GAVI Vaccine Investment Strategy Rabies Analysis Final October

GAVI Vaccine Investment Strategy Rabies Analysis Final October 27, 2008

Rabies CONTENTS • Disease Overview • Vaccine Landscape • Vaccination Policy & Strategies • Vaccine Need & Adoption Forecast • Vaccine Cost Analysis • Implementation-Associated Cost Analysis • Analysis Summary • Key Resources 2

Rabies Disease Overview DISEASE PATHOGEN, TRANSMISSION & TARGET POPULATION 1* • Disease Pathogen – • • Transmission – Usually dog bites in developing countries; animal to person via saliva from a bite – Incubation periods have been as short as 5 -6 days, in the majority of cases is 20 -60 days Geographic Distribution – • Rabies Virus (lyssavirus) Most human cases are in rural areas of Africa and Asia Disease Target Population – All ages are susceptible, however, rabies is most common in ages 5 -15 years old, the majority of whom are male 3

Rabies Disease Overview DISEASE IMPACT 2, 3 • • • Total Morbidity – >10 million suspected cases per year in developing countries – Rural poor and children are most at risk Total Mortality – ~55, 000 cases reported annually and thought to be grossly underreported with the majority of deaths occurring in Asia and Africa – Case fatality rate is 100% once clinical symptoms present (with exception of one known survivor to date) Epidemic Potential – • Human outbreaks can occur as a result of epidemics in the animal population and/or when a rabid animal bites multiple people Disease Sequelae – Permanent neurological sequelae in the one survivor to date 4
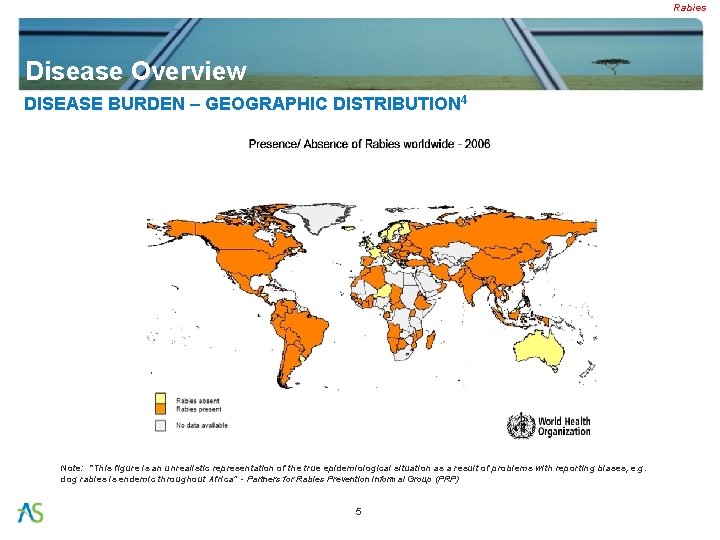
Rabies Disease Overview DISEASE BURDEN – GEOGRAPHIC DISTRIBUTION 4 Note: “This figure is an unrealistic representation of the true epidemiological situation as a result of problems with reporting biases, e. g. dog rabies is endemic throughout Africa” - Partners for Rabies Prevention Informal Group (PRP) 5
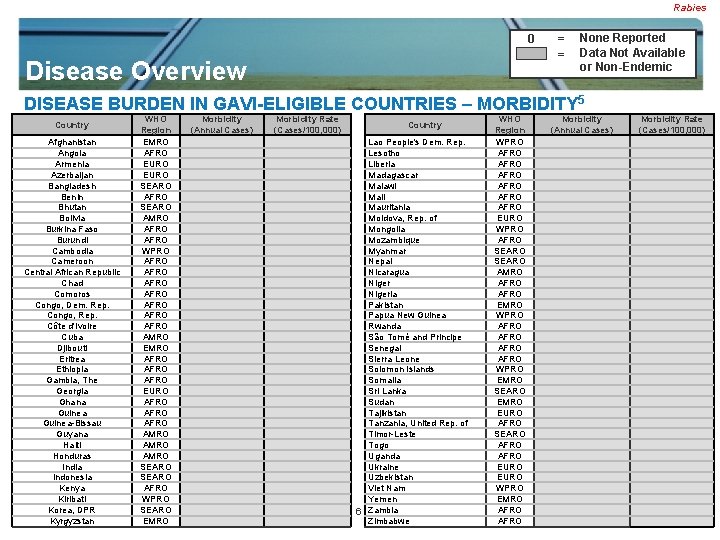
Rabies 0 Disease Overview = = None Reported Data Not Available or Non-Endemic DISEASE BURDEN IN GAVI-ELIGIBLE COUNTRIES – MORBIDITY 5 Country Afghanistan Angola Armenia Azerbaijan Bangladesh Benin Bhutan Bolivia Burkina Faso Burundi Cambodia Cameroon Central African Republic Chad Comoros Congo, Dem. Rep. Congo, Rep. Côte d'Ivoire Cuba Djibouti Eritrea Ethiopia Gambia, The Georgia Ghana Guinea-Bissau Guyana Haiti Honduras India Indonesia Kenya Kiribati Korea, DPR Kyrgyzstan WHO Region EMRO AFRO EURO SEARO AFRO SEARO AMRO AFRO WPRO AFRO AFRO AMRO EMRO AFRO EURO AFRO AMRO SEARO AFRO WPRO SEARO EMRO Morbidity (Annual Cases) Morbidity Rate (Cases/100, 000) Country Lao People's Dem. Rep. Lesotho Liberia Madagascar Malawi Mali Mauritania Moldova, Rep. of Mongolia Mozambique Myanmar Nepal Nicaragua Nigeria Pakistan Papua New Guinea Rwanda São Tomé and Principe Senegal Sierra Leone Solomon Islands Somalia Sri Lanka Sudan Tajikistan Tanzania, United Rep. of Timor-Leste Togo Uganda Ukraine Uzbekistan 4 Viet Nam Yemen 6 Zambia Zimbabwe WHO Region WPRO AFRO AFRO EURO WPRO AFRO SEARO AMRO AFRO EMRO WPRO AFRO WPRO EMRO SEARO EMRO EURO AFRO SEARO AFRO EURO WPRO EMRO AFRO Morbidity (Annual Cases) Morbidity Rate (Cases/100, 000)
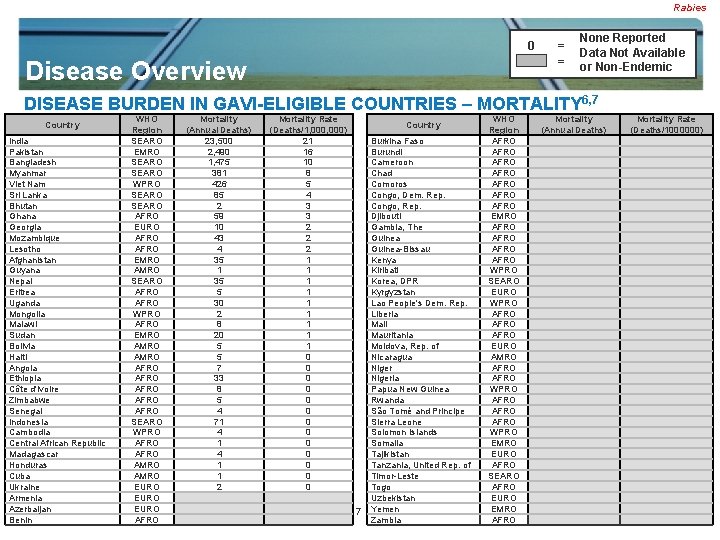
Rabies 0 Disease Overview = = None Reported Data Not Available or Non-Endemic DISEASE BURDEN IN GAVI-ELIGIBLE COUNTRIES – MORTALITY 6, 7 WHO Mortality Region (Annual Deaths) India SEARO 23, 500 Pakistan EMRO 2, 490 Bangladesh SEARO 1, 475 Myanmar SEARO 381 Viet Nam WPRO 426 Sri Lanka SEARO 85 Bhutan SEARO 2 Ghana AFRO 59 Georgia EURO 10 Mozambique AFRO 43 Lesotho AFRO 4 Afghanistan EMRO 35 Guyana AMRO 1 Nepal SEARO 35 Eritrea AFRO 5 Uganda AFRO 30 Mongolia WPRO 2 Malawi AFRO 8 Sudan EMRO 20 Bolivia AMRO 5 Haiti AMRO 5 Angola AFRO 7 Ethiopia AFRO 33 Côte d'Ivoire AFRO 8 Zimbabwe AFRO 5 Senegal AFRO 4 Indonesia SEARO 71 Cambodia WPRO 4 Central African Republic AFRO 1 Madagascar AFRO 4 Honduras AMRO 1 Cuba AMRO 1 Ukraine EURO 2 Armenia EURO Azerbaijan GAVI Vaccine Investment Strategy EURO Vaccine Landscape Analysis_Cholera_Apr 08 Benin AFRO Country Mortality Rate (Deaths/1, 000) 21 16 10 8 5 4 3 3 2 2 2 1 1 1 1 1 0 0 0 0 Country Burkina Faso Burundi Cameroon Chad Comoros Congo, Dem. Rep. Congo, Rep. Djibouti Gambia, The Guinea-Bissau Kenya Kiribati Korea, DPR Kyrgyzstan Lao People's Dem. Rep. Liberia Mali Mauritania Moldova, Rep. of Nicaragua Nigeria Papua New Guinea Rwanda São Tomé and Principe Sierra Leone Solomon Islands Somalia Tajikistan Tanzania, United Rep. of Timor-Leste 4 Togo Uzbekistan 7 Yemen Zambia WHO Region AFRO AFRO EMRO AFRO WPRO SEARO EURO WPRO AFRO EURO AMRO AFRO WPRO AFRO WPRO EMRO EURO AFRO SEARO AFRO EURO EMRO AFRO Mortality (Annual Deaths) Mortality Rate (Deaths/1000000)

