Gases Why does a plastic bottle left in

Gases • Why does a plastic bottle left in a car overnight look crushed on a chilly morning? • How hard is air pushing on your body? • How long will the Sun shine? © 2014 Pearson Education, Inc.

Be sure you know how to: • Draw force diagrams (Section 2. 1). • Use Newton's second and third laws to analyze interactions of objects (Section 2. 8). • Use the impulse-momentum principle (Section 5. 3). © 2014 Pearson Education, Inc.

What's new in this chapter • When we studied energy, we found that in many processes some of the mechanical energy of a system is transformed into internal energy, resulting in a change in the temperature of the interacting objects. • One goal of this chapter is to investigate the connection between temperature and internal energy. – The key to this connection lies in understanding the internal structure of matter. © 2014 Pearson Education, Inc.

Structure of matter • Imagine that you dip a cotton ball in rubbing alcohol and wipe it across a piece of paper. – The wet alcohol strip disappears gradually, with the edges of the strip disappearing first. • Because the alcohol disappeared gradually, it is reasonable to suggest that it is made of "pieces" too small to be seen. – If the alcohol were composed of one piece, it would disappear all at once. © 2014 Pearson Education, Inc.

Three possible mechanisms that explain how the alcohol disappears • The little pieces of liquid move to the inside of the paper and are still there, even though the paper looks dry. • The air surrounding the paper somehow pulls the liquid pieces out of the paper. • The pieces of liquid are moving—they bump into each other and slowly bump each other out of the paper one by one. © 2014 Pearson Education, Inc.

Testing various mechanisms for the drying of wet objects © 2014 Pearson Education, Inc.

Testing various mechanisms for the drying of wet objects © 2014 Pearson Education, Inc.

Testing various mechanisms for the drying of wet objects © 2014 Pearson Education, Inc.

Conclusions from the testing experiments • Based on these experiments, it is reasonable to assume that alcohol and other liquids are composed of smaller objects, called particles, that move randomly in all directions. – These particles need empty space between them so that particles of other materials can move between them. – This model of the internal structure of alcohol can be used to explain many other phenomena that we encounter. © 2014 Pearson Education, Inc.

The particle model explains how we can smell things even when we are not near them • Suppose we open a bottle of perfume. Several minutes later, people all over the room can smell the perfume. • According to the particle model, the particles of perfume leave the bottle and gradually disperse, eventually arriving at our nostrils. • Because everyone in the room eventually smells the perfume, the perfume particles must move in all directions. © 2014 Pearson Education, Inc.

Testing the hypothesis that air has mass • Take two rigid metal cylinders—one from which the air has been evacuated with a vacuum pump, and the other filled with air. Weigh the cylinders. – If the hypothesis that air has mass is correct, the evacuated jar should weigh less than the unevacuated jar. © 2014 Pearson Education, Inc.

Gases, liquids, and solids • Gases are easy to compress, whereas liquids and solids are almost incompressible. • The particle model helps us explain this difference: the amount of empty space between the particles is different in solids, liquids, and gases. © 2014 Pearson Education, Inc.

Gases, liquids, and solids • Gases tend to occupy whatever volume is available. • In contrast, if we move the liquid filling a small container to a much larger container, the liquid volume remains the same independent of the container's shape. • Solids maintain not only their volume but also their shape. © 2014 Pearson Education, Inc.
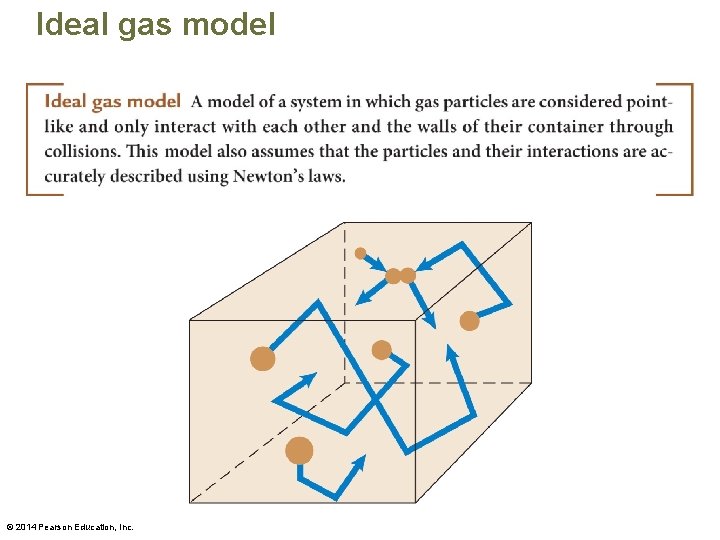
Ideal gas model © 2014 Pearson Education, Inc.

