Gel Permeation Or Size exclusion Chromatography Size exclusion











- Slides: 20

Gel Permeation Or Size exclusion Chromatography

Size exclusion Chromatography Also called: Gel filtration chromatography (MP water): desalting Gel permeation chromatography (MP organic solvent) Molecules are separated according to their size Small molecules penetrate the small pores in the stationary phase, while large molecules do not. Large molecules are eluted first.

History Dates to the 1960’s. Porath and Flodin describe the separation of water soluble macromolecules on cross-linked polydextran gels. Applications It separates according to the size of species in solution (hydrodynamic volume) Species can be: • single molecules • polymer coils • aggregates • micelles • etc…

Applications Typical SEC applications: • analysis of synthetic polymers and oligomers • lipids • proteins • cellulose derivatives • crude oil alkanes SEC methods can also be used to: • hydrolysis • refolding of proteins • polymerisation • aggregation • etc

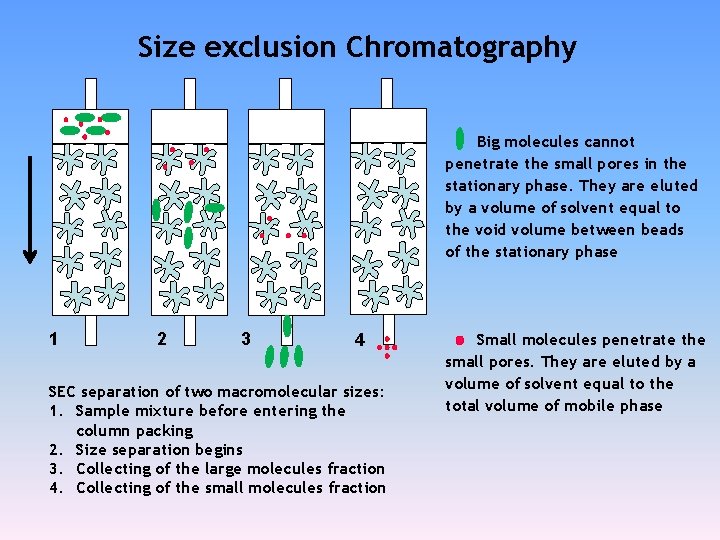
Size exclusion Chromatography Big molecules cannot penetrate the small pores in the stationary phase. They are eluted by a volume of solvent equal to the void volume between beads of the stationary phase 1 2 3 4 SEC separation of two macromolecular sizes: 1. Sample mixture before entering the column packing 2. Size separation begins 3. Collecting of the large molecules fraction 4. Collecting of the small molecules fraction Small molecules penetrate the small pores. They are eluted by a volume of solvent equal to the total volume of mobile phase

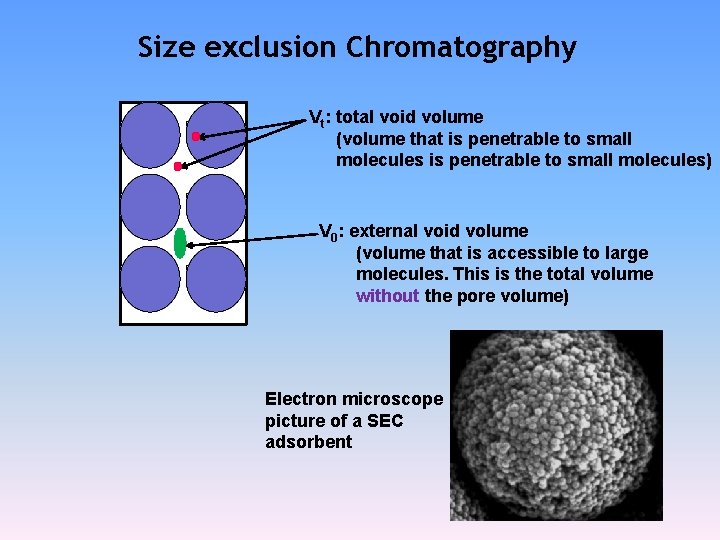
Size exclusion Chromatography Vt: total void volume (volume that is penetrable to small molecules) V 0: external void volume (volume that is accessible to large molecules. This is the total volume without the pore volume) Electron microscope picture of a SEC adsorbent

