General Organic and Biological Chemistry Fourth Edition Karen

General, Organic, and Biological Chemistry Fourth Edition Karen Timberlake Chapter 1 Chemistry and Measurements 1. 8 Writing Conversion Factors © 2013 Pearson Education, Inc. Lectures

Equalities § use two different units to describe the same measured amount. § are written for relationships between units of the metric system; between U. S. units or between metric and U. S. units. Examples: © 2013 Pearson Education, Inc. Chapter 1, Section 8 2

Equalities and Conversion Factors Equalities are § written as a fraction. § used as conversion factors. § can be represented with one equality in the numerator and the second equality in the denominator. Examples: © 2013 Pearson Education, Inc. Chapter 1, Section 8 3
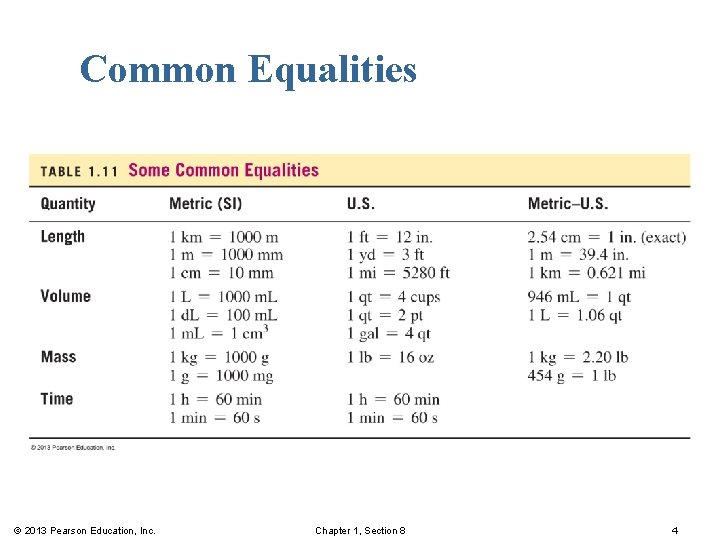
Common Equalities © 2013 Pearson Education, Inc. Chapter 1, Section 8 4

Exact and Measured Numbers in Equalities between units of § the same system are definitions with numbers that are exact. § different systems (metric and U. S. ) are measurements with numbers that have significant figures. The equality of 2. 54 cm = 1 in. is an exception and considered to be exact. © 2013 Pearson Education, Inc. Chapter 1, Section 8 5

Metric Conversion Factors We can write equalities as conversion factors. © 2013 Pearson Education, Inc. Chapter 1, Section 8 6

Metric-US Conversion Factors Metric–US conversion factors are written as a ratio with a numerator and denominator. Example: © 2013 Pearson Education, Inc. Chapter 1, Section 8 7

Equalities on Food Labels The contents of packaged foods § in the U. S. are listed in both metric and U. S. units. § indicate the same amount of a substance in two different units. © 2013 Pearson Education, Inc. Chapter 1, Section 8 8

Learning Check Write equalities and conversion factors for each pair of units. 1. liters and m. L 2. hours and minutes 3. meters and kilometers © 2013 Pearson Education, Inc. Chapter 1, Section 8 9
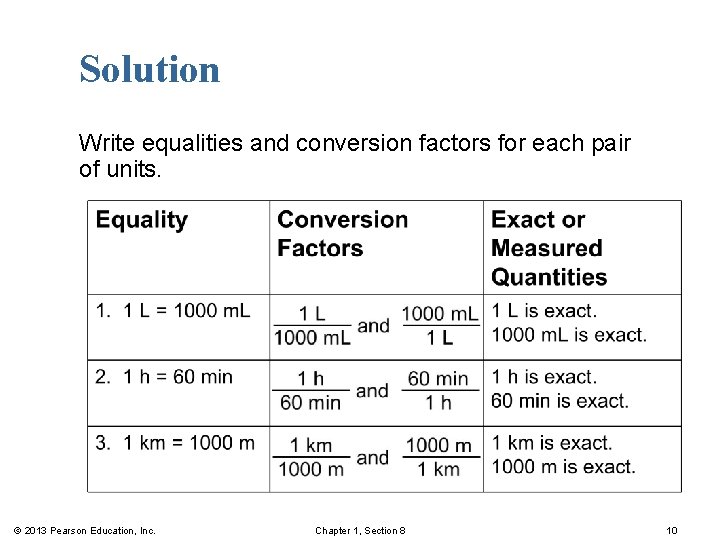
Solution Write equalities and conversion factors for each pair of units. © 2013 Pearson Education, Inc. Chapter 1, Section 8 10