Rabies Disease Overview NON-VACCINE PREVENTION & TREATMENT INTERVENTIONS 8 • Non-Vaccine Preventions – • Control and vaccination of domestic dogs and wild carnivores Treatment Interventions – Post-exposure prophylaxis rabies vaccination and passive immunoglobulin administration around the wound (to neutralize the virus before it enters the CNS) – After symptoms ensue there is no recognized effective treatment 8
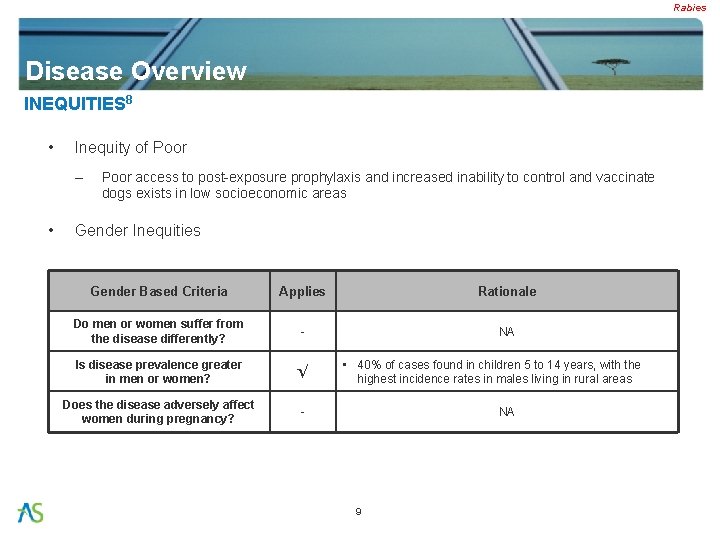
Rabies Disease Overview INEQUITIES 8 • Inequity of Poor – • Poor access to post-exposure prophylaxis and increased inability to control and vaccinate dogs exists in low socioeconomic areas Gender Inequities Gender Based Criteria Applies Rationale Do men or women suffer from the disease differently? - NA Is disease prevalence greater in men or women? √ Does the disease adversely affect women during pregnancy? - • 40% of cases found in children 5 to 14 years, with the highest incidence rates in males living in rural areas NA 9

