Gene Therapy Presented by Professor Mohamed Abd Ellatif




Gene Therapy


Presented by Professor Mohamed Abd Ellatif Professor of Medical Biochemistry And Molecular Biology Faculty of Medicine, Mansoura University Mansoura, Egypt

Definition of Gene therapy is an experimental technique that uses genes to treat or prevent disease.

Approaches of gene therapy § Replacing a mutated gene that causes disease with a healthy copy of the gene § Inactivating, or “knocking out” a mutated gene that is functioning improperly. § Introducing a new gene into the body to help fight a disease.

Principle n Introduction of genetic material into cells to compensate for abnormal genes or to make a beneficial protein.

Two approaches for delivering genetic material exist • In vivo gene therapy • Ex vivo gene therapy


In vivo gene therapy l Direct delivery of DNA (usually via a viral vector) to resident cells of the target tissue. l There are two requirements for such a strategy: 1. The target cells is easily accessible for infusion or injection of virus. 2. The transfer vector infects, integrates, and then expresses therapeutic gene in target cells (not surrounding cells) at effective levels for extended time periods.

Approach of in vivo gene therapy o. The vector can be injected or given intravenously directly into a specific tissue in the body, where it is taken up by individual cells.


Vectors used in gene therapy • Viruses • Non-viruses

Viruses used are: Retroviruses Adeno-associated viruses Lentivirus


Disadvantages of viruses as vector in gene therapy n In all viral types, the vectors tend not to disperse well in a targeted tissue. Even when injected directly into a tumor, they are prone to miss some of the targeted cells. n In addition, their use does not allow long- term gene expression.

Non-viral vectors • Direct introduction of therapeutic DNA into target cells. • Creation of an artificial lipid sphere with an aqueous core (liposome).

• Chemically linking the DNA to a molecule that will bind to special cell receptors. • Human artificial chromosomes (HACs)

Liposome

Definition n are artificial vesicles with a phospholipid bilayer membrane. n It is self-closing spherical particles where one or several lipid membranes encapsulate part of the solvent in which they freely float in their interior.


Size l liposomes are typically 5 -10 µm in diameter with the phosopholipid bilayer about 3 nm thick


Mechanism of formation n self-assembly process that is driven by the amphipathic nature of phospholipid molecules



Function § A liposome can be used to deliver drugs, proteins or nucleic acids (short stretches of DNA and plasmids encoding therapeutic genes) to a cell.

Mechanism of action of liposome n First the outer layer of the liposome fuses with the outer layer of the plasma membrane. n Second, the two fused membranes coalesce as the inner layer of the liposome approaches the inner layer of the plasma membrane. n Finally, the two inner layers fuse so that the drug has access to the cytoplasm.
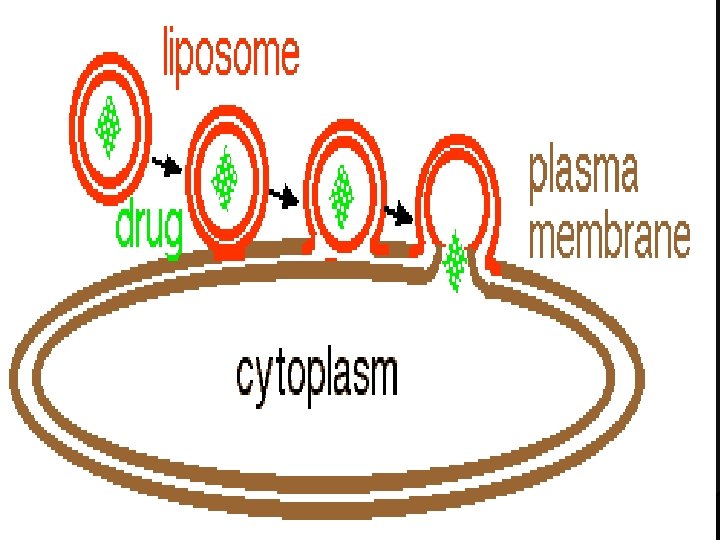

