General overview of brazilian regulation on drugs and

General overview of brazilian regulation on drugs and medical devices products Brazilian Health Surveillance Agency (Anvisa) Balbiana Verazez Sampaio Oliveira Dalmo Luiz Faria Pires Aniceto February, 2015 K-Pharma Academy 2015

The Brazilian Health Surveillance Agency (Anvisa) Regulatory Agency: Administrative Independence and finance autonomy • Linked to the Ministry of Health • Management Contract (indicators and targets) • Stability of the Directors (mandate) • Board of Directors – 5 Directors named by the President of Republic, for a mandate of 3 years, renewed once for another 3 years. • 16 years since its creation (Lei 9. 782/1999)
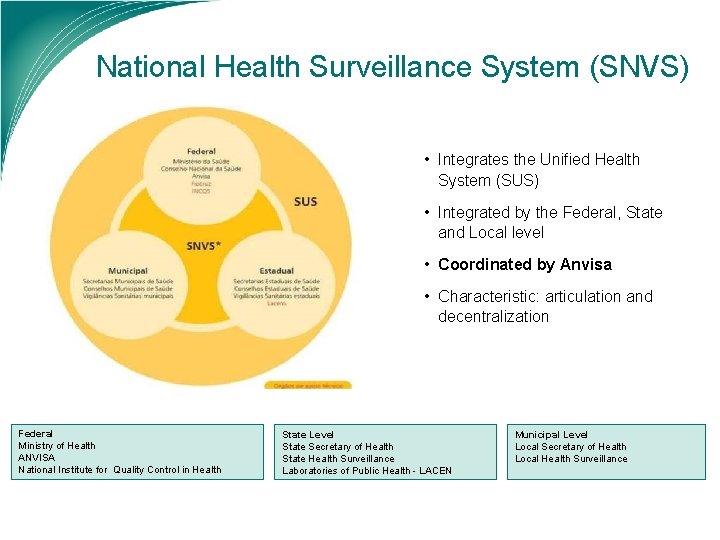
National Health Surveillance System (SNVS) • Integrates the Unified Health System (SUS) • Integrated by the Federal, State and Local level • Coordinated by Anvisa • Characteristic: articulation and decentralization Federal Ministry of Health ANVISA National Institute for Quality Control in Health State Level State Secretary of Health State Health Surveillance Laboratories of Public Health - LACEN Municipal Level Local Secretary of Health Local Health Surveillance
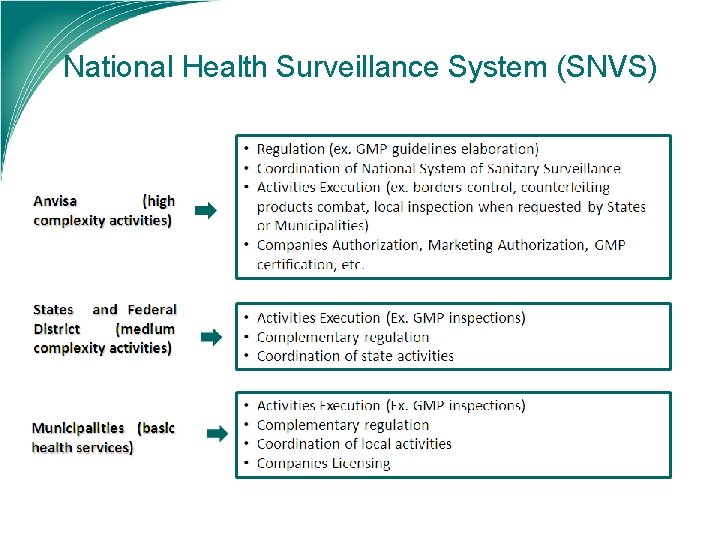
National Health Surveillance System (SNVS)

