General Biology lab Lab 2 Molecules of living

General Biology lab Lab 2 Molecules of living things

Objective • Detection of Carbohydrate by using general test and specific test. • Detection of lipids by using Emulsification test. • Detection of proteins by using Biuret test.

I. Carbohydrates • A carbohydrate : – is an organic compound with the general formula Cm(H 2 O)n, that is, consists only of carbon, hydrogen and oxygen. • Are the most abundant and diverse class of organic compounds occurring in nature. • The main function of carbohydrates is to act as a source of energy for vital activities. • In general, the names of most carbohydrates are recognizable by an -ose -suffix.

• Carbohydrates are classified into three groups according to the number of sugar : 1. Monosaccharide – is the simplest kind of carbohydrate. – It consists of a single sugar molecule, – such as fructose or glucose

2. Disaccharide • Consists of two sugar molecules joined by a glycosidic linkage. • Some common disaccharides : 1. glucose + fructose = sucrose (table sugar) 2. glucose + galactose = lactose (the sugar in milk) 3. glucose + glucose = maltose

3. Polysaccharide • Are formed of large number of monosaccharaides joined by a glycosidic linkage. • The following examples of polysaccharides may contain thousands of glucose monomers: – Starch : is a polymer of α-glucose molecules. – Glycogen : is a polymer of α-glucose. It differs from starch by its pattern of polymer branching. – Cellulose : is a polymer of β-glucose molecules.
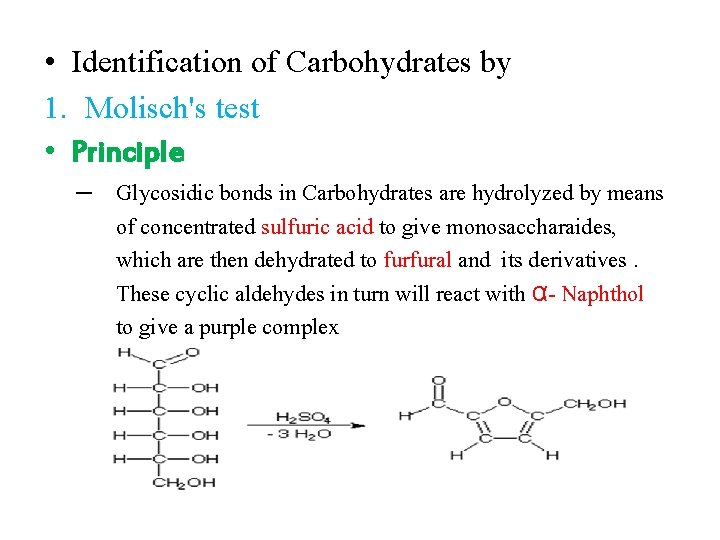
• Identification of Carbohydrates by 1. Molisch's test • Principle – Glycosidic bonds in Carbohydrates are hydrolyzed by means of concentrated sulfuric acid to give monosaccharaides, which are then dehydrated to furfural and its derivatives. These cyclic aldehydes in turn will react with α- Naphthol to give a purple complex

• Materials 1. Sulfuric acid H 2 SO 4 2. α- Naphthol 3. Solution of glucose, starch, sucrose • Procedure 1. In a clean test tube , place 2 ml of sugar solution 2. Add 3 drops of alcoholic α- Naphthol 3. Mix thoroughly, incline the tube and allow about 2 ml of concentrated sulfuric acid to flow down the side of the tube, thus forming a layer of the acid beneath the tested solution. 4. Carefully observe a color change at the junction of the tow liquids. 5. A purple ring is indicative of carbohydrates.
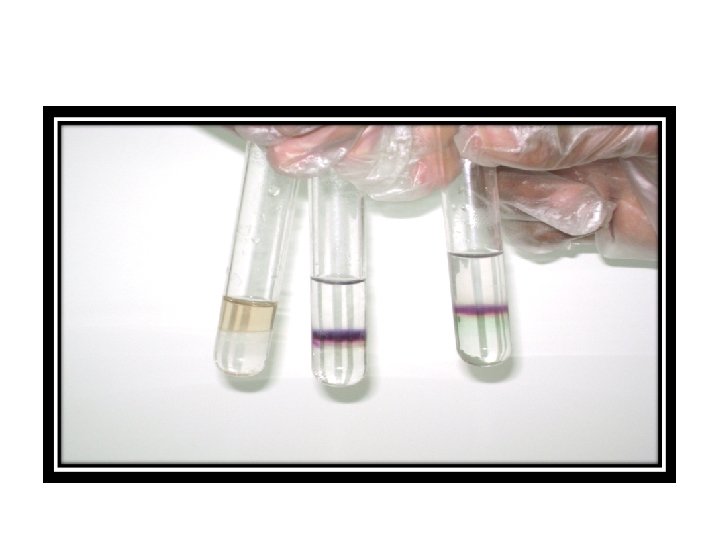

