General Organic and Biological Chemistry Fourth Edition Karen

General, Organic, and Biological Chemistry Fourth Edition Karen Timberlake Chapter 6 Chemical Reactions and Quantities 6. 9 Energy Changes in Chemical Reactions © 2013 Pearson Education, Inc. Lectures

Reaction Conditions There are three conditions required for a chemical reaction to occur. 1. Collision: The reactants must collide. 2. Orientation: The reactants must align properly to. break and form bonds. 3. Energy: The collision must provide the energy of. activation. © 2013 Pearson Education, Inc. Chapter 6, Section 9 2

Activation Energy § Activation energy is the amount of energy required to break the bonds between atoms of the reactants. § If the energy of a collision is less than the activation energy, the molecules bounce apart without reacting. § Many collisions occur, but only a few actually lead to the formation of product. © 2013 Pearson Education, Inc. Chapter 6, Section 9 3
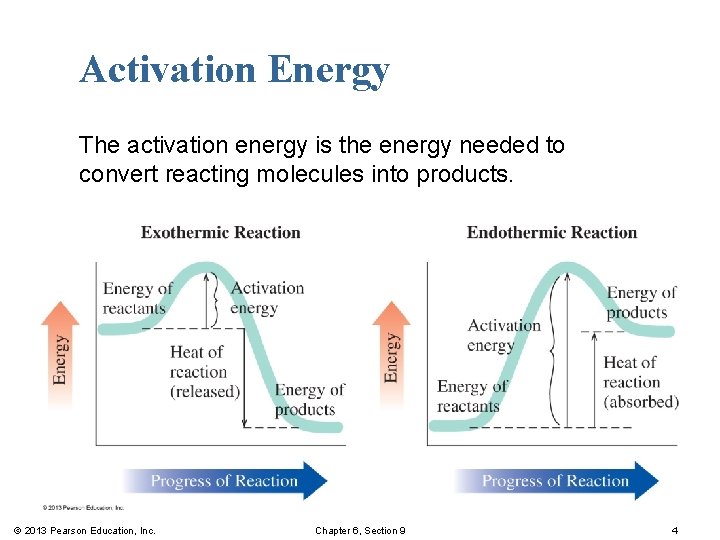
Activation Energy The activation energy is the energy needed to convert reacting molecules into products. © 2013 Pearson Education, Inc. Chapter 6, Section 9 4
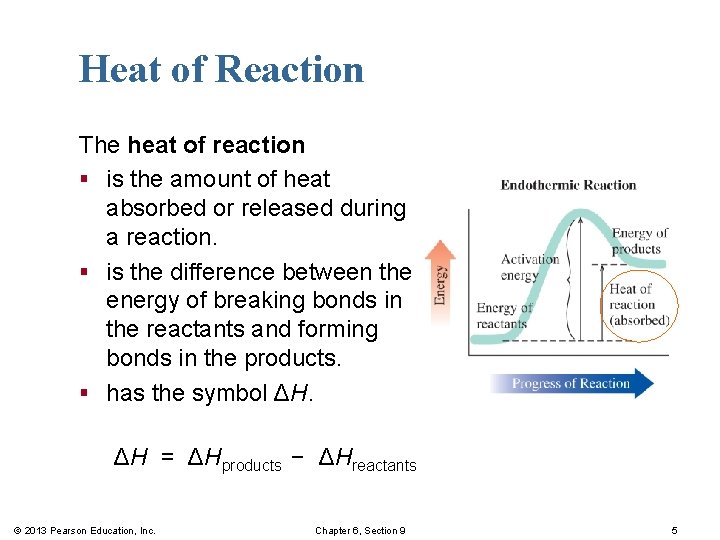
Heat of Reaction The heat of reaction § is the amount of heat absorbed or released during a reaction. § is the difference between the energy of breaking bonds in the reactants and forming bonds in the products. § has the symbol ΔH. ΔH = ΔHproducts − ΔHreactants © 2013 Pearson Education, Inc. Chapter 6, Section 9 5

