General Organic and Biological Chemistry Fourth Edition Karen

General, Organic, and Biological Chemistry Fourth Edition Karen Timberlake Chapter 1 Chemistry and Measurements 1. 10 Density © 2013 Pearson Education, Inc. Lectures

Density § compares the mass of an object to its volume. § is the mass of a substance divided by its volume. § are measured in g/L for gases. § are measured in g/cm 3 or g/m. L for solids and liquids. Density expression: © 2013 Pearson Education, Inc. Chapter 1, Section 10 2
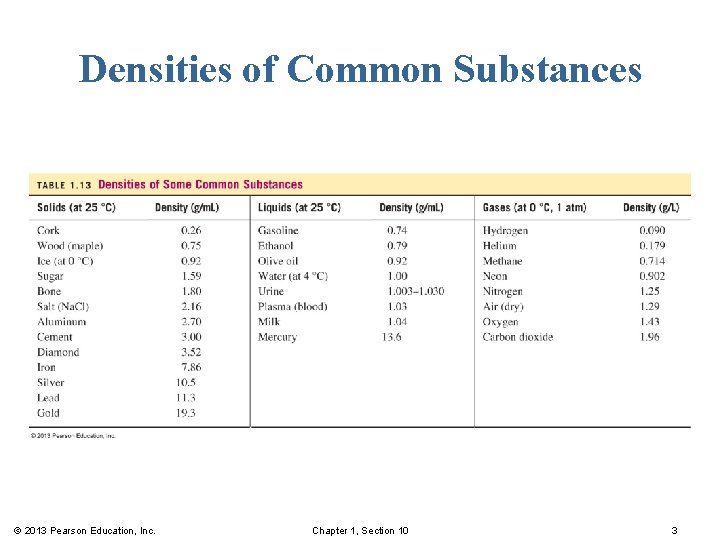
Densities of Common Substances © 2013 Pearson Education, Inc. Chapter 1, Section 10 3

Density Calculations © 2013 Pearson Education, Inc. Chapter 1, Section 10 4

Calculating Density If a 0. 258 -g sample of HDL has a volume of 0. 215 cm 3, what is the density, in g/cm 3, of the HDL sample? Step 1 State the given and needed quantities. Analyze the Problem. Given Need 0. 258 g HDL density in g/cm 3 of HDL 0. 215 cm 3 HDL Step 2 Write the density expression. © 2013 Pearson Education, Inc. Chapter 1, Section 10 5

Calculating Density If a 0. 258 -g sample of HDL has a volume of 0. 215 cm 3, what is the density, in g/cm 3, of the HDL sample? Step 3 Express mass in grams and volume in milliliters (m. L) or cm 3. Step 4 Substitute mass and volume into the density expression and calculate the density. © 2013 Pearson Education, Inc. Chapter 1, Section 10 6

Learning Check Osmium is a very dense metal. What is its density in g/cm 3 if 50. 0 g of osmium has a volume of 2. 22 cm 3? A. 2. 25 g/cm 3 B. 22. 5 g/cm 3 C. 111 g/cm 3 © 2013 Pearson Education, Inc. Chapter 1, Section 10 7

Solution Osmium is a very dense metal. What is its density in g/cm 3 if 50. 0 g of osmium has a volume of 2. 22 cm 3. Step 1 State the given and needed quantities. Analyze the Problem Given 50. 0 g osmium 2. 22 cm 3 Need density g/cm 3 Step 2 Write the density expression. © 2013 Pearson Education, Inc. Chapter 1, Section 10 8

Solution Osmium is a very dense metal. What is its density in g/cm 3 if 50. 0 g of osmium has a volume of 2. 22 cm 3. Step 3 Express mass in grams and volume in cm 3. Step 4 Substitute into the density expression. © 2013 Pearson Education, Inc. Chapter 1, Section 10 9

Measuring Density of Solids Volume displacement is the volume of a solid calculated from the volume of water displaced when it is submerged. © 2013 Pearson Education, Inc. Chapter 1, Section 10 10

Measuring Density Measure the mass of the solid before submerging it in water to determine its volume. The density of this zinc object is calculated from its mass and volume. © 2013 Pearson Education, Inc. Chapter 1, Section 10 11
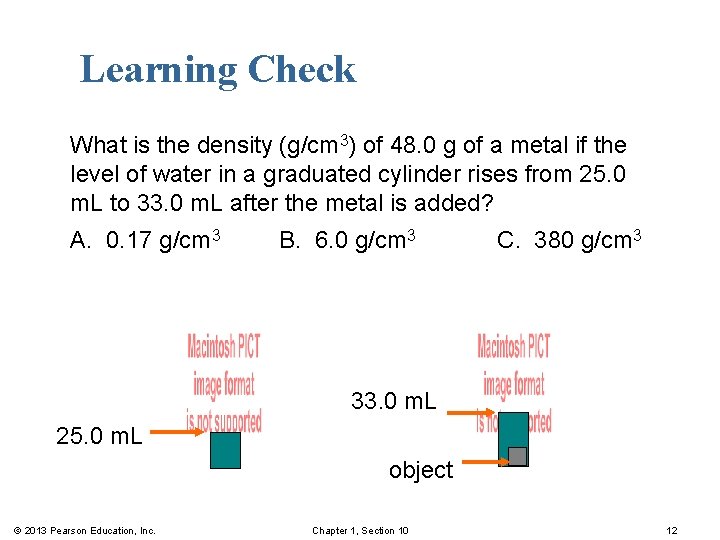
Learning Check What is the density (g/cm 3) of 48. 0 g of a metal if the level of water in a graduated cylinder rises from 25. 0 m. L to 33. 0 m. L after the metal is added? A. 0. 17 g/cm 3 B. 6. 0 g/cm 3 C. 380 g/cm 3 33. 0 m. L 25. 0 m. L object © 2013 Pearson Education, Inc. Chapter 1, Section 10 12