• Methods for Enhancing The Efficiency of Liposome-Based Transfection
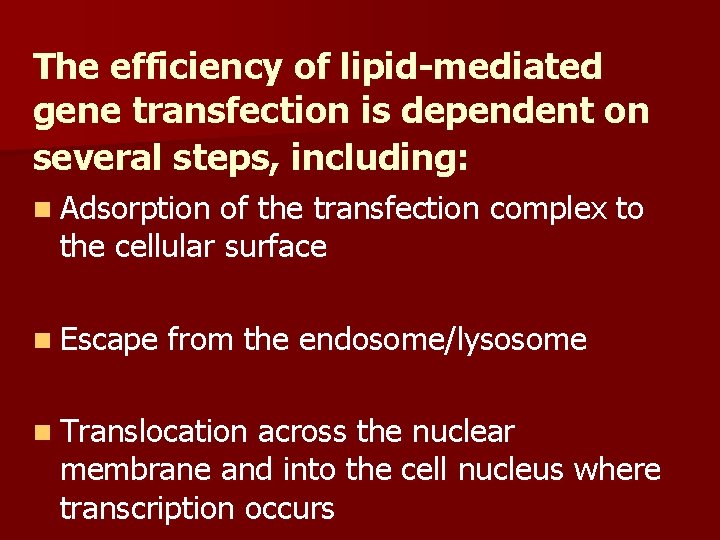
The efficiency of lipid-mediated gene transfection is dependent on several steps, including: n Adsorption of the transfection complex to the cellular surface n Escape from the endosome/lysosome n Translocation across the nuclear membrane and into the cell nucleus where transcription occurs

To improve translocation across the nuclear membrane nuclear localization signals (NLS) are used: • classical NLS • non-classical M 9 NLS

Artificial chromosomes

Advantages of adding an entirely separate artificial chromosome n Eliminates the risk of DNA landing in a wrong place on cell's genome, which can trigger cancer. n The ability to deliver multiple therapeutic genes, but viruses can carry only short sequences of DNA.

§ This artificial chromosome behaves like a normal one in mice: it is duplicated when cells divide and is passed from generation to generation. § The human artificial chromosome survived for as long as 6 months in cells, retaining its integrity while replicating during many cell divisions.

Structure n Capping the ends of chromosomes are telomeres, which is brief repeating sequences of DNA. n Origins of replication, which is DNA sequences that initiate the replication of a chromosome during cell division.

§ At the center of each chromosome is the mysterious centromere, which plays a vital role in the chromosome's segregation in a dividing cell. § Ignorance of the structure, and the size, of human centromeres has been the main reason of inability to create human artificial chromosomes.

Disadvantages of in vivo gene therapy:

n The introduced gene will integrate in the genome and cause interruption or disruption of other gene functions, causing mutation and possibly disease.

Ex vivo gene therapy o. Approach
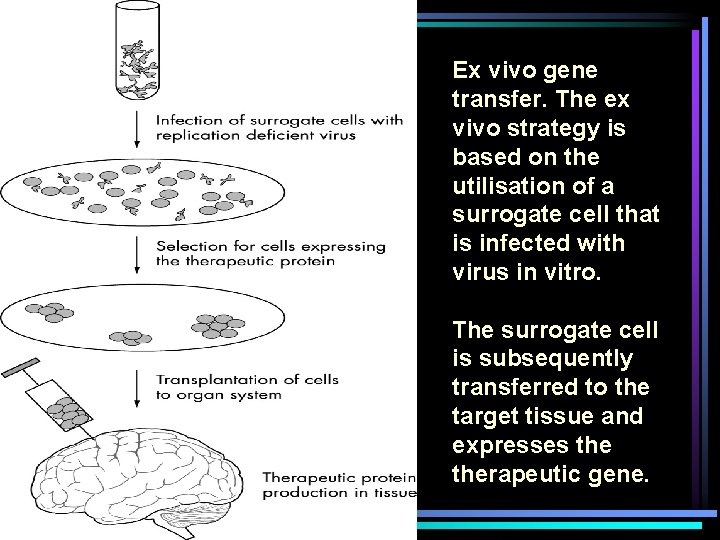
Ex vivo gene transfer. The ex vivo strategy is based on the utilisation of a surrogate cell that is infected with virus in vitro. The surrogate cell is subsequently transferred to the target tissue and expresses therapeutic gene.