Brazilian Health Surveillance Agency GOAL Promote and protect the health of the population and intervene at the risks involved in the production and use of products and services subject to sanitary surveillance, in coordinated action with states, municipalities and the Federal District, in accordance with the principles of the Unified Health System, to improve the quality of life of the Brazilian population. VISION Be legitimized by society as a member institution of the Unified Health System, agile, modern and transparent, with national and international reference in regulation and sanitary control. VALUES ► Ethics and responsability as public agent ►Capacity of articulation and integration ► Excellence in management ► Knowlegde as a source for action ► Transparency ► Accountability

Anvisa • ANVISA’s headquarter is located in Brasília, Brazilian capital. • Presence in all States and Federal District: 83 Posts of Health Surveillance in Brazil. • Currently ANVISA has approximately 3. 000 employees working through the country, most of them is located in Brasília.

Anvisa Regulation Fields Foods Cosmetics Health Service Medicines Pharmacovigilance Advertisement Sanitizer Medical devices Tobacco Laboratórios Official Laboratories Ports, airports and International affairs borders Pesticides Blood, Tissues and e organs SNVS coordination

Anvisa Organization Chart

Brazilian Legal Framework • Anvisa has competency to edit legislation related to health surveillance subjects – Resolution of the Board of Directors (RDC) • RDC – elaborated following the Good Regulatory Practices Process • Regulatory Agenda – annual prevision of priority themes to be regulated • Transparency: legislation is published on the Official Newspaper (Diário Oficial da União) and is on ANVISA’s website

Federal Law and Decrees • Law 6360/76 (beyond other purposes, gives legal provision for product registration); • Law 6437/77 (sets violations of federal health legislation and establishes their respective sanctions); • Law 8080/90 (defines the Unique Health System); • Law 9782/99 (establishes Anvisa’s roles and responsibilities, defines the National Health Surveillance System); • Law 8077/13 (has replaced Decree 79094/77 - gives interpretation of Act 6360/76).

Marketing Authorization / Registration: drugs and medical devices • Premarket Activities Company Authorization (AFE) GMP Certification Registration/Notification • Post-market Activities Post-aprovall changes Renewal (each 5 years) Monitoring Programs Postmarket surveillance

Drug Registration • In Brazil, drugs need to be registered. • The registration must be renewed every five years. • Categories of Drugs registered in Brazil: “New” drugs (innovative and others) Synthetic and semi-synthetic drugs Biologicals (includes biossimilars) Herbal medicines “Copies” (Synthetic and semi-synthetic drugs) Generic Drugs Similar Drugs (“brand generic”)