Ideal gas model • Ideal in this context does not mean perfect, but rather simplified. The ideal gas model is a simplified model with certain assumptions. – Whether this model can be used to represent a real gas needs to be determined via testing experiments. – We need physical quantities to represent the features and behavior of the model. Then we can use it to develop descriptions, explanations, and predictions of new phenomena. © 2014 Pearson Education, Inc.

Pressure • As air particles move randomly in space, they eventually collide with the solid surfaces of any objects in that space. In each of these collisions, the particle exerts an impulsive force on the object—like a tennis ball hitting a practice wall. © 2014 Pearson Education, Inc.
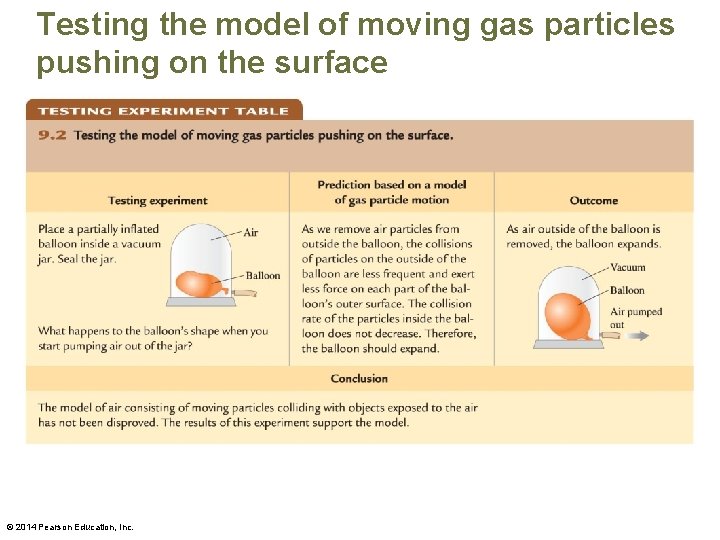
Testing the model of moving gas particles pushing on the surface © 2014 Pearson Education, Inc.

Pressure • The SI unit of pressure is the pascal (Pa), where 1 Pa = 1 N/m 2. © 2014 Pearson Education, Inc.
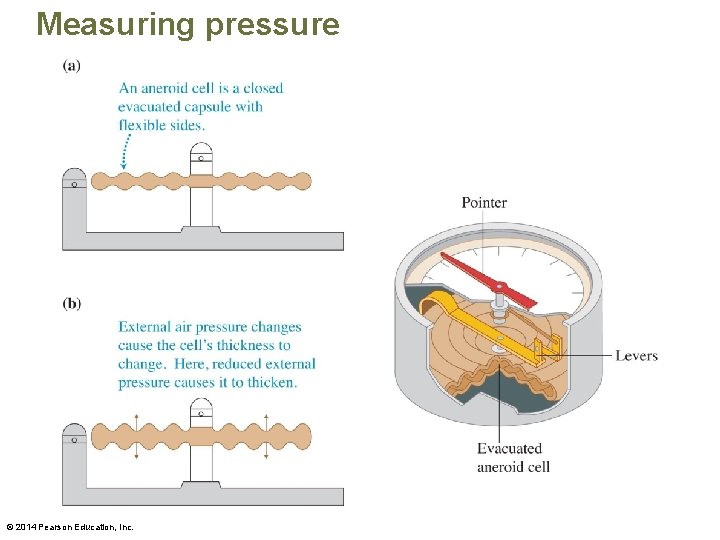
Measuring pressure © 2014 Pearson Education, Inc.