Rabies CONTENTS • Disease Overview • Vaccine Landscape • Vaccination Policy & Strategies • Vaccine Need & Adoption Forecast • Vaccine Cost Analysis • Implementation-Associated Cost Analysis • Analysis Summary • Key Resources 10
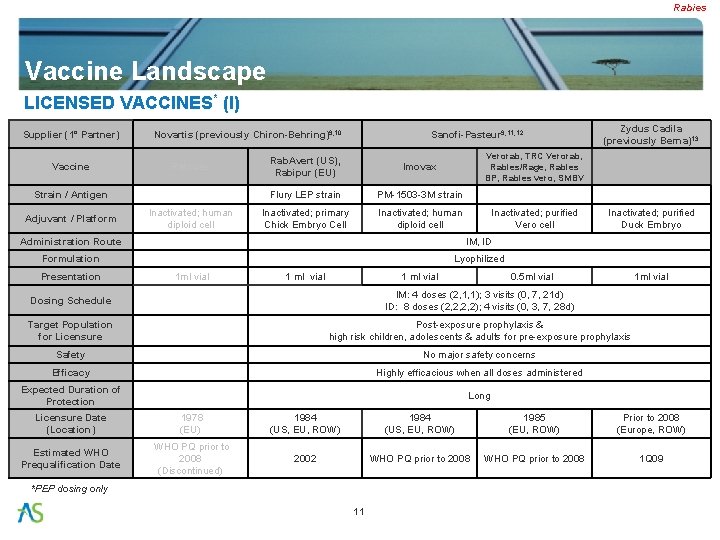
Rabies Vaccine Landscape LICENSED VACCINES* (I) Supplier (1° Partner) Vaccine Novartis (previously Chiron-Behring)9, 10 Rabivac Strain / Antigen Adjuvant / Platform Inactivated; human diploid cell Sanofi-Pasteur 9, 11, 12 Verorab, TRC Verorab, Rabies/Rage, Rabies BP, Rabies vero, SMBV Rab. Avert (US), Rabipur (EU) Imovax Flury LEP strain PM-1503 -3 M strain Inactivated; primary Chick Embryo Cell Inactivated; human diploid cell Inactivated; purified Vero cell Administration Route IM, ID Formulation Lyophilized Presentation 1 ml vial 1 ml vial Zydus Cadila (previously Berna)13 1 ml vial Inactivated; purified Duck Embryo 0. 5 ml vial 1 ml vial Dosing Schedule IM: 4 doses (2, 1, 1); 3 visits (0, 7, 21 d) ID: 8 doses (2, 2, 2, 2); 4 visits (0, 3, 7, 28 d) Target Population for Licensure Post-exposure prophylaxis & high risk children, adolescents & adults for pre-exposure prophylaxis Safety No major safety concerns Efficacy Highly efficacious when all doses administered Expected Duration of Protection Long Licensure Date (Location) 1978 (EU) 1984 (US, EU, ROW) 1985 (EU, ROW) Prior to 2008 (Europe, ROW) Estimated WHO Prequalification Date WHO PQ prior to 2008 (Discontinued) 2002 WHO PQ prior to 2008 1 Q 09 *PEP dosing only 11
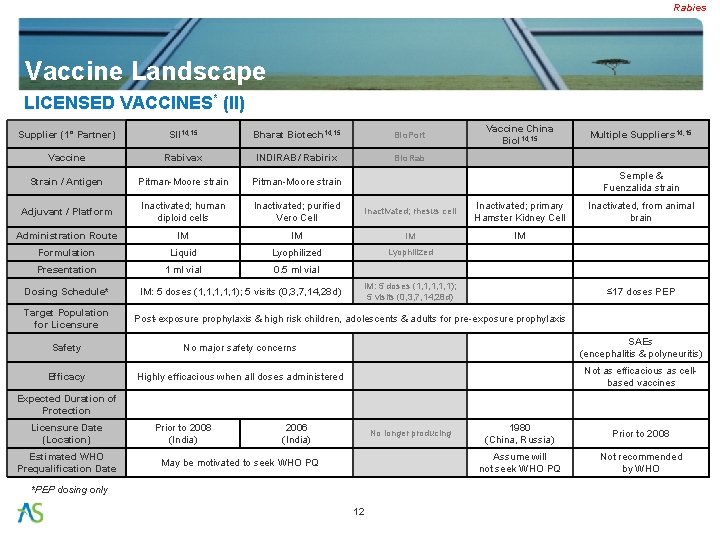
Rabies Vaccine Landscape LICENSED VACCINES* (II) Vaccine China Biol 14, 15 Supplier (1° Partner) SII 14, 15 Bharat Biotech 14, 15 Bio. Port Vaccine Rabivax INDIRAB/ Rabirix Bio. Rab Strain / Antigen Pitman-Moore strain Adjuvant / Platform Inactivated; human diploid cells Inactivated; purified Vero Cell Inactivated; rhesus cell Inactivated; primary Hamster Kidney Cell Administration Route IM IM Formulation Liquid Lyophilized Presentation 1 ml vial 0. 5 ml vial Dosing Schedule* Target Population for Licensure IM: 5 doses (1, 1, 1); 5 visits (0, 3, 7, 14, 28 d) Multiple Suppliers 14, 15 Semple & Fuenzalida strain IM: 5 doses (1, 1, 1); 5 visits (0, 3, 7, 14, 28 d) Inactivated, from animal brain ≤ 17 doses PEP Post-exposure prophylaxis & high risk children, adolescents & adults for pre-exposure prophylaxis Safety No major safety concerns SAEs (encephalitis & polyneuritis) Efficacy Highly efficacious when all doses administered Not as efficacious as cellbased vaccines Expected Duration of Protection Licensure Date (Location) Estimated WHO Prequalification Date Prior to 2008 (India) 2006 (India) No longer producing 1980 (China, Russia) Assume will not seek WHO PQ May be motivated to seek WHO PQ *PEP dosing only 12 Prior to 2008 Not recommended by WHO
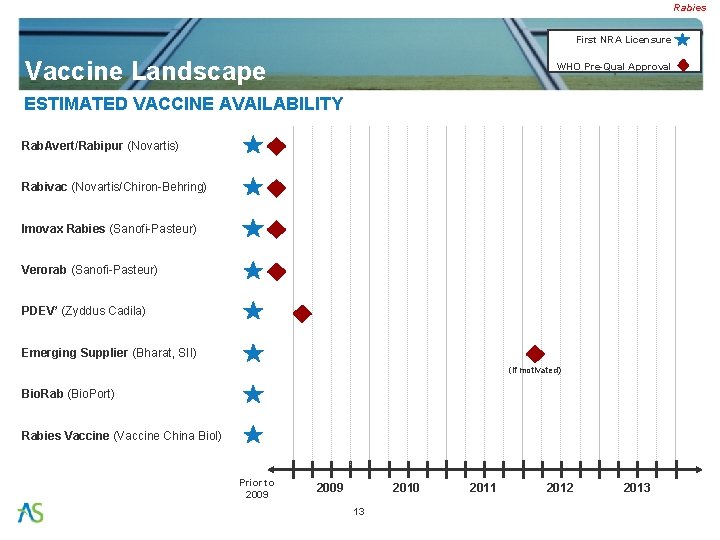
Rabies First NRA Licensure Vaccine Landscape WHO Pre-Qual Approval ESTIMATED VACCINE AVAILABILITY Rab. Avert/Rabipur (Novartis) Rabivac (Novartis/Chiron-Behring) Imovax Rabies (Sanofi-Pasteur) Verorab (Sanofi-Pasteur) PDEV* (Zyddus Cadila) Emerging Supplier (Bharat, SII) (if motivated) Bio. Rab (Bio. Port) Rabies Vaccine (Vaccine China Biol) Prior to 2009 2010 13 2011 2012 2013

RABIES Vaccine Landscape Analysis COST EFFECTIVENESS LITERATURE SUMMARY • Post-exposure rabies prophylaxis is estimated to prevent 330, 304 (90% CI: 141, 844 - 563, 515) deaths in Asia and Africa. Rabies is responsible for an estimated 1. 74 million (90% CI: 0. 25 M – 4. 57 M) DALYs. Global expenditure for rabies prevention is over $1 billion. 16 • A 1996 study demonstrated that pre-exposure rabies prophylaxis for the international traveler was worth $275, 000 per case averted. 17 • Cost comparison of rabies pre-exposure vaccination to post-exposure vaccination in Thai children showed that pre-exposure vaccination was cost-effective when the dog bite incidence is 2% to 30% of the population. 18 14

Rabies CONTENTS • Disease Overview • Vaccine Landscape • Vaccination Policy & Strategies • Vaccine Need & Adoption Forecast • Vaccine Cost Analysis • Implementation-Associated Cost Analysis • Analysis Summary • Key Resources 15
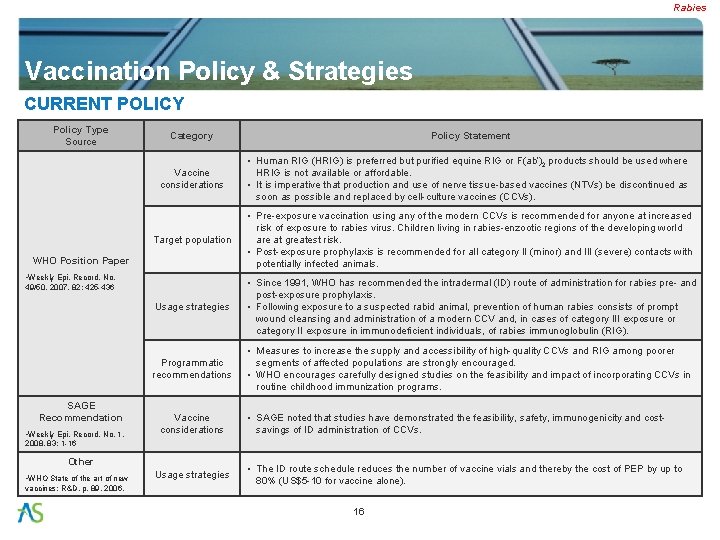
Rabies Vaccination Policy & Strategies CURRENT POLICY Policy Type Source Category Policy Statement Vaccine considerations • Human RIG (HRIG) is preferred but purified equine RIG or F(ab’)2 products should be used where HRIG is not available or affordable. • It is imperative that production and use of nerve tissue-based vaccines (NTVs) be discontinued as soon as possible and replaced by cell-culture vaccines (CCVs). Target population • Pre-exposure vaccination using any of the modern CCVs is recommended for anyone at increased risk of exposure to rabies virus. Children living in rabies-enzootic regions of the developing world are at greatest risk. • Post-exposure prophylaxis is recommended for all category II (minor) and III (severe) contacts with potentially infected animals. Usage strategies • Since 1991, WHO has recommended the intradermal (ID) route of administration for rabies pre- and post-exposure prophylaxis. • Following exposure to a suspected rabid animal, prevention of human rabies consists of prompt wound cleansing and administration of a modern CCV and, in cases of category III exposure or category II exposure in immunodeficient individuals, of rabies immunoglobulin (RIG). Programmatic recommendations • Measures to increase the supply and accessibility of high-quality CCVs and RIG among poorer segments of affected populations are strongly encouraged. • WHO encourages carefully designed studies on the feasibility and impact of incorporating CCVs in routine childhood immunization programs. WHO Position Paper • Weekly Epi. Record, No. 49/50, 2007, 82: 425 -436 SAGE Recommendation • Weekly Epi. Record, No. 1, Vaccine considerations • SAGE noted that studies have demonstrated the feasibility, safety, immunogenicity and costsavings of ID administration of CCVs. 2008, 83: 1 -16 Other • WHO State of the art of new vaccines: R&D, p. 89, 2006. Usage strategies • The ID route schedule reduces the number of vaccine vials and thereby the cost of PEP by up to 80% (US$5 -10 for vaccine alone). 16
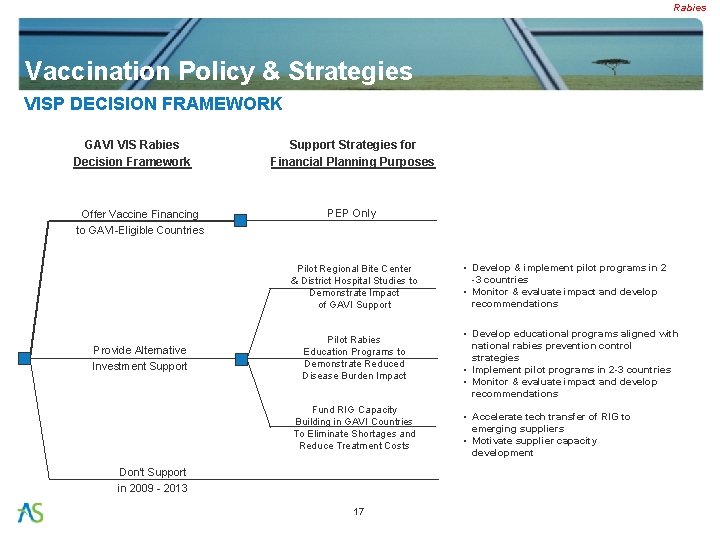
Rabies Vaccination Policy & Strategies VISP DECISION FRAMEWORK GAVI VIS Rabies Decision Framework Offer Vaccine Financing to GAVI-Eligible Countries Support Strategies for Financial Planning Purposes PEP Only Pilot Regional Bite Center & District Hospital Studies to Demonstrate Impact of GAVI Support Provide Alternative Investment Support Pilot Rabies Education Programs to Demonstrate Reduced Disease Burden Impact Fund RIG Capacity Building in GAVI Countries To Eliminate Shortages and Reduce Treatment Costs Don’t Support in 2009 - 2013 17 • Develop & implement pilot programs in 2 -3 countries • Monitor & evaluate impact and develop recommendations • Develop educational programs aligned with national rabies prevention control strategies • Implement pilot programs in 2 -3 countries • Monitor & evaluate impact and develop recommendations • Accelerate tech transfer of RIG to emerging suppliers • Motivate supplier capacity development