Solution What is the density (g/cm 3) of 48. 0 g of a metal if the level of water in a graduated cylinder rises from 25. 0 m. L to 33. 0 m. L after the metal is added? Step 1 State the given and needed quantities. Analyze the Problem. Step 2 © 2013 Pearson Education, Inc. Write the density expression. Chapter 1, Section 10 13

Solution What is the density (g/cm 3) of 48. 0 g of a metal if the level of water in a graduated cylinder rises from 25. 0 m. L to 33. 0 m. L after the metal is added? Step 3 Express mass in grams and volume in cm 3. © 2013 Pearson Education, Inc. Chapter 1, Section 10 14

Solution What is the density (g/cm 3) of 48. 0 g of a metal if the level of water in a graduated cylinder rises from 25. 0 m. L to 33. 0 m. L after the metal is added? Step 4 Substitute mass and volume into density expression. Answer: B © 2013 Pearson Education, Inc. Chapter 1, Section 10 15

Sink or Float Ice floats in water because the density of ice is less than the density of water. Aluminum sinks in water because its density is greater than the density of water. © 2013 Pearson Education, Inc. Chapter 1, Section 10 16

Problem Solving using Density can be written as a conversion factor. © 2013 Pearson Education, Inc. Chapter 1, Section 10 17

Problem Solving using Density can be used as a conversion factor. A density of 3. 8 g/m. L, can be written as an equality, or written as conversion factors. © 2013 Pearson Education, Inc. Chapter 1, Section 10 18

Problem: Density as a Conversion Factor If the density of milk is 1. 04 g/m. L, how many grams of milk are in 0. 50 qt of milk? Step 1 State the given and needed quantities. Analyze the Problem. © 2013 Pearson Education, Inc. Chapter 1, Section 10 19

Problem: Density as a Conversion Factor If the density of milk is 1. 04 g/m. L, how many grams of milk are in 0. 50 qt of milk? Step 2 Write a plan to calculate needed quantity. volume US–Metric factor density factor mass Step 3 Write equalities and conversion factors. © 2013 Pearson Education, Inc. Chapter 1, Section 10 20

Problem: Density as a Conversion Factor If the density of milk is 1. 04 g/m. L, how many grams of milk are in 0. 50 qt of milk? Step 4 Set up the problem to calculate the needed quantity. © 2013 Pearson Education, Inc. Chapter 1, Section 10 21

Learning Check The density of octane, a component of gasoline, is 0. 702 g/m. L. What is the mass, in kg, of 875 m. L of octane? A. 0. 614 kg B. 614 kg C. 1. 25 kg © 2013 Pearson Education, Inc. Chapter 1, Section 10 22

Solution The density of octane, a component of gasoline, is 0. 702 g/m. L. What is the mass, in kg, of 875 m. L of octane? Step 1 State the given and needed quantities. Analyze the Problem © 2013 Pearson Education, Inc. Chapter 1, Section 10 23

Solution The density of octane, a component of gasoline, is 0. 702 g/m. L. What is the mass, in kg, of 875 m. L of octane? Step 2 Write a plan to convert the given unit to the needed unit. Density m. L Factor © 2013 Pearson Education, Inc. Metric Factor Chapter 1, Section 10 kg 24

Solution The density of octane, a component of gasoline, is 0. 702 g/m. L. What is the mass, in kg, of 875 m. L of octane? Step 3 State the equalities and conversion factors. © 2013 Pearson Education, Inc. Chapter 1, Section 10 25

Solution The density of octane, a component of gasoline, is 0. 702 g/m. L. What is the mass, in kg, of 875 m. L of octane? Step 4 Set up the problem to cancel units and calculate the answer. Answer: B © 2013 Pearson Education, Inc. Chapter 1, Section 10 26

Specific Gravity (sp gr) § is the relationship between the density of a substance and the density of water. § is determined by dividing the density of the sample by the density of water. § is a unitless quantity. © 2013 Pearson Education, Inc. Chapter 1, Section 10 27

Specific Gravity is measured by an instrument called a hydrometer. © 2013 Pearson Education, Inc. Chapter 1, Section 10 28

Learning Check Which of the following samples of metals will displace the greatest volume of water? A 25 g of aluminum 2. 70 g/m. L © 2013 Pearson Education, Inc. B 45 g of gold 19. 3 g/m. L Chapter 1, Section 10 C 75 g of lead 11. 3 g/m. L 29

Solution The correct answer is A. 25 g of aluminum 2. 70 g/m. L Plan: Calculate the volume for each metal, and select the metal sample with the greatest volume. © 2013 Pearson Education, Inc. Chapter 1, Section 10 30
- Slides: 30