For successful ex vivo, The cells used should be: • • Readily available and relatively easily obtained. Able to survive for long periods of time in vivo. Able to express a transgene at high levels for extended durations. Not elicit a host mediated immune reaction.
The advantages of using an ex vivo approach ÚSelection of the modified cell population before transplantation. ÚSubclone cells and produce monoclonal populations that produce high levels of therapeutic protein.

• The ability to screen populations and exclude the presence of helper viruses, transformational events, or other deleterious properties obtained after or during the modification process. • So, Viral vectors of low transfection efficiency can be used, because uninfected cells can be selected out of the transplant population.

Currently, autologous primary cell cultures are used. Autologous means that the donor and recipient organism are the same

The advantage of using primary, autologous cell cultures include: • Lack of antigenicity. • Decreased risk of malignant transformation.

Disadvantages of using primary, autologous cell cultures include n Difficulty in harvesting some types of primary cells, maintaining them in culture, and effectively expressing transgenes through current transfection techniques.

Ø Another complication arises when primary cells are transferred to non -host tissue. Ø for example, primary fibroblasts transplanted to the CNS will often produce collagen and other skin appropriate products that interfere with normal CNS functioning. Ø This problem may be overcome with the use of stem cells.

Stem cell ex vivo gene therapy: • • Bone marrow derived stem cell. Hepatocytes. CNS stem cells Fetal derived stem cells.
Human embryo at 4 cell stage
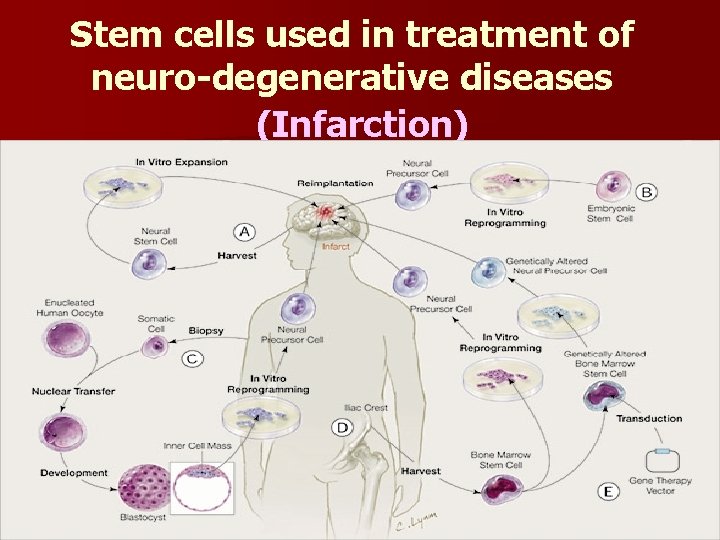
Stem cells used in treatment of neuro-degenerative diseases (Infarction)
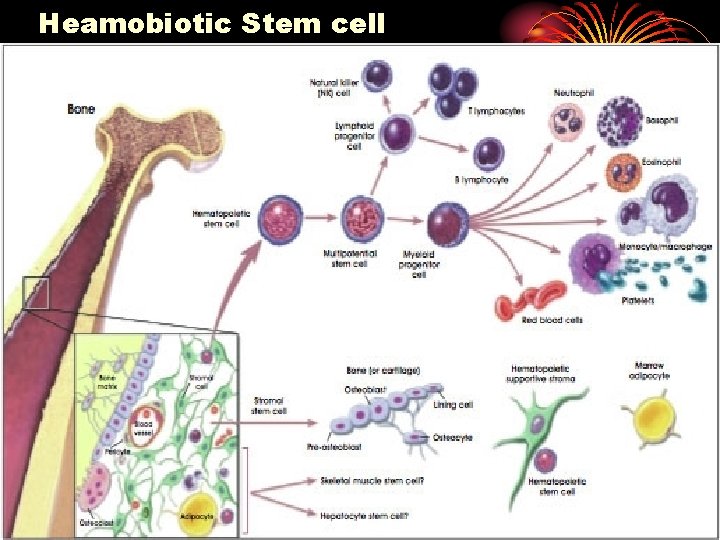
Heamobiotic Stem cell



