General Organic and Biological Chemistry Fourth Edition Karen

General, Organic, and Biological Chemistry Fourth Edition Karen Timberlake Chapter 8 Solutions 8. 6 Properties of Solutions © 2013 Pearson Education, Inc. Lectures

Mixtures: Solution, Colloidal Dispersion or Suspension Particle size, distribution and polarity help us to further classify mixtures as a § solution, § colloidal dispersion, or a § suspension. © 2013 Pearson Education, Inc. Chapter 8, Section 6 2

Solutions In a solution, § the solute is dissolved as small particles that are uniformly dispersed throughout the solvent to give a homogeneous solution. § the solute and solvent particles cannot be visually distinguished and the solution appears transparent. § solute particles are so small that they go through filters and semipermeable membranes. Semipermeable membranes allow solvent and very small solute particles to pass through. © 2013 Pearson Education, Inc. Chapter 8, Section 6 3

Colloids § have much larger solute particles than solutions. § are homogeneous mixtures that do not separate or settle out. § have solute particles that pass through filters but not semipermeable membranes. © 2013 Pearson Education, Inc. Chapter 8, Section 6 4
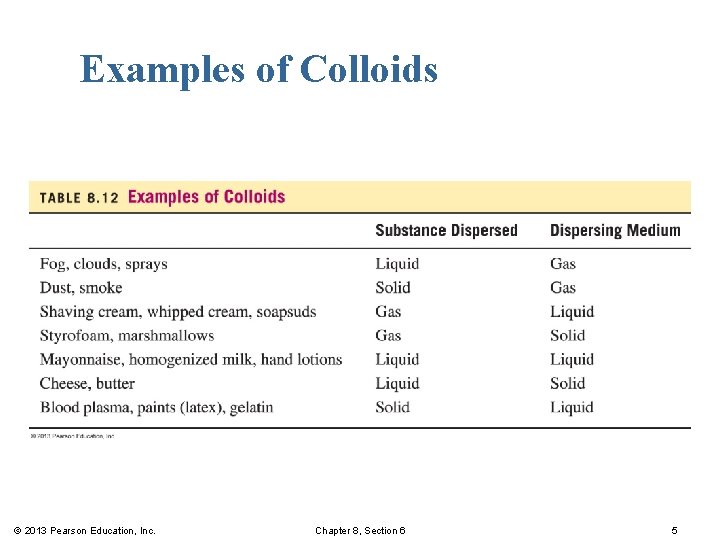
Examples of Colloids © 2013 Pearson Education, Inc. Chapter 8, Section 6 5

Suspensions § are heterogeneous nonuniform mixtures. § have very large particles that can be trapped by filters or semipermeable membranes. § settle out soon after mixing. § must be stirred to stay suspended. Examples of suspensions include medications, such as Kaopectate, calamine lotion, antacid mixtures, and liquid penicillin. © 2013 Pearson Education, Inc. Chapter 8, Section 6 6
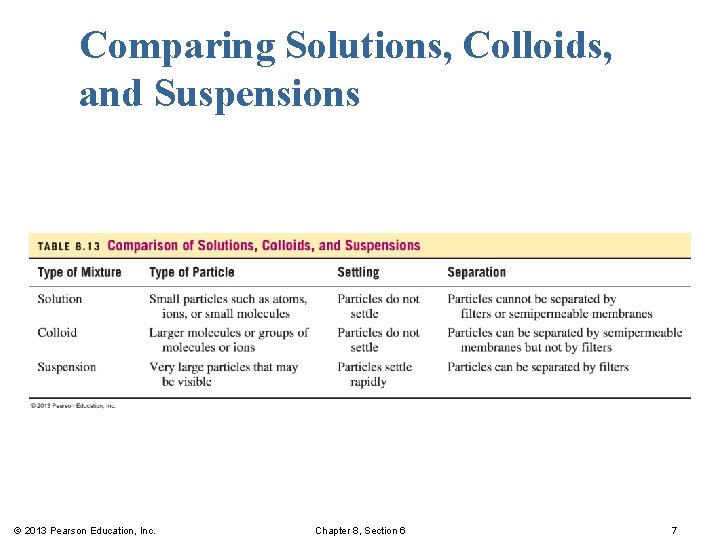
Comparing Solutions, Colloids, and Suspensions © 2013 Pearson Education, Inc. Chapter 8, Section 6 7

