General Chemistry Principles and Modern Applications Petrucci Harwood




















- Slides: 51

General Chemistry Principles and Modern Applications Petrucci • Harwood • Herring 8 th Edition Chapter 12: Liquids, Solids and Intermolecular Forces Philip Dutton University of Windsor, Canada N 9 B 3 P 4 Prentice-Hall © 2002

Contents 12 -1 12 -2 12 -3 12 -4 12 -5 12 -6 12 -7 12 -8 Intermolecular Forces and some Properties of Liquids Vaporization of Liquids: Vapor Pressure Some Properties of Solids Phase Diagrams Van der Waals Forces Hydrogen Bonding Chemical Bonds as Intermolecular Forces Crystal structures Energy Changes in the Formation of Ionic Crystals Focus on Liquid Crystals Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 2 of 35

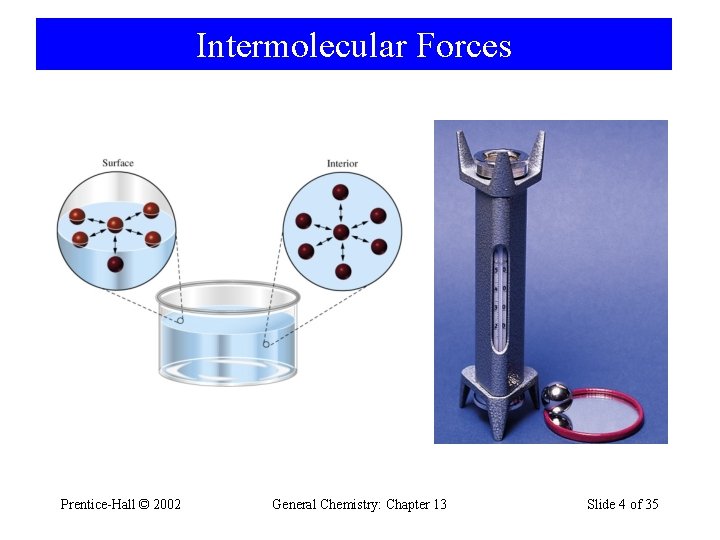
12 -1 Intermolecular Forces and Some Properties of Liquids • Cohesive Forces – Intermolecular forces between like molecules. • Adhesive Forces – Intermolecular forces between unlike molecules. • Surface Tension – Energy or work required to increase the surface area of a liquid. • Viscosity – A liquids resistance to flow Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 3 of 35

Intermolecular Forces Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 4 of 35

Intermolecular Forces Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 5 of 35

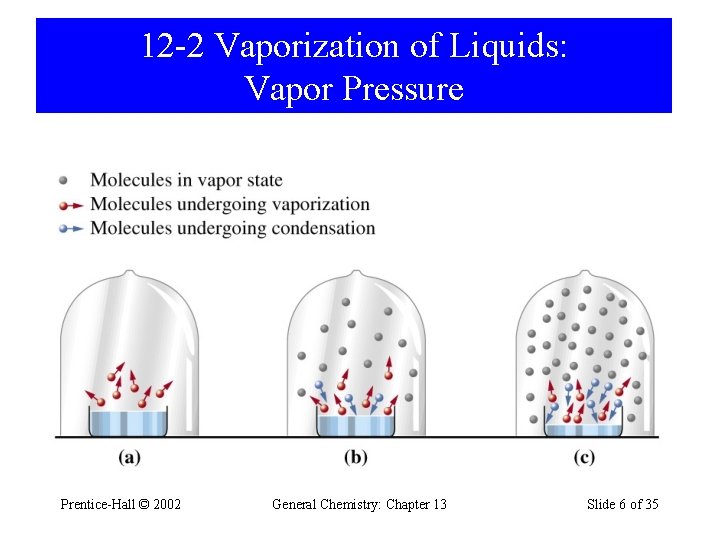
12 -2 Vaporization of Liquids: Vapor Pressure Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 6 of 35

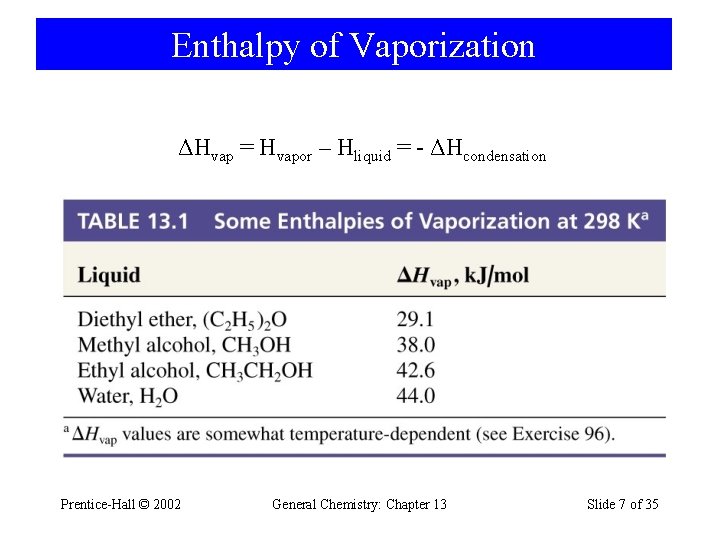
Enthalpy of Vaporization ΔHvap = Hvapor – Hliquid = - ΔHcondensation Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 7 of 35