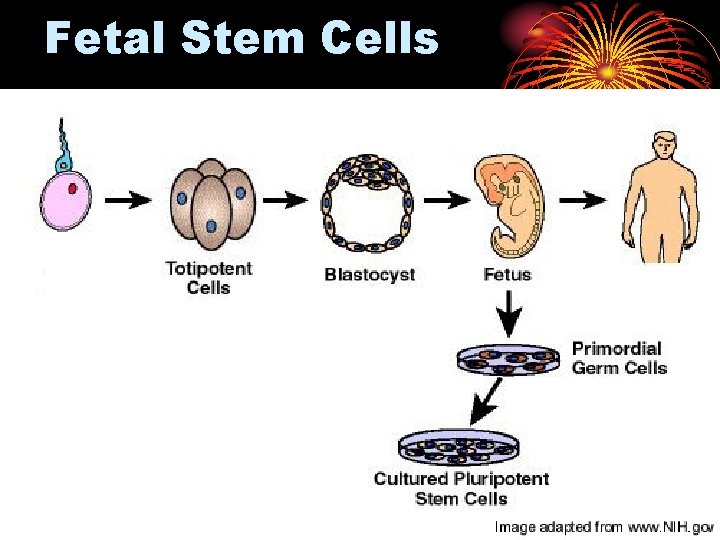
Fetal Stem Cells
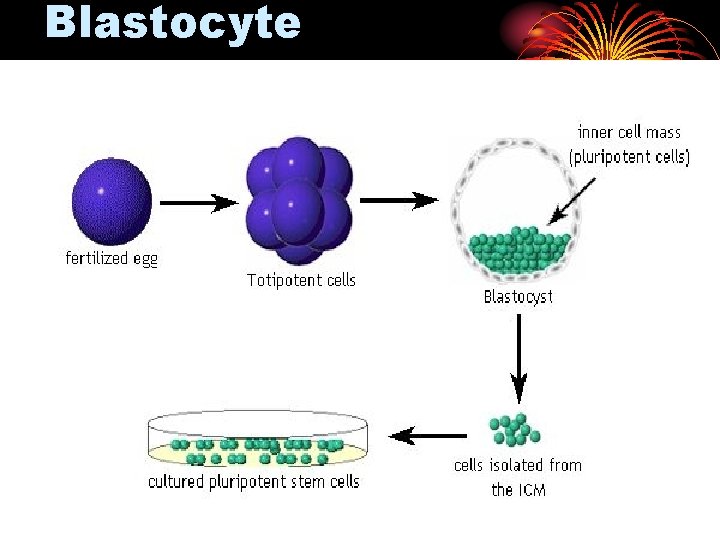
Blastocyte

Approach Stem cell ex vivo gene therapy l Peripheral derived haematopoietic stem cells are of particular interest as a potential surrogate cell. l Haemopoietic stem cells are easily obtained through I. V route, harvested systemically, modified in vitro, re-infused into the peripheral blood with subsequent homing to damaged target tissue such as brain or myocardium.

Advantages § Adult stem cells completely incorporate into any host tissue and transform into a mature cell of that organ. § This ability ensures long term survival of grafted cells. § So these cells could be used to carry therapeutic proteins, and also to repopulate organs with damaged or depleted cell numbers.

Disadvantages n Low viral transfection efficiency. n Technical difficulties in isolating, culturing, and maintaining these cells.

• How safety is the gene therapy

n Gene therapy have very serious health risks, such as toxicity, inflammation, and cancer which is due to the administration of retrovirus, which incorporates randomly into the genome and can lead to insertional mutagenesis and malignant transformation.

Factors have kept gene therapy from becoming an effective treatment for genetic disease

• Short-lived nature of gene therapy • Immune response • Problems with viral vectors as toxicity, immune responses, gene control, targeting tissues and reactivation of virus.

• Lack of viral specificity • Multigene disorders as Alzheimer’s disease, DM and heart disease. • Inefficient gene transfer

Inability to control gene expression: Ú Induction of inflammation for treating such diseases as cancer may be useful, but once the cancer is cured the inflammation continues if cells are expressing the inciting transgene. Chronic inflammation of a specific tissue is undesirable.