Rabies CONTENTS • Disease Overview • Vaccine Landscape • Vaccination Policy & Strategies • Vaccine Need & Adoption Forecast • Vaccine Cost Analysis • Implementation-Associated Cost Analysis • Analysis Summary • Key Resources 18
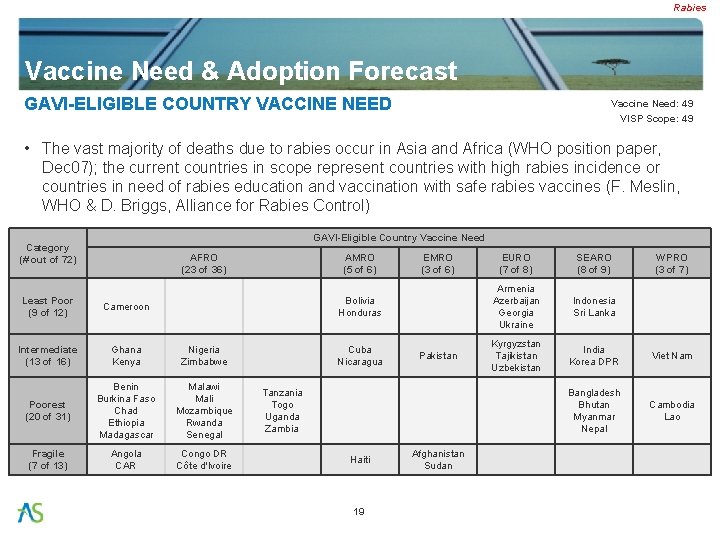
Rabies Vaccine Need & Adoption Forecast GAVI-ELIGIBLE COUNTRY VACCINE NEED Vaccine Need: 49 VISP Scope: 49 • The vast majority of deaths due to rabies occur in Asia and Africa (WHO position paper, Dec 07); the current countries in scope represent countries with high rabies incidence or countries in need of rabies education and vaccination with safe rabies vaccines (F. Meslin, WHO & D. Briggs, Alliance for Rabies Control) GAVI-Eligible Country Vaccine Need Category (# out of 72) AFRO (23 of 36) Least Poor (9 of 12) Cameroon Intermediate (13 of 16) Ghana Kenya Nigeria Zimbabwe Poorest (20 of 31) Benin Burkina Faso Chad Ethiopia Madagascar Malawi Mali Mozambique Rwanda Senegal Fragile (7 of 13) Angola CAR Congo DR Côte d'Ivoire AMRO (5 of 6) EMRO (3 of 6) Bolivia Honduras Cuba Nicaragua Pakistan Tanzania Togo Uganda Zambia Haiti 19 Afghanistan Sudan EURO (7 of 8) SEARO (8 of 9) WPRO (3 of 7) Armenia Azerbaijan Georgia Ukraine Indonesia Sri Lanka Kyrgyzstan Tajikistan Uzbekistan India Korea DPR Viet Nam Bangladesh Bhutan Myanmar Nepal Cambodia Lao
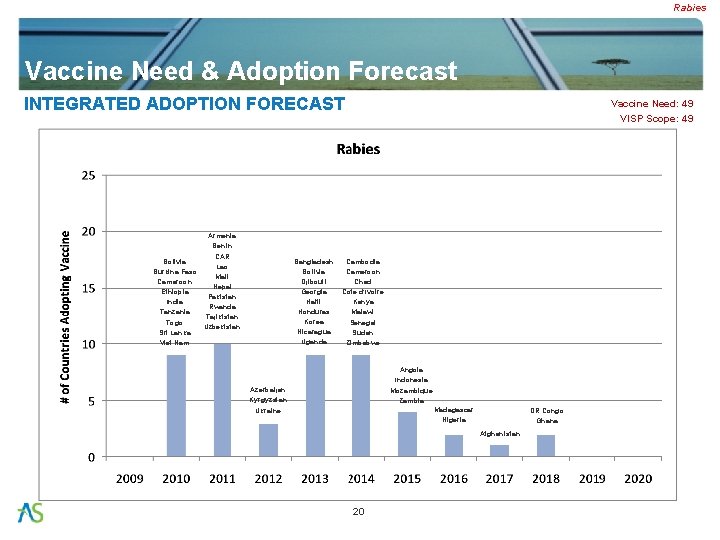
Rabies Vaccine Need & Adoption Forecast INTEGRATED ADOPTION FORECAST Armenia Benin CAR Bolivia Lao Burkina Faso Mali Cameroon Nepal Ethiopia Pakistan India Rwanda Tanzania Tajikistan Togo Uzbekistan Sri Lanka Viet Nam Bangladesh Bolivia Djibouti Georgia Haiti Honduras Korea Nicaragua Uganda Vaccine Need: 49 VISP Scope: 49 Cambodia Cameroon Chad Cote d’Ivoire Kenya Malawi Senegal Sudan Zimbabwe Angola Indonesia Mozambique Zambia Azerbaijan Kyrgyzstan Ukraine Madagascar Nigeria DR Congo Ghana Afghanistan 20
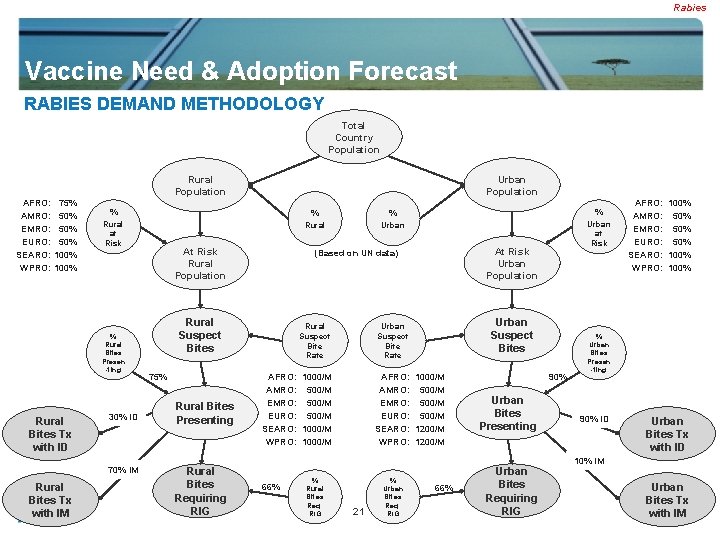
Rabies Vaccine Need & Adoption Forecast RABIES DEMAND METHODOLOGY Total Country Population Rural Population AFRO: 75% AMRO: 50% EURO: 50% SEARO: 100% WPRO: 100% % % Rural at Risk Rural Urban at Risk % Rural Bites Presen -ting Rural Bites Tx with ID 30% ID 70% IM Rural Bites Tx with IM Urban Population At Risk Rural Population Rural Suspect Bites 75% Rural Bites Presenting Rural Bites Requiring RIG At Risk Urban Population (Based on UN data) Rural Suspect Bite Rate AFRO: 1000/M AMRO: 500/M EURO: 500/M SEARO: 1000/M WPRO: 1000/M 66% % Rural Bites Req. RIG Urban Suspect Bites Urban Suspect Bite Rate AFRO: 1000/M AMRO: 500/M EURO: 500/M SEARO: 1200/M WPRO: 1200/M 21 % Urban Bites Req. RIG 66% 90% Urban Bites Presenting Urban Bites Requiring RIG AFRO: 100% AMRO: 50% EURO: 50% SEARO: 100% WPRO: 100% % Urban Bites Presen -ting 90% ID Urban Bites Tx with ID 10% IM Urban Bites Tx with IM