Drug Registration – Category of medicines NEW DRUGS SIMILAR SPECIFIC DRUGS BIOLOGICALS CATEGORY OF DRUGS HERBAL MEDICINES GENERIC DRUGS NOTIFIED HOMEOPHATICS
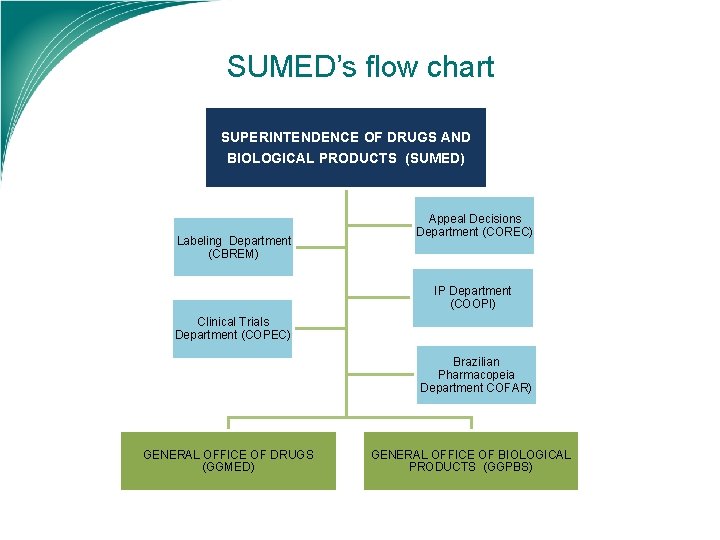
SUMED’s flow chart SUPERINTENDENCE OF DRUGS AND BIOLOGICAL PRODUCTS (SUMED) Labeling Department (CBREM) Appeal Decisions Department (COREC) IP Department (COOPI) Clinical Trials Department (COPEC) Brazilian Pharmacopeia Department COFAR) GENERAL OFFICE OF DRUGS (GGMED) GENERAL OFFICE OF BIOLOGICAL PRODUCTS (GGPBS)
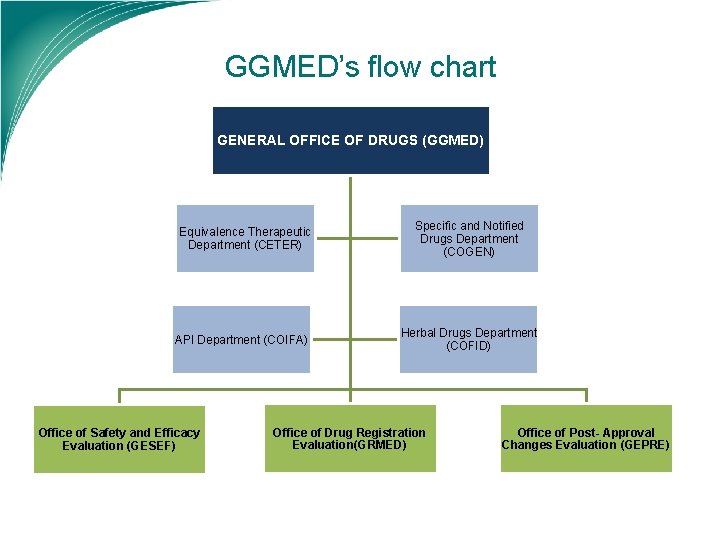
GGMED’s flow chart GENERAL OFFICE OF DRUGS (GGMED) Equivalence Therapeutic Department (CETER) API Department (COIFA) Office of Safety and Efficacy Evaluation (GESEF) Specific and Notified Drugs Department (COGEN) Herbal Drugs Department (COFID) Office of Drug Registration Evaluation(GRMED) Office of Post- Approval Changes Evaluation (GEPRE)

Drug Registration QUALITY EFICACY DRUG REGISTRATION SAFETY LEGAL DOCUMENTATION Each of categories have their specific legislations and requirements.

Drug Registration – main requirements • Company Authorization (AFE) • Good Manufacturing Practices (GMP certificates) • Information about API used – drug master file / DCB (Common Brazilian Nomination) • Production Report (quanti/qualitative formula, role of each excipiente, production details, sumarized validation report) • Quality Control Report • Analythical Method Validation • Stability studies of 3 pilot batches (Zone IVb) • Pharmaceutical equivalence and bioequivalence for “copies” (Center Certified by Anvisa) • Clinical trials for “news”