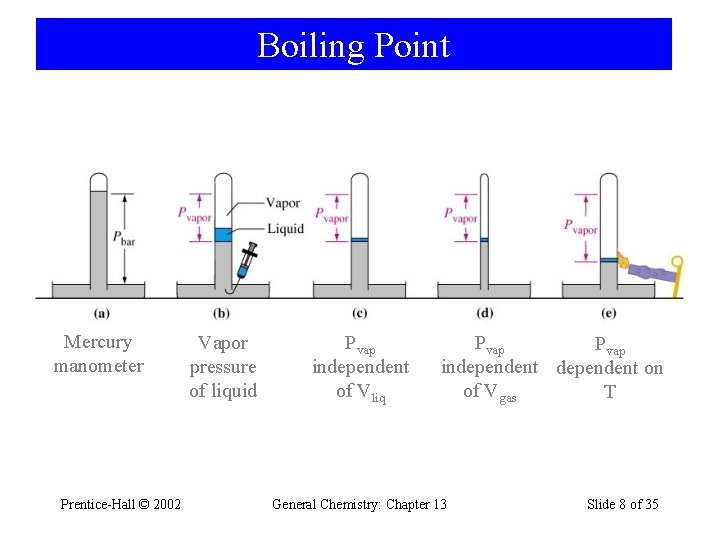
Boiling Point Mercury manometer Prentice-Hall © 2002 Vapor pressure of liquid Pvap independent of Vliq Pvap independent on of Vgas T General Chemistry: Chapter 13 Slide 8 of 35

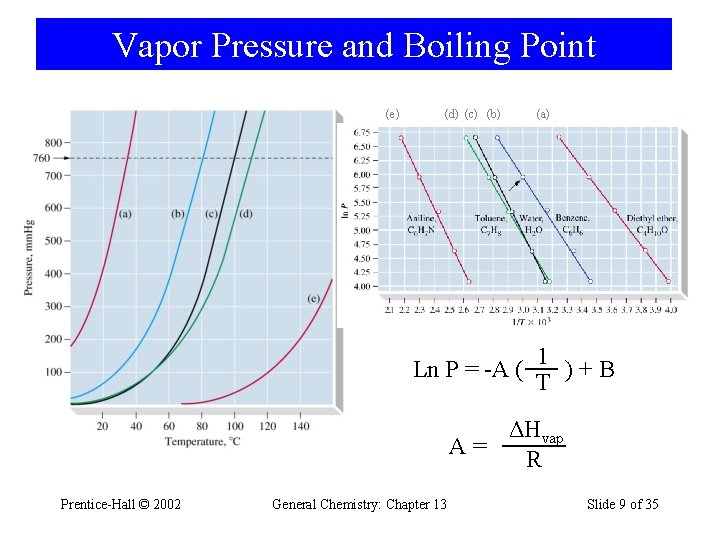
Vapor Pressure and Boiling Point (e) (d) (c) (b) (a) 1 Ln P = -A ( )+B T ΔHvap A= R Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 9 of 35

Clausius-Clapeyron Equation Ln P = -A ( 1 )+B T ΔHvap P 2 1 1 Ln =( ) P 1 T 2 R Example 12 -1: The vapor pressure is 5. 08 k. Pa at 18. 1 °C, the vaporization enthalpy is 41. 9 k. J/mol. What is the vapor pressure at 61. 1 °C? Example 12 -2: A compound has a 5. 258 k. Pa vapor pressure at 15. 80 °C. At 40. 35 °C it has 45. 61 k. Pa vapor pressure. What is the molar enthalpy of vaporization in J mol-1 of this compound? Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 10 of 35

12 -3 Some Properties of Solids Freezing Point Melting Point ΔHfus(H 2 O) = +6. 01 k. J/mol Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 11 of 35

Sublimation ΔHsub = ΔHfus + ΔHvap = -ΔHdeposition Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 12 of 35

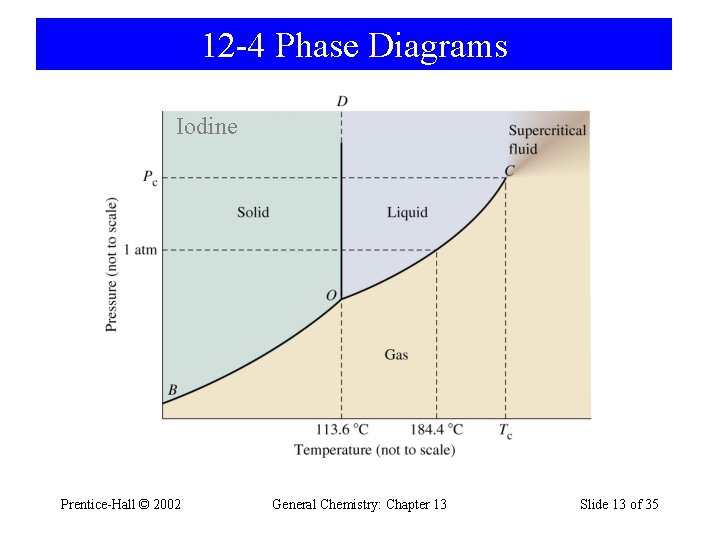
12 -4 Phase Diagrams Iodine Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 13 of 35

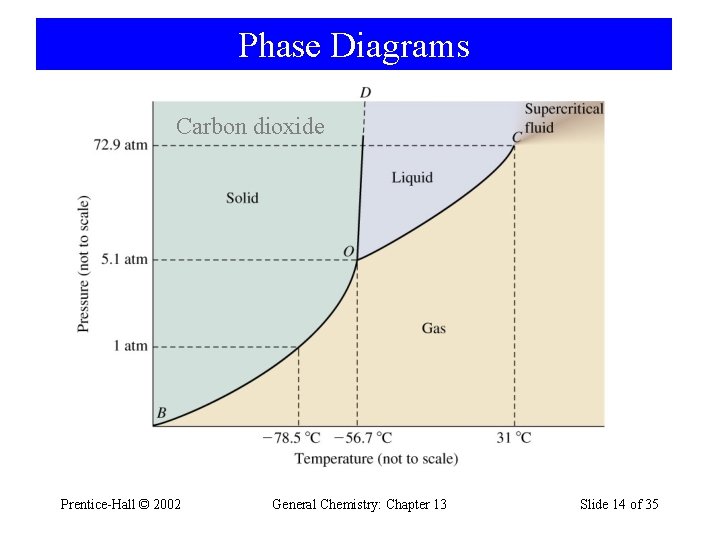
Phase Diagrams Carbon dioxide Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 14 of 35