Solutions, Colloids, and Suspensions Properties of different types of mixtures: (a) suspensions settle out; (b) suspensions are separated by a filter; (c) solution particles go through a semipermeable membrane, but colloids and suspensions do not. © 2013 Pearson Education, Inc. Chapter 8, Section 6 8

Learning Check A mixture that has solute particles that do not settle out, but are too large to pass through a semipermeable membrane is called a A. solution. B. colloid. C. suspension. © 2013 Pearson Education, Inc. Chapter 8, Section 6 9

Solution A mixture that has solute particles that do not settle out, but are too large to pass through a semipermeable membrane is called a B. colloid. © 2013 Pearson Education, Inc. Chapter 8, Section 6 10

Colligative Properties Colligative properties § are changes in the properties of a solvent when solute particles are added. § depend on the number of solute particles in solution. § lower freezing points. § increase boiling points. © 2013 Pearson Education, Inc. Chapter 8, Section 6 11

Freezing Point Lowering and Boiling Point Elevation When 1 mole of solute particles is added to 1000 g of water, the § freezing point of water decreases by 1. 86 C (from 0 C to – 1. 86 C). § boiling point of water increases by 0. 52 C (from 100 C to 100. 52 C). © 2013 Pearson Education, Inc. Chapter 8, Section 6 12

Freezing Point Lowering The salt Ca. Cl 2 used to melt ice on roads and sidewalks produces 3 moles of particles from 1 mole of Ca. Cl 2: 1 mole of Ca 2+ and 2 moles of Cl−. © 2013 Pearson Education, Inc. Chapter 8, Section 6 13

Electrolytes and Nonelectrolytes A solute that is a nonelectrolyte dissolves as molecules, whereas a solute that is a strong electrolyte dissolves entirely as ions. § One mole of ethylene glycol, a nonelectrolyte dissolves in water, breaking up into one mole of molecules. § One mole of calcium chloride, a strong electrolyte dissolves in water, breaking up into three moles of ions, one mole of Ca 2+ ions and two moles of Cl− ions. © 2013 Pearson Education, Inc. Chapter 8, Section 6 14

Moles of Particles The number of moles of particles depends on the type of solute. § Nonelectrolytes dissolve as molecules. 1 mole of nonelectrolyte = 1 mole of particles in water § Strong electrolytes dissolve as ions. 1 mole of electrolyte = 2 to 4 moles of particles in water © 2013 Pearson Education, Inc. Chapter 8, Section 6 15
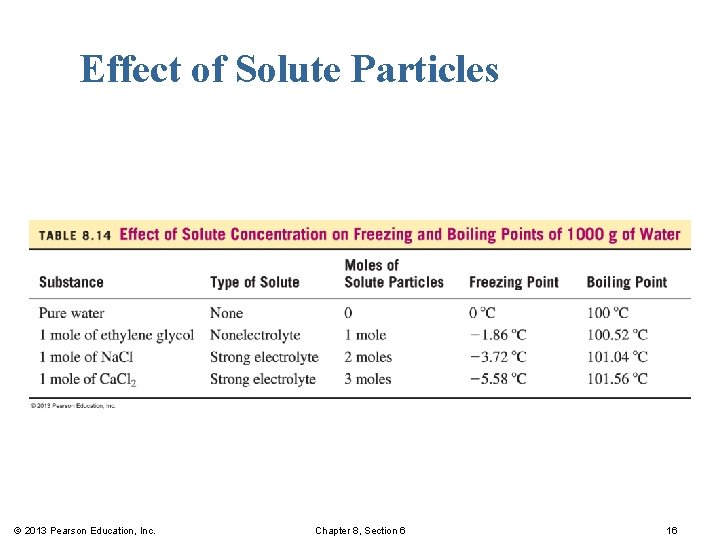
Effect of Solute Particles © 2013 Pearson Education, Inc. Chapter 8, Section 6 16

Learning Check Identify the solution that will have a lower freezing point. A. 1 mole Ca(NO 3)2 dissolved in 1 liter of water B. 1 mole KOH dissolved in 1 liter of water C. 1 mole C 6 H 12 O 6 dissolved in 1 liter of water © 2013 Pearson Education, Inc. Chapter 8, Section 6 17