Gauge pressure • When you use a tire gauge to measure the air pressure in a car tire, you are comparing the pressure inside the tire to the pressure of the atmosphere outside the tire. © 2014 Pearson Education, Inc.

Density • For gases, a much more useful physical quantity than mass of the individual particles is the mass of one unit of volume—density. © 2014 Pearson Education, Inc.

Tip © 2014 Pearson Education, Inc.

Mass and size of particles • In 1811, an Italian scientist named Avogadro proposed that equal volumes of different types of gas, when at the same temperature and pressure, contain the same number of gas particles. • The mass in grams of any substance that has exactly Avogadro's number of particles is equal to the atomic mass of that substance. © 2014 Pearson Education, Inc.

Avogadro's number and the mole © 2014 Pearson Education, Inc.

Quantitative analysis of an ideal gas • We need more simplifying assumptions. – Assume that the particles do not collide with each other—they collide only with the walls of the container, exerting pressure on the walls. This is a reasonable assumption for a gas of low density. – Assume that the collisions of particles with the walls are elastic. This makes sense, because the pressure of the gas in a closed container remains constant, which would not happen if the particles' kinetic energy decreased. © 2014 Pearson Education, Inc.
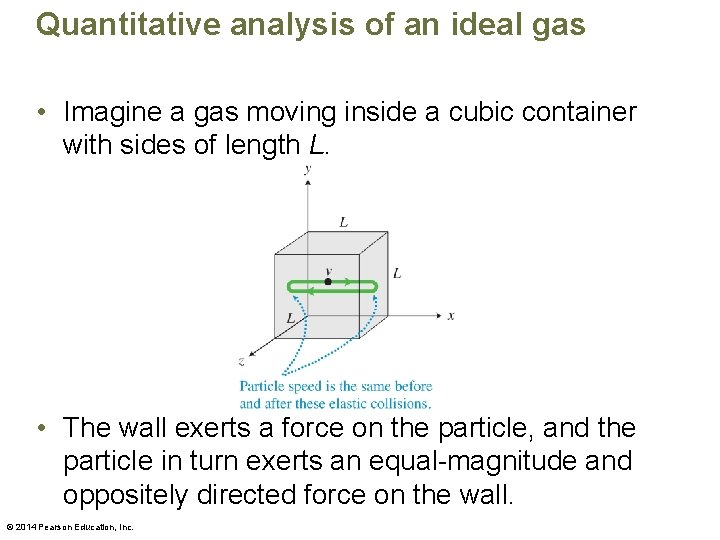
Quantitative analysis of an ideal gas • Imagine a gas moving inside a cubic container with sides of length L. • The wall exerts a force on the particle, and the particle in turn exerts an equal-magnitude and oppositely directed force on the wall. © 2014 Pearson Education, Inc.
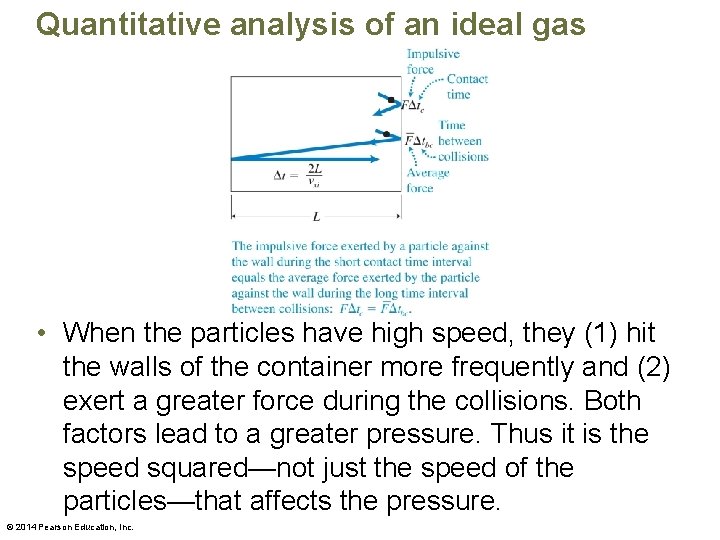
Quantitative analysis of an ideal gas • When the particles have high speed, they (1) hit the walls of the container more frequently and (2) exert a greater force during the collisions. Both factors lead to a greater pressure. Thus it is the speed squared—not just the speed of the particles—that affects the pressure. © 2014 Pearson Education, Inc.