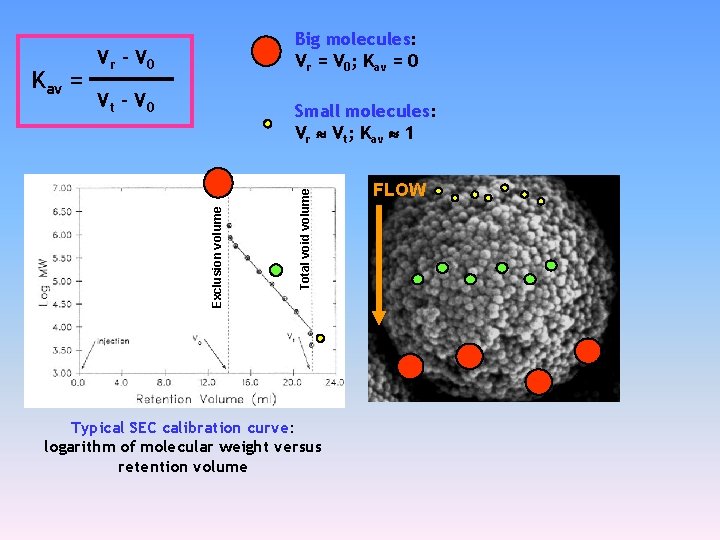
Size exclusion Chromatography Retention of molecules in SEC is characterized by partition coefficient Kav = Vr – V 0 Vt – V 0 Vr: retention volume for a solute V 0: external void volume Vt : total void volume (total volume of the mobile phase in a column

Vr – V 0 Vt – V 0 Total void volume Small molecules: Vr Vt; Kav 1 Exclusion volume Kav = Big molecules: Vr = V 0; Kav = 0 Typical SEC calibration curve: logarithm of molecular weight versus retention volume FLOW

Stationary Phase Polymers of glucose: • Cellulose • Dextran (If cross-link is glycerin: Sephadex) Polymers of agarose Polyacrylamides Crossed-linked gels Small pore size Exclude molecules with MM 700 Crossed-linked gels Big pore size Exclude molecules with MM 108 • The finer the particle, the greater the resolution, the slower the flow rate of the column • Particles with different pore sizes can be mixed to give a wider molecular size separation range • In general, two types of molecules can be separated provided that there is a 10% difference in their molecular sizes

Columns Unlike the other modes of liquid chromatography, the separation comes from the stationary phase only. The mobile phase should have no effect as long as the sample is well dissolvable. Usual size of SEC columns: 7 -8 mm diameter (analytical) 20 -25 mm diameter (preparative) Separation is carried out on the pores which typically equals 40% of the total column volume. Length 20 -60 cm As a result long columns, or several columns are required. • porous silica • cross-linked organic gels Packing:

Columns Selecting SEC columns: Handling SEC columns: • Separation range selected carefully • A column set should be always run in the same mobile phase • Small particle size (5µm) provides more theoretical plates • Should not be operated in backward position • Small particle size more sensitive to contamination • No air bubbles in columns during injection and assembly • Small particle size can result on shear degradation of large polymers • Always use a pre-column when in doubt about the quality of the sample • Combination of packings with different separation range can be achieved by: columns of different porosity or mixed bed columns • Chemical nature of column packing crucial Handbook of Size Exclusion Chromatography and Related Techniques: Revised And Expanded By Chi-san Wu Edition: 2, illustrated, revised Published by CRC Press, 2004 694 pages

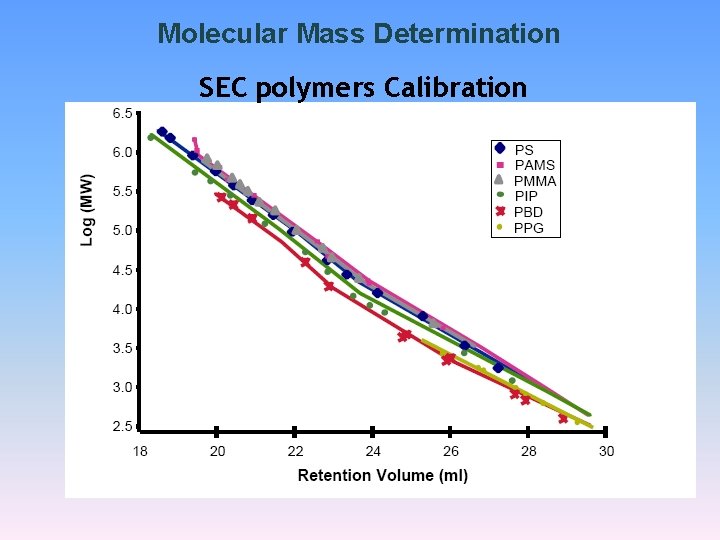
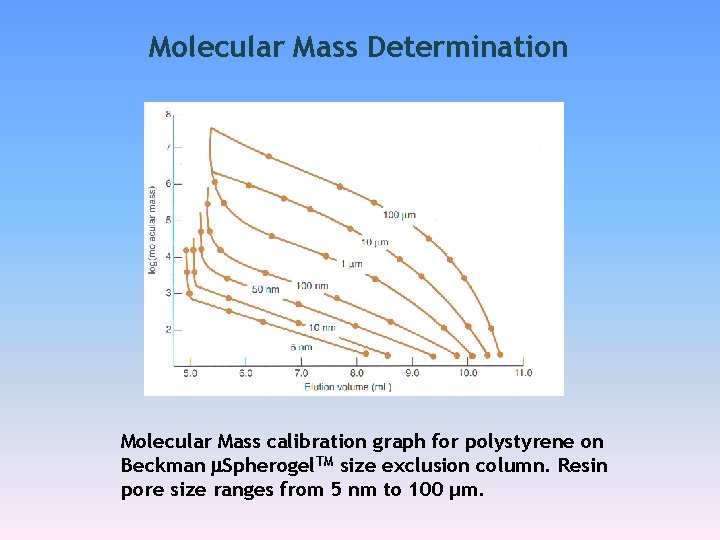
Molecular Mass Determination SEC polymers Calibration