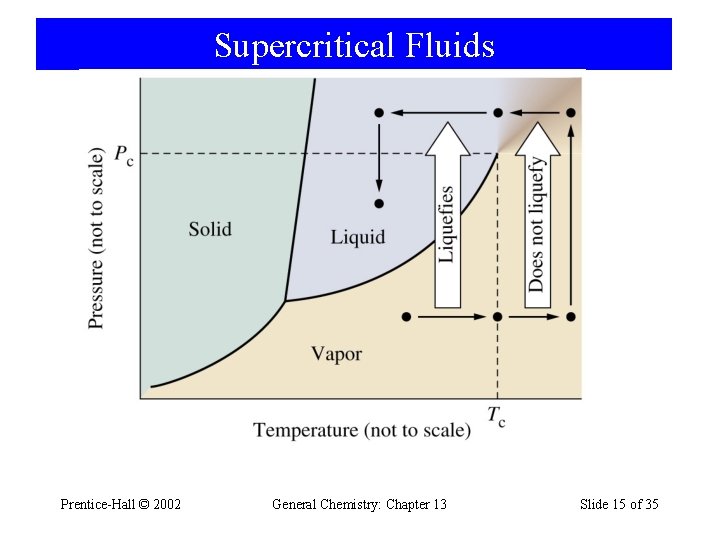
Supercritical Fluids Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 15 of 35

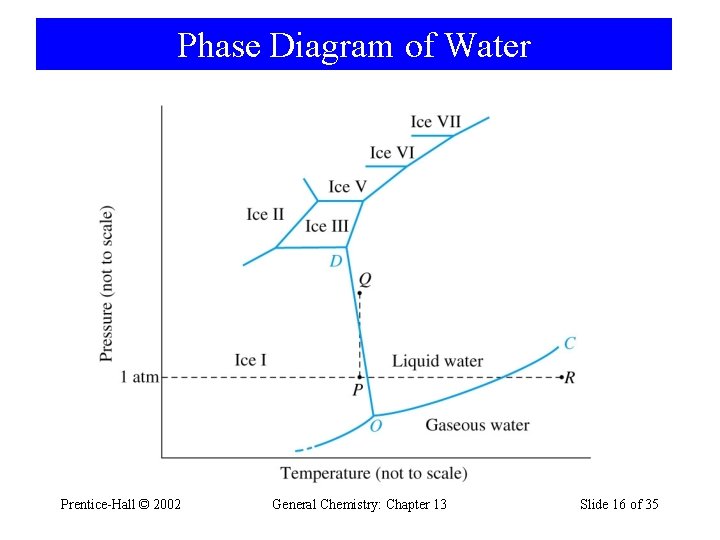
Phase Diagram of Water Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 16 of 35

The Critical Point Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 17 of 35

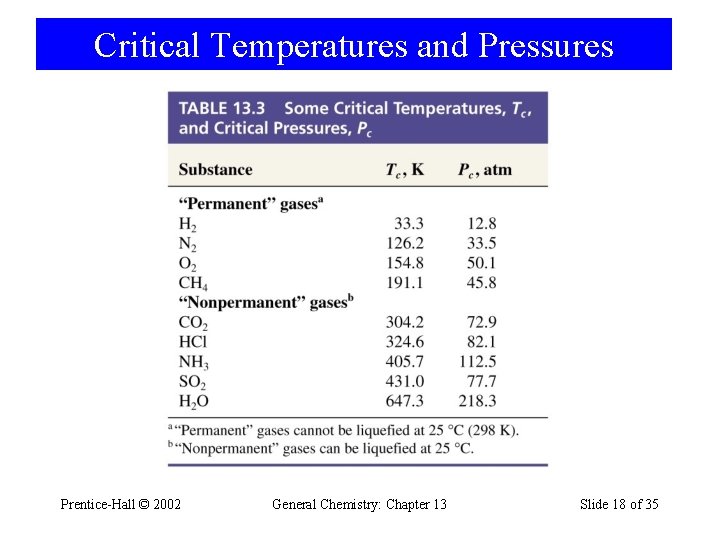
Critical Temperatures and Pressures Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 18 of 35

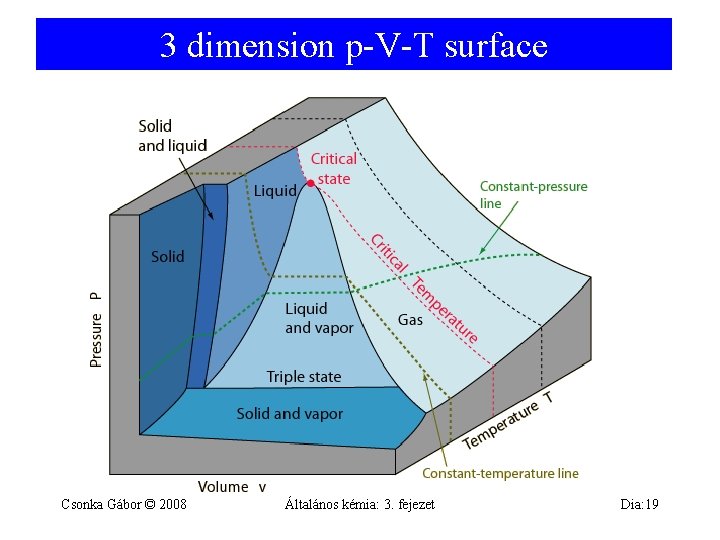
3 dimension p-V-T surface Csonka Gábor © 2008 Általános kémia: 3. fejezet Dia: 19