The use of growth factors n Uncontrolled growth factor expression and function is intimately involved in the malignant transformation processes. n The contineous expression of a growth factor predisposes to malignancy. n It is essential to be able to turn off growth factor expression if malignancy is detected, or if treatment is toxic or no longer useful or necessary.

Recent developments in gene therapy research • Injection of genes into the brain using liposomes coated in a polymer call polyethylene glycol (PEG). Viral vectors are too big to cross the blood brain barrier. This is important in treating Parkinson’s disease • RNA interference or gene silencing may be a new way to treat Huntington's.

• New gene therapy approach repairs errors in messenger RNA derived from defective genes. • Gene therapy for treating children with XSCID (sever combined immunodeficiency) or the "bubble boy" disease is stopped when the treatment causes leukemia in one of the patients.
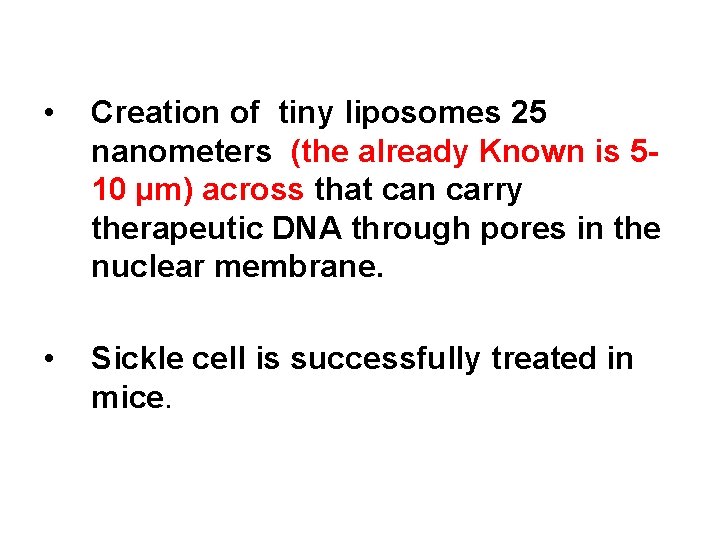
• Creation of tiny liposomes 25 nanometers (the already Known is 510 µm) across that can carry therapeutic DNA through pores in the nuclear membrane. • Sickle cell is successfully treated in mice.

Approaches of gene therapy • Replacing a mutated gene that causes disease with a healthy copy of the gene • Inactivating, or “knocking out” a mutated gene that is functioning improperly. • Introducing a new gene into the body to help fight a disease.

Two new techniques of gene therapy n RNA interference (post-transcriptional gene silencing). n Exon skipping.

RNA interference (posttranscriptional gene silencing): l Briefly, double stranded RNA, homologous to the gene targeted for suppression, is introduced into cells where it is cleaved into small fragments of double stranded RNA named short interfering RNAs (si. RNA).

These si. RNAs decrease gene expression by: • Guiding the enzymatic destruction of the homologous RNA, preventing translation to active protein. • Priming RNA polymerase to synthesis more si. RNA, and resulting in persistent gene suppression.

• To effectively silence specific genes in mammalian cells, a short hairpin RNA (sh. RNA) was designed. • These sequences, result in the transcription of a double stranded RNA brought together by a hairpin loop structure. • These sh. RNAs effectively mimic si. RNA and result in specific and persistent gene suppression in mammalian cells.


Exon skipping n The technique allows selected exons to be deleted from the final protein. n This occur by using short sequences of RNA that are complementary to exon recognition sequences or exon splicing enhancer sequences. n The expressed complementary RNA will bind to these regions of the gene and prevent the splicing of intron and exon at that site. The result of the altered posttranscriptional processing is the removal of a target exon from the final protein.




Germ line gene therapy

Definition • It is gene therapy which is targeted to egg and sperm cells (germ cells), and would allow the inserted gene to be passed on to future generations.

Principle • Addition of the transferred gene to the nuclear genome and its stable transmission to subsequent generations in a Mendelian fashion.

Importance n Genes could be "corrected" in the egg or sperm you are using to conceive. n The child that results would be spared certain genetic problems that might otherwise have occurred.

Disadvantages Ú Germline gene transfer might affect germ cells by making changes that could disrupt the development of the embryo or fetus in unexpected ways. Ú It is not right to make changes to a germ line, because some of the people who will be affected are not even born yet and therefore cannot give their consent.