Drug Registration – legislation Registration Resolution Synthetic and semi-synthetic drugs (news, generics and similars) RDC Nº. 60/2014 Homeopathic Drug RDC Nº. 26/2007 Herbal Medicines RDC Nº. 26/2014 Biological Drug RDC Nº. 55/2010 Specific Drugs RDC Nº 24/2011 Notified RDC Nº 199/2006 Post-Registation Resolution Drug Post Registration RDC Nº 48/2009 Drug Post Registration for biologicals RDC Nº 24/2013
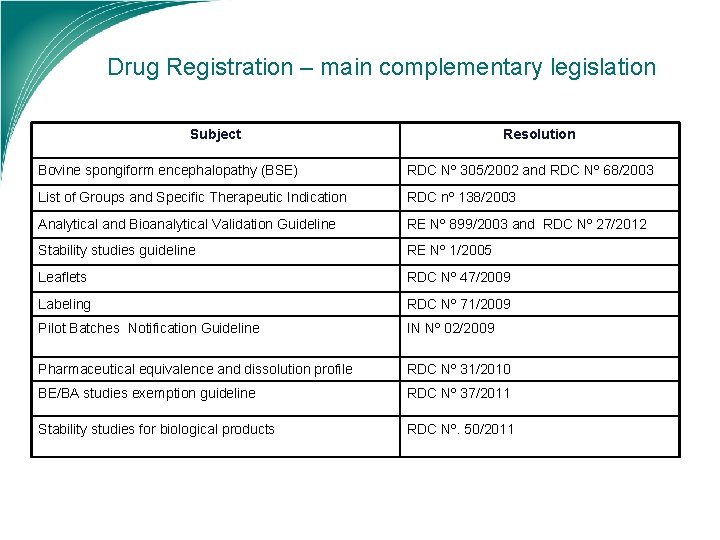
Drug Registration – main complementary legislation Subject Resolution Bovine spongiform encephalopathy (BSE) RDC Nº 305/2002 and RDC Nº 68/2003 List of Groups and Specific Therapeutic Indication RDC nº 138/2003 Analytical and Bioanalytical Validation Guideline RE Nº 899/2003 and RDC Nº 27/2012 Stability studies guideline RE Nº 1/2005 Leaflets RDC Nº 47/2009 Labeling RDC Nº 71/2009 Pilot Batches Notification Guideline IN Nº 02/2009 Pharmaceutical equivalence and dissolution profile RDC Nº 31/2010 BE/BA studies exemption guideline RDC Nº 37/2011 Stability studies for biological products RDC Nº. 50/2011

Drug Registration – GMP legislation Subject Resolution RDC GMP guide for Drug Products RDC Nº 17/2010 GMP guide for APIs RDC Nº 69/2014 GMP Certificate RDC Nº 39/2013

Medical Devices Registration – Classification • For regulatory purposes MD are separated into: Non IVD: e. g. equipment, software, articles, and materials. IVD: e. g. reagents, calibrators, standards and controls. Each of them have their specific legislations and requirements, including classification.
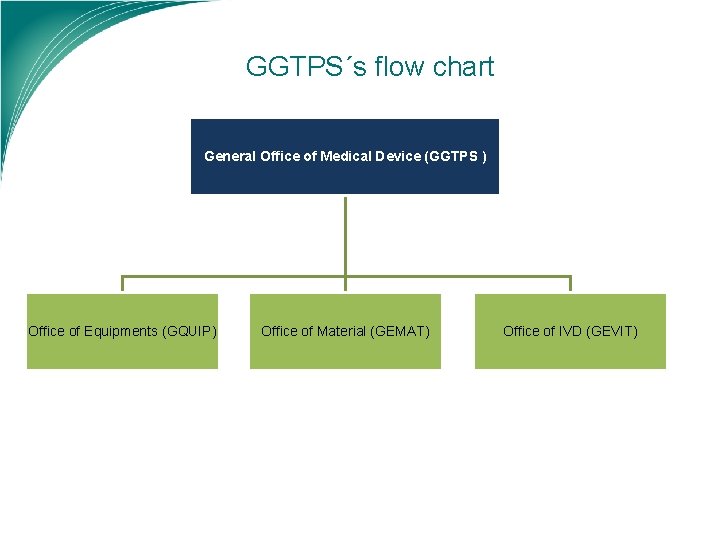
GGTPS´s flow chart General Office of Medical Device (GGTPS ) Office of Equipments (GQUIP) Office of Material (GEMAT) Office of IVD (GEVIT)

Non IVD Classification Established by RDC 185/2001: • Four Classes: – Class I: low risk; – Class II: medium risk; – Class III: high risk; – Class IV: maximum risk. 18 Rules are used to classify the device according to the duration of contact with the patient, the degree of invasiveness and the body part affected by the use of the device.

IVD Classification • Four Classes according to RDC 61/2011: – Class I - low risk devices to the individual and low risk to public health (e. g. sample collection receptacles, culture media); – Class II - medium risk devices to the individual and/or low risk to public health (e. g. cholesterol quantitative test); – Class III - high risk devices to the individual and/or medium risk to public health (e. g. syphilis test); and – Class IV - high-risk devices to the individual and high risk to public health (e. g. HIV 1/2 Ab assay). 9 Rules are used to classify the IVDs according to their intended use.