Rabies Vaccine Need & Adoption Forecast CURRENT COUNTRY SUPPORT AS PERCENT OF TOTAL NEED • Rabies experts provided estimate of current country-by-county vaccine usage as percent of total need* • Approximately 35% of total GAVIeligible country vaccine requirements are currently being supported by countries themselves 22 * F. Meslin, WHO & D. Briggs, Alliance for Rabies Control

Rabies Vaccine Need & Adoption Forecast GAVI VACCINE FINANCING POLICY • As a matter of policy, GAVI does not replace country funds already allocated to rabies vaccines • However, most countries are under resourcing rabies vaccination due to: – – High cost of vaccine and rabies immunoglobulin Vaccine and immunoglobulin supply shortages • Therefore, GAVI would only plan to financially support the difference between actual vaccine need and percent of need currently covered by country • This incremental need assessment will be complicated given a country’s level of funding in any given year is influenced by many factors and may differ year on year • This analysis is focused on GAVI-eligible countries’ total need 23
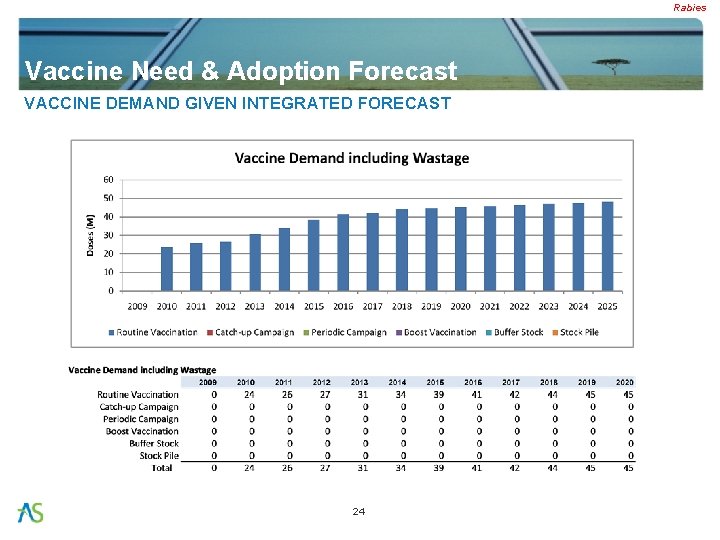
Rabies Vaccine Need & Adoption Forecast VACCINE DEMAND GIVEN INTEGRATED FORECAST 24

Rabies CONTENTS • Disease Overview • Vaccine Landscape • Vaccination Policy & Strategies • Vaccine Need & Adoption Forecast • Vaccine Cost Analysis • Implementation-Associated Cost Analysis • Analysis Summary • Key Resources 25
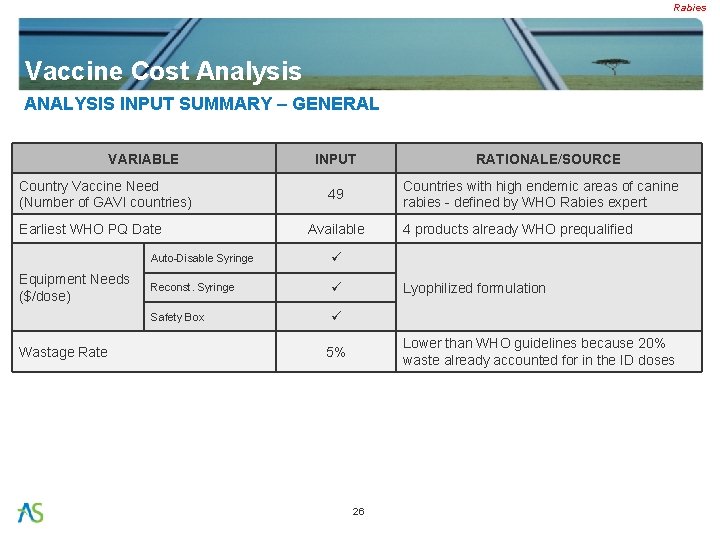
Rabies Vaccine Cost Analysis ANALYSIS INPUT SUMMARY – GENERAL VARIABLE Country Vaccine Need (Number of GAVI countries) Earliest WHO PQ Date Equipment Needs ($/dose) Wastage Rate INPUT Countries with high endemic areas of canine rabies - defined by WHO Rabies expert 49 Available Auto-Disable Syringe Reconst. Syringe Safety Box RATIONALE/SOURCE 4 products already WHO prequalified Lyophilized formulation Lower than WHO guidelines because 20% waste already accounted for in the ID doses 5% 26
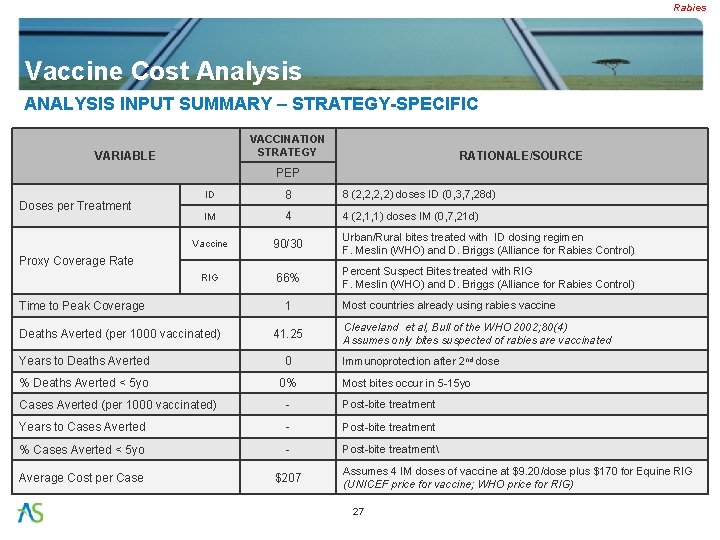
Rabies Vaccine Cost Analysis ANALYSIS INPUT SUMMARY – STRATEGY-SPECIFIC VACCINATION STRATEGY VARIABLE RATIONALE/SOURCE PEP Doses per Treatment ID 8 8 (2, 2, 2, 2) doses ID (0, 3, 7, 28 d) IM 4 4 (2, 1, 1) doses IM (0, 7, 21 d) Vaccine 90/30 Urban/Rural bites treated with ID dosing regimen F. Meslin (WHO) and D. Briggs (Alliance for Rabies Control) RIG 66% Percent Suspect Bites treated with RIG F. Meslin (WHO) and D. Briggs (Alliance for Rabies Control) Proxy Coverage Rate Time to Peak Coverage Deaths Averted (per 1000 vaccinated) 1 41. 25 Years to Deaths Averted 0 % Deaths Averted < 5 yo 0% Most countries already using rabies vaccine Cleaveland et al, Bull of the WHO 2002; 80(4) Assumes only bites suspected of rabies are vaccinated Immunoprotection after 2 nd dose Most bites occur in 5 -15 yo Cases Averted (per 1000 vaccinated) - Post-bite treatment Years to Cases Averted - Post-bite treatment % Cases Averted < 5 yo - Post-bite treatment Average Cost per Case $207 Assumes 4 IM doses of vaccine at $9. 20/dose plus $170 for Equine RIG (UNICEF price for vaccine; WHO price for RIG) 27