Ooplasmic transfer is a process in which some cytoplasm from a healthy donor egg is injected into an egg from a woman with fertility problems. This procedure increases the ability of the recipient egg to be fertilized and develop into a healthy embryo.

n Mitochondrial DNA may has a role in this management. n The children born following this procedure have three genetic parents, since they carry mitochondrial DNA from the donor mother and nuclear DNA from the mother and father. n This procedure represents the first instances of germline gene transfer in humans.
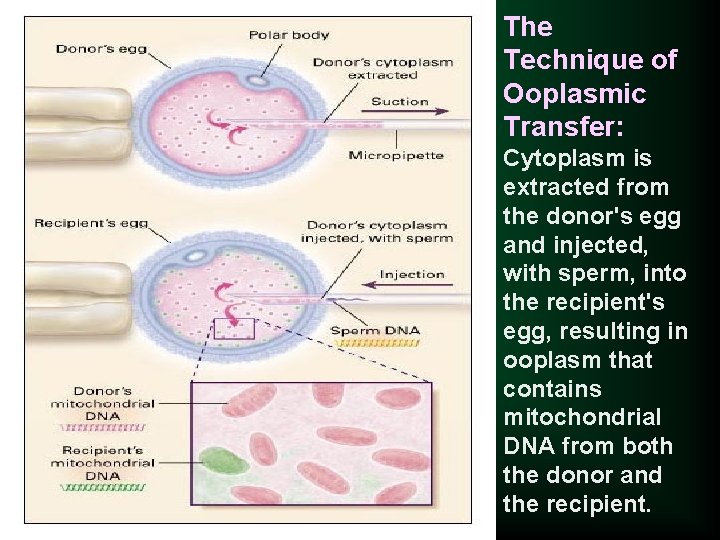
The Technique of Ooplasmic Transfer: Cytoplasm is extracted from the donor's egg and injected, with sperm, into the recipient's egg, resulting in ooplasm that contains mitochondrial DNA from both the donor and the recipient.

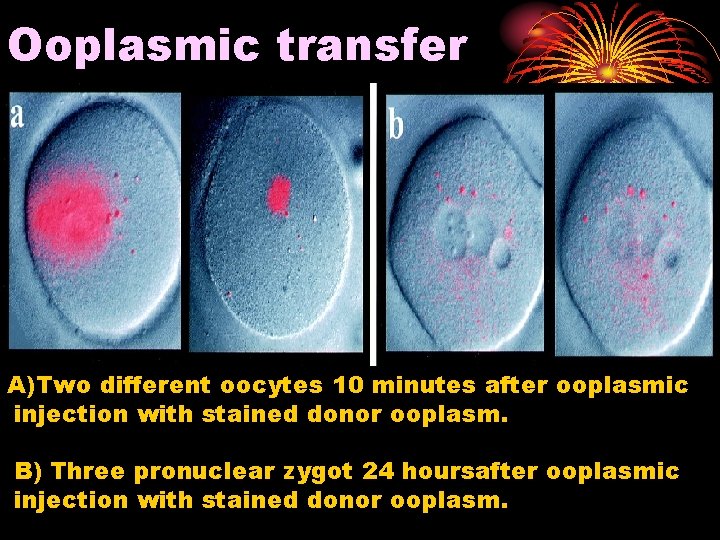
Ooplasmic transfer A)Two different oocytes 10 minutes after ooplasmic injection with stained donor ooplasm. B) Three pronuclear zygot 24 hoursafter ooplasmic injection with stained donor ooplasm.

Pronuclear microinjection § Microinjection is technique for introducing a solution of DNA into a cell using a fine microcapillary pipette. § Pronuclear microinjection is an technique used for gene transfer at the embryonic stage.

Principle • A fine glass needle is used to inject a purified double stranded DNA sequence into the nucleus of a fertilized mammalian oocyte. • This process leads to the integration of the sequence (transgene) into the genome. As a result, the animal is born with a copy of the new sequence in every cell.