2. Benedict's test for reducing sugars • Principle • Benedict's solution test for presence of the two functional groups aldehyde and ketone present in many but not all sugar. Both functional groups contain an oxygen atom double-bond to carbon. sugar that contain these groups are called reducing sugar. • cupric ions, which in an alkaline environment, oxidize the aldehyde group to a carboxylic acid. Cupric ions are reduced to cuprous oxide, which forms a red color. • RCHO + 2 Cu 2+ + 4 OH- > RCOOH + Cu 2 O + 2 H 2 O

• Material 1. Benedict's solution 2. Solution of glucose, starch, sucrose. • Procedure 1. Label 4 test tube (glucose, starch, sucrose , distilled water). 2. Add 3 ml of Benedict solution to each tube 3. Place the tubes in a boiling water bath for 2 -4 minutes


II. Lipids – consists mostly of hydrocarbons – that are insoluble in water (and other polar solvents) – but are soluble in nonpolar substances (like ether or chloroform). • There are three major groups of lipids 1. Triglycerides 2. phospholipid 3. Steroids
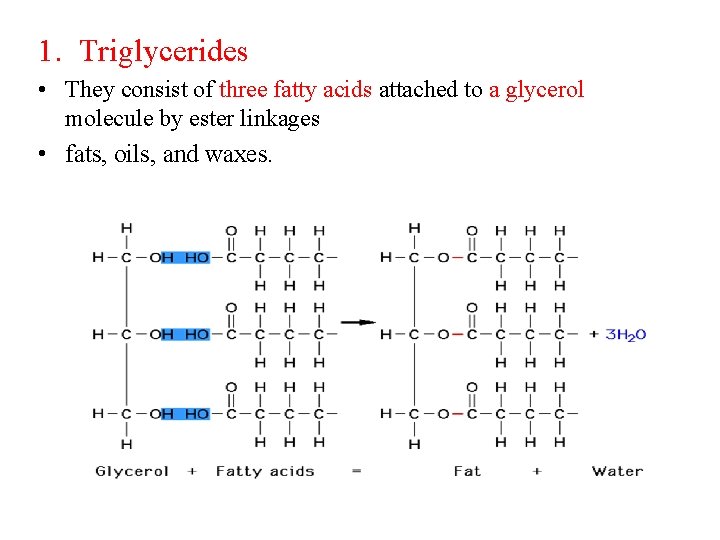
1. Triglycerides • They consist of three fatty acids attached to a glycerol molecule by ester linkages • fats, oils, and waxes.
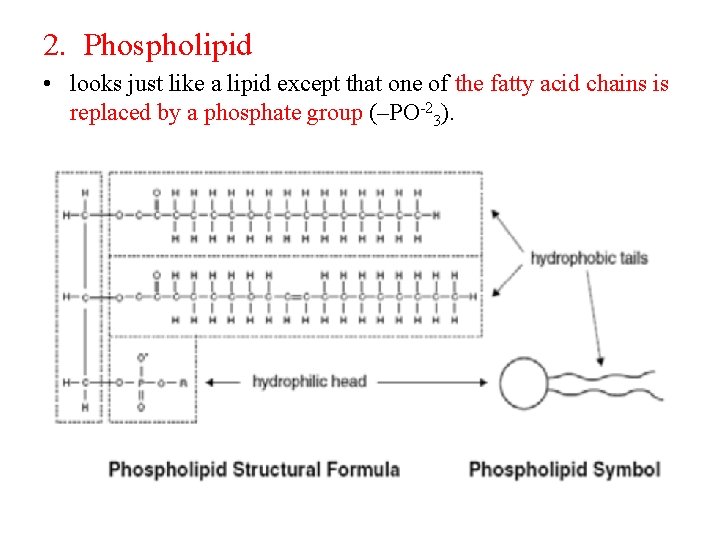
2. Phospholipid • looks just like a lipid except that one of the fatty acid chains is replaced by a phosphate group (–PO-23).
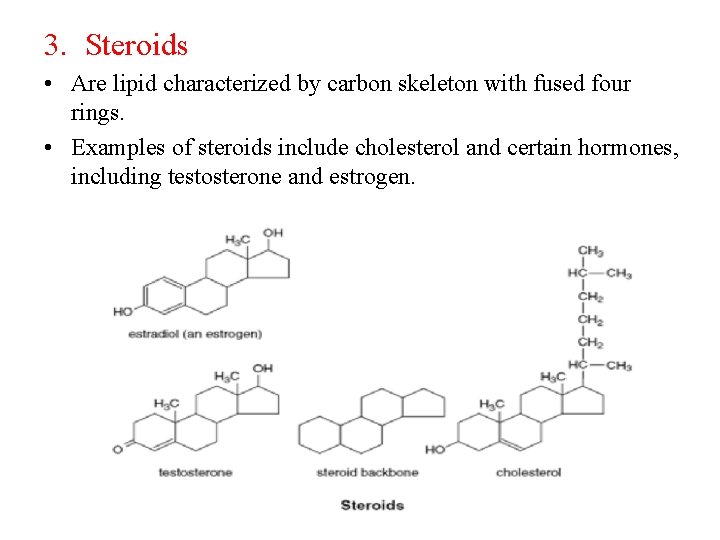
3. Steroids • Are lipid characterized by carbon skeleton with fused four rings. • Examples of steroids include cholesterol and certain hormones, including testosterone and estrogen.

