Gases Volume and Moles Avogadros Law General Organic



















- Slides: 20

Gases Volume and Moles (Avogadro’s Law) General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 1

Avogadro's Law: Volume and Moles Avogadro’s law states that § the volume of a gas is directly related to the number of moles (n) of gas § T and P are constant V 1 = V 2 n 1 n 2 General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 2

Learning Check If 0. 75 mole of helium gas occupies a volume of 1. 5 L, what volume will 1. 2 moles of helium occupy at the same temperature and pressure? V 1 = V 2 n 1 n 2 General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 3

Solution Answer : 2. 4 L STEP 1 Conditions 2 Know Predict V 1 = 1. 5 L V 2 = ? V increases n 1 = 0. 75 mole n 2 = 1. 2 moles n increases STEP 2 Solve for unknown V 2: V 2 = V 1 x n 2 n 1 STEP 3 Substitute values and solve for V 2: V 2 = 1. 5 L x 1. 2 moles He = 2. 4 L 0. 75 mole He General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 4

STP The volumes of gases can be compared at STP (Standard Temperature and Pressure) when they have § the same temperature Standard temperature (T) § the same pressure Standard pressure (P) = 0 °C or 273 K = 1 atm (760 mm. Hg) General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 5

Molar Volume The molar volume of a gas § is measured at STP (standard temperature and pressure) § is 22. 4 L for 1 mole of any gas General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 6

Molar Volume as a Conversion Factor The molar volume at STP § has about the same volume as 3 basketballs § can be used to form 2 conversion factors: 22. 4 L and 1 mole 22. 4 L General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 7

Guide to Using Molar Volume General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 8

Using Molar Volume What is the volume occupied by 2. 75 moles of N 2 gas at STP? STEP 1 Given: 2. 75 moles of N 2 Need: Liters of N 2 STEP 2 Write a plan: Use the molar volume to convert moles to liters. General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 9

Using Molar Volume (continued) STEP 3 Write equalities and conversion factors: 1 mole of gas = 22. 4 L 1 mole gas and 22. 4 L 1 mole gas STEP 4 Substitute data and solve: 2. 75 moles N 2 x 22. 4 L = 61. 6 L of N 2 1 mole N 2 General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 10

Learning Check What is the volume at STP of 4. 00 g of CH 4? General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 11

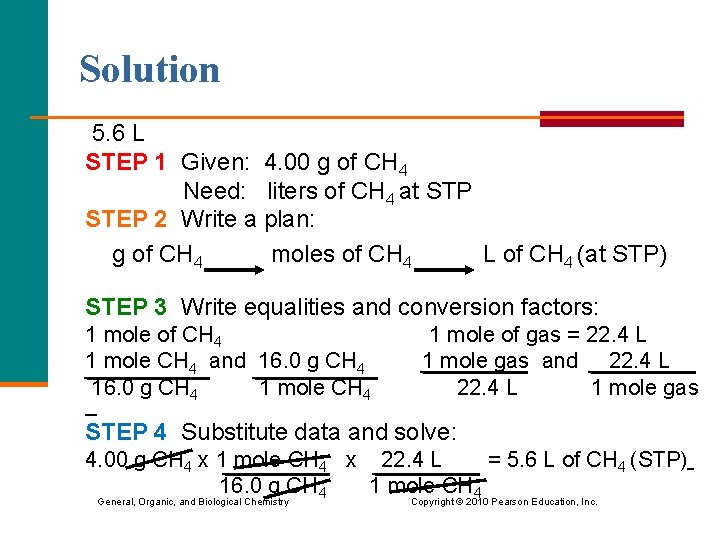
Solution 5. 6 L STEP 1 Given: 4. 00 g of CH 4 Need: liters of CH 4 at STP STEP 2 Write a plan: g of CH 4 moles of CH 4 L of CH 4 (at STP) STEP 3 Write equalities and conversion factors: 1 mole of CH 4 1 mole of gas = 22. 4 L 1 mole CH 4 and 16. 0 g CH 4 1 mole gas and 22. 4 L 16. 0 g CH 4 1 mole CH 4 22. 4 L 1 mole gas STEP 4 Substitute data and solve: 4. 00 g CH 4 x 1 mole CH 4 x 22. 4 L = 5. 6 L of CH 4 (STP) 16. 0 g CH 4 1 mole CH 4 General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc.

Learning Check How many grams of He are present in 8. 00 L of He at STP? General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 13

Solution 1. 43 g of He STEP 1 Given: 8. 00 L of He Need: grams of He at STP STEP 2 Write a plan: L of He moles of He g of He (at STP) STEP 3 Write equalities and conversion factors: 1 mole of gas = 22. 4 L 1 mole of He = 4. 00 g of He 1 mole gas and 22. 4 L 1 mole He and 4. 00 g He 22. 4 L 1 mole gas 4. 00 g He 1 mole CH 4 STEP 4 Substitute data and solve: 8. 00 L He x 1 mole He x 4. 00 g He = 1. 43 g of He 22. 4 L He 1 mole He General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 14

Gases in Chemical Reactions The volume or amount of a gas at STP in a chemical reaction can be calculated from § STP conditions § mole factors from the balanced equation General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 15

Guide to Problem Solving Reactions Involving Gases General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 16

Example of Gases in Equations What volume (L) of O 2 gas is needed to completely react with 15. 0 g of aluminum at STP? 4 Al(s) + 3 O 2 (g) 2 Al 2 O 3(s) General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 17

Solution for Example of Gases in Equations What volume (L) of O 2 gas is needed to completely react with 15. 0 g of aluminum at STP? 4 Al(s) + 3 O 2 (g) 2 Al 2 O 3(s) STEP 1 Find moles of Al using molar mass. STEP 2 Determine moles of O 2 using mole–mole factor. STEP 3 Convert moles of O 2 to L using molar volume. (STEP 1) (STEP 2) (STEP 3) 15. 0 g Al x 1 mole Al x 3 moles O 2 x 22. 4 L (STP) 27. 0 g Al 4 moles Al 1 mole O 2 = 9. 33 L of O 2 (at STP) General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 18

Learning Check What mass of Fe will react with 5. 50 L of O 2 at STP? 4 Fe(s) + 3 O 2(g) 2 Fe 2 O 3(s) General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 19

Solution STEP 1 Find moles of O 2 using molar volume. STEP 2 Determine moles of Fe using mole–mole factor. STEP 3 Convert moles of Fe to grams using molar mass. (STEP 1) (STEP 2) (STEP 3) 5. 50 L O 2 x 1 mole O 2 x 4 moles Fe x 55. 9 g Fe 22. 4 L O 2 3 moles O 2 1 mole Fe = 18. 3 g of Fe General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 20