Solution Identify the solution that will have a lower freezing point. A. 1 mole Ca(NO 3)2 dissolved in 1 liter of water For every 1 mole of Ca(NO 3)2, 3 moles of particles are produced, 1 mole Ca 2+ and 2 moles NO 3−. B. 1 mole KOH dissolved in 1 liter of water For every 1 mole of KOH, 2 moles of particles are produced, 1 mole K+ and 1 mole OH−. C. 1 mole C 6 H 12 O 6 dissolved in 1 liter of water For every 1 mole of C 6 H 12 O 6, 1 mole of particles are produced. Answer is A; it produces the largest number of particles. © 2013 Pearson Education, Inc. Chapter 8, Section 6 18

Osmosis In osmosis, § water (solvent) flows from the lower solute concentration into the higher solute concentration across a semipermeable membrane. § the level of the solution with the higher solute concentration rises. § the concentrations of the two solutions become equal with time. © 2013 Pearson Education, Inc. Chapter 8, Section 6 19

Osmosis Water flows into the solution with a higher solute concentration until the concentration of both solutions are equal. © 2013 Pearson Education, Inc. Chapter 8, Section 6 20

Example of Osmosis A semipermeable membrane separates a 4% starch solution from a 10% starch solution. Starch is a colloid and cannot pass through the membrane, but water can. What happens? 4% starch 10% starch H 2 O semipermeable membrane © 2013 Pearson Education, Inc. Chapter 8, Section 6 21

Example of Osmosis § The 10% starch solution is diluted by the flow of water out of the 4% solution, and its volume increases. § The 4% solution loses water, and its volume decreases. § Eventually, the water flow between the two becomes equal. 7% starch H 2 O © 2013 Pearson Education, Inc. Chapter 8, Section 6 22

Osmotic Pressure Osmotic pressure § is produced by the solute particles dissolved in a solution. § is the pressure that prevents the flow of additional water into the more concentrated solution. § increases as the number of dissolved particles in the solution increases. © 2013 Pearson Education, Inc. Chapter 8, Section 6 23

Learning Check A semipermeable membrane separates a 10% (m/v) starch solution (A) from a 5% (m/v) starch solution (B). If starch is a colloid, fill in the blanks in the statements below. 1. Solution ____ has the greater osmotic pressure. 2. Water initially flows from ___ into ___. 3. The level of solution ____ will be lower. © 2013 Pearson Education, Inc. Chapter 8, Section 6 24

Solution A semipermeable membrane separates a 10% (m/v) starch solution (A) from a 5% (m/v) starch solution (B). If starch is a colloid, fill in the blanks in the statements below. 1. Solution A has the greater osmotic pressure. 2. Water initially flows from B into A. 3. The level of solution B will be lower. © 2013 Pearson Education, Inc. Chapter 8, Section 6 25

Osmotic Pressure of the Blood Red blood cells § have cell walls that are semipermeable membranes. § maintain an osmotic pressure that cannot change without damage occurring. § must maintain an equal flow of water between the red blood cell and its surrounding environment. © 2013 Pearson Education, Inc. Chapter 8, Section 6 26

Isotonic Solutions An isotonic solution § exerts the same osmotic pressure as red blood cells. § of 5. 0% (m/v) glucose or 0. 9% (m/v) Na. Cl are examples of isotonic solutions. A red blood cell placed in an isotonic solution retains its volume because there is an equal flow of water into and out of the cell. © 2013 Pearson Education, Inc. Chapter 8, Section 6 27

Hypotonic Solutions A hypotonic solution § has a lower osmotic pressure than red blood cells (RBCs). § causes water to flow into RBCs. § causes hemolysis (RBCs swell and may burst). © 2013 Pearson Education, Inc. Chapter 8, Section 6 28

Hypertonic Solutions A hypertonic solution § has a higher osmotic pressure than RBCs. § causes water to flow out of RBCs. § causes crenation (RBCs shrink in size). © 2013 Pearson Education, Inc. Chapter 8, Section 6 29