• Identification of lipids by Emulsification test. • Materials 1. Neutral olive oil 2. Solution of Na 2 Co 3 • Procedure • TO 5 ml water in a test tube add 2 to 3 drops of Na 2 Co 3. introduce into this alkaline solution a drop of neutral olive oil and shake.
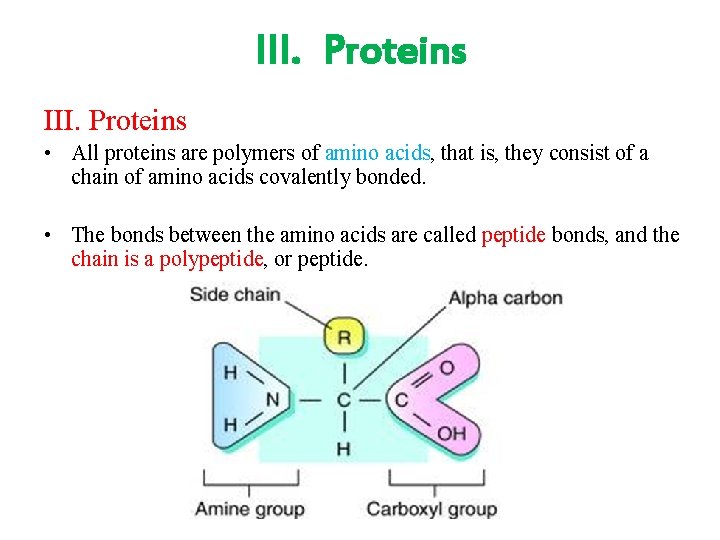
III. Proteins • All proteins are polymers of amino acids, that is, they consist of a chain of amino acids covalently bonded. • The bonds between the amino acids are called peptide bonds, and the chain is a polypeptide, or peptide.

• Identification of protein by Biuret test. • Principle – The copper atoms of Biuret solution (Cu. SO 4 ) in a basic environment will react with peptide bonds (-CO ---NH) to form a chelate of a deep violet color, indicating the presence of proteins. • Materials 1. Copper sulfate Cu. So 4 2. Sodium hydroxide Na. OH 3. Protein solution (Albumin and Gelatin)

• Procedure 1. Place 2 -3 ml of the protein solution in a test tube. 2. Add equal volume of Na. OH solution and mix thoroughly 3. Add Cu. So 4 solution drop by drop, mix between drops.
- Slides: 20