Tip © 2014 Pearson Education, Inc.

Time interval between collisions • Why does it take 5 to 10 minutes for the smell of perfume to travel across a room if the average speed of air particles is v = 480 m/s? – The average distance between particles in a gas is about D = 3. 3 x 10– 7 cm. – Perhaps we cannot assume that the gas particle collisions can be ignored—in fact, they collide about 109 times per second. – Even though the gas particles are moving fast, their migration from one place to another is slow. © 2014 Pearson Education, Inc.
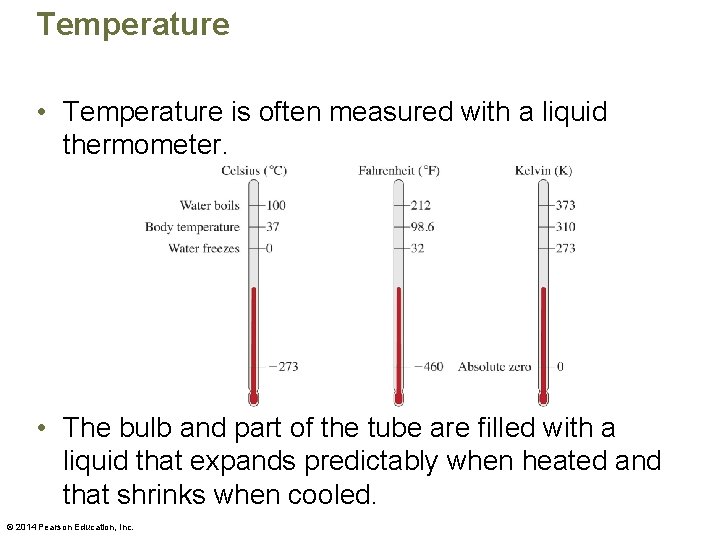
Temperature • Temperature is often measured with a liquid thermometer. • The bulb and part of the tube are filled with a liquid that expands predictably when heated and that shrinks when cooled. © 2014 Pearson Education, Inc.

Connecting properties of matter to its temperature © 2014 Pearson Education, Inc.

Connecting properties of matter to its temperature • In the experiment, we can hypothesize that the ball expands because the impulses of the particles against the inside walls are larger when the gas is warm. – This would happen if the particles were moving faster. – Based on this reasoning, we can hypothesize that the temperature of a gas is related to the speed of the random motion of its particles. © 2014 Pearson Education, Inc.
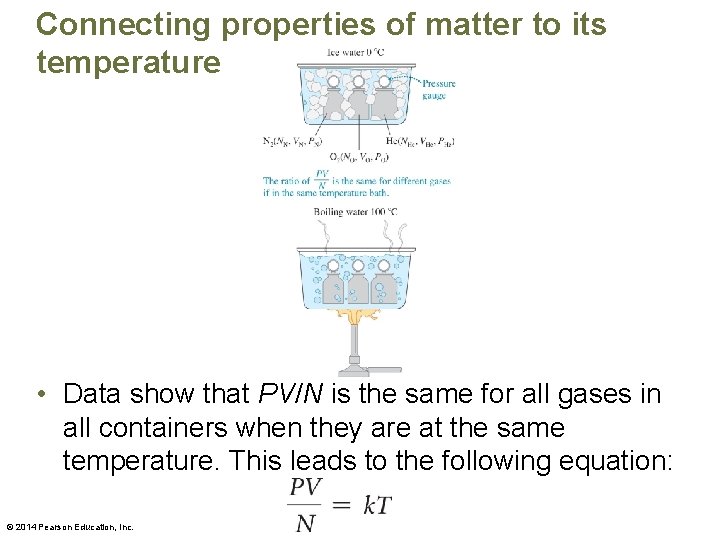
Connecting properties of matter to its temperature • Data show that PV/N is the same for all gases in all containers when they are at the same temperature. This leads to the following equation: © 2014 Pearson Education, Inc.