Conversion Factors in a Problem A conversion factor § may be obtained from information in a word problem. § is written for that problem only. Example 1: The motorcycle was traveling at a speed of 85 km/h. Example 2: One tablet contains 500 mg vitamin C. © 2013 Pearson Education, Inc. Chapter 1, Section 8 11

Percent as a Conversion Factor A percent factor § uses a ratio of the parts to the whole in a fraction. § uses the same units for the parts and whole. § uses the value 100 for the whole. § can be written as two factors. Example: A food contains 18% (by mass) fat. © 2013 Pearson Education, Inc. Chapter 1, Section 8 12

Parts per Million, Parts per Billion Parts per Million, ppm is the same as mg per kilogram mg/kg of substance. Parts per Billion, ppb is the same as g per kilogram g/kg of substance. Example: The maximum dose of lead in pottery glaze is 2 ppm. © 2013 Pearson Education, Inc. Chapter 1, Section 8 13

Chemistry Link to Health Toxicology and Risk-Benefit Assessment One measure of toxicity is the LD 50 or lethal dose, which is the concentration of the substance that causes death in 50% of the test animals. A dose is typically measured in ppm (mg/kg) of body mass or ppb (μg/kg). © 2013 Pearson Education, Inc. Chapter 1, Section 8 14
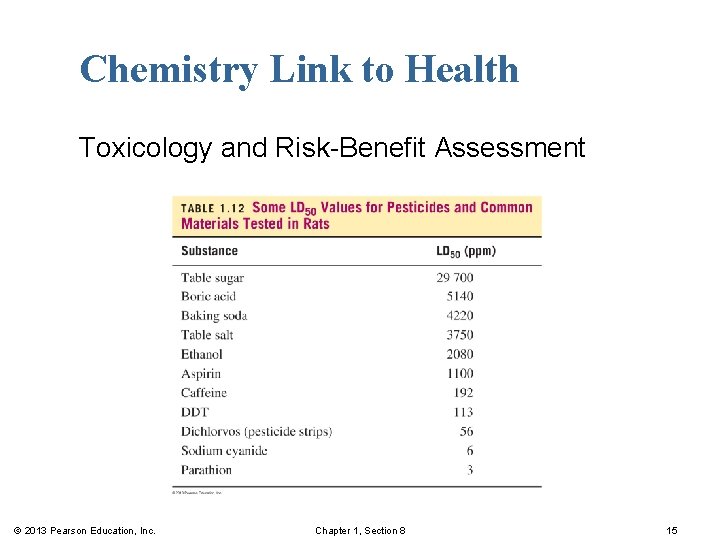
Chemistry Link to Health Toxicology and Risk-Benefit Assessment © 2013 Pearson Education, Inc. Chapter 1, Section 8 15

Learning Check Write the equality and conversion factors for each of the following. 1. meters and centimeters 2. jewelry that contains 18% gold 3. one liter of gas is $ 0. 95 © 2013 Pearson Education, Inc. Chapter 1, Section 8 16

Solution Write the equality and conversion factors for each of the following. © 2013 Pearson Education, Inc. Chapter 1, Section 8 17
- Slides: 17