ and hydroxide ions OH- are mixed, the resulting solution is neutral. The resulting reaction will produce a salt precipitate and water Na. OH (aq) + HCl(aq) Na. Cl + H 2 O base acid salt water Ca(OH)2 + 2 HCl Ca. Cl 2 + 2 H 2 O base acid salt water

a. HBr + Ca(OH)2 → b. Sulfuric acid combines with ammonium hydroxide c. Sodium Hydroxide plus Carbonic Acid
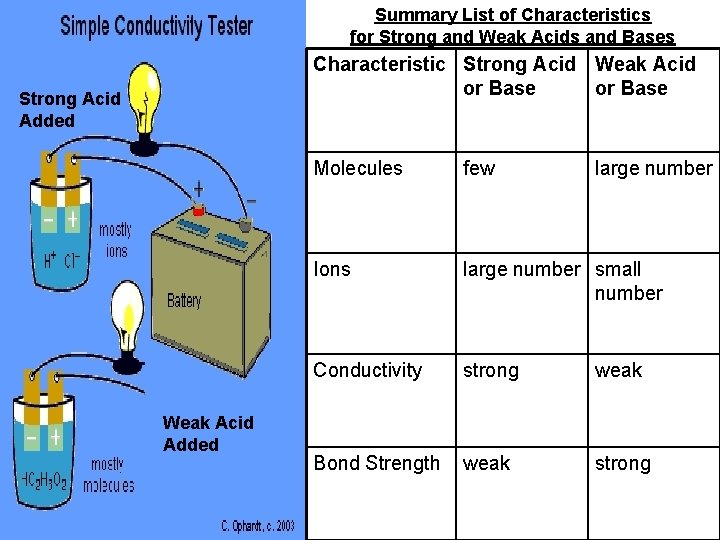
Summary List of Characteristics for Strong and Weak Acids and Bases Characteristic Strong Acid Weak Acid Strong Acid Added Weak Acid Added or Base Molecules few large number Ions large number small number Conductivity strong weak Bond Strength weak strong

The term p. H was derived from the French word pouvoir hydrogene meaning “Hydrogen Power” p. H is defined as the negative logarithm of the hydrogen ion concentration. The equation is: p. H = - log [H+] similarly, p. OH = - log [OH-] 1 x 10 -p. H M

The p. H scale, (0 - 14), is the full set of p. H numbers which indicate the concentration of H+ and OH-ions in water. From 0 – 6 the substance is Acidic. 7 is neutral. From 8 -14 the substance is Basic. The lower the number the more acidic, the higher the number the more basic.

Three Common Indicators: Litmus (Vegetable dye) Paper: It is red below p. H 4. 5 and blue above p. H 8. 2. Phenolphthalein: indicator of acids (colorless) and bases (pink). Hydrion Paper: Ranges in color, Acids/Red, Bases/Blue
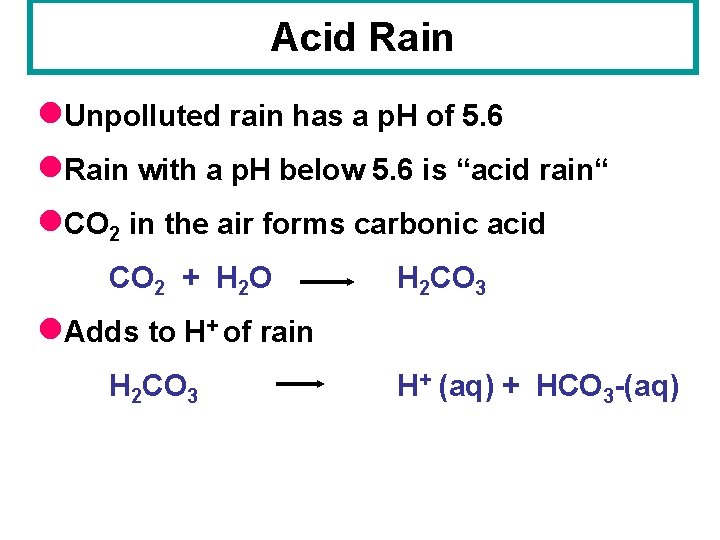
Acid Rain l. Unpolluted rain has a p. H of 5. 6 l. Rain with a p. H below 5. 6 is “acid rain“ l. CO 2 in the air forms carbonic acid CO 2 + H 2 O H 2 CO 3 H+ (aq) + HCO 3 -(aq) l. Adds to H+ of rain H 2 CO 3
- Slides: 18