Rabies CONTENTS • Disease Overview • Vaccine Landscape • Vaccination Policy & Strategies • Vaccine Need & Adoption Forecast • Vaccine Cost Analysis – Post-Exposure Prophylaxis (PEP) – Additional Investment Alternatives • Implementation-Associated Cost Analysis • Analysis Summary • Key Resources 28
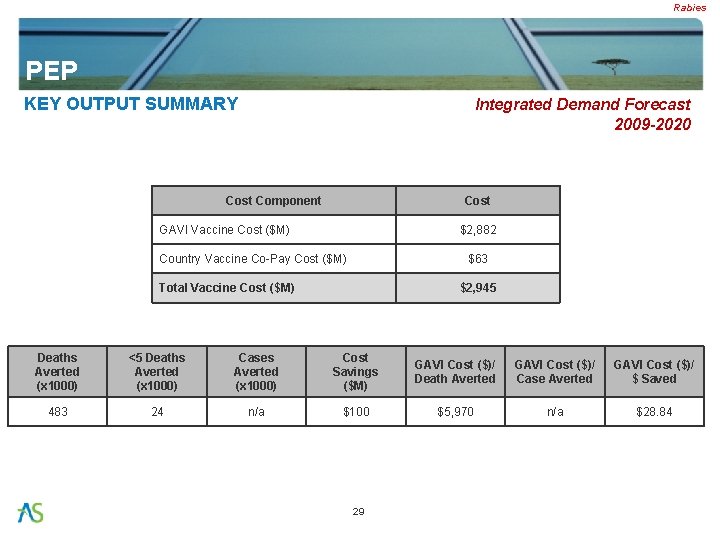
Rabies PEP KEY OUTPUT SUMMARY Integrated Demand Forecast 2009 -2020 Cost Component Cost GAVI Vaccine Cost ($M) $2, 882 Country Vaccine Co-Pay Cost ($M) $63 Total Vaccine Cost ($M) $2, 945 Deaths Averted (x 1000) <5 Deaths Averted (x 1000) Cases Averted (x 1000) Cost Savings ($M) GAVI Cost ($)/ Death Averted GAVI Cost ($)/ Case Averted GAVI Cost ($)/ $ Saved 483 24 n/a $100 $5, 970 n/a $28. 84 29
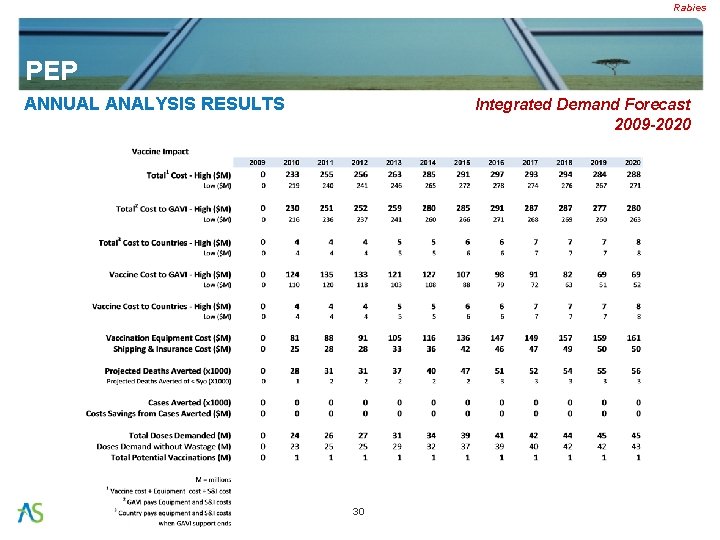
Rabies PEP ANNUAL ANALYSIS RESULTS Integrated Demand Forecast 2009 -2020 30

Rabies CONTENTS • Disease Overview • Vaccine Landscape • Vaccination Policy & Strategies • Vaccine Need & Adoption Forecast • Vaccine Cost Analysis – Post-Exposure Prophylaxis – Additional Investment Alternatives • Implementation-Associated Cost Analysis • Analysis Summary • Key Resources 31

Rabies Additional Investment Opportunities BACKGROUND OF UNDERSTANDING • Although rabies has the highest case fatality rate of any disease known to mankind, preventing human rabies in reality is not a complicated issue as it is one of the easiest diseases to prevent • There are three basic steps to preventing human rabies: 1. Increase educational awareness activities to reduce exposures, increase professional expertise and improve diagnoses and surveillance 2. Administer prompt and appropriate post-exposure prophylaxis (PEP) when exposures do occur 3. Conduct preventative immunization (Pre. P) to protect the relatively few numbers of the populace that are living in high-risk, remote geographic locations with no access to PEP • It has been proven in many countries that if and when these three steps are instituted the number of human rabies deaths quickly and dramatically decreases 32

Rabies Additional Investment Opportunities OPTION A* • Pilot PEP in Select Countries To Demonstrate GAVI Impact Experience over two decades has proven that reduced dose regimens for PEP are both efficacious and cost-effective – Updated Thai Red Cross (TRC) intradermal (ID) regimen or “ 2 -2 -2 -0 -2” is most cost effective PEP regimen recommended by WHO – TRC has increased access to vaccines in poor countries and has reduced the PEP cost per patient from 40 to 80% • Awareness as to the utilization and benefit of reduced dose regimens is lacking in most poor countries where it could be of great value • By piloting ID PEP in 4 select countries (2 in Asia & 2 in Africa), GAVI can demonstrate the health impact and cost reduction potential of this approach – With pilot PEP data, GAVI can then decide whether to extend support to all relevant GAVI-eligible countries • Estimated Total 5 -Year Budget: $6 M ($300 K/year/country) – Vaccine + RIG: $2 million – Service Delivery: $400, 000 annually – Social Mobilization/Education: $400, 000 annually 33 * Potentially eligible for GAVI Operations Research funding, if available

Rabies Additional Investment Opportunities OPTION B Support Rabies Education Programs to Reduce Disease Burden • Rabies is one of few diseases that can be dramatically reduced through increased education • Activities to improve educational awareness is inadequate in most poor developing countries • Educational initiatives specifically targeted for national governments, professional public health staff and the general public are clearly the least expensive of investments that would result in the highest return in lives saved within the shortest amount of time. • Education for professionals in poor developing countries would include: – – Use of intradermal (ID) administration for PEP Dissemination of latest WHO recommendations Increased awareness of source of rabies Training on modern diagnostic tools currently available for rabies confirmation • Estimated 5 -Year Budget per Country: $1. 2 M – Education materials development: $200, 000 – Education and training: $200, 000 annually 34

Rabies Additional Investment Opportunities OPTION C Fund RIG Capacity Building To Eliminate Shortages and Reduce costs • Rabies immunoglobulin (RIG) is the most expensive component of rabies treatment needed for approximately 66% of all suspect bites • Strengthening the production of equine RIG and supporting the development of alternative technologies to replace equine RIG is critical for ensuring adequate supplies and lowering the current price • Estimated 5 -Year Budget: $4 M – Equine RIG: $3 M – Monoclonal antibody preclinical and clinical testing: $1 M 35