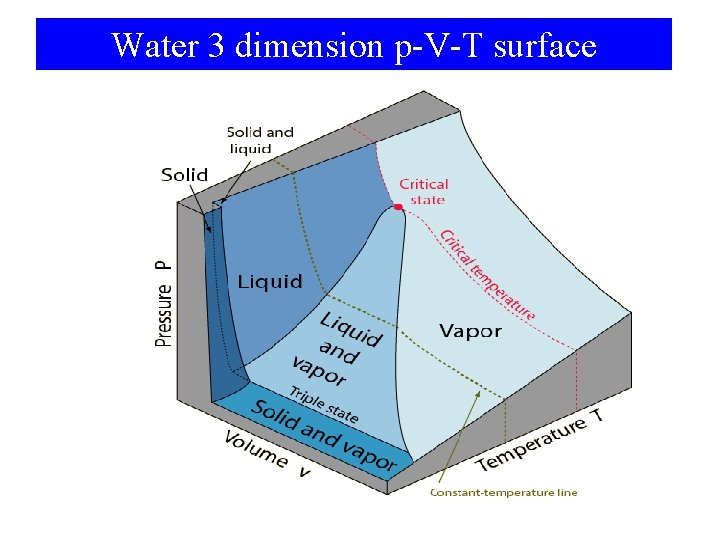
Water 3 dimension p-V-T surface

Van der Waals Constants • The Van der Waals constants can be obtained from the critical data: a = 3 pc Vc 3 Csonka Gábor © 2008 and Általános kémia: 3. fejezet b = Vc/3 Dia: 21

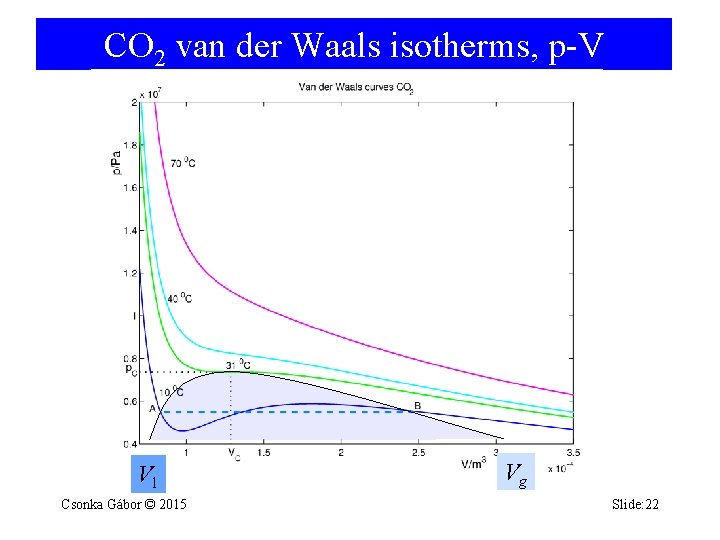
CO 2 van der Waals isotherms, p-V Vl Csonka Gábor © 2015 Vg Slide: 22

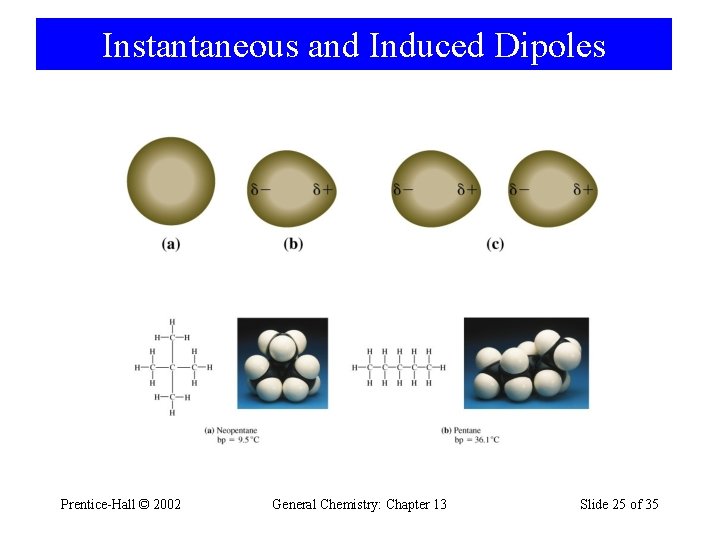
12 -5 Van der Waals Forces • Instantaneous dipoles. – Electrons move in an orbital to cause a polarization. • Induced dipoles. – Electrons move in response to an outside force. • Dispersion or London forces. – Instaneous dipole – induced dipole attraction. – Related to polarizability. Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 23 of 35

Phenomenon of Induction Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 24 of 35

Instantaneous and Induced Dipoles Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 25 of 35

London dispersion in hydrocarbons C 5 H 12 C 15 H 32 C 18 H 38 Slide 26 of 35

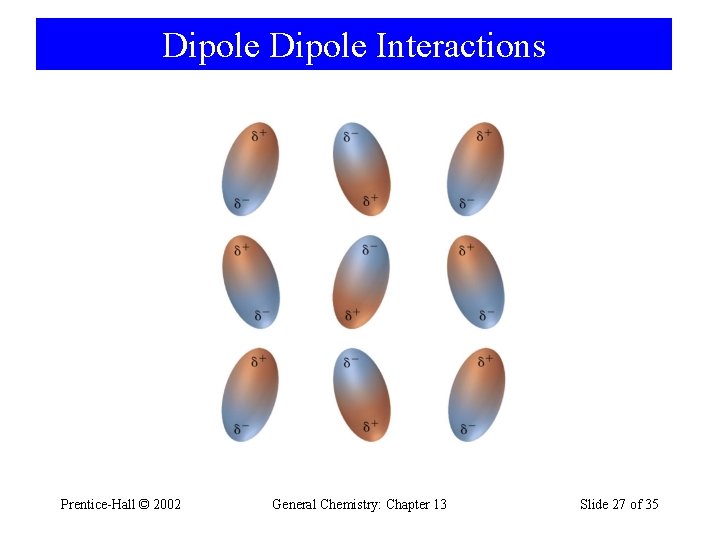
Dipole Interactions Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 27 of 35