Molecular Mass Determination Molecular Mass calibration graph for polystyrene on Beckman Spherogel. TM size exclusion column. Resin pore size ranges from 5 nm to 100 µm.

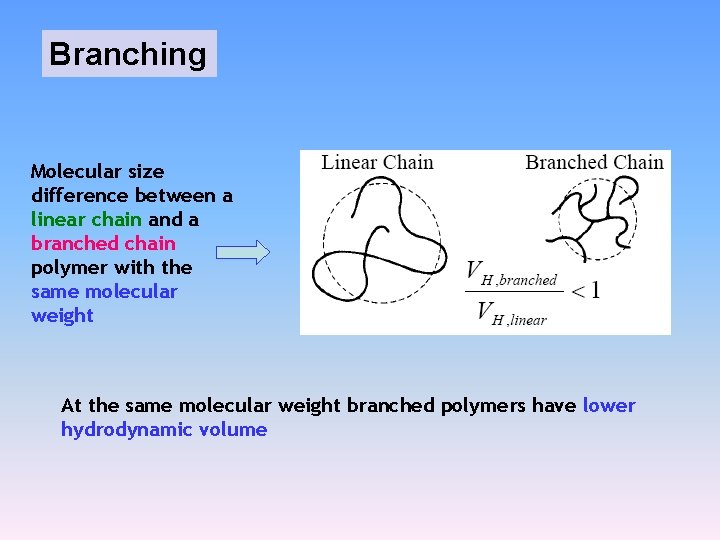
Molecular Mass Determination Caution: • Branching • Possible adsorption due to occasional charges on the gel • Other adsorption mechanisms (silica must be coated with a hydrophilic phase to minimise solute adsorption)

Branching Molecular size difference between a linear chain and a branched chain polymer with the same molecular weight At the same molecular weight branched polymers have lower hydrodynamic volume

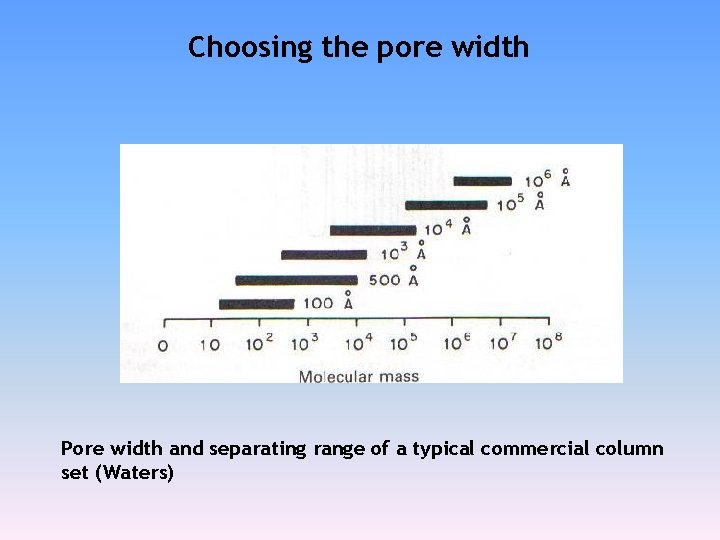
Choosing the pore width Pore width and separating range of a typical commercial column set (Waters)