Finding the constant "k" • We now have two equations with two unknowns: the constant k and the water temperature T. • We subtract the first equation from the second to get k = 1. 38 x 10– 23 J/degree. © 2014 Pearson Education, Inc.

Absolute (Kelvin) temperature scale and ideal gas law • We need a scale on which the zero point is the lowest possible temperature. That way, all temperatures will be positive. • The lowest possible temperature on the new scale is 0; on the Celsius scale, it would be – 270 °C. • This temperature scale is called the absolute temperature scale or the Kelvin scale. It was invented by William Thomson, Lord Kelvin, in 1848. © 2014 Pearson Education, Inc.

Ideal gas law • Because one mole has Avogadro's number of particles, N = n. NA. Substituting this, we get: © 2014 Pearson Education, Inc.

Ideal gas law © 2014 Pearson Education, Inc.

Temperature and particle motion • We can now connect the average kinetic energy of the gas particles to the absolute temperature of the gas: • The temperature of a gas is an indication of the average random translational kinetic energy of the particles in the ideal gas. © 2014 Pearson Education, Inc.

Temperature and particle motion • You have two containers holding identical gases that have been sitting in the same room for a long time. One container is large and the other one is small. Which one has a higher temperature? – Because the average kinetic energy per particle is the same in each container, the temperatures of the two gases are the same. – The total kinetic energy of the particles in the large container is larger because it contains more particles. © 2014 Pearson Education, Inc.

Temperature and particle motion • What will happen if you mix a container of hot gas with a container of cold gas? – The faster-moving particles of the hot gas will collide with the slower-moving particles of the cold gas. Following a collision, on average the faster-moving particle will be moving more slowly than before, and the slower particle will be moving more rapidly. – Eventually, the particles of the two gases will have the same average kinetic energy and, therefore, the same temperature. – This is called thermal equilibrium. © 2014 Pearson Education, Inc.

Tip © 2014 Pearson Education, Inc.
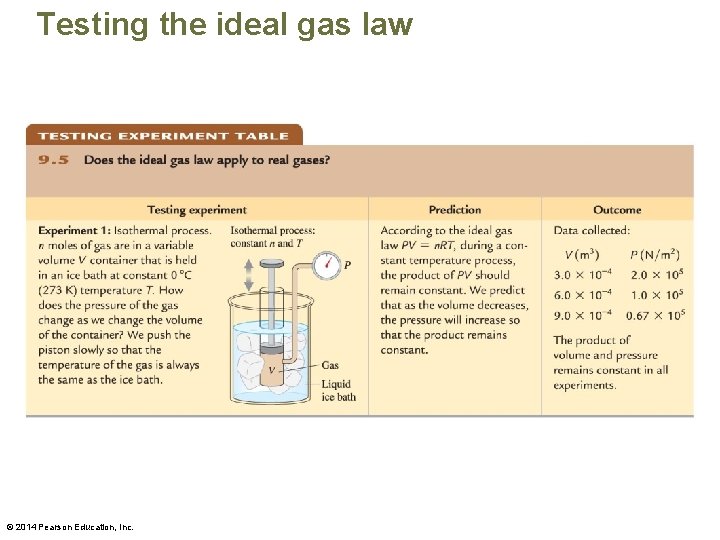
Testing the ideal gas law © 2014 Pearson Education, Inc.

Testing the ideal gas law © 2014 Pearson Education, Inc.

Testing the ideal gas law © 2014 Pearson Education, Inc.

A summary of ideal gas law processes © 2014 Pearson Education, Inc.

Reflection on the process of construction of knowledge for the ideal gas law • The first step was to construct a simplified model of a system that could represent a real gas—the ideal gas model. – This involved making assumptions about the internal structure of gases. – The model was based partly on observations and partly on our knowledge of particle motion and interactions. • We used this model to devise a mathematical description of the behavior of gases, the ideal gas law. © 2014 Pearson Education, Inc.