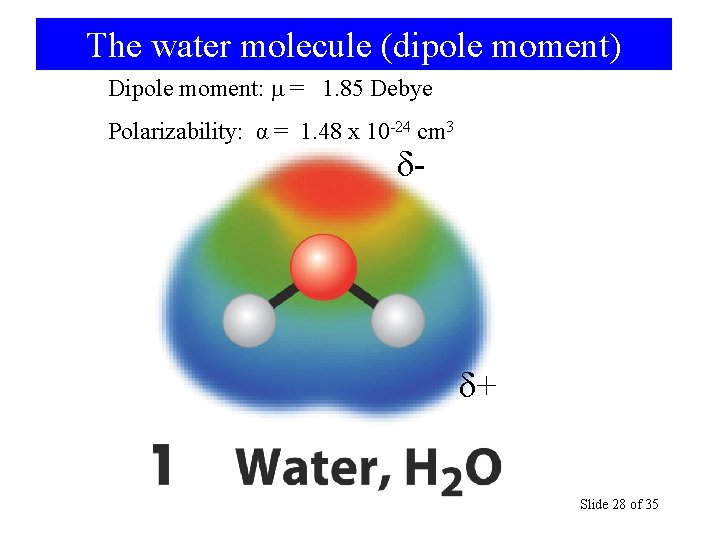
The water molecule (dipole moment) Dipole moment: = 1. 85 Debye Polarizability: α = 1. 48 x 10 -24 cm 3 δ- δ+ General Chemistry: Chapter 13 Slide 28 of 35

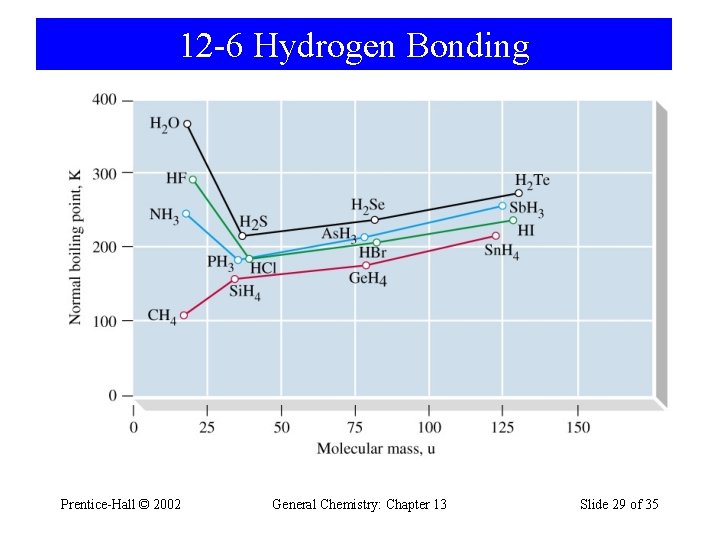
12 -6 Hydrogen Bonding Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 29 of 35

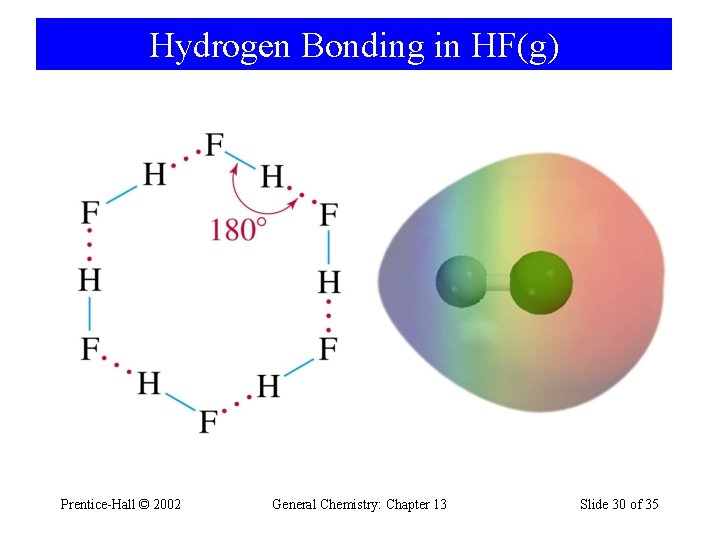
Hydrogen Bonding in HF(g) Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 30 of 35

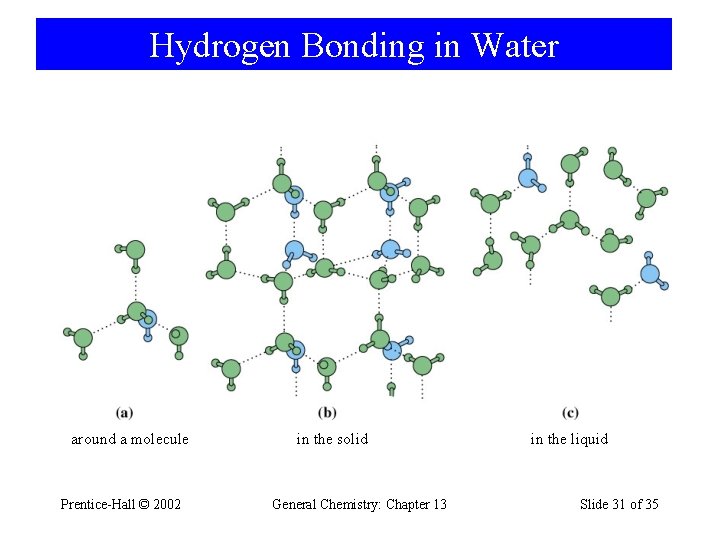
Hydrogen Bonding in Water around a molecule Prentice-Hall © 2002 in the solid General Chemistry: Chapter 13 in the liquid Slide 31 of 35