Medical Device Registration • There are two types of premarket applications: – Registration: Classes III and IV devices (n. IVD) and Classes II, III and IV (IVD); – Notification: Classes I and II (n. IVD) and Classe I (IVD). • Both are valid for 5 years and must be renewed after this period.

Medical Device Registration– main requirements • Company Authorization (AFE) • GMP Certification • Requests a comprehensive documentation, including all technical data and tests about the device, and for some types of devices, clinical data are also requested. • Requirements defined by RDC 185/01 (n. IVD) and RDC 206/06 (IVD).

Medical Device Notification – main requirements • Company Authorization (AFE) • Does not request GMP certification. However the manufacturer shall comply with GMP requirements and may the subject to a regulatory inspection at anytime. • Requests only summarized documentation and specific forms; • Requirements defined by RDC 24/09 (n. IVD) and RDC 206/06 (IVD);
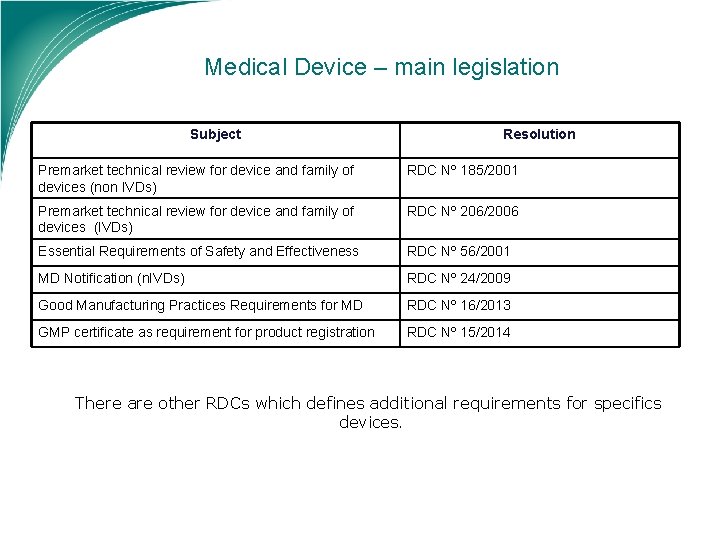
Medical Device – main legislation Subject Resolution Premarket technical review for device and family of devices (non IVDs) RDC Nº 185/2001 Premarket technical review for device and family of devices (IVDs) RDC Nº 206/2006 Essential Requirements of Safety and Effectiveness RDC Nº 56/2001 MD Notification (n. IVDs) RDC Nº 24/2009 Good Manufacturing Practices Requirements for MD RDC Nº 16/2013 GMP certificate as requirement for product registration RDC Nº 15/2014 There are other RDCs which defines additional requirements for specifics devices.

Information about registered products Information on the dossiers analysis queue at ANVISA website Talk with the Agency http: //portal. anvisa. gov. br/wps/portal/anvisa/home

Face to face meetings (no fee is required) • • Clarifying Anvisa’s technical requirements; New product presentation; Regulatory and technical advice; Enterprises associations representatives. http: //www 10. anvisa. gov. br/Parlatorio/login. seam? cid=184
Legislation http: //portal 2. saude. gov. br/saudelegis/LEG_NORMA_PESQ_CONSULTA. CFM

Anvisa Headquarter BRASÍLIA D. F

THANK YOU! Contacts Agência Nacional de Vigilância Sanitária - Anvisa SIA Trecho 5 - Área especial 57 - Lote 200 Zip code: 71205 -050 Brasília - DF Phone: + 55 61 3462 6000 www. anvisa. gov. br www. twitter. com/anvisa_oficial Anvisa Atende: 0800 -642 -9782 ouvidoria@anvisa. gov. br
- Slides: 33