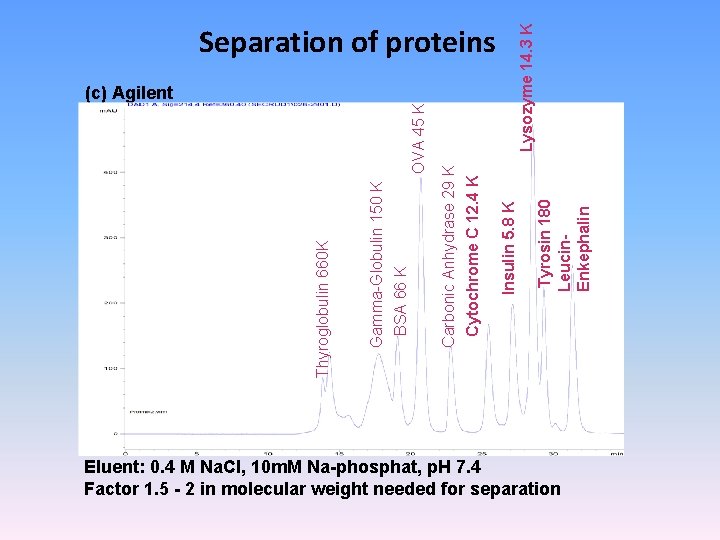
Tyrosin 180 Leucin. Enkephalin Insulin 5. 8 K (c) Agilent Lysozyme 14. 3 K OVA 45 K Carbonic Anhydrase 29 K Cytochrome C 12. 4 K Gamma-Globulin 150 K BSA 66 K Thyroglobulin 660 K Separation of proteins Eluent: 0. 4 M Na. Cl, 10 m. M Na-phosphat, p. H 7. 4 Factor 1. 5 - 2 in molecular weight needed for separation

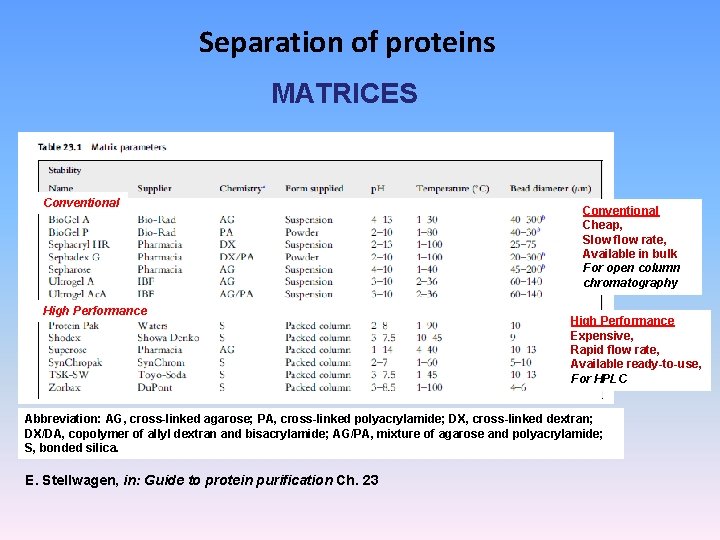
Separation of proteins MATRICES Conventional High Performance Conventional Cheap, Slow flow rate, Available in bulk For open column chromatography High Performance Expensive, Rapid flow rate, Available ready-to-use, For HPLC Abbreviation: AG, cross-linked agarose; PA, cross-linked polyacrylamide; DX, cross-linked dextran; DX/DA, copolymer of allyl dextran and bisacrylamide; AG/PA, mixture of agarose and polyacrylamide; S, bonded silica. E. Stellwagen, in: Guide to protein purification Ch. 23

Separation of proteins Mobile Phases Separation in SEC mostly depends on the stationary phase and does not depend on the properties of an eluent. However precautions must be taken to minimize the binding of proteins to the matrix by electrostatic or by van der Waals interactions. Enzymatic hydrolysis of proteins is another danger. To face these problems - the ionic strength of the chromatographic solvent should be at least 0. 2 M - proteolytic inhibitors need be present in the chromatographic solvent - addition of insecticides like 0. 02% Na. N 3 is desirable to prevent the growth of microorganisms

Hint SEC has a relatively low resolution power. Fewer than 10 proteins can be resolved from one another in the effluent from any sizeexclusion column. Significant enhancement in purification (-fold) by SEC can be expected if the desired protein has a molecular weight either considerably larger or smaller than that of the majority of proteins in a mixture. Accordingly, it is wise to perform size-exclusion chromatography relatively late in a purification procedure when the numbers of other proteins are small and when the preceding step has fractionated the protein mixture on the basis of a completely different property. E. Stellwagen, in: Guide to protein purification Ch. 23