Rabies CONTENTS • Disease Overview • Vaccine Landscape • Vaccination Policy & Strategies • Vaccine Need & Adoption Forecast • Vaccine Cost Analysis • Implementation-Associated Cost Analysis • Analysis Summary • Key Resources 36
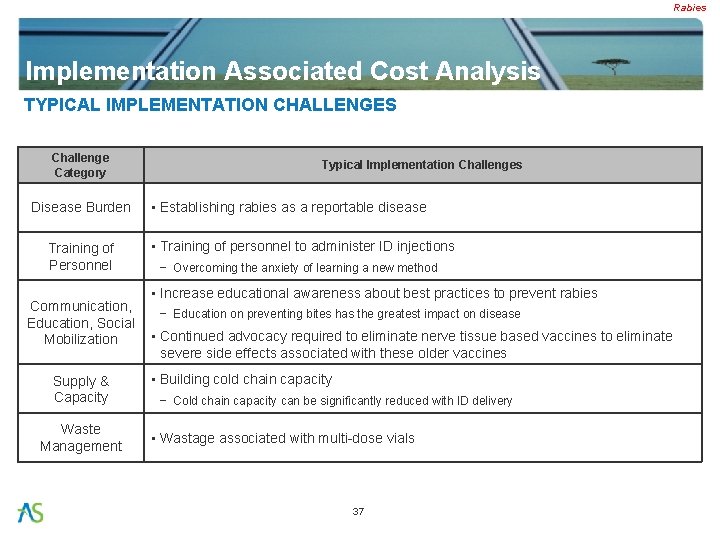
Rabies Implementation Associated Cost Analysis TYPICAL IMPLEMENTATION CHALLENGES Challenge Category Disease Burden Training of Personnel Typical Implementation Challenges • Establishing rabies as a reportable disease • Training of personnel to administer ID injections − Overcoming the anxiety of learning a new method • Increase educational awareness about best practices to prevent rabies Communication, − Education on preventing bites has the greatest impact on disease Education, Social • Continued advocacy required to eliminate nerve tissue based vaccines to eliminate Mobilization severe side effects associated with these older vaccines Supply & Capacity Waste Management • Building cold chain capacity − Cold chain capacity can be significantly reduced with ID delivery • Wastage associated with multi-dose vials 37

Rabies Implementation Associated Cost Analysis UNIQUE IMPLEMENTATION CHALLENGES Challenge Category Unique Implementation Challenges • Non-EPI vaccination – animal (mostly dog) bite driven vaccination strategy • Potential need to integrate vaccination strategy with other rabies control programs • Vaccination strategy needs to include patient access via rabies ‘dog bite treatment centers at central as well as district hospitals Vaccination Strategy • Continued advocacy required to support promotion of: − The use of economical and safe multisite ID post exposure prophylaxis − Discontinuation of production and use of nerve tissue based vaccines • Major vaccine shortage for safe cell culture based vaccines • Severe bite cases require vaccination and rabies immunoglobulin (RIG) – Major RIG shortage • Significant facility and staff training upgrades required (e. g. wound wash stations) Vaccination Access Via Rabies • Need to promote and train medics overcome resistance to ID vs. IM vaccine Bite Centers administration 38
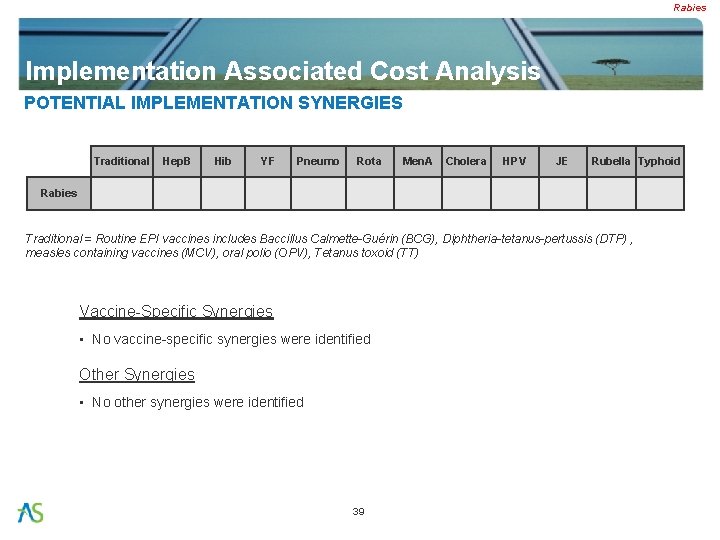
Rabies Implementation Associated Cost Analysis POTENTIAL IMPLEMENTATION SYNERGIES Traditional Hep. B Hib YF Pneumo Rota Men. A Cholera HPV JE Rubella Typhoid Rabies Traditional = Routine EPI vaccines includes Baccillus Calmette-Guérin (BCG), Diphtheria-tetanus-pertussis (DTP) , measles containing vaccines (MCV), oral polio (OPV), Tetanus toxoid (TT) Vaccine-Specific Synergies • No vaccine-specific synergies were identified Other Synergies • No other synergies were identified 39
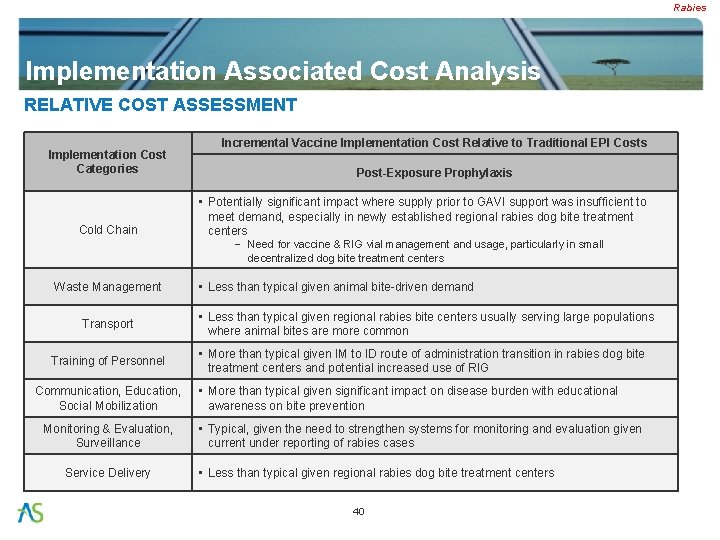
Rabies Implementation Associated Cost Analysis RELATIVE COST ASSESSMENT Implementation Cost Categories Cold Chain Incremental Vaccine Implementation Cost Relative to Traditional EPI Costs Post-Exposure Prophylaxis • Potentially significant impact where supply prior to GAVI support was insufficient to meet demand, especially in newly established regional rabies dog bite treatment centers − Need for vaccine & RIG vial management and usage, particularly in small decentralized dog bite treatment centers Waste Management Transport Training of Personnel Communication, Education, Social Mobilization Monitoring & Evaluation, Surveillance Service Delivery • Less than typical given animal bite-driven demand • Less than typical given regional rabies bite centers usually serving large populations where animal bites are more common • More than typical given IM to ID route of administration transition in rabies dog bite treatment centers and potential increased use of RIG • More than typical given significant impact on disease burden with educational awareness on bite prevention • Typical, given the need to strengthen systems for monitoring and evaluation given current under reporting of rabies cases • Less than typical given regional rabies dog bite treatment centers 40
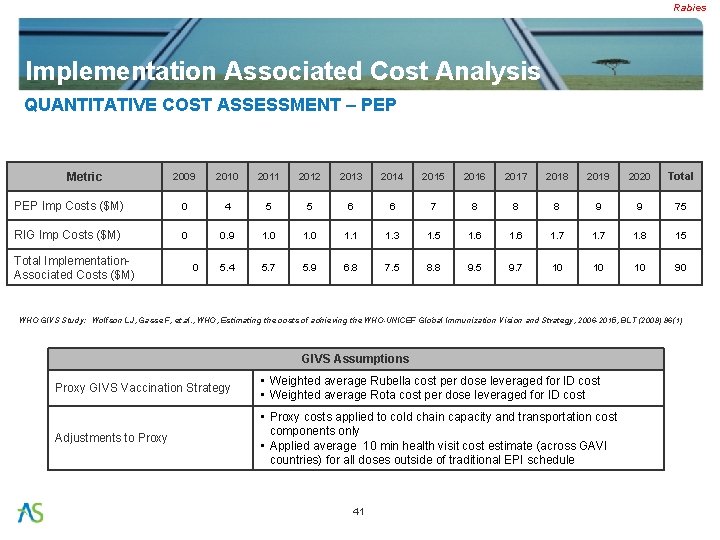
Rabies Implementation Associated Cost Analysis QUANTITATIVE COST ASSESSMENT – PEP Metric 2009 2010 2011 2012 2013 2014 2015 2016 2017 2018 2019 2020 Total PEP Imp Costs ($M) 0 4 5 5 6 6 7 8 8 8 9 9 75 RIG Imp Costs ($M) 0 0. 9 1. 0 1. 1 1. 3 1. 5 1. 6 1. 7 1. 8 15 0 5. 4 5. 7 5. 9 6. 8 7. 5 8. 8 9. 5 9. 7 10 10 10 90 Total Implementation. Associated Costs ($M) WHO GIVS Study: Wolfson LJ, Gasse F, et. al. , WHO, Estimating the costs of achieving the WHO-UNICEF Global Immunization Vision and Strategy, 2006 -2015, BLT (2008) 86(1) GIVS Assumptions Proxy GIVS Vaccination Strategy • Weighted average Rubella cost per dose leveraged for ID cost • Weighted average Rota cost per dose leveraged for ID cost Adjustments to Proxy • Proxy costs applied to cold chain capacity and transportation cost components only • Applied average 10 min health visit cost estimate (across GAVI countries) for all doses outside of traditional EPI schedule 41
Rabies CONTENTS • Disease Overview • Vaccine Landscape • Vaccination Policy & Strategies • Vaccine Need & Adoption Forecast • Vaccine Cost Analysis • Implementation-Associated Cost Analysis • Analysis Summary • Key Resources 42
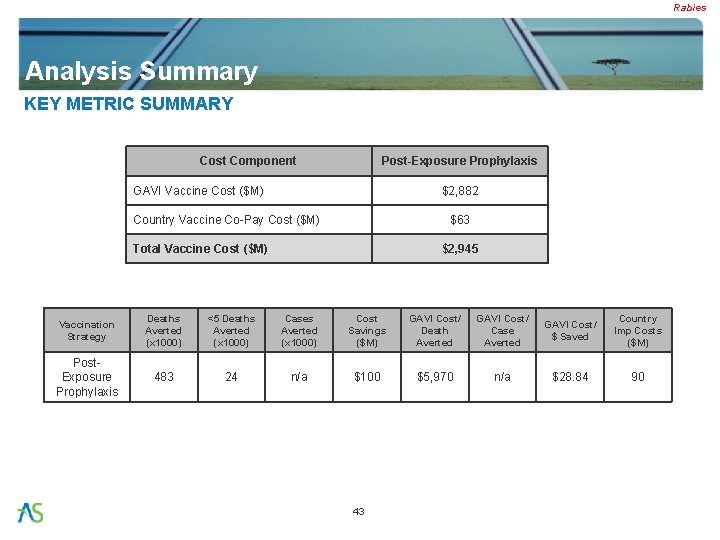
Rabies Analysis Summary KEY METRIC SUMMARY Cost Component Post-Exposure Prophylaxis GAVI Vaccine Cost ($M) $2, 882 Country Vaccine Co-Pay Cost ($M) $63 Total Vaccine Cost ($M) $2, 945 Vaccination Strategy Deaths Averted (x 1000) <5 Deaths Averted (x 1000) Cases Averted (x 1000) Cost Savings ($M) GAVI Cost/ Death Averted GAVI Cost/ Case Averted GAVI Cost/ $ Saved Country Imp Costs ($M) Post. Exposure Prophylaxis 483 24 n/a $100 $5, 970 n/a $28. 84 90 43
Rabies CONTENTS • Disease Overview • Vaccine Landscape • Vaccination Policy & Strategies • Vaccine Need & Adoption Forecast • Vaccine Cost Analysis • Implementation-Associated Cost Analysis • Analysis Summary • Key Resources 44