Reflection on the process of construction of knowledge for the ideal gas law (continued) • We then tested the model's applicability to real gases by using it to predict how macroscopic quantities describing the gas would change during specific processes. – Macroscopic quantities: temperature, pressure, volume, and the amount of gas – Processes: isothermal, isobaric, and isochoric – Ideal gas law: used to construct equations that described those processes • These predictions were consistent with the outcomes of the new testing experiments. © 2014 Pearson Education, Inc.
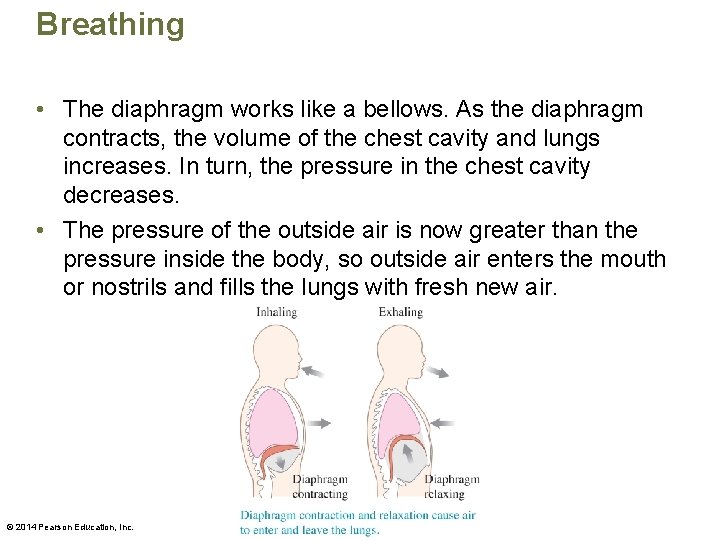
Breathing • The diaphragm works like a bellows. As the diaphragm contracts, the volume of the chest cavity and lungs increases. In turn, the pressure in the chest cavity decreases. • The pressure of the outside air is now greater than the pressure inside the body, so outside air enters the mouth or nostrils and fills the lungs with fresh new air. © 2014 Pearson Education, Inc.
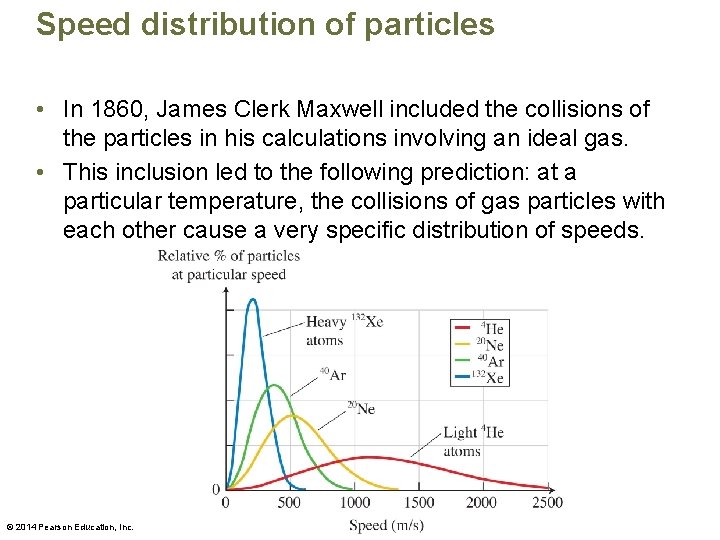
Speed distribution of particles • In 1860, James Clerk Maxwell included the collisions of the particles in his calculations involving an ideal gas. • This inclusion led to the following prediction: at a particular temperature, the collisions of gas particles with each other cause a very specific distribution of speeds. © 2014 Pearson Education, Inc.
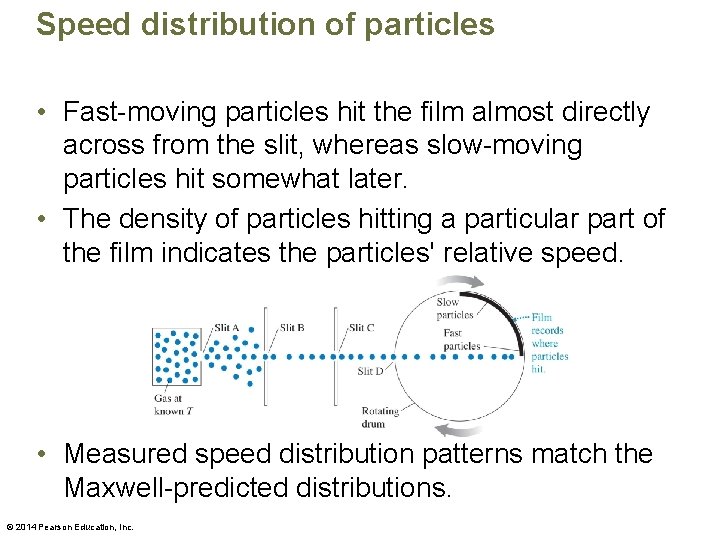
Speed distribution of particles • Fast-moving particles hit the film almost directly across from the slit, whereas slow-moving particles hit somewhat later. • The density of particles hitting a particular part of the film indicates the particles' relative speed. • Measured speed distribution patterns match the Maxwell-predicted distributions. © 2014 Pearson Education, Inc.