Disadvantages It is suggested that embryo gene transfer is unsafe, as its use results in random integration of the transgene, a lack of control of the number of gene copies inserted, significant rearrangements of host genetic material, and insertional mutagenesis. So, this approach is not applied on humans.
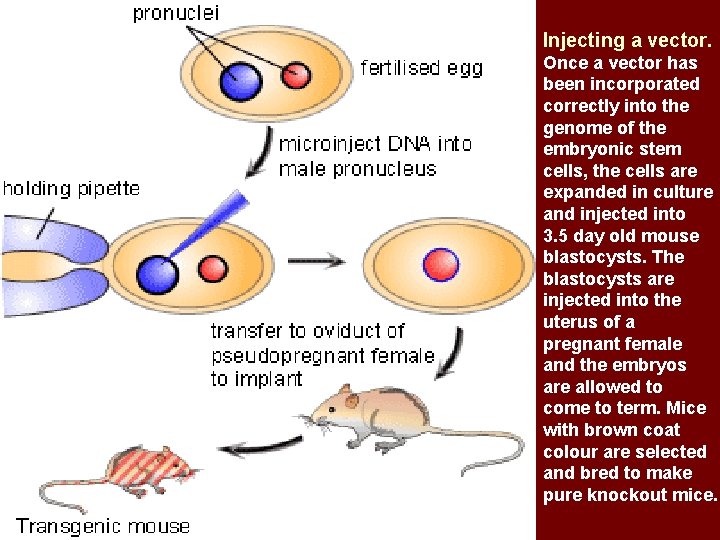
Injecting a vector. Once a vector has been incorporated correctly into the genome of the embryonic stem cells, the cells are expanded in culture and injected into 3. 5 day old mouse blastocysts. The blastocysts are injected into the uterus of a pregnant female and the embryos are allowed to come to term. Mice with brown coat colour are selected and bred to make pure knockout mice.
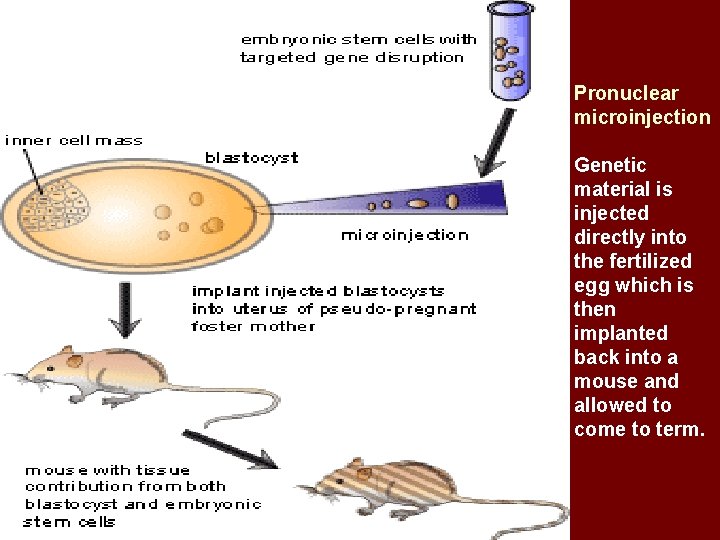
Pronuclear microinjection Genetic material is injected directly into the fertilized egg which is then implanted back into a mouse and allowed to come to term.

Somatic Cell Nuclear Transfer (SCNT) n A somatic cell is fused with an enucleated oocyte. n The nucleus of the somatic cell provides the genetic information, while the oocyte provides the nutrients and other energyproducing materials that are necessary for development of an embryo. n Once fusion has occurred, the cell is totipotent, and eventually develops into a blastocyst, at which point the inner cell mass is isolated. The pluripotent stem cell line is then established.

• Pluripotent stem cells derived from SCNT were capable of differentiating into all cell types, including gametes. • Because SCNT involves cloning, there are many ethical concerns in using this technique in humans. • For this reason, experiments of this nature have only been conducted in mice.

Ethical issues surrounding gene therapy Ú How can “good” and “bad” uses of gene therapy be distinguished? Ú Who decides which traits are normal and which constitute a disability or disorder? Ú Will the high costs of gene therapy make it available only to the wealthy?

n n Could the widespread use of gene therapy make society less accepting of people who are different? Should people be allowed to use gene therapy to enhance basic human traits such as height, intelligence, or athletic ability?

- Slides: 108