Rabies Key Resources EXPERT CONSULTATION • Vaccine Experts – Francois Meslin, Head of Zoonotic Disease, WHO – Deborah Briggs, Board of Directors, Alliance for Rabies Control – Sarah Cleaveland, Board of Directors, Alliance for Rabies Control – Charles Rupprecht, Chief of the CDC Rabies Program, CDC – Herve Bourhy, Director of the WHO Collaborating Center for Rabies, Institute Pasteur • Suppliers – Michael Attlan, Marketing Director, Sanofi – Shawn Gilchrist, Sanofi – Ferdinando Borgese, Global Brand Manager, Novartis Vaccine – John-Kenneth Billingsley, Executive Director, Novartis – Olga Popova, Director Government Affairs, Crucell 45

RABIES Appendix REFERENCES (I) 1. Plotkin et al, Vaccines, 5 th Edition, Chap. 27, 2008. 2. Weekly Epi Record, No. 49/50, 2007, 82, 425 -436, 7 Dec 07. 3. WHO, Disease Outbreak News, Rabies, www. who. int/csr/don/archive/disease/Rabies_disease/en/, Mar 2008. 4. WHO, Essential rabies maps, www. who. int/rabies_maps/en/index. html. 5. WHO, Rab. Net, “Human rabies, number of people bitten by suspected dogs per 100, 000 population, ” www. who. int/globalatlas/default. asp, Mar 08 (data is incomplete or not reported). 6. WHO, Rab. Net, Human rabies deaths, www. who. int/globalatlas/default. asp, Mar 08 [Mortality = Average of reported deaths between 2000 -2007]. 7. UN Population Division, World Population Prospects: The 2006 revision population database, esa. un. org/unpp/index. asp? panel=2 [Mortality Rate = Mortality/(Population) x 1, 000]. 8. Plotkin et al, Vaccines, 5 th Edition, Chap. 27, 2008. 46

RABIES Appendix REFERENCES (II) 9. WHO Prequalified Vaccines, www. who. int/immunization_standards/vaccine_quality/pq_suppliers/en/index. html. 10. Rab. Avert Package Insert, www. novartis-vaccines. com/products/Rabavert_PI_0404. pdf; www. novartisvaccines. com/products/travel. shtml. 11. Imovax Product Insert, www. vaccineshoppe. com/image. cfm? doc_id=5983&image_type=product_pdf. 12. Travel Med Infect Dis. 2007 Nov; 5(6): 327 -48. Epub 2007 Sep 17; www. ncbi. nlm. nih. gov/pubmed/17983973. 13. Berna Biotech recently transferred to Cadila Health Ltd, an Indian manufacturer; Zyddus Cadila is currently seeking WHO prequalification according to Partners for Rabies Prevention Informal Group; RIG = rabies immunoglobulin; TRC Verorab (Thai Red Cross) 14. Plotkin et al, Vaccines, 5 th Edition, Chap. 27, 2008. 15. Correspondence with WHO and Partners for Rabies Prevention Informal Group (PRP), June-August 2008. 47

RABIES Appendix REFERENCES (III) 16. WHO, Weekly Epidemiology Record, No. 49/50, 2007, 82, 425 -436, 7 Dec 07. 17. Le. Guerrier P, et al, Pre-exposure rabies prophylaxis for the international traveler: a decision analysis; Vaccine, 14(2): 167 -176, 1996. 18. Chulasugandha P, et al, Cost comparison of rabies pre-exposure vaccination with postexposure treatment in Thai children, Vaccine, 24(9): 1478 -1482, 2006. 48
- Slides: 48