General Biology lab Lab 6 Diffusion and Osmosis

General Biology lab Lab 6 Diffusion and Osmosis

• Solution : – is a homogeneous mixture of two or more components. • Solvent: – the substance present in the largest amount in a solution. • Solute : – substance present in smaller amount.
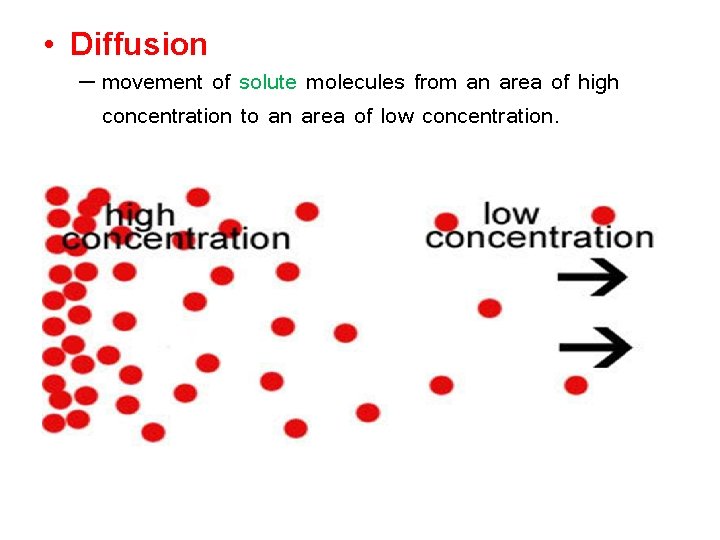
• Diffusion – movement of solute molecules from an area of high concentration to an area of low concentration.
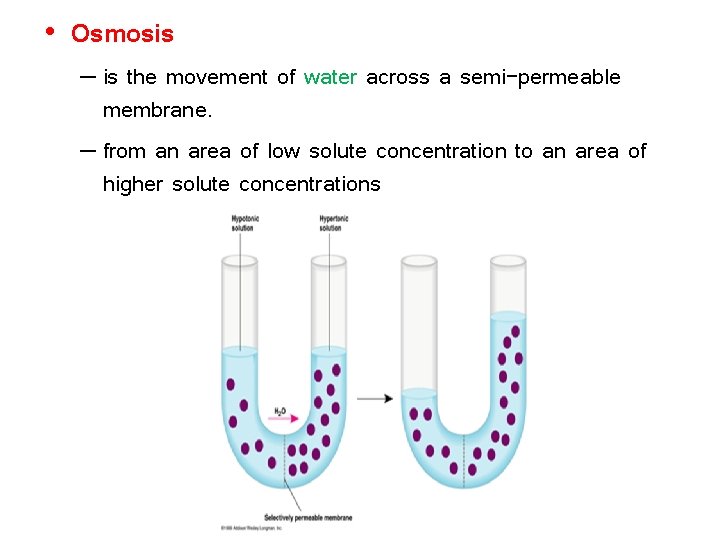
• Osmosis – is the movement of water across a semi-permeable membrane. – from an area of low solute concentration to an area of higher solute concentrations

Procedures diffusion and molecular weight 1. In one region of the a petri dish (upper portion ) place a few of drops of green stain (MW= 150 g/mole) 2. Then add in the opposite region, a few drops of red stain ( MW= 400 g/mole) 3. Examine what happened every 15 minutes for one hour.

Diffusion across a differentially permeable membrane 1. Fill one bag (Dialysis membranes) with phenolphthalein (a p. H indicator that turns red in basic/alkaline conditions ) 2. Then submerge the bag into a beaker containing sodium hydroxide (Na. OH – very alkaline solution ) • Dialysis membranes – are differentially permeable (only molecules that are small enough can pass through the membrane ).

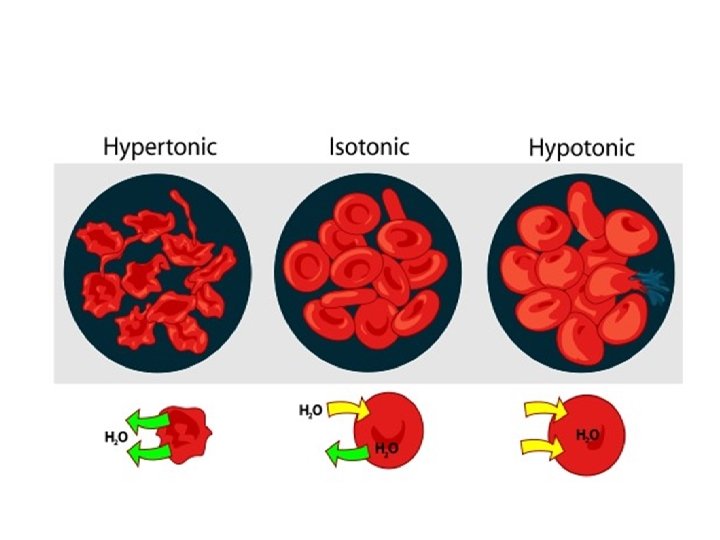
Osmosis and Cells • The movement of liquids in and out cells is dependant on the concentration of the solution surrounding it. • There are 3 types of situations in which this could vary: 1. Isotonic: – Here the external solution concentration and the internal concentration of the organism are the same.

2. Hypotonic: – Here the external solution concentration is less than the concentration of the organism. – In this case water will rush into the organism. 3. Hypertonic: – Here the external solution concentration is greater than the concentration of the organism. – In this case the water will rush out of the organism.

Effects of Osmosis on Plant Tissues Plasmolysis • It refers to the shrinking of the cytoplasm of a cell in response to diffusion of water out of the cell. • This may occur when cells are placed in a (hypertonic ) solution.

procedure • Cut 3 pieces of potato to the same size • Put the first into 0% Na. CL. – the second in 5% Na. Cl. – the third 15% Na. Cl. • Leave them for 15 - 20 minutes.

Normal cells Plasmolysed cells

Effects of Osmosis on RBCs Hemolysis • meaning "blood loosing" is the rupturing of red blood cells and the release of their contents into surrounding fluid • Hemolysis occurs when red blood cells are placed in hypotonic solutions.

Crenation • A process resulting from osmosis in which red blood cells, in a hypertonic solution, undergo shrinkage.

procedure • Label three clean serological tube 1 to 3 and arrange them sequentially in a test tube rack. • deliver 2 ml of each solution (Solution 0. 6 M Na. Cl 0. 9 M Na. Cl, 1. 4 M Na. Cl) to the appropriate tube. • Dispense 2 drops of blood to each tube using a syringe. Shake each tube from side to mix , then let stand for 5 minutes

• Hold the rack of tubes up to the light and compare them – if the solution is transparent, hemolysis has occurred. If you are unable to see through the tube, no hemolysis has occurred and the cells should be intact. – If crenation has occurred, the appearance will be somewhat between the clarity of hemolysis and the opacity of osmotic equilibrium.

- Slides: 18