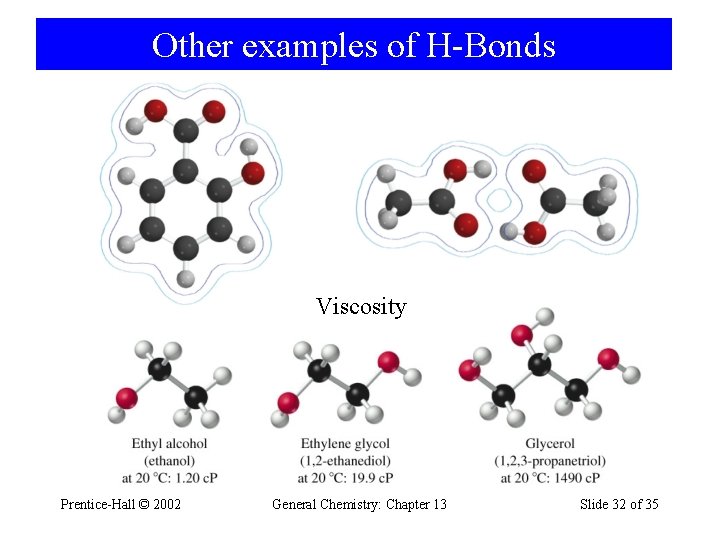
Other examples of H-Bonds Viscosity Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 32 of 35

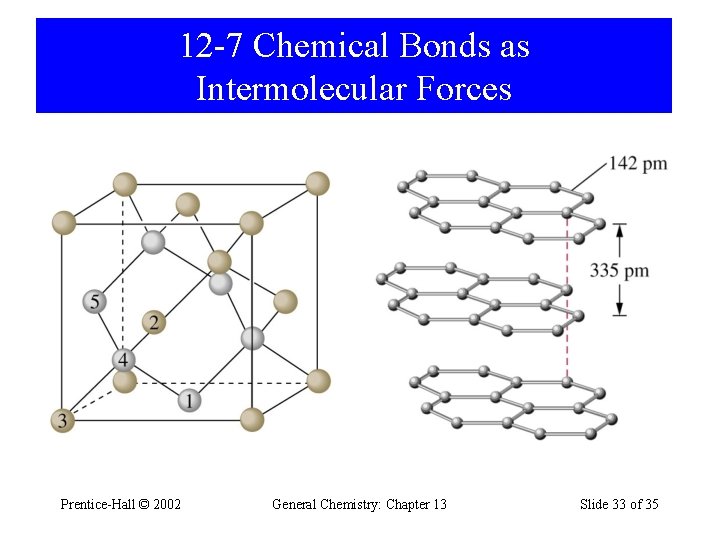
12 -7 Chemical Bonds as Intermolecular Forces Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 33 of 35

Other Carbon Allotropes Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 34 of 35

Interionic Forces Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 35 of 35

12 -8 Crystal Structures Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 36 of 35

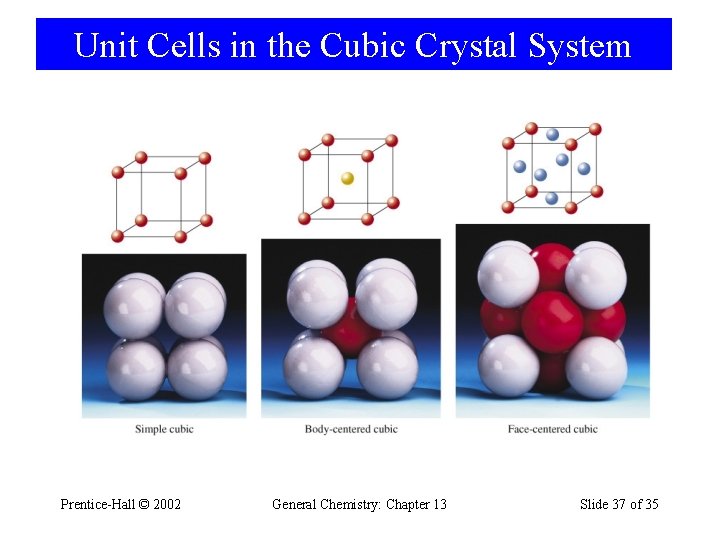
Unit Cells in the Cubic Crystal System Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 37 of 35

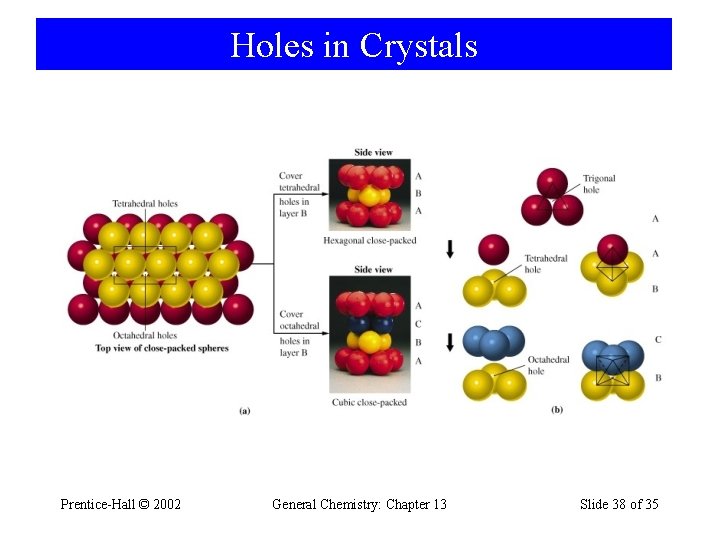
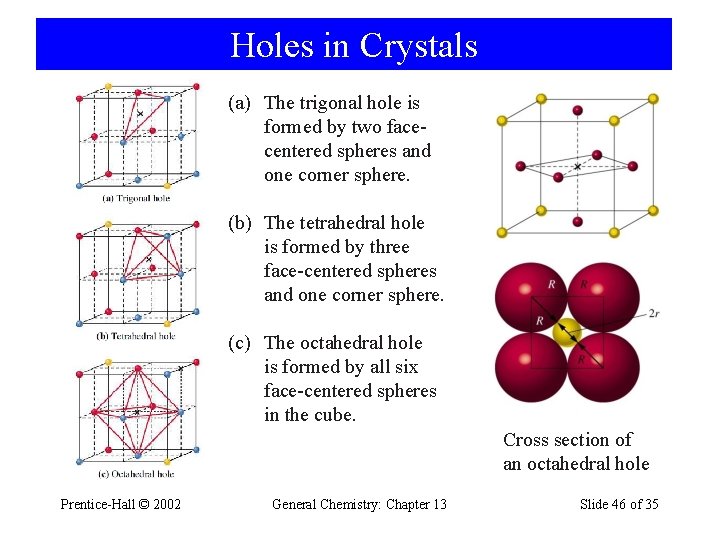
Holes in Crystals Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 38 of 35