Exothermic Reactions In an exothermic reaction, § the energy of the reactants is greater than that of the products and heat is released along with the products. § the heat of reaction (ΔH) value is written with a negative sign (–) indicating heat is released. © 2013 Pearson Education, Inc. Chapter 6, Section 9 6
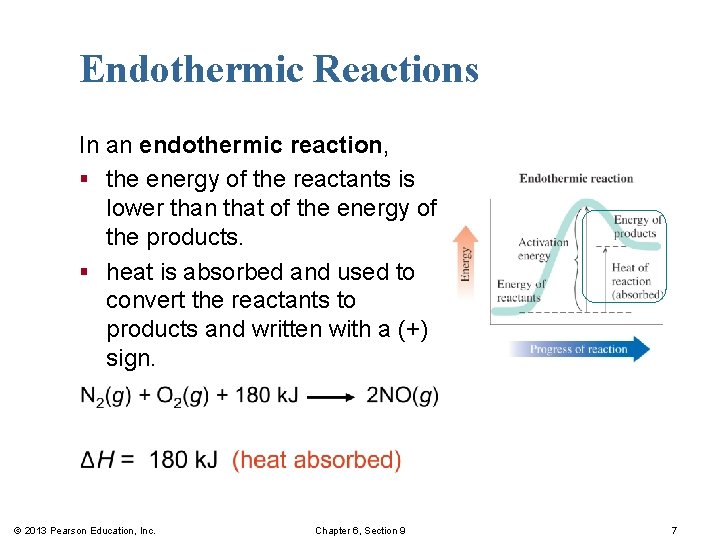
Endothermic Reactions In an endothermic reaction, § the energy of the reactants is lower than that of the energy of the products. § heat is absorbed and used to convert the reactants to products and written with a (+) sign. © 2013 Pearson Education, Inc. Chapter 6, Section 9 7

Heat of Reaction Summary Reaction Type Endothermic Exothermic © 2013 Pearson Education, Inc. Energy Change Heat absorbed Heat released Heat in Reaction Reactant side Product side Chapter 6, Section 9 Sign of ΔH + – 8

Learning Check Identify each of the following reactions as exothermic or endothermic. © 2013 Pearson Education, Inc. Chapter 6, Section 9 9

Solution Identify each of the following reactions as exothermic or endothermic. Heat is a product. The reaction is exothermic. Heat is a reactant. The reaction is endothermic. Heat is a product. The reaction is exothermic. © 2013 Pearson Education, Inc. Chapter 6, Section 9 10

Guide to Calculations Using Heat of Reaction ( H) © 2013 Pearson Education, Inc. Chapter 6, Section 9 11

Calculating the Heat in a Reaction If 15. 0 g of NO are produced according to the following reaction, how many k. J were absorbed? Step 1 State the given and needed quantities. Analyze the Problem. © 2013 Pearson Education, Inc. Chapter 6, Section 9 12

Calculating the Heat in a Reaction If 15. 0 g of NO are produced according to the following reaction, how many k. J were absorbed? Step 2 Write a plan using heat of reaction and any molar mass needed. g of NO molar mass moles of NO mass © 2013 Pearson Education, Inc. Heat of heat of reaction kilojoules reaction Chapter 6, Section 9 13

Calculating the Heat in a Reaction If 15. 0 g of NO are produced according to the following reaction, how many k. J were absorbed? Step 3 Write the conversion factors including heat of reaction. © 2013 Pearson Education, Inc. Chapter 6, Section 9 14

Calculating the Heat in a Reaction If 15. 0 g of NO are produced according to the following reaction, how many k. J were absorbed? Step 4 Set up the problem to calculate the heat. © 2013 Pearson Education, Inc. Chapter 6, Section 9 15

Learning Check How many grams of O 2 reacted if 306 k. J were released in the following reaction? A. 91. 9 g of O 2 B. 46. 0 g of O 2 C. 2. 87 g of O 2 © 2013 Pearson Education, Inc. Chapter 6, Section 9 16

Solution How many grams of O 2 reacted if 306 k. J were released in the following reaction? Step 1 State the given and needed quantities. Analyze the Problem. © 2013 Pearson Education, Inc. Chapter 6, Section 9 17

Solution How many grams of O 2 reacted if 306 k. J were released in the following reaction? Step 2 Write a plan using heat of the reaction and any molar mass needed. kilojoules © 2013 Pearson Education, Inc. Heat of heat of reaction molar moles of O 2 molar mass Chapter 6, Section 9 grams of O 2 18

Solution How many grams of O 2 reacted if 306 k. J were released in the following reaction? Step 3 Write the conversion factors including heat of reaction. © 2013 Pearson Education, Inc. Chapter 6, Section 9 19

Solution How many grams of O 2 reacted if 306 k. J were released in the following reaction? Step 4 Set up the problem to calculate mass of O 2. Answer is A. © 2013 Pearson Education, Inc. Chapter 6, Section 9 20
- Slides: 20