Limitations of the ideal gas law • For real gases such as air, measurements of pressure and volume at conditions of normal pressure and temperature are consistent with predictions made by the ideal gas law. – At very high pressures or very low temperatures, real measurements differ from those predictions. – The ideal gas law describes gases accurately only over certain temperature and pressure ranges. © 2014 Pearson Education, Inc.

Tip © 2014 Pearson Education, Inc.

Tip © 2014 Pearson Education, Inc.

Thermal energy and the Sun: Putting it all together • The maximum possible lifetime for the Sun if it converts its thermal energy into forms of radiation is 40 million years. – Yet we know that the geological age of Earth 4. 5 billion years! – This simple estimate suggests either that the Sun's material cannot be modeled as an ideal gas or that another energy source within the Sun far exceeds thermal energy present. © 2014 Pearson Education, Inc.

Summary © 2014 Pearson Education, Inc.

Summary © 2014 Pearson Education, Inc.

Summary © 2014 Pearson Education, Inc.

Summary © 2014 Pearson Education, Inc.

Summary © 2014 Pearson Education, Inc.

Summary © 2014 Pearson Education, Inc.

Summary © 2014 Pearson Education, Inc.
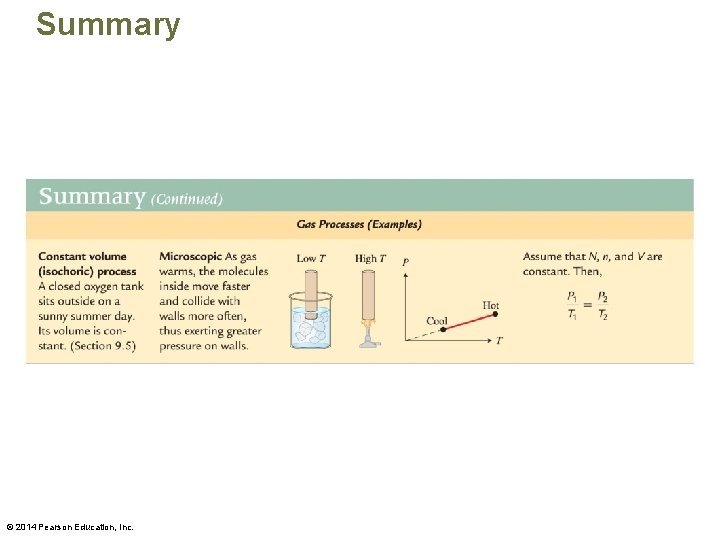
Summary © 2014 Pearson Education, Inc.

Summary © 2014 Pearson Education, Inc.
- Slides: 63