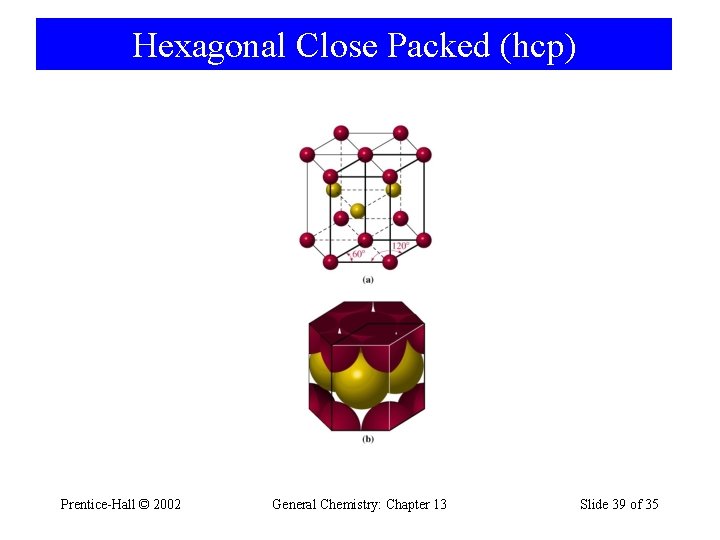
Hexagonal Close Packed (hcp) Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 39 of 35

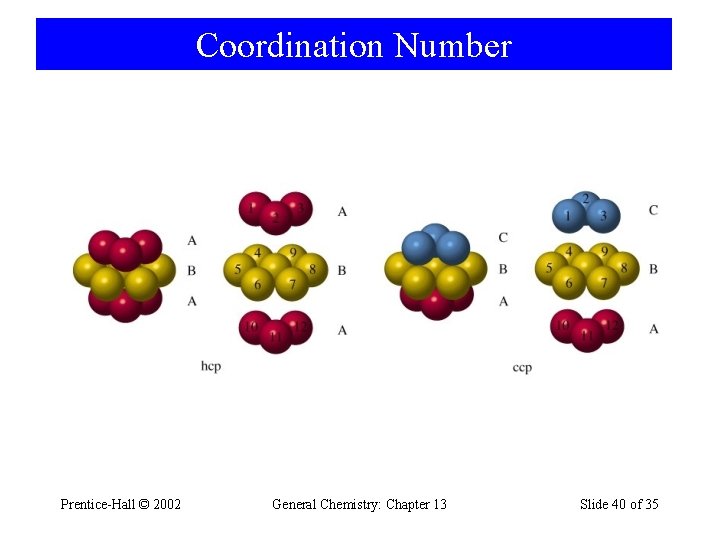
Coordination Number Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 40 of 35

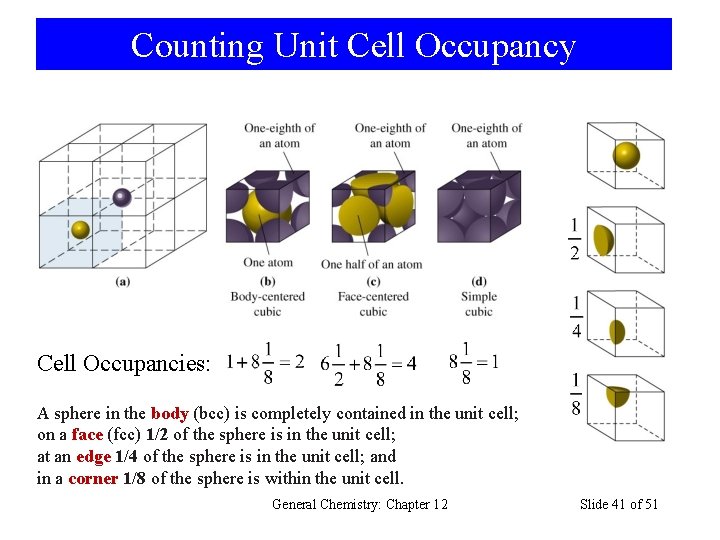
Counting Unit Cell Occupancy Cell Occupancies: A sphere in the body (bcc) is completely contained in the unit cell; on a face (fcc) 1/2 of the sphere is in the unit cell; at an edge 1/4 of the sphere is in the unit cell; and in a corner 1/8 of the sphere is within the unit cell. General Chemistry: Chapter 12 Slide 41 of 51