Isotonic, Hypertonic and Hypotonic Solutions § In an isotonic solution, a red blood cell retains its normal volume. § In a hypotonic solution, water flows into a red blood cell, causing it to swell and burst (hemolysis). § In a hypertonic solution, water leaves the red blood cell, causing it to shrink (crenation). © 2013 Pearson Education, Inc. Chapter 8, Section 6 30
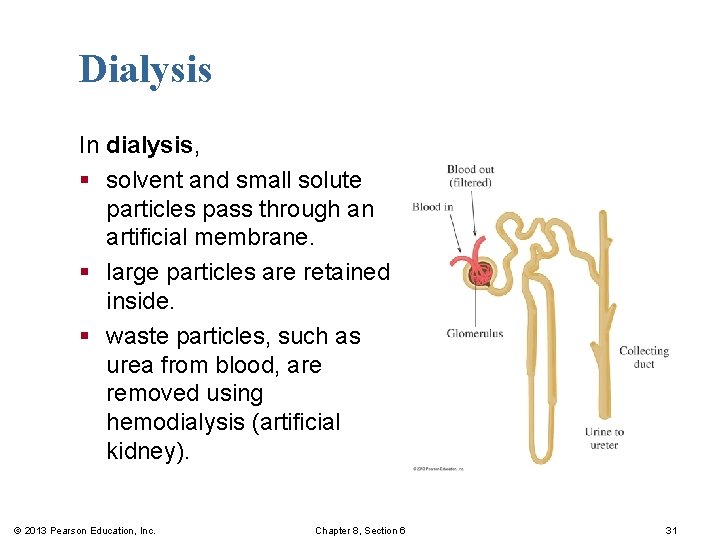
Dialysis In dialysis, § solvent and small solute particles pass through an artificial membrane. § large particles are retained inside. § waste particles, such as urea from blood, are removed using hemodialysis (artificial kidney). © 2013 Pearson Education, Inc. Chapter 8, Section 6 31

Learning Check Indicate if each of the following solutions is A. isotonic. B. hypotonic. C. hypertonic. 1. ____ 2% (m/v) Na. Cl solution 2. ____ 1% (m/v) glucose solution 3. ____ 0. 5% (m/v) Na. Cl solution 4. ____ 5% (m/v) glucose solution © 2013 Pearson Education, Inc. Chapter 8, Section 6 32

Solution Indicate if each of the following solutions is A. isotonic. B. hypotonic. C. hypertonic. 1. ____ C 2% (m/v) Na. Cl solution 2. ____ B 1% (m/v) glucose solution 3. ____ B 0. 5% (m/v) Na. Cl solution A 5% (m/v) glucose solution 4. ____ © 2013 Pearson Education, Inc. Chapter 8, Section 6 33

Learning Check When placed in each of the following, indicate if a red blood cell will A. not change. B. hemolyze. C. crenate. 1. ____ 5% (m/v) glucose solution 2. ____ 1% (m/v) glucose solution 3. ____ 0. 5% (m/v) Na. Cl solution 4. ____ 2% (m/v) Na. Cl solution © 2013 Pearson Education, Inc. Chapter 8, Section 6 34

Solution When placed in each of the following, indicate if a red blood cell will A. not change. B. hemolyze. C. crenate. A 5%(m/v) glucose solution 1. ____ B 1%(m/v) glucose solution 2. ____ 3. ____ B 0. 5%(m/v) Na. Cl solution 4. ____ C 2%(m/v) Na. Cl solution © 2013 Pearson Education, Inc. Chapter 8, Section 6 35
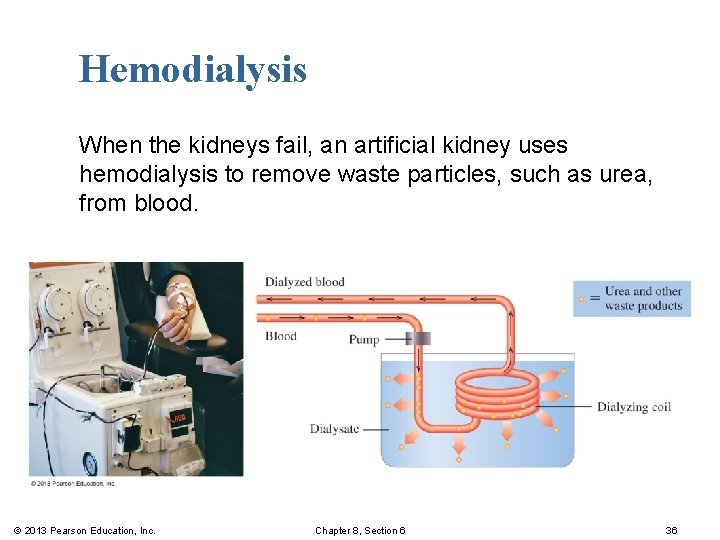
Hemodialysis When the kidneys fail, an artificial kidney uses hemodialysis to remove waste particles, such as urea, from blood. © 2013 Pearson Education, Inc. Chapter 8, Section 6 36
- Slides: 36