General Organic and Biological Chemistry Fourth Edition Karen

General, Organic, and Biological Chemistry Fourth Edition Karen Timberlake Chapter 2 Energy and Matter 2. 3 Classification of Matter © 2013 Pearson Education, Inc. Lectures

Matter § is anything that has mass and occupies space. § makes up the things we see everyday, such as water, wood, cooking pans, clothes, and shoes. § can be classified as a pure substance or mixture. Mixture Matter Pure Substance © 2013 Pearson Education, Inc. Chapter 2, Section 3 2

Pure Substances A pure substance § has a fixed or constant composition. § can be classified as an element or compound. Element – one Pure Substance type of atom Compound – two or more elements chemically combined © 2013 Pearson Education, Inc. Chapter 2, Section 3 3

Elements are pure substances that contain atoms of only one type. Examples: copper (Cu) lead (Pb) aluminum (Al) © 2013 Pearson Education, Inc. Chapter 2, Section 3 4

Compounds contain two or more elements that are chemically combined in a definite ratio. Examples: salt (Na. Cl) table sugar (C 12 H 22 O 11) water (H 2 O) © 2013 Pearson Education, Inc. Chapter 2, Section 3 5

Elements in a Compound Table salt is a compound that contains the elements sodium and chlorine. © 2013 Pearson Education, Inc. Chapter 2, Section 3 6

Mixtures A mixture is matter that consists of § two or more substances that are physically mixed, not chemically combined. § a composition that is not constant. § substances that can be separated by physical methods. © 2013 Pearson Education, Inc. Chapter 2, Section 3 7

Physical Separation of A Mixture The physical separation of a mixture § involves only physical changes. § occurs when pasta and water are separated with a strainer. © 2013 Pearson Education, Inc. Chapter 2, Section 3 8

Separation of A Mixture in Lab The physical separation of a mixture in a chemistry lab may include: § filtration § chromatography © 2013 Pearson Education, Inc. Chapter 2, Section 3 9

Types of Mixtures are § classified by their composition. § called homogeneous when they are uniform. § called heterogeneous when they are not uniform. Mixtures Homogeneous Mixture – Uniform Heterogeneous Mixture – Not uniform © 2013 Pearson Education, Inc. Chapter 2, Section 3 10

Homogeneous Mixtures In a homogeneous mixture, the § composition is uniform throughout, and § different parts of the mixture are not visible. © 2013 Pearson Education, Inc. Chapter 2, Section 3 11

Heterogeneous Mixtures In a heterogeneous mixture, the § composition is not uniform; it varies from one part of the mixture to another, and § different parts of the mixture are visible. © 2013 Pearson Education, Inc. Chapter 2, Section 3 12
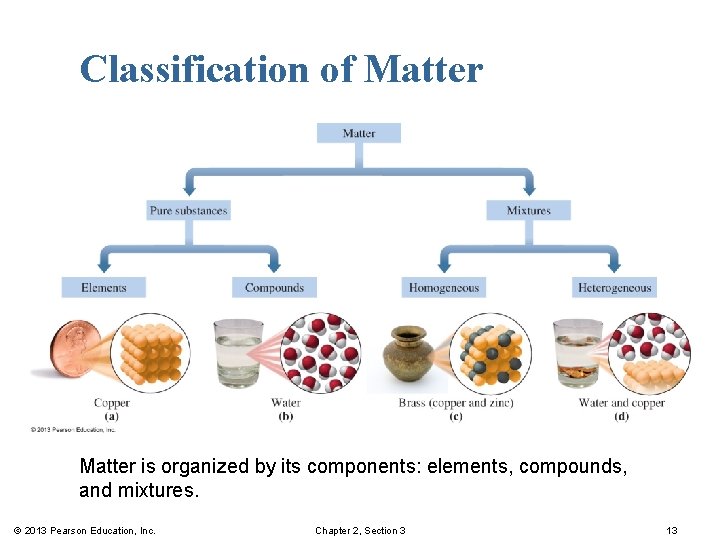
Classification of Matter is organized by its components: elements, compounds, and mixtures. © 2013 Pearson Education, Inc. Chapter 2, Section 3 13

Learning Check Identify each of the following as a pure substance or a mixture. A. pasta and tomato sauce B. aluminum foil C. helium D. air © 2013 Pearson Education, Inc. Chapter 2, Section 3 14

Solution Identify each of the following as a pure substance or a mixture. A. pasta and tomato sauce mixture B. aluminum foil pure substance C. helium pure substance D. air mixture © 2013 Pearson Education, Inc. Chapter 2, Section 3 15

Learning Check Identify each of the following as a homogeneous or heterogeneous mixture. A. hot fudge sundae B. a clear shampoo C. sugar water D. peach pie © 2013 Pearson Education, Inc. Chapter 2, Section 3 16

Solution Identify each of the following as a homogeneous or heterogeneous mixture. A. hot fudge sundae heterogeneous mixture B. a clear shampoo homogeneous mixture C. sugar water homogeneous mixture D. peach pie heterogeneous mixture © 2013 Pearson Education, Inc. Chapter 2, Section 3 17
- Slides: 17