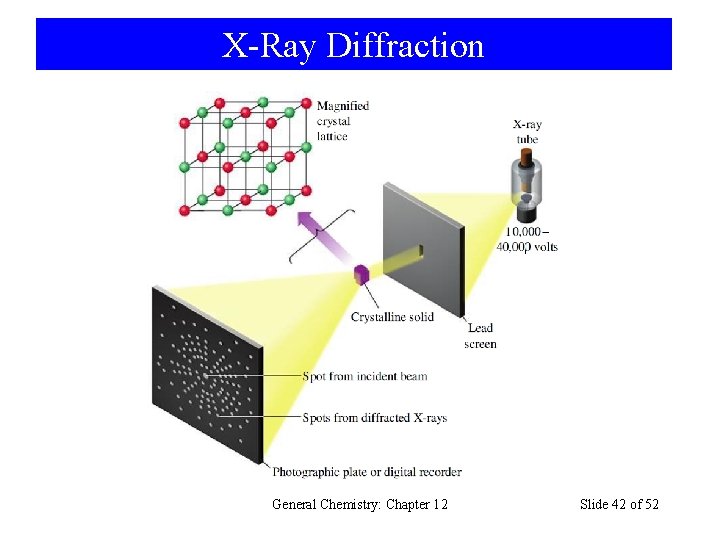
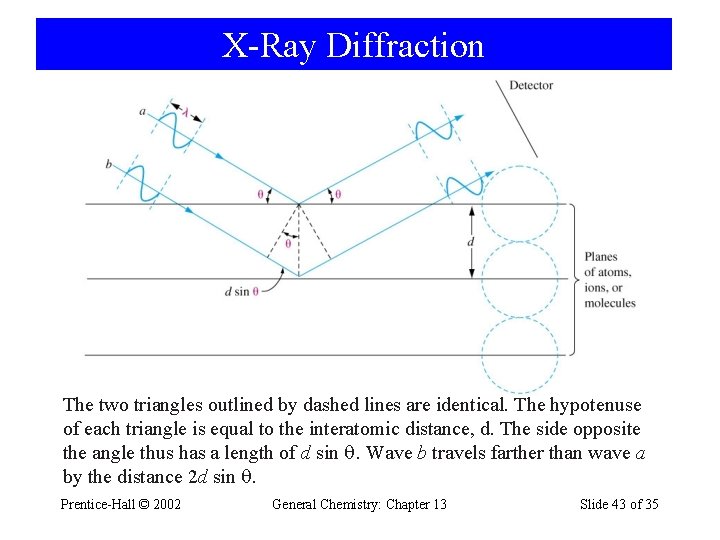
X-Ray Diffraction General Chemistry: Chapter 12 Slide 42 of 52

X-Ray Diffraction The two triangles outlined by dashed lines are identical. The hypotenuse of each triangle is equal to the interatomic distance, d. The side opposite the angle thus has a length of d sin . Wave b travels farther than wave a by the distance 2 d sin . Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 43 of 35

Atomic Radii from Crystal Structures At room temperature, iron crystallizes in a bcc structure. By X -ray diffraction, the edge of the cubic cell is found to be 286. 7 pm. What is the radius of an iron atom? The length of the cube diagonal is four times the atomic radius. r = 124. 1 pm Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 44 of 35

Relating Density to Crystal Structure Data Calculate the density of iron (example 12 -10, p 537): • we need the mass of the unit cell (in grams) and its volume (in cm 3) • there are two Fe atoms per bcc unit cell, which we can use to calculate the mass: m = 2 MFe / NA • the length of the unit cell is l = 286. 7 pm = 2. 867 10 -8 cm, which we can use to calculate the volume: V = l 3, and the density: d = m/V. • Density of Fe: 7. 871 g/cm 3 General Chemistry: Chapter 12 Slide 45 of 52

Holes in Crystals (a) The trigonal hole is formed by two facecentered spheres and one corner sphere. (b) The tetrahedral hole is formed by three face-centered spheres and one corner sphere. (c) The octahedral hole is formed by all six face-centered spheres in the cube. Cross section of an octahedral hole Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 46 of 35

Ionic Crystal: Cesium Chloride (1) Some of the ions are positively charged and some are negatively charged, and (2) the cations and anions are of different sizes. (3) The relative sizes of cations and anions are important in establishing a particular packing arrangement. Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 47 of 35

Sodium Chloride Unit Cell Oppositely charged ions are in contact. We can think of this structure as an fcc lattice of Cl- ions, with Na+ ions filling the octahedral holes. 4 Na+ ions and 4 Cl- ions, total 8 ions are in the unit cell. General Chemistry: Chapter 13 Slide 48 of 35

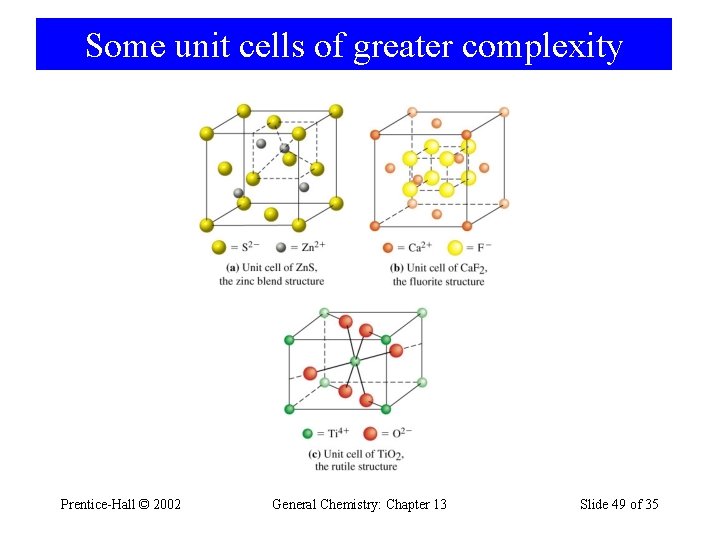
Some unit cells of greater complexity Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 49 of 35

12 -9 Enthalpy diagram for the Formation of Na. Cl Ionic Crystal Step 1. Sublime one mole of solid Na. Step 2. Dissociate 0. 5 mole Cl 2(g) of into one mole of Cl(g). Step 3. Ionize one mole of Na(g) to Na+(g). Step 4. Convert one mole of Cl(g) to Cl-(g). Step 5. Allow the Na+(g) and Cl(g) to form one mole of Na. Cl(s). Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 50 of 35

Chapter 13 Questions 1, 3, 4, 13, 24, 26, 31, 45, 52, 61, 94, 107 Prentice-Hall © 2002 General Chemistry: Chapter 13 Slide 51 of 35